Abstract
Background
Although colonoscopy is considered the most effective tool for reducing colorectal cancer-related morbidity, the age at which average-risk individuals begin colonoscopic screening is undetermined. This study aimed to compare the adenoma and advanced adenoma detection rates according to age and sex in a large average-risk population in the rural areas of Eastern China.
Methods
This observational, single-center, retrospective study included patients with average colorectal cancer risk and examined the adenoma and advanced adenoma detection rates using age intervals of 5 years. We also compared the size and age of patients with and without advanced adenoma.
Results
We included 18 928 patients with a median age of 54 years (range 15–90 years), including 10 143 men and 8785 women. The adenoma and advanced adenoma detection rates were 17.08% and 5.24%, respectively, and increased with age in the whole population. The adenoma detection rates increased from 8.97% (aged 40–44) to 14.98% (aged 45–49) and 6.24% (aged 45–49) to 11.00% (aged 50–54) in men and women (both
Conclusion
The adenoma and advanced adenoma detection rates increased significantly in average-risk population aged 45 years and older, especially in men.
Keywords
Introduction
Colorectal cancer (CRC) is one of the most common cancers worldwide, with significantly increased morbidity and mortality rates in China over the last few decades.1,2 The etiopathogenesis of most CRCs follows a sequence of events that have been described as the adenoma-carcinoma sequence.3,4 This sequence typically takes more than 10 years to complete in sporadic cancers and can be effectively disrupted via screening. 5 Colonoscopy is considered the most efficacious screening method for reducing CRC incidence and mortality because it allows for the early detection and removal of precancerous polyps and the detection of early-stage CRC.1,6 The US Multi-Society Task Force (MSTF) for colorectal cancer recommends initiating CRC screening at 50 years of age in average-risk populations, except African Americans, who should begin screening at 45 years of age. 5 The National Cancer Center of China also recommends screening for CRC starting at the age of 50 years for average-risk individuals, but the screening options vary widely. 7 Owing to the discomfort, complications, and cost of a colonoscopy, it is rarely used for initial screening in the average-risk population. 7 Previous study showed that colorectal adenoma and advanced neoplasia detection rates increase from 45 years of age in the European population. 8 However, reliable data on the risk of precancerous polyps and early-stage CRC in other populations are rare, especially in average-risk individuals under 50.5,7 This makes it difficult to recommend an appropriate age for colonoscopic screening in average-risk populations.
The incidences of CRC and adenoma vary according to age, sex, and region.9-11 To improve the time- and cost-effectiveness of colonoscopy and reduce complications, it is worth discussing the age at which screening using colonoscopy is initiated in different regions and sexes.
This study aimed to determine the adenoma detection rate (ADR) and advanced adenoma detection rate (AADR) according to age and sex in a large average-risk population of consecutive patients admitted to our digestive endoscopy unit for colonoscopy in the rural areas of Eastern China.
Materials and Methods
This observational, single-center, continuous retrospective study was conducted at our hospital from January 1, 2019, to March 31, 2022. This study conformed to STROBE guidelines. 12 All patients were informed about the use of their endoscopic data for clinical research purposes. Data were retrospectively collected from the medical patient management software. All patient details have been de-identified. All authors declare that they have access to the study data and have reviewed and approved the final manuscript.
Patients
All consecutive patients scheduled for colonoscopy at our hospital from January 1, 2019, to December 31, 2021, were included. Based on the Chinese guidelines for the screening, early detection, and treatment of CRC, 7 patients who were at average risk of CRC were included. Patients with none of the following risk factors were considered average-risk patients for CRC: 1. first-degree relatives with CRC (including a family history of non-hereditary CRC and family history of hereditary CRC); 2. a history of CRC, 3. a history of colorectal adenoma, 4. a history 8–10 years of active inflammatory bowel disease; and 5. positive fecal immunochemical test results. The exclusion criteria were as follows: 1. patients who did not undergo cecal intubation and 2. patients with polyps showing no pathological findings.
Data Collection
The following data were collected: age, sex, polyp size, polyp histopathology, and the number of adenomas. If a patient had more than one polyp, the maximum diameter of the largest polyp was recorded. If a patient had both adenomas and non-adenomatous polyps, the maximum diameter of the adenoma was recorded.
Colonoscopy Considerations
The following colonoscopes were used: H260AI, Q260AZI, CF-Q260AI, CF-HQ290I, and HQ290I (Olympus, Tokyo, Japan).
Histopathological Considerations
An adenoma was defined as a tubular, tubulovillous, or villous adenoma. 5 An advanced adenoma was defined as an adenoma with a size of ≥10 mm, with tubulovillous or villous histology, or with high-grade dysplasia in the absence of invasive CRC. 13 Sessile serrated lesions (SSLs) are hyperplastic polyps, sessile serrated adenomas/polyps, or traditional serrated adenomas. Sessile serrated adenomas/polyps and traditional serrated adenomas are also known as serrated adenomas. 5 Inflammatory polyp was defined as non-neoplastic polypoid lesion of intestinal mucosal secondary to chronic nonspecific inflammatory diseases of the colorectal. 14
Statistical Analysis
Statistical analysis was performed using IBM SPSS Statistics for Windows, version 27.0. Quantitative and qualitative variables are expressed as median (interquartile range; IQR) and numbers (percentages), respectively. Continuous variables were compared using the Wilcoxon-Mann–Whitney U test or Kruskal–Wallis test. Categorical variables were compared using the chi-square test, CALIBRATION chi-square test, or Fisher’s exact test, as required. A
Results
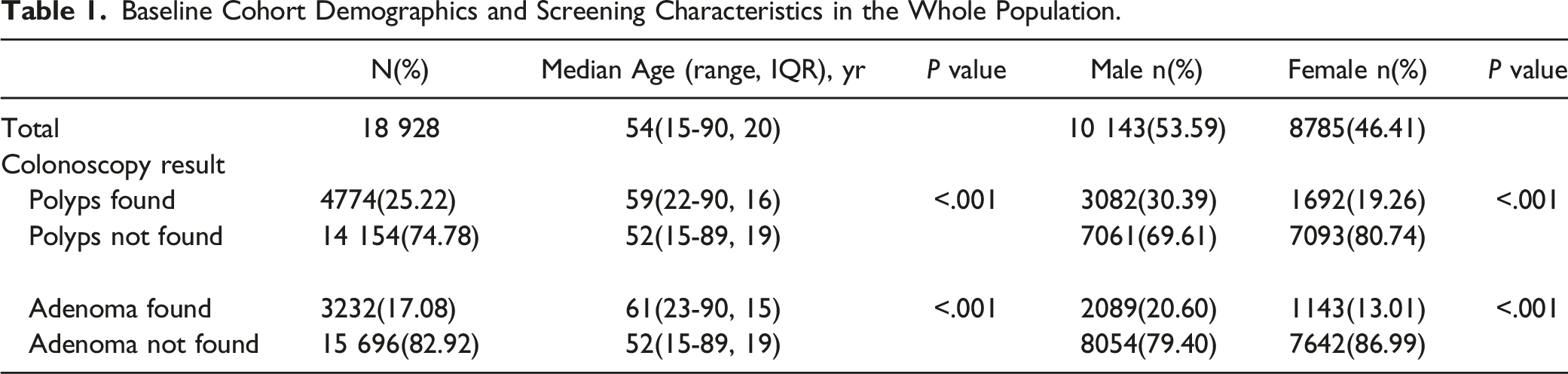
Baseline Cohort Demographics and Screening Characteristics in the Whole Population.
Results in the Whole Population
Of the 18 929 patients, polyps were detected in 4774 patients with a median age of 59 years (range 22–90, IQR 16). Polyps were not detected in the remaining 14 154 patients with a median age of 52 years (range 15–89, IQR 19). The polyp detection rate (PDR) was 25.22%. The age of the patients with polyps was significantly higher than that of those without polyps (
Comparative Analysis of Advanced and Non-advanced Adenomas.

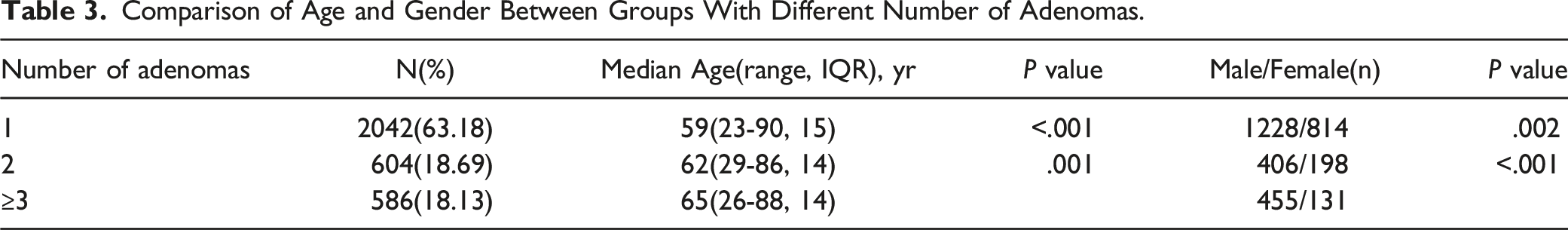
Comparison of Age and Gender Between Groups With Different Number of Adenomas.
Results According to Age Intervals and Gender
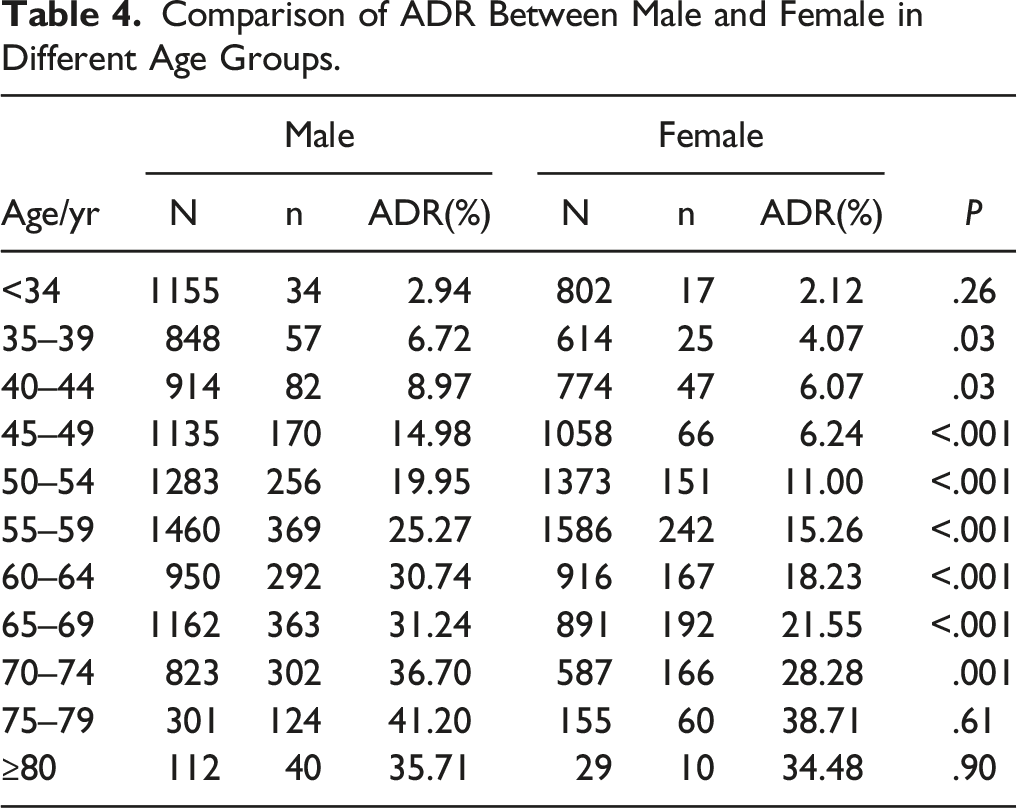
We examined ADR and AADR according to age using age intervals of 5 years. Both ADR and AADR increased significantly with age in the whole population in men and women (Figure 1, Supplementary Figures 1 and 2). After the age of 45 years, the growth rates of ADR and AADR increased significantly in the entire population (Figure 1). The ADR markedly increased from 8.97% to 14.98% in male patients aged 40–44 and 45–49 years ( (A) Adenoma detection rate according to age in the whole population. (B) Advanced adenoma detection rate according to age in the whole population. (A) Adenoma detection rate in the 35–39, 40–44, and 45–49 age groups in men. (B) Adenoma detection rate in the 40–44, 45–49, and 50–54 age groups in women. (C) Advanced adenoma detection rate in the 35–39, 40–44, and 45–49 age groups in men. (D) Advanced adenoma detection rate in the 40–44, 45–49, and 50–54 age groups in women.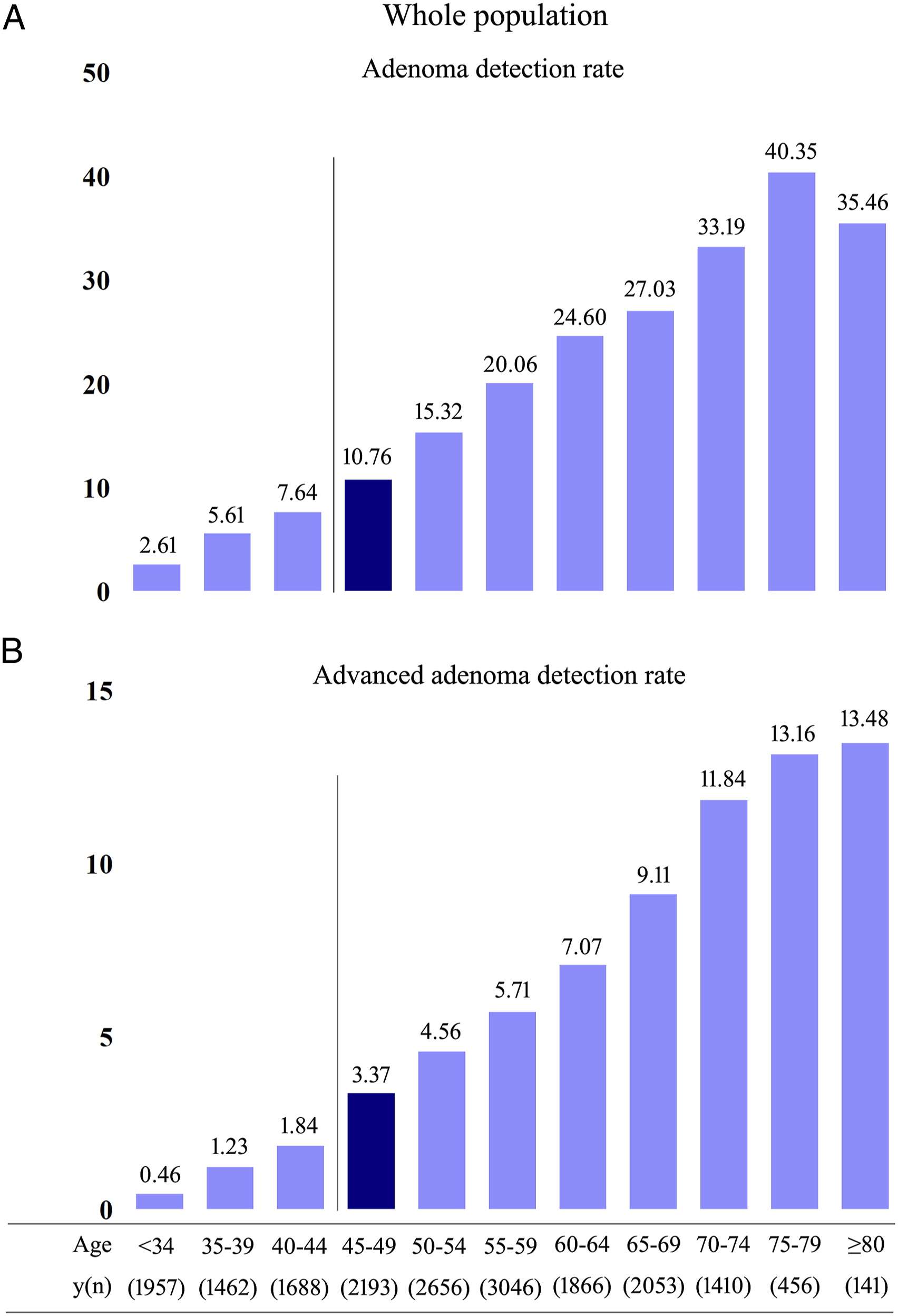
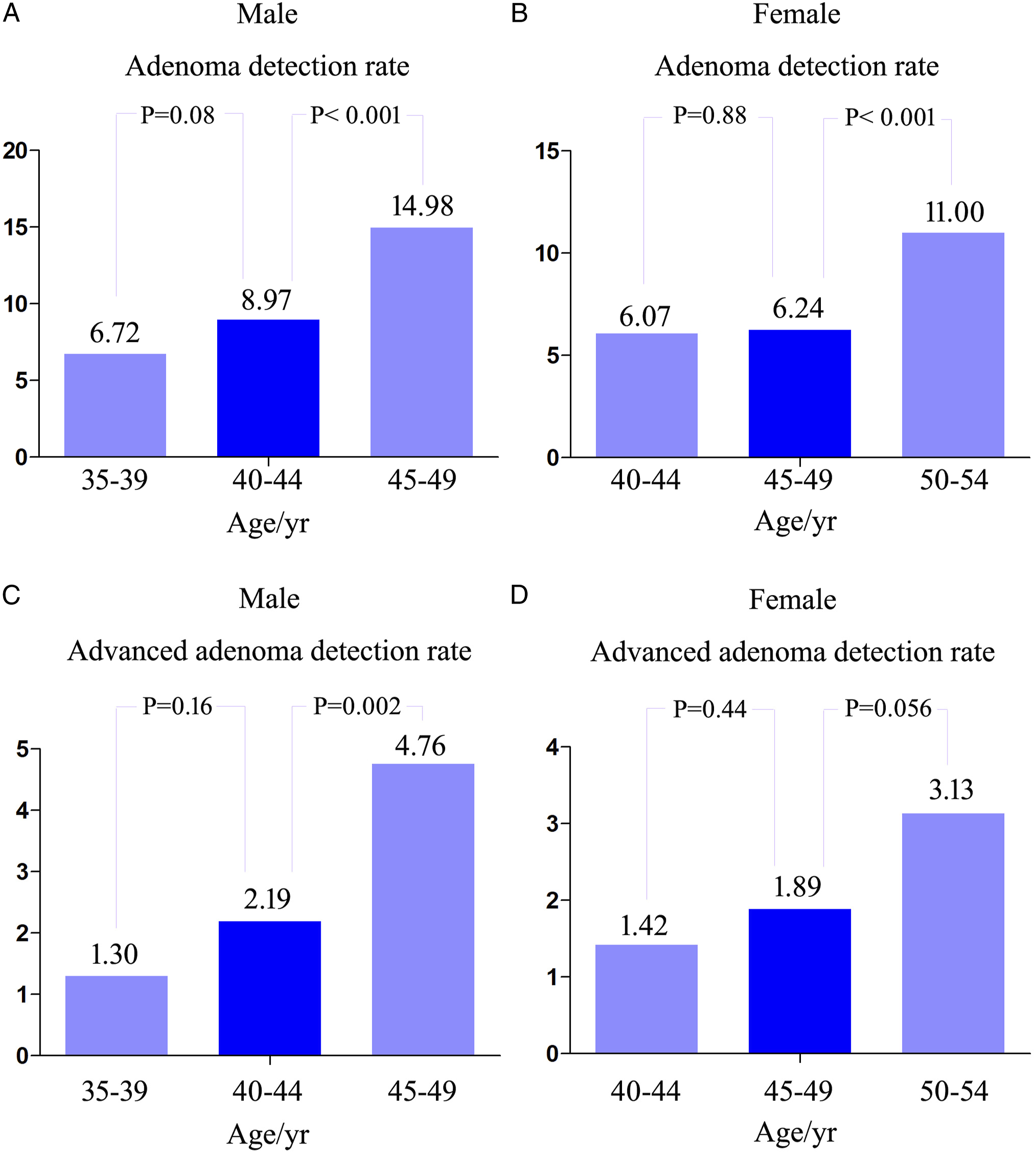
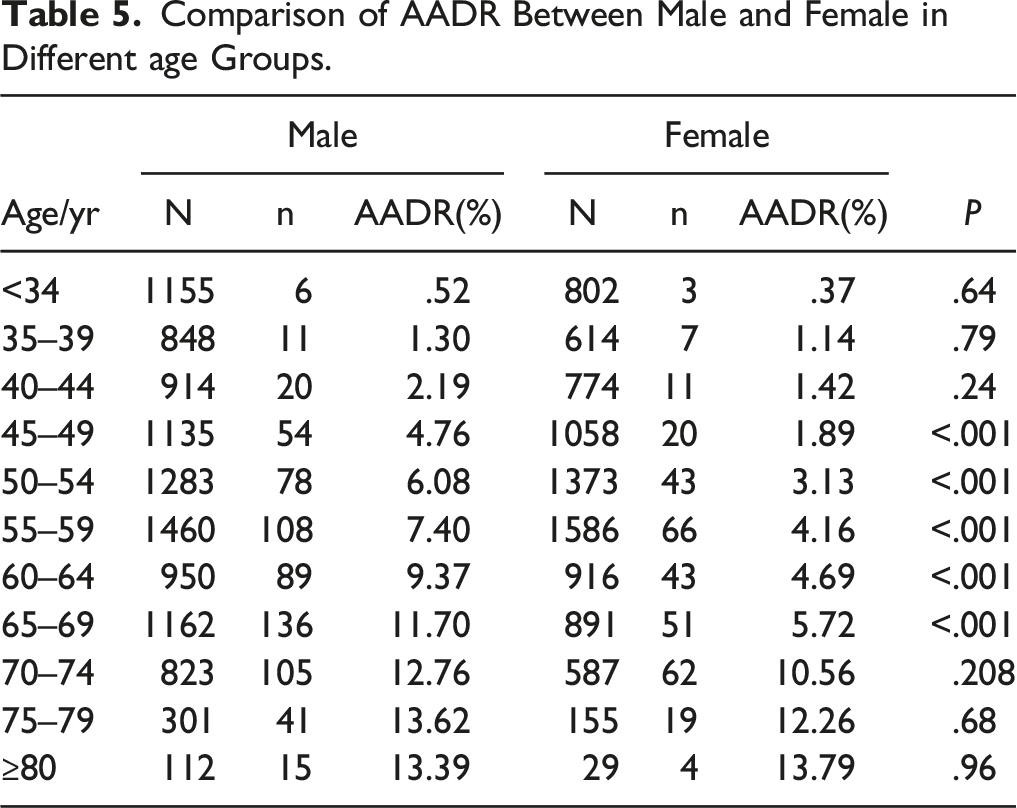
The AADR markedly increased from 2.19% to 4.76% between male patients aged 40–44 and 45–49 years (
Comparison of ADR Between Male and Female in Different Age Groups.
Comparison of AADR Between Male and Female in Different age Groups.
Results According to Polyp Histopathology
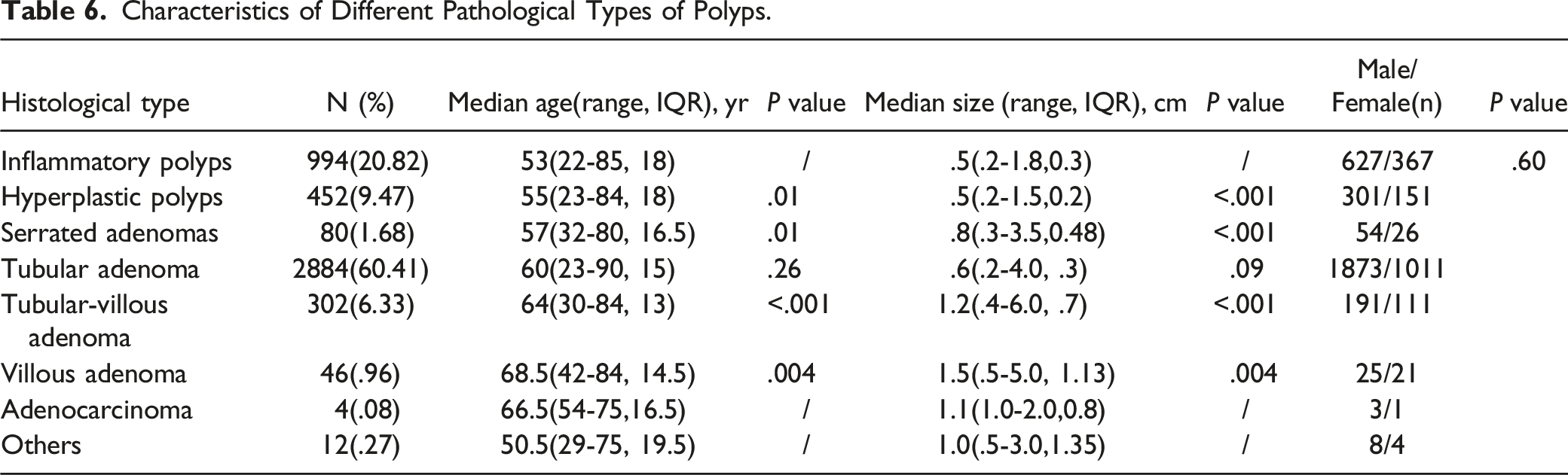
Characteristics of Different Pathological Types of Polyps.
Discussion
Our study demonstrated that both the ADR and AADR increase with age in an average-risk population and their growth rate starts to increase in individuals aged 45 years and older, especially in men. Karsenti D et al 8 also showed that ADR started to increase from 45 years of age, but this study only explored the change rule of ADR in the entire population and did not conduct a comparative study between the rates in men and women. We found that the ADR of male patients aged 45−49 was significantly higher than that of female patients, with a 2.4-fold increase, whereas this gap was not as large at other age intervals. The AADR showed a similar trend. The essential reason for this phenomenon is that ADR and AADR start to increase markedly from the age of 45 years in men, while they start to increase markedly from the age of 50 years in women. Coincidentally, the average age of menopause in women is approximately 50 years. 15 This coincidence led us to consider whether menopause increases the incidence of colorectal adenomas. Although previous studies16,17 have confirmed that postmenopausal estrogen and progesterone replacement therapy can reduce the risk of CRC, whether menopause increases the incidence of colorectal adenomas remains unclear. Although many previous studies have confirmed that the male sex is associated with higher ADR,18-20 to the best of our knowledge, our study is the first to find that ADR increases markedly at an earlier age of approximately 5 years in average-risk of men compared to women. We also found the same trend in AADR, although it was not statistically significant. The National Cancer Center of China recommends that colonoscopy screening starts at the age of 40 years in a high-risk population or 10 years earlier than in the youngest person with CRC in a first-degree relative. 7 However, the age at which average-risk individuals begin colonoscopic screening has not yet been determined. Consistent with the existing literature, 8 our study provides strong evidence for the initiation of colonoscopy screening at 45 years of age in an average-risk population, especially in men.
Many quality indicators have been proposed for colonoscopy; however, ADR and cecal intubation rate are the most commonly used and recognized.21,22 ADR has been validated as predictors of cancer occurring after colonoscopy. 23 The currently recommended minimal thresholds for detection are ≥25% overall or should be ≥ 30% for male patients and should be ≥ 20% for female patients. 5 In our study, the overall ADR was 17.08% (20.60% in men, 13.01% in women). This lower ADR than the recommended value could be due to the following reasons: First, all included patients were at average risk for CRC. Previous studies have shown that high-risk populations of CRC may have higher ADR.8,24 Secondly, half of the patients included aged ≤54 years, in particular, more than half of the women were ≤54 years. Previous studies have shown that ADRs are strongly age-related and continue to increase with increasing age.24,25 Third, the incidence and prevalence of colorectal cancer in China are lower than those in the United States. 10 However, these standards are based on the recommendations of the US MSTF on colorectal cancer. 5 More research on CRC is needed to develop standards suitable for China, especially for young average-risk populations. Fourth, most patients included in our study resided in rural areas of China and had a lower rate of CRC. 11
In this study, we found that advanced adenomas were significantly larger than non-advanced adenomas, and patients with advanced adenomas were significantly older than those with non-advanced adenomas. The proportion of villous components in adenomas positively correlated with patient age and adenoma size. This suggests that cancer risk in patients with colorectal adenoma is positively correlated with age and adenoma size. These findings are similar to those of previously published data.26,27 Our study showed that tubular adenoma was the most common pathological type of colorectal polyp, followed by an inflammatory polyp, hyperplastic polyp, and tubular-villous adenoma. These results are similar to those of previous studies.28,29 Other pathological types, such as villous adenomas, serrated adenomas, juvenile polyps, and adenocarcinomas, are relatively rare. To some extent, our clinical research data reflect the characteristics of the pathological composition of polyps in this region in the average-risk population of CRC.
Serrated adenomas, including sessile serrated adenomas (also known as sessile serrated polyps) and traditional serrated adenomas, have emerged as important lesions in colorectal carcinogenesis through the serrated polyp neoplasia pathway and have been recognized as precursor lesions of interval cancer. 30 The proportion of serrated adenomas was lower than that in other published data (found in 8%–9% of screening colonoscopies performed by expert detectors).31,32 This may be because the histological classification of serrated lesions varies widely among pathologists, and some serrated adenomas are judged as hyperplastic polyps or conventional adenomas. Previous reports also found a poor interobserver agreement between pathologists in the differentiation of hyperplastic polyps from sessile serrated polyps and tubular-villous adenomas from traditional serrated adenomas.30,33,34 More educational training among endoscopists and pathologists is essential for identifying serrated lesions.
Our study has some limitations. First, this was not a multicenter study and a limited number of patients were included. Therefore, the results of this study are not representative of the entire East China region. Second, this was a retrospective study, and information on smoking status, body mass index, medication use, and dietary habits were lacking. Bowel preparation quality and withdrawal time could not be controlled. These factors may have biased our results. Therefore, a prospective multicenter cohort study to remove these factors and obtain reliable results will be conducted.
Conclusions
In conclusion, our study confirmed that the ADR and AADR increased significantly in whole average-risk population from 45 years of age, and is the first to show that the ADR increased significantly in average-risk men aged 45 years and older, 5 years earlier than in women. This suggests that colonoscopy screening may begin at 45 years of age in the average-risk population for CRC, especially in men.
Supplemental Material
Supplemental Material - Adenoma Detection Rate in Average-Risk Population: An Observational Consecutive Retrospective Study
Supplemental Material for Adenoma Detection Rate in Average-Risk Population: An Observational Consecutive Retrospective Study by Xiaoyan He, Xiangyin Lv, Binbin Zhang, Xiaoxuan Ying, Chiyu Hu, Xiaoying Zhou and Jianwen Hu in Cancer Control
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
*The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the grants from the National Natural Science Foundation of China (82100594) and the medical and health research project of Zhejiang province (2022KY429).
Ethical Approval
The study was approved by the Medical Ethics Committee of Dongyang People's Hospital, under number DRY 2021-YX-240.
Informed Consent
Informed consent was obtained from all subjects or their legal guardians. All methods were carried out in accordance with relevant guidelines and regulations.
Supplemental Material
Supplemental material for this article is available online.
Appendix
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
