Abstract
Introduction
Trifluridine-tipiracil (FTD/TPI) is approved as monotherapy and in combination with bevacizumab for the treatment of metastatic colorectal cancer (mCRC). This UK real-world retrospective analysis aimed to evaluate the use of granulocyte colony stimulating factor (G-CSF) to prevent FTD/TPI induced neutropenia in patients with mCRC.
Methods
Retrospective data were collected across five UK Healthcare sites. Consecutive patients with mCRC having received at least 2 cycles of FTD/TPI in addition to G-CSF administered as primary or secondary prophylaxis were included. The study assessed the timing and duration of G-CSF treatment, incidence of neutropenia, and subsequent dose delays/reductions. Time on FTD/TPI treatment, Progression free Survival (PFS), and Overall Survival (OS) were also calculated for this patient cohort.
Results
The data set included 55 mCRC patients in total; 25 receiving primary prophylaxis, and 30 receiving secondary prophylaxis. 68% of G-CSF prophylaxis commenced on day 15, with 99% initiated between days 14 and 22. Following G-CSF use, 25% of patients experienced a dose delay and 18% a dose reduction due to neutropenia. 14.5% of patients reported grade ≥3 neutropenia. In patients receiving primary prophylaxis a mean of 18.4 weeks of FTD/TPI were completed, whereas in those receiving secondary prophylaxis the mean was 28.8 weeks. Primary prophylaxis patients exhibited a median PFS of 3 months, and a median OS of 9.7 months. In secondary prophylaxis patients the median PFS was 7.2 months and the median OS 17.5 months.
Conclusion
G-CSF prophylaxis reduced the incidence of neutropenia, improved dose intensity, and extended PFS and OS in this real-world study of mCRC patients on FTD/TPI. G-CSF prophylaxis was commonly administered as a 5-day course of filgrastim starting on day 15.
Keywords
Introduction
Trifluridine/tipiracil (FTD/TPI) is a combination product, containing an antineoplastic thymidine-based nucleoside analogue, trifluridine, and the thymidine phosphorylase inhibitor, tipiracil hydrochloride. In the United Kingdom (UK) and European Union, it is approved both as monotherapy and in combination with bevacizumab for the treatment of metastatic colorectal cancers (mCRC).1,2 The current study was conducted prior to the approval of FTD/TPI in combination with bevacizumab.
Clinical experience with FTD/TPI shows that it is well tolerated, but around 53% of patients will experience neutropenia 3 leading to dose reductions, delays or treatment cessation. Research shows that neutropenia can impair patient’s mental well-being, causing anxiety, depression, fear, anger, and frustration. 4 These findings were confirmed in our pre-study patient interviews. Delays due to neutropenia also increase the frequency of blood testing and place additional demand on clinical services.
In the RECOURSE FTD/TPI monotherapy trial, which led to the first mCRC license, 38% of patients in the treatment arm developed Grade ≥3 neutropenia, 4% were reported to have developed febrile neutropenia, and 67% of patients developing neutropenia of any grade. There were low levels (9%) of G-CSF usage throughout the course of the trial. 5
Neutropenia rates increase when FTD/TPI is used in combination with bevacizumab, with Grade ≥3 neutropenia occurring at a rate of 43.1% in the SUNLIGHT study. However, febrile neutropenia occurred in just one patient in the combination group and six patients in the FTD/TPI group. Prescribing of G-CSF was more common, with 19.5% of patients in the combination arm receiving G-CSF. 6
Despite the awareness that neutropenia is a common side effect of FTD/TPI, with associated consequences for treatment, there is no published guidance specific to the use of prophylactic G-CSF for patients in the palliative, metastatic setting. This is the case for patients receiving both FTD/TPI and similar therapies in later lines of treatment. Evidence is sparse and clinical practice is thought to vary significantly.
Available guidance concentrates on the risk and management of febrile neutropenia, rather than neutropenia in the absence of fever. The European Society of Medical Oncology (ESMO) and the European Organization of Research and Treatment of Cancer (EORTC) clinical guidelines categorize risk of febrile neutropenia with chemotherapy regimens into high (≥20%), intermediate (10%–20%) and low (<10%) risk groups. They recommend supporting high-risk patients with prophylactic G-CSF. In regimes with intermediate risk, contributing factors for febrile neutropenia should be considered and patient’s individual risk assessed.7,8 The American Society of Clinical Oncology (ASCO) recommend prophylactic use of G-CSF when the risk of febrile neutropenia is >20% and no other equally effective treatments with a lower risk of febrile neutropenia are available. 9 The RECOURSE and SUNLIGHT data suggests that the risk of febrile neutropenia with FTD/TPI monotherapy and combination would be less than the intermediate risk group, therefore prophylactic G-CSF is not recommended by ESMO, EORTC or ASCO.5,6,8-10
In our experience, management of neutropenia, in the absence of fever, in patients receiving chemotherapy with palliative intent has mainly been managed using dose delay and dose reduction. Given the lack of definitive guidance plus the fact that, to our knowledge, no published data exists to determine the safety or efficacy of the FTD/TPI and G-CSF combination, we undertook this exploratory analysis.
We elected to review the use of prophylactic G-CSF in patients receiving FTD/TPI in the identified centers to gain insights regarding timing and duration of dosing, to help standardize practice, maintain dose schedule and ensure that outcomes were beneficial for patients.
Methods
This observational study utilized data collected retrospectively as part of routine clinical care. These patients received treatment in an oncology out-patient clinic setting across five Healthcare Trusts in England.
All patient data was anonymized. Patients were allocated a number for the purposes of data collection, known only to the investigator and not disclosed. Sites were included based on their experience of prophylactic prescribing of G-CSF with FTD/TPI, which was not routine practice in the UK at that time.
Consecutive patients having initiated FTD/TPI monotherapy and received at least two cycles of treatment in addition to prophylactic G-CSF between 31 August 2019 and 1 September 2021 were included. All these patients had received at least one dose of prophylactic G-CSF. There were no patients at these sites who had received only one cycle of FTD/TPI with G-CSF during this period, so these data are inclusive of all relevant patients. Data cut off for follow-up was 1 April 2022. Data were retrospectively collected using a standardized Microsoft Excel spreadsheet, ensuring the consistency of data collection across sites and for individual patients. Details recorded included anonymized patient details, risk factors, comorbidities, concomitant medications, investigations, treatment decisions and adverse events. Patients were stratified according to whether they had received G-CSF as primary or secondary prophylaxis for neutropenia. Primary prophylaxis was defined as G-CSF prescribed alongside FTD/TPI without a previous episode of neutropenia. Secondary prophylaxis with G-CSF was prompted by patients experiencing neutropenia due to treatment with FTD/TPI. There was no distinction made between neutropenia in the absence of fever and febrile neutropenia.
Under normal circumstances, the recommended dose schedule of FTD/TPI is 35 mg/m2 twice daily on days 1-5 and days 8-12 of each 28-day cycle. The full blood count (FBC) is checked prior to day 1 of each cycle of FTD/TPI and neutrophils are required to be ≥1.5 × 109/L. If the neutrophils are below this limit, treatment is delayed until the count has recovered. Dose reductions for neutropenia are recommended only in the case of febrile neutropenia or grade 4 neutropenia that results in more than I week’s delay in start of new cycle. 3
This was a single arm study, lacking a control group, due to the complexities of finding a sufficiently matched population within the limited patient pool and timelines. Due to the exploratory, retrospective nature of the investigation statistical power was not calculated.
The intention was to describe patterns, and identify any potential associations rather than test specific, pre-defined hypotheses. In terms of analysis, descriptive data were used to summarize the majority of data. For survival data, all time-to-event endpoints were analyzed using the Kaplan-Meier estimator. For the outcome of progression-free survival (PFS), patients who did not progress or die by the data cut-off date were censored at the last adequate assessment date. For the outcome of overall survival (OS), patients who did not die by the data cut-off date were censored at the date of last clinic visit. An indirect comparison with the RECOURSE study 5 was employed as a reference point for discussion.
The aims of the review were to investigate the timing and duration of supportive G-CSF regimens used and the resulting outcomes relating to the incidence of neutropenia and associated dose delays/reductions. Additional objectives were duration of FTD/TPI therapy, progression free survival (PFS) and overall survival (OS).The reporting of this study conforms to STROBE guidelines. 10
Results
Fiftyfive consecutive patients in total were identified across 5 UK sites, who had received prophylactic treatment with G-CSF to prevent FTD/TPI related neutropenia within the defined period of data collection (31 August 2019 to 1 September 2021). Twenty five patients received primary G-CSF prophylaxis and 30 secondary G-CSF prophylaxis in total. During analysis, four patients were found to have started treatment outside of the identified inclusion period. Three patients started treatment before the inclusion date, with one starting after. One patient who started before the inclusion date was recorded as a death after the cut-off. It was agreed by the investigators to include these patients in the results, due to the limited patient numbers.
Patient Demographics and Characteristics
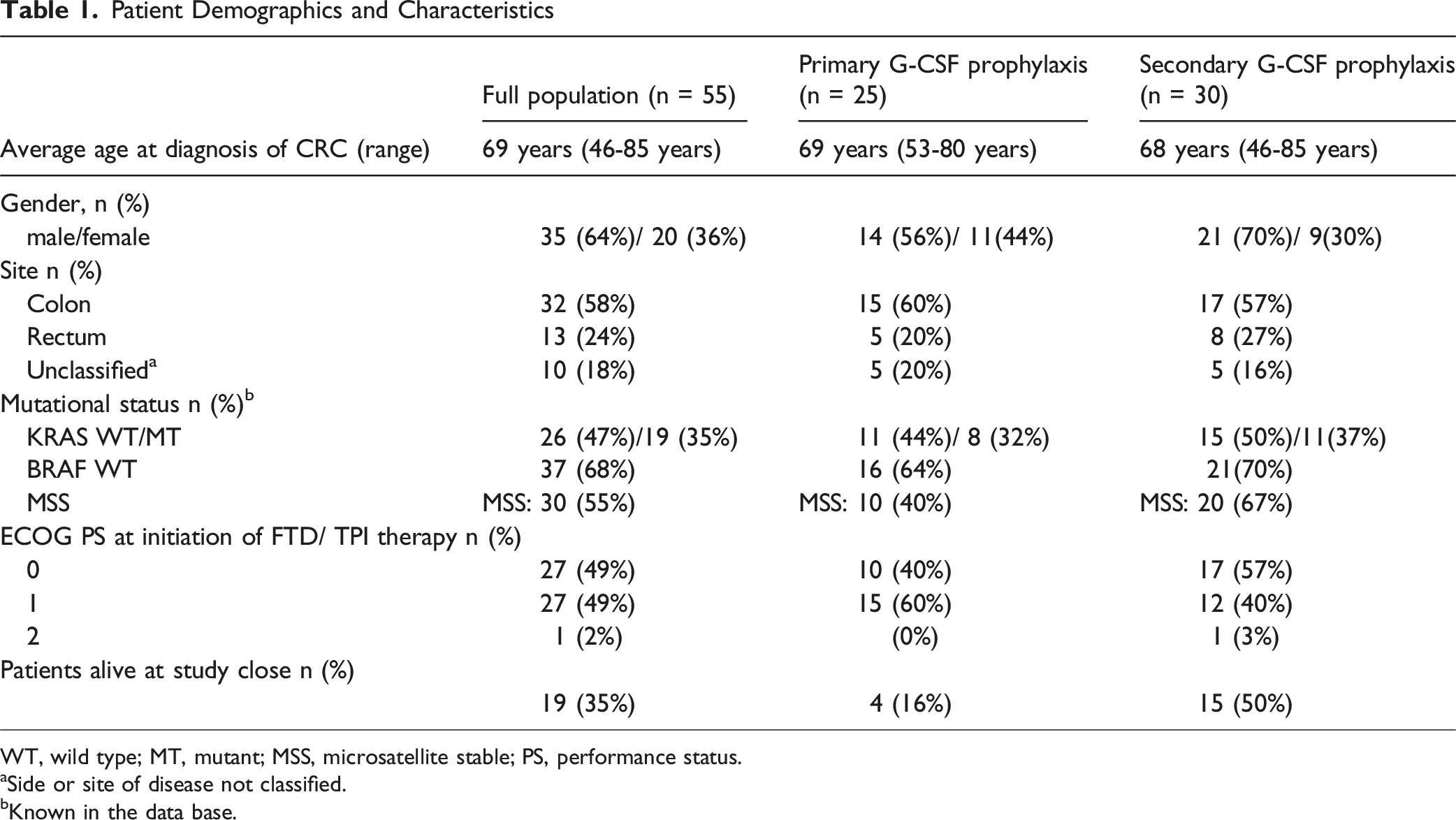
WT, wild type; MT, mutant; MSS, microsatellite stable; PS, performance status.
aSide or site of disease not classified.
bKnown in the data base.
The average age of patient population was 69 but ranged from 46-85 yrs of age. Male sex predominated (64%), as did colon disease (58%) rather than rectal (24%). Due to the observational nature of the study, site of disease was not recorded in 18% of patient notes. Mutational status recording was also not consistent, but 35% of the total population were RAS mutant. Performance status was very evenly split between 0 and 1 (49% each).
As regards the baseline characteristics between the primary and secondary prophylaxis groups, the secondary prophylaxis group had a higher male component (70% vs 56%) with more patients with good performance status (PS 0: 57% vs 40%).
Baseline Characteristics
Site of Metastases

In line with the current license approval, 71% of patient received two previous treatments for metastatic disease (Figure 1), whilst 22% had received three prior lines of treatment. Number of Previous mCRC Treatments (%)
When comparing the secondary prophylactic group with the primary prophylaxis group, metastases were less common in the liver (63%) and lung (47%) but more common in the peritoneum (23%). Similar percentages of patients had ≤2 metastatic sites, but more secondary prophylaxis patients exhibited rapid progression since diagnosis of first metastases (37% vs 28%). Number of previous treatments did not differ markedly between the two groups.
G-CSF Administration
In relation to choice of G-CSF, 85% of patients received filgrastim and 15% received peg-filgrastim (Figure 2). 68% of G-CSF prophylaxis was commenced on day 15 of the cycle, with 99% initiated between days 14 and 22. Choice of G-CSF and schedules were as per local protocols. Recorded Administrations of G-CSF by Day of First Dose
Neutropenia Following G-CSF Use

*Plot shows the total recorded administrations of G-CSF by first day given. Each patient will have contributed multiple data points, one data point per cycle of G-CSF received.
Dose Delays and Reductions Post G-CSF Administration

Number of Cycles of Trifluridine/Tipiracil Complete

The mean number of cycles that patients received was greater in the secondary prophylaxis group, compared to the primary prophylaxis group (7.2 vs 6 cycles). The minimum number of cycles completed was 2, with a maximum of 20 cycles completed within the allocated timescales for the secondary prophylaxis group.
Survival Data


Progression-free Survival

Overall Survival
Number at Risk
Number at Risk
19 out of 55 (35%) patients were still alive in the total population at the time of the study cut-off.
Discussion
This retrospective observational review of 55 patients suggests that the use of G-CSF prophylaxis to reduce FTD/TPI induced neutropenia may be achievable, with 15% of patients experiencing grade 3 or 4 neutropenia in subsequent cycles following the administration of G-CSF. There were no reported incidents of patients experiencing febrile neutropenia and requiring hospitalization. Consistency in the timing and duration of G-CSF use across the multiple sites was observed, which was most commonly delivered as a 5-day course of filgrastim, starting on day 15.
Our real-world data of FTD/TPI use with G-CSF prophylaxis in patients with mCRC showed that patients completed an average of 6 cycles of FTD/TPI, with a mPFS of 4 months and mOS of 10.1 months. In the primary prophylaxis group, mPFS was 3.1 months with median OS of 9.7 months. In the secondary prophylaxis group, mPFS was 7.2 months, mOS 17.5 months.
As this was purely an exploratory analysis, it is not possible to draw definitive conclusions regarding the duration of treatment and survival data. However, both primary and secondary prophylaxis groups demonstrated a longer duration of treatment and PFS/OS than that observed in the RECOURSE trial, where the mean number of cycles complete was 3.3, median PFS was reported as 2 months and median OS as 7.1 months. 5
We believe that there may be a similar benefit in reducing the incidence of neutropenia and maintaining dose intensity with monotherapy, as seen in the combination of FTD/TPI with bevacizumab. 6
Good prognostic factors, relating to overall survival in mCRC patients treated with FTD/TPI monotherapy, have shown to be adequate organ function, ECOG performance status 0-1, <3 metastatic sites when commencing treatment and ≥18 months from diagnosis of first metastases. 11 Age, KRAS status, ethnicity and regional differences are not thought to affect prognosis. 12
Comparing the primary and secondary prophylaxis patient populations, the secondary prophylaxis group had better baseline performance status (PS 0 57% vs 40%). There were similarities in the proportion of patients with <3 metastatic sites (80% secondary prophylaxis vs 76% primary prophylaxis). The percentage of patients with more than 18 months since the diagnosis of first metastasis, hence less aggressive disease, was 63% in the secondary prophylaxis group, compared to 72% in the primary prophylaxis group.
The number of lines of therapy are similar between the different groups; 72% of patients received 1 or 2 previous lines of treatment in the secondary prophylaxis group compared to 70% in the primary prophylaxis group. There were 2 patients in the secondary prophylaxis group who were prescribed FTD/TPI beyond fourth line, suggesting that the number of treatments received by the patient before starting FTD/TPI is not predictive of duration of therapy.
Given the secondary prophylaxis population did not exhibit markedly superior prognostic factors, except for performance status, the occurrence of neutropenia in these secondary prophylaxis patients may possibly have been a marker of trifluridine tipiracil efficacy. This in turn may have led to longer duration of treatment and associated PFS/OS. In RECOURSE, patients who were treated with FTD/TPI and demonstrated higher FTD exposure, as determined by area under the curve, showed an increased risk of chemotherapy induced neutropenia CIN. CIN appeared to be primarily a surrogate of effective dosing but also associated with improved outcomes over those with lower FTD exposure. The onset of any-grade CIN during cycles 1 and 2 represented an independent predictive marker of significantly longer OS, PFS, and time to ECOG PS ≥2. 13
In comparison to the RECOURSE study, use of prophylactic G-CSF appeared to facilitate maintaining dose intensity. In the FTD/TPI group who began at least two cycles of treatment in RECOURSE, 53% had a delay of 4 days or more in beginning their next cycle owing to toxicity; the delay in approximately half of this subgroup extended for 8 days or more. 5 Our data suggested a lower overall rate of dose delay (25%), with half of these extending for 7 days or more.
The manufacturer recommendation relating to dose reduction in the event of neutropenia is to reduce FTD/TPI only when grade 4 neutropenia results in treatment delays of more than 7 days. 3 In this patient cohort, although 15% of patients experienced a grade 3/4 neutropenia, 18% patients were recorded as having a dose reduction. Three patients in total had a grade 4 neutropenia, with delays of more than 7 days to recover in just two patients (4% of total population). This suggests that clinicians employed dose reduction more frequently than that recommended by the protocol for the RECOURSE trial. 14 This may be representative of a real-world patient population and clinical practice, compared to protocol driven trial results.
Since completing the current study, the interventional LONGBOARD study has been published. 15 This study provides a more robust assessment of the efficacy of secondary prophylaxis of neutropenia using G-CSF to maintain dose intensity in patients receiving FTD/TPI monotherapy. The primary endpoint was the rate of patients free from dose reduction or cycles postponement >7 days at 6 months (RFRP-6m). LONGBOARD achieved its primary objective with RFRP-6m rates of 92.5%. Median overall survival and progression-free survival at 12.3 and 3.6 months were comparable to our full patient population. The authors concluded that the occurrence of grade ≥3 neutropenia seems to be associated with FTD/TPI efficacy.
The time period of our study was prior to the approval of combination therapy with FTD/TPI and bevacizumab. It has been shown in a subsequent retrospective analysis, that administration of pegfilgrastim to metastatic colorectal patients receiving combination treatment with FTD/TPI plus bevacizumab enabled the management of severe neutropenia without the requirement for dose delay. The authors also believed that there may be the potential for the management of neutropenia to confer a survival benefit in this patient cohort. 16
Although we found G-CSF to be well tolerated, it has the potential to increase the overall toxicity profile of treatment. Despite no reported adverse events during the study, we do know that bone pain is the most commonly reported adverse effect in practice. Although usually responsive to anti-inflammatory drugs, this can have a potential impact on daily functioning. 17 In addition, rare cases of splenomegaly and splenic rupture have been reported following administration of filgrastim. 18 However, there were no reported adverse events associated with G-CSF during the study. Based on this study’s observations, the introduction of G-CSF does not add significant toxicity to patient treatment.
In an effort to minimize the occurrence of neutropenia without the use of G-CSF, additional research has been performed in biweekly dosing schedules of FTD/TPI both as monotherapy and in combination therapy with bevacizumab. This alternative dosing schedule demonstrated a reduction in myelosuppression, whilst maintaining efficacy.19,20
It is recognized by the authors that this is a small, retrospective review, with some associated limitations. The study had a restrictive sample size and was not powered to make conclusive findings. Comparisons with published data from RECOURSE should be treated with caution due to the different study design. Data collection included patients who received treatment during the COVID pandemic, which may have led to more frequent and less selective use of primary prophylaxis with G-CSF. Due to the line of treatment, disease state and pre-treatment of patients, there is some degree of interpatient variability that we had to accept. Performance status was fairly consistent, but the nature of cancer, having been pre-treated with a number of different lines, may behave differently. Both filgrastim and pegfilgrastim were utilized, but no separate analysis was possible due to sample size. Longer follow-up would have been advantageous to better determine mature OS rates.
Conclusion
In conclusion, this exploratory analysis suggests that G-CSF prophylaxis may be a viable strategy to manage neutropenia in patients receiving FTD/TPI, contributing to better treatment outcomes in terms of maintenance of dose intensity, prolonged PFS and OS.
Footnotes
Acknowledgements
An adapted MS Excel spreadsheet was supplied by Servier to facilitate data collection. Data analysis was conducted by third-party company, Delta-Hat. The corresponding author had final responsibility for the decision to submit for publication.
Ethical Considerations
UK National Health Service Research Ethics Committee review was not required for this study.
Consent to Participate
The participants in the study were not randomised to different groups, the study protocol did not demand changing treatment/ patient care from accepted standards for any of the patients involved and finally, and the results are not intended to be generalisable. The study was registered at participating centres under local guidance for clinical audit. Owing to the retrospective nature of the study, individual consent for this analysis was waived. All patient details were de-identified.
Consent for Publication
All authors have approved the final version of this article and given their consent for its publication.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by Servier, which provided funding to the respective Healthcare Organizations for data collection. The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Vanessa Potter: Consultancy fee from Servier 2022, Victoria Kunene: Financial support for educational meetings from Servier, Conference attendance and professional development support from Servier, Ian Purcell: Conference attendance support, speaker fee Servier, 2023. Sharath Gangadhara: Conference attendance support, Servier, 2023. Laura Bell: Employee of Servier, in a medicine science liaison role.
Data Availability Statement
Supporting data available on request from corresponding author.
