Abstract
The aim is to estimate the prognostic value of lactate dehydrogenase (LDH) in patients undergoing surgical resection for laryngeal squamous cell carcinoma (LSCC). A total of 640 resected LSCC patients were included. Preoperative lactate dehydrogenase (LDH) was assessed. Kaplan-Meier survival analysis and Cox regression analysis were conducted for overall survival (OS) and recurrence-free survival (RFS). Kaplan-Meier analysis, univariate analysis and multivariate analysis demonstrated significant prognostic value for preoperative LDH. Although LDH was predictor of OS, it failed to be a predictor of RFS. The univariate HR and 95% CI of LDH were 0.484 and 0.357-0.658 (P < 0.0001). The multivariate analysis showed that LDH (HR = 0.518, 95% CI: 0.380-0.705, p < 0.0001) was related to OS. Elevated preoperative LDH >132 IU/L was significantly associated with better survival. Preoperative LDH might be an independent prognostic marker of OS in LSCC patients undergoing surgical resection.
Introduction
Laryngeal cancer is one of the most common cancers of the head and neck. 1 Laryngeal squamous carcinoma accounts for 85-95% of laryngeal carcinomas. 2 Surgery and/or radiotherapy and chemotherapy are currently recognized as treatment options for laryngeal cancer. 3 Local recurrence and distant metastasis are the main causes of treatment failure in patients with laryngeal cancer. Many clinical and histopathological parameters have been studied as possible prognostic factors for laryngeal cancer. In recent years, in-depth research on biomarkers has contributed to improving the early diagnosis, treatment, and prognosis of malignant tumors.
Lactate dehydrogenase (LDH) plays an important role by catalyzing the mutual conversion of pyruvate to lactate. 4 LDH is overexpressed in many cancers and is considered to play an important role in tumor maintenance and progression. 5,6 Meta-analysis studies of patients with advanced tumors showed that there was a highly significant association between elevated serum LDH levels and survival rates in patients with solid tumors such as renal cell carcinoma, prostate cancer, gastric cancer, melanoma, nasopharyngeal cancer and lung cancer. Furthermore, LDH is a prognostic biomarker for advanced cancer. 7 A meta-analysis of colorectal cancer and urinary tract cancer also showed a relationship between LDH level and poor overall survival. 8,9 Serum LDH levels combined with TNM staging can more accurately predict disease risk and can be used as independent, robust and reliable biomarkers to predict overall survival (OS), disease-free survival (DFS) and distant metastasis-free survival (DMFS)[10]. Serum LDH is also an independent predictor of progression-free survival in patients with thymic cancer and high-grade osteosarcoma. 10,11 In these patients, it has been found to be a powerful predictor of bone metastasis. The purpose of this study was to evaluate the prognostic value of LDH for overall survival (OS) and recurrence-free survival (RFS) in LSCC patients undergoing surgical resection. Therefore, we conducted a multicenter retrospective study of 640 LSCC patients undergoing surgical resection.
Patients and Methods
Research Population
Between January 2013 and December 2014, a retrospective study involving the medical records of patients undergoing laryngeal surgery (total or partial laryngectomy) was conducted at the Head and Neck Surgery Department of the Cancer Hospital of Harbin Medical University and the Cancer Hospital of the Chinese Academy of Medical Sciences. This study was examined and approved by the ethics committee of the Cancer Hospital of Harbin Medical University and the Cancer Hospital of the Chinese Academy of Medical Sciences. The ethical committee approval number is 2020-17-SJ.The study was carried out according to the principles of the Helsinki Declaration and its amendments. All participants provided informed consent to participate in the study. All patients underwent open laryngeal surgery, and further radiotherapy and chemotherapy were performed according to the pathology and lymph node metastasis after the operation (the specific plan was formulated according to NCCN guidelines). On the basis of meeting the following inclusion and exclusion criteria, we collected a total of 640 patients with laryngeal cancer, including 530 male and 110 female patients.
The inclusion criteria were as follows: (1) laryngeal squamous cell carcinoma confirmed by pathology; (2) laryngeal resection (total or partial laryngectomy); and (3) complete laboratory examination, imaging examination and other clinical data.
The exclusion criteria were as follows: (1) patients with recurrence of the primary tumor; (2) patients with a history of preoperative chemotherapy and/or radiotherapy; (3) patients with positive surgical margins; (4) patients with severe liver and kidney dysfunction; (5) patients with severe heart disease and (6) other exclusion criteria included unavailable blood sample, unavailable follow-up data, or death within the 30 days following surgery. Patients who met the inclusion criteria were followed up for 3 years or more and were reviewed regularly. The flow diagram of assessment for eligibility according to the inclusion and exclusion criteria was as follow (Figure 1).

The flow diagram of assessment for eligibility according to the inclusion and exclusion criteria.
Data Collection
For each patient, extensive demographic, clinicopathological and treatment information were extracted from the electronic medical record system. The demographic data and clinicopathological characteristics of the patients were collected from the database of our institute, including sex, age, body mass index, smoking history, drinking history, tumor location, tumor differentiation degree and tumor TNM stage. The conventional TNM system for laryngeal cancer established by the American Joint Committee on Cancer was used to stage tumors. 12 Lactate dehydrogenase (LDH) was obtained from blood tests before surgery. The blood samples were taken from each patient 1 week before operation.
The primary outcome was overall survival (OS) from diagnosis to death and the second outcome was recurrence free survival (RFS) from CRC diagnosis to disease recurrence or metastasis or death, whichever came first. Outcomes were observed during the follow-up period through December 31, 2019 via an established protocol. Postoperative patients were followed up at 3-6 months intervals for the first year and then annually. The median follow-up time was 57.3 months (range: 1.0-126.1 months).
LDH level is associated with LSCC patient prognosis in our study, we estimated the sample size according to a Cox regression model. A sample size of 508 patients was needed to achieve 90% power (at the 5% level of statistical significance) to detect a hazard ratio (HR) of 0.75 with an overall event rate of 25% in this cohort. Finally, we included approximately 25% more patients and targeted a total sample size of 640 patients.The sample size was calculated using PASS (version 11.0.7, NCSS LLC., USA).
Statistical Analysis
We reported means and standard deviations or counts and frequencies for continuous or categorical variables, respectively. Differences in continuous and categorical covariates between patients with higher versus lower LDH levels were compared with Student’s t tests and chi-square (χ2) tests, respectively.
We first categorized patients into 2 groups according to the optimal cut-off LDH level, which was determined by the ROC method with case-control status as the dependent variable (0, alive; 1, dead). We then conducted univariate and multivariate Cox regression analyses and reported hazard ratios (HRs) and 95% confidence intervals (CIs) to assess the association between LDH levels and the prognosis of laryngeal carcinoma patients. Kaplan-Meier curves and the log-rank test were then conducted to compare OS and RFS between groups. Two-sided statistical significance was defined as P < 0.05. ROC analyses were performed with MedCalc version 12.6.1.0, and all other statistical analyses were performed with SPSS Statistics version 23.0 (IBM, Inc., USA).
Results
Cutoff Value of LDH
According to the ROC curve, the area under the curve (AUC) and 95% CI of preoperative LDH was 0.553 (95% CI 0.513-0.592, P = 0.029), and the optimal cutoff value was 132 (U/L) (Figure 2).

Receiver operating characteristic (ROC) curve analysis for LDH.
Baseline Characteristics of the Patients
A total of 640 patients were enrolled in this study, including 530 males and 110 females. All patients were diagnosed with laryngeal squamous cell carcinoma without distant metastases. The grouped clinicopathological parameters according to LDH are shown in Table 1. The basic information of the 2 groups of patients was compared. The sex (P = 0.996), age (P = 0.502), BMI (P = 0.132), smoking status (P = 0.105), alcohol consumption (P = 0.703), histology (P = 0.937), T stage (P = 0.112), N stage (P = 0.485) and TNM stage (P = 0.286) of the patients were not significantly different between the 2 groups. The level of LDH may be related to tumor location (P = 0.038).
Baseline characteristics of patients grouped by LDH levels.

Overall Survival According to Preoperative LDH
At the last follow-up, 51 and 165 patients died in the LDH ≤132 and LDH >132 groups, and the 5-year OS rates were 45.1% and 69.8%, respectively (p < 0.001) (Figure 3). The univariate HR and 95% CI of LDH were 0.484 (0.357-0.658, P < 0.0001). The survival results were also significantly different for patient age (HR = 1.370, 95% CI: 1.106-1.769, p = 0.016), BMI (HR = 0.572,95% CI: 0.426-0.768, p = 0.0002), alcohol consumption (HR = 1.266, 95% CI: 1.001-1.669, p = 0.049), tumor location (glottic) (HR = 0.511, 95% CI: 0.384-0.679, p < 0.0001), histology (high) (HR = 0.655, 95%, CI: 0.496-0.865, p = 0.003), TNM stage 3 (HR = 2.556, 95% CI: 1.693-3.858, p < 0.0001) and TNM stage 4 (HR = 4.051, 95% CI: 2.740-5.991, p < 0.0001) (Table 2). However, the multivariate analysis only supported that patient age (HR = 1.395, 95% CI: 1.074-1.811, p = 0.013), BMI (HR = 0.639, 95% CI: 0.474-0.861, p = 0.003), alcohol consumption (HR = 1.323, 95% CI: 1.019-1.718, p = 0.036), TNM stage 3 (HR = 2.309, 95% CI: 1.503-3.546, p < 0.0001), TNM stage 4 (HR = 3.552, 95% CI: 2.331-5.414, p < 0.0001) and LDH (HR = 0.518, 95% CI: 0.380-0.705, p < 0.0001) were independent prognostic factors of OS in LSCC patients (Table 2).

Kaplan–Meier estimates of overall survival for patients with LSCC in the LDH >132 (solid line) and LDH≤132 (dotted line) groups. (dotted line) groups. There was a significant difference in survival between the 2 groups (P = 0.010; stratified log-rank test).
Univariate and multivariate analyses of the prognositic effect of LDH on overall survival of patients with laryngeal squamous cell carcinoma.

Abbreviations: HR, hazard ratio; CI, confidence interval; LDH, lactate dehydrogenase.
* Statistically significant p < 0.05.
Recurrence-Free Survival According to Preoperative LDH
There were 117 patients who had evidence of cancer recurrence. The 5-year RFS rates were 75.8% and 78.6% in the LDH ≤132 and LDH >132 groups, respectively (p > 0.05) (Figure 4). Histology (HR = 0.594, 95% CI: 0.403-0.876, p = 0.009), TNM stage 2 (HR = 1.844, 95% CI: 1.094-3.109, p = 0.022), TNM stage 3 (HR = 2.462, 95% CI: 1.414-4.285, p = 0.001) and TNM stage 4 (HR = 2.989, 95% CI: 1.713-5.215, P < 0.0001) were associated with decreased RFS in univariate analysis (Table 3). Significant differences in survival curves were not found in the analysis of LDH (HR = 0.847, 95% CI: 0.520-1.380, p = 0.0505) (Table 3).

Kaplan–Meier estimates of RFS for patients with LSCC in the LDH >132 (solid line) and LDH≤132 (dotted line) groups. There was no significant difference in RFS between the 2 groups (P = 0.010; stratified log-rank test).
In multivariate analysis, histology (HR = 0.647, 95% CI: 0.433-0.966, p = 0.033), TNM stage 2 (HR = 1.83, 95% CI: 1.089-3.100, p = 0.023), TNM stage 3 (HR = 2.233, 95% CI: 1.274-3.913, p = 0.005) and TNM stage 4 (HR = 2.648, 95% CI: 1.503-4.665, P = 0.001) had significant predictive values for RFS (Table 3). LDH (HR = 0.883, 95% CI: 0.540-1.444, p = 0.621) was not found to have significant predictive value for RFS (Table 3).
Univariate and multivariate analyses of the prognositic effect of LDH on recurrence-free survival of patients with laryngeal squamous cell carcinoma.
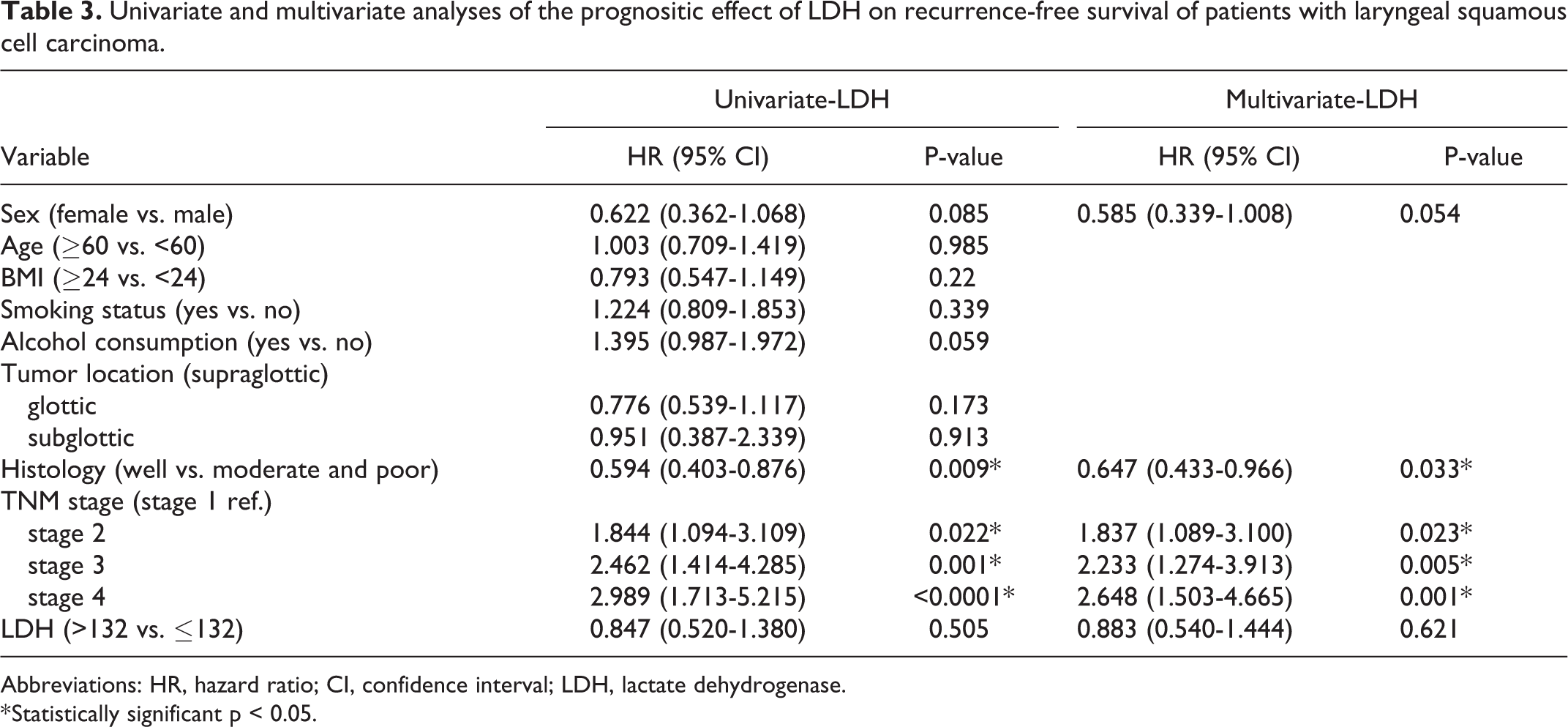
Abbreviations: HR, hazard ratio; CI, confidence interval; LDH, lactate dehydrogenase.
* Statistically significant p < 0.05.
Discussion
The tumor microenvironment (TME) is a complex dynamic cellular environment that consists of tumor cells, stromal cells, blood vessels, extracellular matrix (ECM), growth factors and cellular metabolites. 13 Cancer cells preferentially convert glucose into lactic acid through aerobic glycolysis, also known as the “Warburg effect.” 14 Lactate dehydrogenase (LDH), one of the key metabolic enzymes present in the TME, plays a crucial role in the conversion of pyruvate to lactate, making it an important participant in cancer metabolism. 15
Serum LDH was first studied as a clinical biomarker in malignant lymphoma. 16 Relevant studies have shown that LDH is a significant adverse prognostic factor for invasive B cell lymphoma, mantle cell lymphoma, and follicular lymphoma. 17 -19 Serum lactate dehydrogenase (LDH) levels are one of the most useful independent prognostic factors in metastatic melanoma. 20 Some researchers believe that LDH is an independent prognostic factor of lung cancer and is closely related to the TNM stage, therapeutic effect and survival of lung cancer. 21,22 Additionally, serum LDH levels can be used as prognostic indicators for malignant tumors such as colorectal cancer, 23 nasopharyngeal carcinoma, 24 prostate cancer 25 and breast cancer. 26 Our study showed that LDH >132 IU/L was associated with better prognosis in LSCC patients. There was a significant correlation between preoperative serum LDH levels and overall tumor survival (OS) but not with recurrence-free survival (RFS). The survival rate of patients with preoperative serum LDH levels higher than 132 IU/L was significantly higher (Figure 3). This result may be associated with the tumor location, which is closely related to the prognosis of LSCC patients. The prognosis of glottic laryngeal carcinoma is significantly better than that of supraglottic laryngeal carcinoma and subglottic laryngeal carcinoma. Our study showed that the proportion of glottic laryngeal carcinoma in the LDH >132 IU/L group was much higher than that in the LDH≤132 IU/L group (P = 0.038). Thus, preoperative serum LDH levels combined with TNM staging can be a more accurate predictor of overall survival (OS) in laryngeal squamous cell carcinoma. In addition, our research is a multicenter clinical study, and each research center obtained consistent results to avoid the possible limitations of single-center research. Therefore, our conclusion has more extensive significance and greater credibility.
LDH was also studied in cultured tumor cells and tumor tissues in vitro, and these studies suggested that LDH plays an important role in early clinical tumor diagnosis. The results showed that LDH was expressed in breast tissue, prostate cancer and colon cancer, and it was a sign of anaerobic metabolism changes in cells, facilitating the proliferation of cancer cells under a hypoxic microenvironment. 27 -29 Ki-67-positive cancer cells were significantly reduced, and the apoptosis rate was enhanced in LDH-positive tumor samples. Approximately 23.9% of LDH enzyme activity in breast cancer tissue was higher than that in radiodense tissue (ACR4), which plays an important role in the diagnosis of breast cancer. 30 The increase in LDH levels in the tumor tissue of the juvenile breast cancer cell line was related to high cell proliferation. 31 One study with oxidized SiHa human cervical adenocarcinoma cells as the main model showed that LDH controls early tumor progression and the number of cancer cells and has a negative impact on the survival of patients. LDH silenced by sird HB-2 can reduce the number of cells in cervical cancer cell lines, the MCF7 human breast cancer cell line, the HCT116 and WiDr human colon cancer cell lines, the SKOV3 human ovarian cancer cell line, and the T98G and U373 human glioblastoma cell lines. Therefore, we will investigate LDH in laryngeal cancer tissue samples and in vitro cultured laryngeal cancer cell lines to reveal the prognostic significance of LDH in laryngeal cancer. In addition, we will study the effect of LDH in vitro and in vivo on the occurrence and development of laryngeal cancer and explore whether LDH can be a potential therapeutic target for laryngeal cancer. The present study has several potential limitations. First, selection bias may have influenced survival data due to the retrospective nature of this study. Second, this study was conducted with a relatively small number of subjects, further studies with adequate statistical power and a larger number of patient subgroups are needed to determine the reliability and accuracy of our study. Third, due to the lack of independent validation cohort, future studies with large sample size are needed.
Conclusion
Overall, preoperative serum LDH is significantly associated with OS in LSCC patients undergoing surgical resection. Preoperative serum LDH may be an independent prognostic marker of OS in LSCC patients undergoing surgical resection. However, further histological and cytological studies are still needed to confirm and extend our results to improve the treatment of LSCC.
Footnotes
Acknowledgments
The authors report no conflicts of interest. This work had no specific funding. This study was examined and approved by the ethics committee of the Cancer Hospital of Harbin Medical University and the Cancer Hospital of the Chinese Academy of Medical Sciences. The study was carried out according to the principles of the Helsinki Declaration and its amendments. All participants provided informed consent to participate in the study.
Author Contributions
These authors have contributed equally to the work. EG, LG, XZ, XY, RC, CX, and CZ participated in the experiments and data collection. EG and LG drafted the manuscript. CA, KS, GW, CD, ZY, JG, JS, HM, and XL did the statistical analysis of data. SM and XM participated in the design of the study and the general supervision of the research group. SM made strict revision of this manuscript and drafted parts of it.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
