Abstract
Introduction
The role of primary prophylaxis (PP) with granulocyte colony-stimulating factor (G-CSF) for patients with metastatic pancreatic adenocarcinoma (MPA) treated with FOLFIRINOX is unknown. We aimed to compare the frequencies of grades 3 or 4 neutropenia (G3/4N) and febrile neutropenia (FN) and survival outcomes according to the use of PP.
Methods
This is a retrospective study. We included patients with pathologically confirmed MPA treated with FOLFIRINOX in first-line. Patients who received primary prophylaxis (PP group) were compared to patients who received secondary or no G-CSF (no-PP group). Overall survival (OS) and progression-free survival (PFS) were evaluated using the standard Cox proportional hazard model. To account for potential biases, we performed sensitivity analyses excluding patients who received secondary prophilaxis and treating G-CSF as a time-dependent covariate in extended Cox proportional hazard models.
Results
The study population consisted of 123 patients. PP was used by 75 patients (61.0%). G3/4 N occurred more frequently among patients without PP (10.7 vs 41.7%; P < .001). There was no difference in the frequency of FN between groups (5.3 vs 8.3%; P = .710). In multivariate analysis, PP was associated with a trend toward improved OS (HR = .66; 95% confidence interval [95% CI] .41 - 1.07; P = .094). In the multivariate model excluding patients with secondary prophylaxis (HR = .54; 95% CI 0.32 - .91; P = .022) and in the time-dependent model (HR = .47; 95% CI 0.28 - .80; P = .005), PP was associated with statistically superior OS.
Conclusions
Despite the reduction in the frequency of G3/4N, the risk of FN among patients treated with FOLFIRINOX without G-CSF is too low to justify its use in a routine basis. However, given the potential of G-CSF to improve survival in this setting, further studies are warranted to assess its role during treatment with FOLFIRINOX for patients with MPA.
Introduction
FOLFIRINOX stands as one of the most important advances in the management of metastatic pancreatic adenocarcinoma (MPA). In the ACCORD11/PRODIGE4 study, it was associated with improved overall survival, progression-free survival, overall response rate, and quality of life compared to single-agent gemcitabine.1,2 However, this chemotherapy regimen is associated with significant toxicities. In this pivotal study, 45.7% of the patients developed grades 3 or 4 neutropenia (G3/4N). Additionally, despite the low rate of febrile neutropenia (FN) in the ACCORD11/PRODIGE4 trial (5.4%), real-world studies have reported rates of FN ranging from 7 to 26%, suggesting the true risk of developing FN is higher than initially appreciated.3-6
As a consequence, two strategies have been employed to try to mitigate the effects of FOLFIRINOX on the white blood cell count. A modified version of FOLFIRINOX, with the omission of bolus 5-FU and lower dose of irinotecan, is widely used in clinical practice based on retrospective studies that show similar survival results and milder toxicity profile, including lower rates of neutropenia.7,8 Another option is the use of primary prophylaxis (PP) with granulocyte colony-stimulating factor (G-CSF). In retrospective series of patients treated for MPA with FOLFIRINOX, 4.9 to 100% of patients have received PP, meaning that its use is not consistent in the literature. This might stem from differences in guideline recommendations. In the most recent version of the National Comprehensive Cancer Network guidelines for the management of neutropenia, 9 patients with MPA treated with FOLFIRINOX are considered to be at high-risk of developing NF, warranting the use of PP. Conversely, in the ESMO guidelines, routine use of PP should be reserved for patients treated with chemotherapy regimens with expected rates of FN of at least 20%. 10
Another controversy arises when evaluating the role of PP in the survival outcomes of patients treated with FOLFIRINOX in the first-line setting. In a single-center retrospective study from Korea, Jung et al showed that patients who received PP had a higher median number of cycles of FOLFIRINOX (9 vs 6; P = .004) and longer median overall survival (14.7 vs 8.8 months; P = .001). 11 Contrariwise, Moriyama et al found no significant differences in median progression-free survival (7.3 vs 4.5 months; P = .173) or median overall survival (16.9 vs 14.2 months; P = .302) between patients with metastatic or recurrent pancreatic cancer treated with or without PP. 12 Nonetheless, the latter study suggests patients who undergo PP might achieve higher objective response (30 vs 6%; P = .06) and disease control (74 vs 41%; P = .04) rates.
Hence, we designed and carried out a retrospective study to evaluate the frequencies of G3/4N and FN among patients with PDAC treated with FOLFIRINOX in the first-line setting with or without PP and to explored the putative role of PP with G-CSF in survival outcomes.
Materials and Methods
This is a retrospective, single-center study. We used routinely collected data from the electronical records of consecutive patients with MPA diagnosed from January 2011 to December 2019 at A.C. Camargo Cancer Center. Given the retrospective nature of this study, the Institution’s Internal Ethics Board Committee waived the need for informed consent and approved the conduct of the study (CAAE 822894.5.0000.5432). The reporting of this study conforms to STROBE guidelines. 13
Patients
We included patients aged 18 years-old and above with pathologically confirmed pancreatic adenocarcinoma diagnosed from January 2011 to December 2019 and pathological or unequivocal radiological evidence of metastatic disease who were treated with first-line FOLFIRINOX at A.C. Camargo Cancer Center. In patients with hyperbilirubinemia, the use of FOLFOX in the first two cycles was allowed. We excluded patients with performance status (PS) measured by the ECOG (Eastern Cooperative Oncology Group) scale ≥3, patients treated with FOLFIRINOX in the setting of localized disease (potentially resectable or locally advanced), and patients treated outside of our institution.
Variables
We collected data on the following baseline clinical features: age at start of FOLFIRINOX, sex, comorbidities, ECOG PS, and BMI (body mass index). We also gathered information on laboratory (baseline total neutrophil count, baseline total lymphocyte count, and CA 19-9 levels) and radiological (primary tumor site and sites of metastatic disease) characteristics. We also recorded data on treatment features (PP use, type of G-CSF, type of FOLFIRINOX, duration of FOLFIRINOX, reason for discontinuation of PP, and further chemotherapy regimens after FOLFIRINOX discontinuation). To evaluate the study’s outcomes, we also collected data on the frequency, grade, and cycle of occurrence of G3/4N and NF, and on the dates of disease progression and death. Baseline Neutrophil-to-Lymphocyte ratio (NLR) was calculated as the ratio of neutrophils to lymphocytes in baseline blood tests before FOLFIRINOX initiation.
Procedures
Patients were allocated according to the use of primary prophylaxis (PP). Group PP included patients who underwent treatment with G-CSF since the beginning of first-line therapy. The use of PP was left to the decision of the treating physician. G-CSF agents used were filgrastim (subcutaneous, 300 μg/day, for three to five days, every two weeks) or peg-filgrastim (subcutaneous, 6 mg, every two weeks). They were started 12 to 24 hours after the end of the 5-fluorouracil 46-hour infusion. Group no-PP included patients for whom only secondary G-CSF or no prophylaxis were implemented. Standard FOLFIRINOX was undertaken in accordance with the PRODIGE4/ACCORD11 trial. 1 For most patients in the modified FOLFIRINOX group, the dose of irinotecan was reduced to 150 mg/m2 and bolus 5-FU was omitted.
A complete blood count was ordered every two weeks, just before the FOLFIRINOX cycle to look for hematological toxicities. Disease response evaluation was carried out every 2 to 3 months with cross-sectional imaging methods (mostly CT scans) and CA 19-9 measurements.
Outcomes
G3/4N was defined according to the Common Terminology Criteria for Adverse Events v5.0. We defined FN as a single temperature ≥38.3°C (101°F) or a temperature ≥38.0°C (100.4°F) sustained over one hour and an absolute neutrophil count ≤500 cells/μL, in accordance with the Infectious Disease Society of America guidelines. 14 Overall survival (OS) was defined as the time from the start of FOLFIRINOX to death (from any cause). Progression-free survival (PFS) was defined as the time from the start of FOLFIRINOX to death or disease progression (whatever took place first). 15 Disease progression was determined according to radiological reports or the treating physician’s impressions recorded in the medical charts. Patients were censored at the last follow-up visit in the absence of an event.
Statistical Analysis
We described the distributions of categorical variables using absolute frequencies and ratios and we compared the distributions of categorical variables between two different groups using Fisher’s exact test. We described the distributions of numerical variables using medians and interquartile ranges (IQR) and we compared the distributions of numerical variables between two different groups using Mann-Whitney’s U test. The occurrence of G3/4N and FN was calculated per patient. To account for potential differences in the incidence of G3/4N and FN according to variations in the duration of treatment with FOLFIRINOX, we also calculated the incidence of such outcomes as the number of episodes adjusted for the duration of treatment with FOLFIRINOX (reported as episodes per 100 patient-months of treatment). Differences in these incidence rates between groups were assessed using univariate exposure-corrected Poisson’s regression.
The Kaplan-Meier estimator was used to calculate median survival times (and respective 95% confidence intervals [95% CI]) and generate survival curves. Differences in time-to-event outcomes were assessed using the log-rank test. To look for potential factors associated with the risk of G3/4N (response variable being patients with grades 3 or 4 neutropenia), we generated univariate and multivariate logistic regression models. In search of putative prognostic factors and to establish the effects of PP on survival, we generated univariate and multivariate Cox proportional hazards models for OS and PFS. All variables assessed in the univariate analyses were used to generate multivariate Cox proportional hazard models for OS and PFS.
Given that some patients in the no PP group received G-CSF as secondary prophylaxis during first-line treatment with FOLFIRINOX, we performed two post-hoc sensitivity analyses to try to address this potential source of bias. In the first one, we generated univariate and multivariate Cox proportional hazard models for both OS and PFS after excluding these patients; hence, in this analysis, only patients who never received G-CSF comprised the no PP group. Additionally, we created extended Cox proportional hazard models for both OS and PFS using G-CSF as a time-dependent covariate (and not presence or absence of primary prophylaxis). 16 In this analysis, patients treated with secondary prophylaxis had their G-CSF status changed once they started such treatment.
Two-tailed tests with P values less than .05 were considered statistically significant. All statistical analyses were done with Stata version 16.0, except for the time-dependent multivariate Cox analyses, which were undertaken with R version 3.4.0 (and the survival package). 17 Further information on the statistical analysis can be found in the supplementary material.
Results
We identified 279 patients diagnosed with pancreatic adenocarcinoma from January 2011 to December 2019 treated with FOLFIRINOX in first-line setting. We excluded 156 patients for the following reasons: treatment with FOLFIRINOX for non-metastatic disease (N = 114) and treatment outside AC Camargo Cancer Center (N = 42). Therefore, a total of 123 patients were included. Seventy-five patients (61.0%) received primary prophylaxis with G-CSF, and were clustered in the PP group. The remaining 45 (39.0%) patients were gathered in the no PP group.
Patients’ Characteristics
Baseline Patients’ Clinical Characteristics.
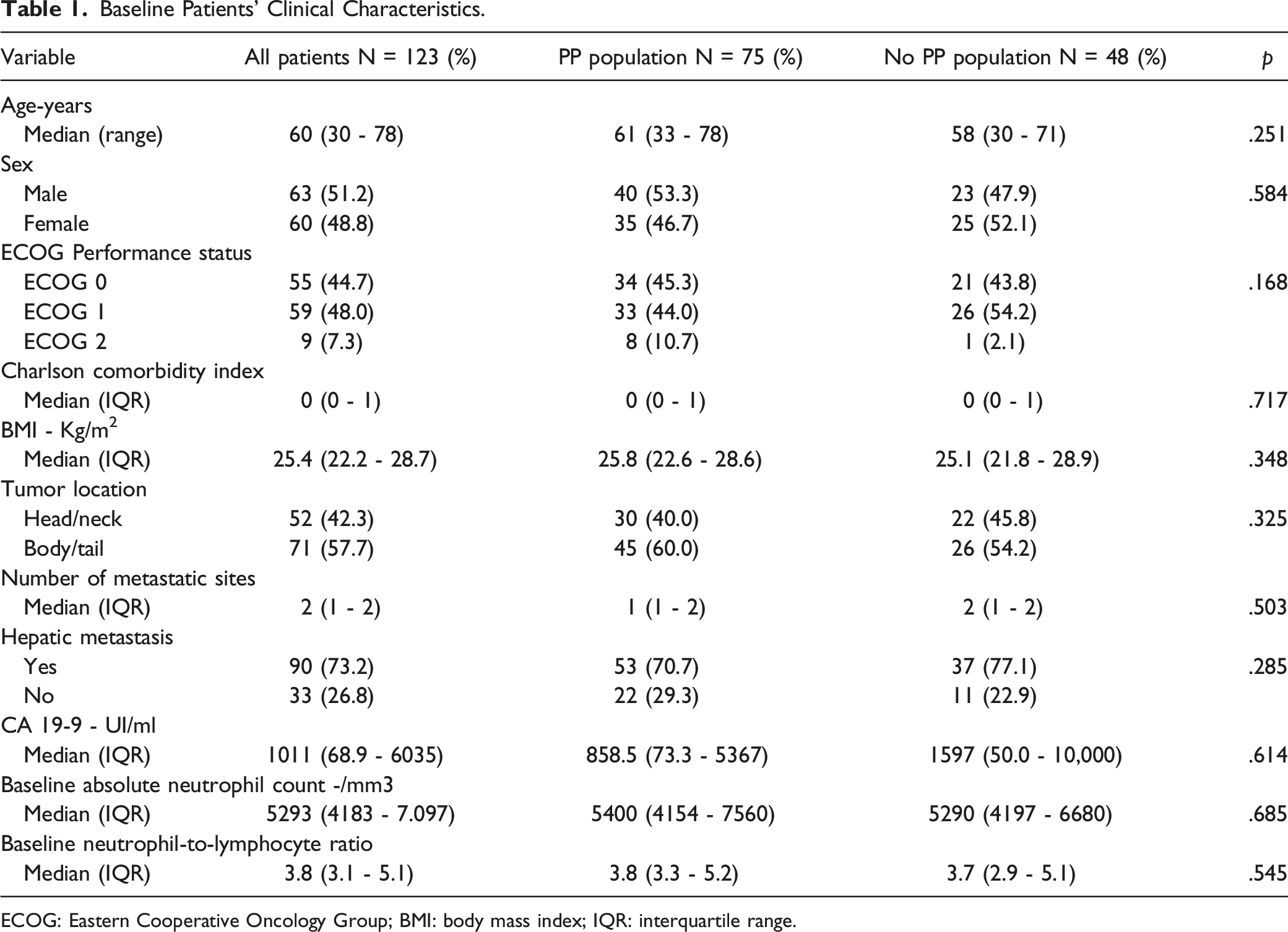
ECOG: Eastern Cooperative Oncology Group; BMI: body mass index; IQR: interquartile range.
Treatment Characteristics
Treatment Characteristics.

G-CSF: granulocyte colony-stimulating factor; IQR: interquartile range.
N = 86.
N = 81.
Grades 3 or 4 Neutropenia
We documented 33 episodes of G3/4N in 28 patients (22.8%). G3/4 N was less common in the PP group (10.7 vs 41.7%; P < .001). In the PP group, eight out of nine episodes of G3/4N occurred during treatment with G-CSG. Among patients of the PP group, the risk of G3/4N was numerically lower amongst those treated with a pegylated formulation of G-CSF (7.0 vs 22.2%; P = .088). In the overall population, 3.3 episodes of G3/4N were observed per 100 patient-months of treatment with FOLFIRINOX. Patients in the PP group also showed a lower incidence rate of G3/4N (1.21 vs 7.13 episodes per 100 patient-months of treatment; P < .001). Additionally, most of the episodes of G3/4N occurred before the eighth cycle of FOLFIRINOX - supplementary figure 1. In the multivariate logistic regression model, the use of G-CSF as primary prophylaxis was associated with a decreased risk of developing G3/4N (OR = .13; 95%CI 0.04 - .39; P < .001) - supplementary Table 1.
Febrile Neutropenia
We documented eight episodes of FN in eight patients (6.5%). There was no difference in the frequency of FN between patients in the PP and no PP groups (5.3 vs 8.3%; P = .710). In the PP group, three out of four episodes of FN occurred during treatment with G-CSG. In the overall population, .79 episodes of FN were observed per 100 patient-months of treatment with FOLFIRINOX. Again, there was no difference in the incidence rate of FN between the PP and no PP groups (.61 vs 1.14 episodes per 100 patient-months of treatment; P = .370). Additionally, nearly half of the episodes of FN occurred before the fifth cycle of FOLFIRINOX - supplementary figure 2.
Survival Analysis
Median follow-up was 75.4 months (95%CI 21.9 - not reached). Eighteen patients (14.6%) were lost to follow-up after disease progression on FOLFIRINOX. Ninety-eight patients (79.7%) died during follow-up. Median OS was 13.3 months (95%CI 11.8 - 15.0). Median OS for patients in PP and no PP groups were 15.2 and 10.8 months (log-rank P = .013), respectively - Figure 1. In multivariate Cox proportional hazards model for OS, the presence of hepatic metastasis was the only factor associated with worse overall survival (HR = 1.89; 95% CI 1.09 - 3.30; P = .024). The use of PP was associated with a non-statistically significant reduction in the hazards of death (HR = .66; 95% CI 0.41 - 1.07; P = .094) Table 3. One hundred seventeen patients either died or experienced disease progression during follow-up. Median PFS was 8.3 months (95% CI 7.3 - 9.4). Median PFS in PP and no PP groups were 9.4 and 6.1 months (log-rank P = .040), respectively - supplementary figure 3. In multivariate Cox proportional hazards model for PFS, the use of PP was not associated with a reduction in the hazards of death or disease progression (HR = .83; 95% CI 0.54 - 1.27; P = .388) Table 4. Also, there was no sign of statistical interactions between the effects of primary prophylaxis on both OS and PFS according to the baseline NLR - supplementary Table 2. Overall survival of patients in the Primary Prophylaxis (PP) and No Primary Prophylaxis (No PP) groups.
In the first sensitivity analysis, we excluded patients who received G-CSF as secondary prophylaxis for FN. In the multivariate Cox proportional hazard model for OS, the presence of hepatic metastasis was associated with inferior overall survival (HR = 2.11; 95% CI 1.14 - 3.89; P = .017), while primary prophylaxis with G-CSF was associated with superior overall survival (HR = .54; 95% CI 0.32 - .91; P = .022) - supplementary Table 3. In the multivariate Cox proportional hazard model for PFS, increasing Charlson comorbidity index was associated with inferior progression-free survival (HR = 1.81; 95% CI 1.19 - 2.74; P = .005), while primary prophylaxis with G-CSF was not associated with statistically superior progression-free survival (HR = .70; 95% CI 0.44 - 1.11; P = .131) - supplementary Table 4. In the second sensitivity analysis, we built Cox proportional hazard models for OS and PFS using G-CSF as a time-dependent covariate. In this analysis, the presence of hepatic metastasis was associated with inferior overall survival (HR = 1.76; 95% CI 1.09 - 2.85; P = .021), while treatment with G-CSF was associated with improved overall survival (HR = .47; 95% CI 0.28 - .80; P = .005). Increasing titers of CA 19-9 were associated with inferior progression-free survival (HR = 1.00; 95% CI 1.00 - 1.00; P = .032), while treatment with G-CSF was associated with a trend toward improved progression-free survival (HR = .67; 95% CI 0.43 - 1.03; P = .067) - supplementary Table 5.
Further Lines of Treatment
Cox Proportional Hazard Models for Overall Survival (N = 109 in the Multivariate Model).

ECOG: Eastern Cooperative Oncology Group; PP: primary prophylaxis; HR: Hazard ratio; CI: Confidence interval.
Global and specific P values for the chi-square tests of Schoenfeld residual were >.05.
Discussion
Cox Proportional Hazard Models for Progression-Free Survival (N = 109 in the Multivariate Model).

ECOG: Eastern Cooperative Oncology Group; PP: primary prophylaxis; HR: Hazard ratio; CI: Confidence interval.
Global and specific P values for the chi-square tests of Schoenfeld residual were >.05.
Selected Studies Evaluating the Incidence of Grades 3 or 4 Neutropenia and Febrile Neutropenia During Treatment with FOLFIRINOX.

Grade 4 only.
Despite being an important toxicity surrogate, many believe that the use of G-CSF during cytotoxic chemotherapy should not be guided by the risk of G3/4N. Indeed, most guidelines suggest PP should be offered to patients undergoing chemotherapy regimens with expected rates of FN of 20% and above.10,34 However, rates of NF during treatment with FOLFIRINOX are highly variable in the literature, ranging from 0 to 26% - Table 5.18-33 Indeed, the higher figures observed in some retrospective studies have led the NCCN guideline to advise in favor of PP for patients treated with FOLFIRINOX. 1 In our study, only 6.5% of the patients experienced FN and even for those who did not receive PP, the frequency of FN was relatively low (8%). It is important to highlight that 23% of the patients in the no PP group eventually received G-CSF, which might have contributed to lower the chances of FN in this group. In any case, our data suggest that G-CSF should not be used routinely as PP in patients with MPA treated with FOLFIRINOX in the first-line setting and it that it could be selectively used as secondary prophylaxis for patients who develop significant white blood cell toxicity.
However, the aforementioned guidelines also suggest patients’ characteristics should considered when deciding whether or not to administer PP to patients undergoing chemotherapy. In this sense, knowledge about patient’s features that render them more susceptible to hematological toxicity could help medical oncologist decide the need for G-CSF at treatment start. During treatment with FOLFIRINOX, Keum et al showed that female sex and overweight were associated with increased risk of grade 4 neutropenia. 35 Regarding the risk of febrile neutropenia, the same study suggested that female sex, poor performance status, overweight, and initial biliary stent placement were associated with increased frequency of FN. In another study, Sasaki et al observed low that absolute neutrophil count, thrombocytopenia, hyperbilirubinemia, location in the pancreatic head, and use of standard FOLFIRINOX were associated with increased risk of FN. 36 In our study, we could not identify any clinical feature associated to the risk of G3/4N. This is likely related to the study’s modest sample size and the high frequency of PP with G-CSF, which might have blurred the relationship between clinical characteristics and the risk of G3/4 neutropenia. Despite these results, we think that clinical characteristics associated with increased risk of developing G3/4N and FN should be factored in the decision-making process. Additionally, patients with increased risk of complicated FN, such as elderly patients, should also be considered for PP when treated with FOLFIRINOX. 37
It is important to highlight that other approaches apart from G-CSF can be used to minimize the toxicity from FOLFIRINOX. Modified FOLFIRINOX has been shown to be as effective as its standard schedule, with lower risk of severe neutropenia.7,8 Indeed, in our study, despite not reaching statistical significance, the use modified FOLFIRINOX was associated with a 44% reduction in the odds of developing G3/4N. Additionally, routine evaluation of polymorphisms in the limiting-rate enzymes involved in the metabolism of 5-fluorouracil and irinotecan can help identify those more susceptible to severe toxicities from FOLFIRINOX. Patients with dihydropyrimidine dehydrogenase (DYPD) deficiency are at higher risk of hematological toxicities from FOLFIRINOX, including neutropenia. 38 Importantly, recent data suggest that the search for DPD deficiency in the setting of adjuvant treatment in colon cancer is cost-effective, 39 raising the question as to whether this would apply to other treatment scenarios. Likewise, patients with polymorphisms affecting the uridine diphosphate glucuronosyltransferase 1A1 (UGT1A1) present increased toxicity when treated with FOLFIRINOX 38 and recent data suggest that irinotecan dose optimization based on UGT 1A1 genotyping might improve FOLFIRINOX safety profile. 40 In our study time span, patients did not undergo DPD and UGT deficiency testing routinely. However, given the evidence in other treatment scenarios and the availability of the test in our institution, we have recently adopted a customary practice of screening patients for DPD polymorphisms associated with decreased enzyme activity. Importantly, both the use of modified FOLFIRINOX and the screening for polymorphisms associated with decreased enzymatic function lead to lower rates of non-hematological toxicities and, potentially, to overall treatment cost reductions.
In this study, we showed that patients who received PP during treatment with FOLFIRINOX experienced longer unadjusted OS. In the standard Cox proportional hazards model with all patients, PP was also associated with OS, but not at the pre-specified significance threshold. It is important to highlight that given the relatively low number of events, we deliberately chose not to perform variable selection in the construction of the multivariable models, which could have artificially narrowed estimates’ confidence intervals and decreased significance tests’ P values. 41 Also, given that patients who received secondary prophylaxis with G-CSF can introduce bias in the survival analysis, we performed two sensitivity analyses, one excluding these patients and another dealing with the exposure to G-CSF as a time-dependent covariate in extended Cox proportional hazards models. In both analyses, the use of G-CSF was associated with improved OS, reinforcing the trend seen in the primary multivariate analysis.
So far only two studies have assessed the relationship between the use of G-CSF during treatment with FOLFIRINOX in advanced pancreatic cancer and OS. In the study by Jung et al, patients who received PP experienced improved OS (14.7 vs 8.8 months; P = .001), even in the multivariate analysis. 11 Contrariwise, in the study by Moriyama et al, there was no statistically significant difference in OS (16.9 vs 14.2 months; P = .302) according to the use of G-CSF either as primary or secondary prophylaxis. 12 However, the latter study had a smaller sample size, included patients with non-metastatic disease, and the authors were also able to show that the use of G-CSF was associated with improved disease control rate (74 vs 41%; P = .04).
A caveat in the putative survival benefit associated with the use of G-CSF in patients treated with FOLFIRINOX for MPA is the lack of a clear underpinning biological mechanism. Theoretically, patients treated with G-CSF can achieve higher dose intensities, and therefore, extract the most from their cytotoxic treatment. However, recent data suggest that FOLFIRINOX dose intensity (either global or drug-specific) is not associated with OS. 42 Indeed, in the trial by Jung et al, PP was associated with improved OS even after adjusting the multivariate model for the dose intensity. 11 Another important issue is the lack of clear improvement in PFS. Across all the models we built, we could not find a robust evidence of an association between the use of G-CSF and PFS. Perhaps this indicates that the use of PP could have a delayed effect on OS. In this sense, we showed that patients treated with PP more often received combination chemotherapy in the second-line setting (not statistically significant). This is in line with randomized studies that showed improved survival with polychemotherapy after progression on gemcitabine-based chemotherapy.43-45
A recent observation is that the effect of G-CSF on the OS of patients with pancreatic cancer might depend on the disease stage. In a study that included patients with operable pancreatic cancer who received neoadjuvant treatment with FOLFIRINOX, those who received G-CSF experienced inferior OS, even after adjustment for other covariates. 46 In localized pancreatic cancer, neutrophils seem to play a role in the mechanism of metastatic dissemination, 47 raising the question as to whether the neutrophilia caused by the use of G-CSF could facilitate this phenomenon. Indeed, even in the metastatic setting, limited evidence suggests that patients who experience G3/4N during treatment with FOLFIRINOX without PP have improved outcomes when compared to those who do not develop this toxicity. 48 Finally, the negative prognostic impact of an increased Neutrophil-to-Lymphocyte ratio in the metastatic setting, 49 regardless of the chemotherapy regimen used, signalizes the need to further understand the interaction between pancreatic cancer and hematopoietic growth factors.
Despite these controversies, G-CSF continues to be frequently administrated in patients treated with FOLFIRNOX for MPA. Additionally, all add-on trials that used FOLFIRINOX as a backbone failed to show improvements in survival outcomes.50-52 Therefore, we believe that a randomized trial evaluating the role of G-CSF in this setting is warranted.
Our study has limitations. Due to its retrospective nature, we found no clear reasons influencing the clinician’s decision to give G-CSF as PP. Also, due to the study’s modest sample size, there were slight unbalances between the two groups in terms of clinical characteristics. We acknowledge such factors might have affected survival outcomes. Moreover, we were not able to gather data on objective response, dose intensity, and other G3/4 toxicities. Patients in the PP group experienced higher rates of treatment de-escalation, which could be secondary to a longer period of treatment without progression, but also higher dose intensity and need to reduce doses due to other non-hematological toxicities. However, in our experience, apart from peripheral neuropathy, non-hematological G3/4 toxicities seldom lead to treatment de-escalation. Additionally, we did not report data on the toxicity of G-CSF, which we found very challenging to retrospectively extract from medical records. Last, we did not assess the cost-effectiveness of the prophylactic use of G-CSF in this scenario. However, we present extensive data on white blood cell toxicity and survival outcomes, including PFS, with a statistically sound approach. Additionally, we portrayed information on further lines of treatment, which could be an important variable in understanding the association between the use of G-CSF and OS.
To conclude, patients treated with FOLFIRINOX and primary prophylaxis for febrile neutropenia experience less grades 3 or 4 neutropenia. Patients without primary prophylaxis have low probability of developing febrile neutropenia, and therefore, routine use of G-CSF is currently not warranted in our setting. Nonetheless, the OS benefit seen in our study highlights the need to assess the role of G-CSF during treatment with FOLFIRINOX in the randomized controlled setting.
Supplemental Material
Supplemental Material - Impact of Granulocyte Colony-Stimulating Factor (G-CSF) on the Outcomes of Patients With Metastatic Pancreatic Adenocarcinoma (MPA) During First-Line Treatment With FOLFIRINOX: A Single-Center Retrospective Analysis
Supplemental Material for Impact of Granulocyte Colony-Stimulating Factor (G-CSF) on the Outcomes of Patients With Metastatic Pancreatic Adenocarcinoma (MPA) During First-Line Treatment With FOLFIRINOX: A Single-Center Retrospective Analysis by Angelo Borsarelli Carvalho de Brito, Rachel P Riechelmann, and Victor Hugo Fonseca de Jesus in Cancer Control
Footnotes
Authors’ Contributions
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
Ethics’ approval number: CAAE 82289418.5.0000.5432.
Supplemental Material
Supplemental material for this article is available online.
Appendix
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
