Abstract
Background
Cervical cancer (CC) is the fourth most common malignancy of the female genital tract. Human Papillomavirus (HPV) is the main cause of precancerous lesions and CC cases worldwide
Objective
We assessed the prevalence and distribution of HPV types and
Methods
HPV genotypes were detected by 3 methods depending on the year of in which the sample was analyzed: MY09/11 RFLPs (1997 to 2010), GP5+/6+ primer systems (2005 to 2010) and INNO-LiPA HPV Genotyping Extra (2010 to 2019) in cervical samples (No-IL: 4445; LSIL: 2464; HSILs: 151 and CC: 253) from women from southern Mexico.
Results
The overall HPV prevalence was 54.17%, and hpv-16 was the most common genotype. In single infection, the high-risk HPV genotypes (group 1) were associated with squamous intraepitelial lesions (LSIL: HPV–39 (OR = 10.58, 95% CI 4.09–27.36, P < .001); HSIL: HPV-31 (OR = 14.76, 95% CI 6.56–33.20, P < .001); and CC: HPV-16 (OR = 25.01, 95% CI 18.83–33.21, P < .001). In multiple infections, the HPV genotypes (HPV-16 and HPV-18) were also associated with a high risk of lesions [LSIL: HPV-18 (OR = 3.45; 95% CI 1.36–8.91; P = .009); HSIL: HPV-18 (OR = 5.12; 95% CI 1.21–21.68; P = .026); and CC: HPV-16 (OR = 3.03; 95% CI 1.72–5.32; P < .001)] compared to single infection. In the analysis adjusted for age, giving birth, and cigarette smoking, a significant increase in the risk of LSIL, HSIL, and CC was maintained.
Conclusions
This study provides current data on the prevalence and distribution of HPV genotypes in women from southern Mexico, which could serve as a valuable reference to guide nationwide CC screening programs and provide scientific evidence that could be useful for vaccine development efforts. Likewise, it was identified that infection with carcinogenic HPV genotypes is an independent risk factor for LSIL, HSIL, and CC.
Keywords
Introduction
Cervical cancer (CC) is the fourth most common cancer in women over 15 years of age, with an estimated 569 681 new cases and 311 299 deaths worldwide in 2018. In Mexico, CC is the third most common cancer in women over 15 years of age; approximately 7868 new CC cases are diagnosed, and 4121 deaths occur annually. 1 In 2015, the Mexican Ministry of Health reported that the state of Guerrero had a CC-related mortality rate of 14.1 deaths per 100 000 women. 2 More than 200 types of human papillomavirus (HPV) have been characterized, and 65 of the HPV types are found predominantly or exclusively in the anogenital tract.3–6 According to the epidemiological classification based on the risk of developing cancer, the types of HPV are grouped as “high risk (HR)” (HPV-16, -18, −31, −33, −35, −39, −45, −51, −52, −56, −58, and −59), “probably high-risk (pHR)” (HPV-26, −34, −53, −66, −67, −68, −69, −70, −73, −82, −85, and −97,“low-risk (LR)” (HPV-6, −11, −13, −40, −42, −43, −44, −54, −61, −72, −81, and −89) and “undetermined-risk” (HPV-2a, −3, −7, −10, −27, −28, −29, −30,−32, −34,−55, −57, −62, −71, −74, −77, −83, −84, −86, −87, −90, and −91).7,8 The International Agency for Research on Cancer (IARC) classifies HPV types into Group 1 (carcinogenic: HPV-16 -18, −31, −33, −35, −39, −45, −51,−52, −56, −58, and −59), Group 2 (probably or possibly carcinogenic: HPV-5, -8, -26, −30, −34, −53, −66, −67,−68, −69, −70, −73, −82, −85, and −97), and Group 3 (low-risk types: 6, 11, 40, 42, 43, 44, 54, 61, 72, 81, 89, other beta and gamma types).9-11
High-risk HPVs, belonging to IARC carcinogenic Group 1, are the cause of virtually 100% of cervical cancer cases, and there is epidemiological evidence that they are also associated with vulvar, vaginal, penile and anal cancer. Worldwide, HPV-16 and HPV-18 contribute to over 70% of all cervical cancer cases. HPV types 31, 33, 35, 45, 52, and 58 cause approximately 20% of cervical cancer cases worldwide.(12) The prevalence of HPV-16 and/or HPV-18 among women with no intraepithelial lesion (No-IL) is 3.9%, with low-grade squamous intraepithelial lesions (LSIL) is 25.8%, with high-grade squamous intraepithelial lesions (HSIL) is 51.9%. 13
Several studies on HPV prevalence in some regions of Mexico show differences in prevalence ranging from 37.6% to 79.2%. A study that included 2956 cervical samples from women from different regions of Mexico with No-IL, LSIL, HSIL, and CC, found that the overall prevalence was 67.1% and HPV-16 was the most frequent among HPV-positive women with No-IL (6.2%), LSIL (22.1%), HSIL (44.3%), and CC (57%). A study conducted in central Mexico, 79.2% of the samples were positive for at least one HPV genotype, and HPV-51 was the most prevalent genotype, followed by HPV-16. 14 In northeastern Mexico, HPV-16 was the most prevalent genotype, followed by HPV-6. In south-central Mexico HPV-52 was the most prevalent, followed by HPV-16. 15 Likewise, in northeast Mexico, 59% of samples were positive for at least one HPV genotype, and HPV-16 was the most prevalent genotype, followed by HPV-18. 16 In a study conducted in healthy women in Acapulco, in the state of Guerrero, located in southern Mexico, the prevalence of HPV was 37.6%. 17 In a previous study conducted in women in the state of Guerrero, an HPV prevalence of 53.9% was found in cervical samples, 40.9% in No-IL, 94.5% in LSIL, 83.5% in HSIL, and 100% in CC. HPV-16 was the most frequent in HSIL and CC. 18
The prevalence of HPV infections differs among countries and can even vary among regions of the same country. This difference is partly influenced by migration, sexual behavior, type of cervical lesion in women sampled, and diagnostic methods, which have an important role. Over the years, diagnostic methods have been improved, aiding in the identification of different types of HPVs in the same patient (multiple infections), which has an important impact on the prevalence of HPVs in women with precursor lesions and CC. In this study, our objective was to update the available information about the prevalence of HPV and its association with precancerous lesions and CC in women of the State of Guerrero, Mexico using data collected over 22 years from women with CC or precursor lesions and women without ILs. This updated information is important for those seeking to develop interventions to prevent CC and vaccines.
Materials and Methods
Study Population
A total of 7313 females aged 14-97 years, residents of the state of Guerrero Mexico, were enrolled in this study from January 1997 to December 2019. (Figure S1). The participants were classified into 4 grades of cervical diagnosis: (1) no intraepithelial lesions (No-IL), (2) low-grade squamous intraepithelial lesions (LSIL), (3) high-grade squamous intraepithelial lesions (HSIL), and (4) cervical cancer (CC). Sexually active women were considered for this study. The exclusion criteria for the study were as follows: patients with other malignancies, patients with a history of chemotherapy or radiotherapy, patients with previous physical cervical treatments, patients with autoimmune diseases, and women without experience of sexual intercourse.
Sample Collection
Sample collection was performed as described by Illades-Aguiar et al, 2009. Briefly, all women included in this study provided exo-endocervical, exfoliated cell samples collected by sampling the ectocervix with an Ayre spatula and endocervix with a cytobrush, ensuring that tissue from the transformation zone was obtained. Smears were utilized for cytomorphological examination using conventional Papanicolaou. For women who were cytologically diagnosed with No-IL or LSIL, a biopsy was not performed. For women with a cytological diagnosis of HSIL or CC, a biopsy was performed to confirm the diagnosis. 19
Pap smears were stained and read by a cytopathologist, and classification was performed according to the Bethesda System. 20 The histological diagnosis was made by a pathologist according to the classification system of the International Federation of Gynecology and Obstetrics (FIGO). 21
HPV Detection and Typing
For HPV detection, cervical cytobrush samples (in participants with No-ILs and LSILs) were placed in lysis buffer (10 mM Tris pH 8.0, 20 mM EDTA pH 8.0, and .5% sodium dodecyl sulfate) to separate any cervical material and stored at −20°C until analysis. To detect HPV in women with HSILs or CC, biopsy specimens were stored in phosphate-buffered saline (PBS) and stored at −70°C until analysis. DNA was purified from cervical samples with the standard SDS–proteinase K–phenol–chloroform method. 22
HPV DNA was detected by 3 methods depending on the year in which the sample was obtained and analyzed as previously described: (1) between 1997 and 2010 (4296 samples; refer to Table S1), HPV detection was performed with the polymerase chain reaction-restriction fragment length polymorphism (PCR-RFLP) system using the MY11/MY09 L1 gene; (2) between 2005 and 2010 (1039 samples; refer to Table S1), detection was performed with the GP5+/6+ PCR system and by sequencing analysis when the samples analyzed with MY09/11 PCR were negative, according to data reported by Illades-Aguiar et al, 2010 18 ; and (3) between 2010 and 2019 (1978 samples; refer to Table S1), detection was performed with the INNO-LiPA HPV Genotyping Extra CE assay. 23
For the PCR-RFLP system involving the MY11/MY09 L1 gene, PCR was carried out in a total volume of 30 μL using 1 μg of template DNA; 1X PCR buffer (10 mM Tris-HCl pH 8.5, 50 mM KCl); 2 mM MgCl2; 150 mM of each deoxynucleotide triphosphate (dATP, dTTP, dCTP, and dGTP); .8 mM of each consensus primer, namely, MY11 (5′GCMCAGGGWCATAAYAATGG3′) and MY09 (5′-CGTCCMARRGGAWACTGATC-3′); and 1.25 U of Ampli Taq GoldTM (Applied Biosystems, Foster City, CA). DNA was amplified in a GeneAmp PCR System 2400 (Applied Biosystems, Foster City, CA) with the following program: 10 min at 95°C, 40 cycles of 60 sec at 95°C, 60 sec at 58°C, and 60 sec at 72°C, with a final extension step at 72°C for 10 min. To check the integrity of the DNA, a region of 268 base pairs of the cellular β-globin gene was amplified using primers GH20 (5′-GAAGAGCCAAGGACAGGTAC-3′) and PC04 (5′-CAACTTCATCCACGTTCACC-3′). 24 The HPV-16 plasmid, CaSki cells and HeLa cells were selected as positive controls, and human DNA without HPV DNA and water were chosen as negative controls. All HPV-positive samples were typed by RFLP analysis. Each restriction reaction was performed separately in a final volume of 20 μL using 2 μL of the MY09/11 PCR product and the restriction enzymes BamHI, DdeI, HaeIII, HinfI, PstI, RsaI, and Sau3AI (Invitrogen, Carlsbad, CA) according to the manufacturer’s instructions. RFLP analysis was performed to identify more than 40 genital HPV types. 25
From 2005 to 2010, when samples analyzed with the PCR-RFLP system using the MY11/MY09 L1 gene were negative or HPV could not be genotyped by RFLPs, the presence of HPV DNA was detected using the general GP5+/6+ PCR system.
PCR was carried out in a total volume of 20 μL containing 1 μL of the first reaction (PCR using the MY11/MY09 L1 gene); 1X PCR buffer (10 mM Tris-HCl pH 8.5, 50 mM KCl); 2 mM MgCl2; 150 mM of each deoxynucleotide triphosphate (dATP, dTTP, dCTP, and dGTP); .8 mM of each consensus primer, namely, GP5+ (5′-TTTGTTACTGTGGTAGATACTAC-3′) and GP6+ (5′-GAAAAArAAACTGTAAATCArATTC-3′); and 1.25 U of Ampli Taq GoldTM (Applied Biosystems, Foster City, CA). DNA was amplified in a GeneAmp PCR System 2400 (Applied Biosystems, Foster City, CA) with the following program: 40 cycles of 60 sec at 4°C, 60 sec at 55°C and 60 sec at 72°C, with a final extension step at 72°C for 5 min. 26
PCR GP5+/GP6+ products were subjected to sequencing analysis. Briefly, PCR products were purified using 75% isopropanol and columns (Centri-Sep Spin Columns, Applied Biosystems, Foster City, CA). Forward and reverse sequencing reactions were carried out using 15 to 30 ng of template DNA from the PCR amplimers, 3.2 pmol of the GP5+ and GP6+ primers (each) and the reagent from the Big Dye Terminator Chemistry Version 3 Cycle Sequencing Kit (Applied Biosystems, Foster City, CA) in a final volume of 20 μL, according to the manufacturer’s instructions. The sequencing rounds were performed using an automated sequencer (310 ABI PRISM Genetic Analyzer, Applied Biosystems, Foster City, CA). The sequences obtained were compared with sequences available in the GenBank database using the BLAST tool. 27
The INNO-LiPA HPV Genotyping Extra CE assay is a line probe assay that is based on the principle of reverse hybridization for the qualitative detection and identification of genotypes of HPV by the detection of specific sequences (65 bp) in the L1 region of the HPV genome using broad-spectrum SPF10 biotinylated primers. PCR was performed in a final reaction volume of 50 μl containing 40 μl of the PCR master mix and 10 μl of the extracted DNA. The amplification program was as follows: 9 min at 94°C, 40 cycles of 30 sec at 94°C, 45 sec at 52°C, and 45 sec at 72°C, with a final extension step at 72°C for 10 min. The PCR amplicons were subsequently analyzed by reverse hybridization on a nitrocellulose strip following the manufacturer’s instructions. The line probe assays were evaluated by 2 independent observers and would have been adjudicated by a third observer if different results were reported, although this approach was not applied in this study. The INNO-LiPA HPV Genotyping Extra CE assay is designed for the identification of HPV from HR-HPVs: 16, 18, 31,33, 35, 39, 45, 51, 52, 56, 58, and 59; pRH-HPVs: 26, 53, 66, 67, 68, 70, 73, and 82; LR-HPVs: 6, 11, 40, 42, 43, 44, 54, 61, 81, and 89); and UR-HPVs: 62 and 83.
HPV types were classified according to the IARC,9–11 Muñoz et al, 7 and Burk et al, 8 HPV classification system. Forty-eight different HPV types were identified in this study: 12 HR genotypes (group 1: HPV-16, 18, 31, 33, 35, 39, 45, 51, 52, 56, 58, and 59), ten pHR genotypes (group 2: HPV-26, 34, 53, 66, 67, 68, 69, 70, 73, and 82), 12 LR genotypes (group 3: HPV-6, 11, 13, 40, 43, 44, 54, 61, 72, 81, and 89), and 14 genotypes of the UR genotypes (HPV-7, 32, 55, 57, 62, 71, 74, 83, 84, 86, 87, 90, 102, and 106).
Statistical Analysis
Statistical analyses were performed using STATA software version 14.0 (StataCorp, College Station, TX, USA). The results are presented as the mean ± standard deviation (SD) or median, 25th and 75th interquartile. The Kolmogorov-Smirnov test was used to assess the distribution of continuous variables (age). Descriptive analysis was performed on the distribution of HPV genotypes using indicators such as frequency and prevalence. Chi-square or Fisher’s exact tests were conducted to determine the differences in HPV prevalence between 2 different groups. To estimate the risk of CC and precursor lesions associated with HPV types and other risk factors, odds ratios (ORs) and 95% confidence intervals (CIs) were calculated by using univariate logistic regression (non-adjusted model). Age (age groups: 14–24, 25–34, 35–44, 45–44, 45–54, and >55), giving birth (0, 1–2, and ≥3), and cigarette smoking (yes or no) were included in a second multivariate logistic analysis (adjusted model) to identify independent risk factor (HPV infection) for risk of precursor lesions and CC. P values <.05 were considered statistically significant.
Results
Characteristics of the Population
Demographic and clinical characteristics of the study population.
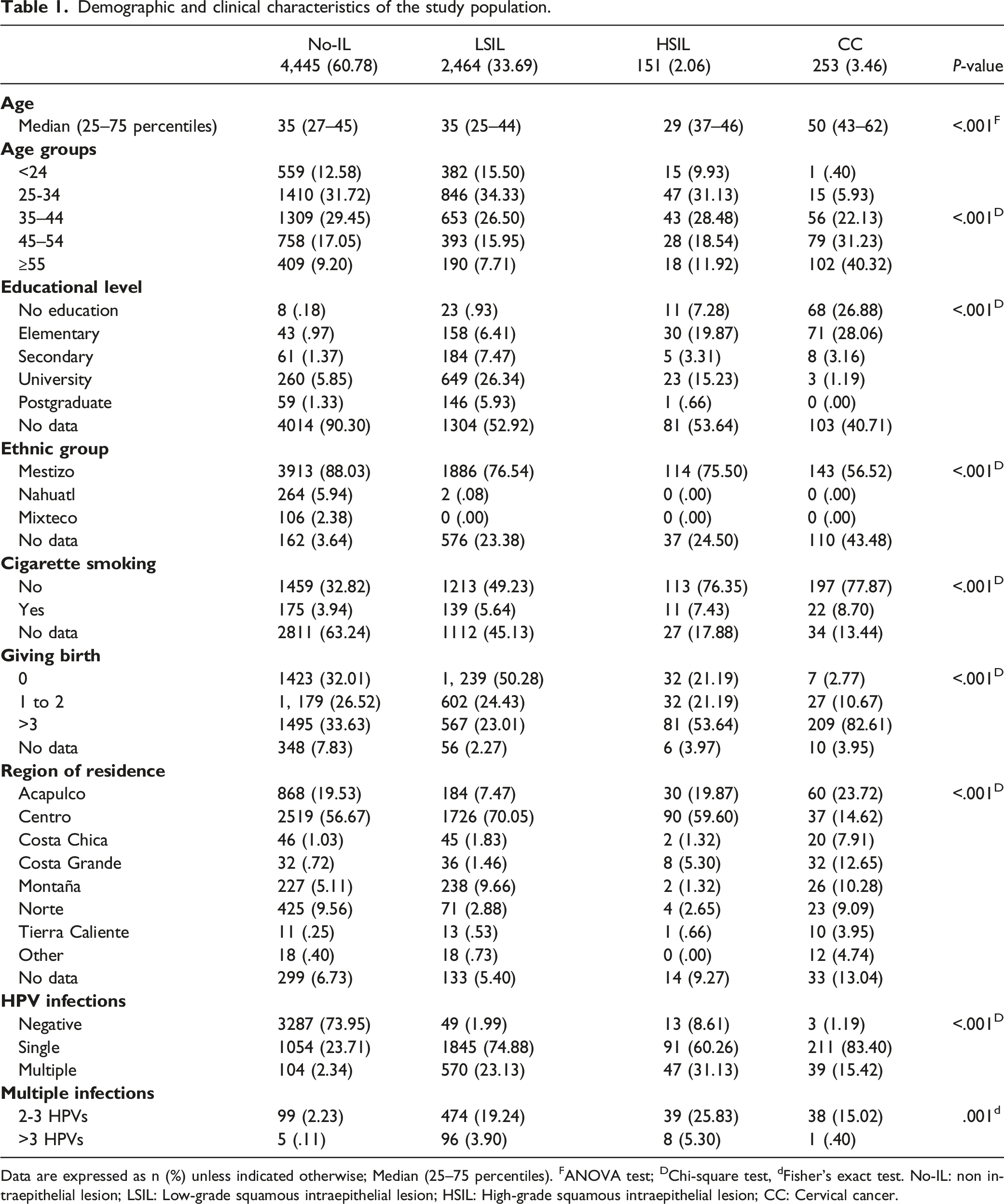
Data are expressed as n (%) unless indicated otherwise; Median (25–75 percentiles). FANOVA test; DChi-square test, dFisher’s exact test. No-IL: non intraepithelial lesion; LSIL: Low-grade squamous intraepithelial lesion; HSIL: High-grade squamous intraepithelial lesion; CC: Cervical cancer.
Some potential risk factors related to the risk of lesions were examined. Table S2 summarizes potential risk factors and the association of these factors with CC. In the non-adjusted model for age, cigarette smoking, and having 3 or more births, an association with the risk of developing CC was observed. In the fully adjusted model, women >55 years of age (OR = 57.76, 95% CI 7.87–423.95, P < .001), cigarette smoking (OR = 3.31, 95% CI 1.96–5.58, P < .001), and having 3 or more births (OR = 6.32, 95% CI, 3.77–10.58, P < .001) showed statistically significant associations with CC (Table S2).
Prevalence of HPV Grouped by Year
The total data of this study ranged from January 1997 to December 2019. Considering only samples that were positive for HPV infection (3961 women), the following results were obtained. HR-HPV infections peaked in 1997 and then fell slightly, peaking in 2004 and slightly increasing in 2008 (Figure 1A). Multiple infections peaked in 2014. Likewise, it was observed that was higher than multiple infection, which peaked in 1997, and decreased slightly to peak again in 2010 and 2014. While infections with 2 or 3 genotypes peaked in 2014 (Figure 1B), HPV-16 in single infections prevailed during the 22 years. Other HPV infections did not change significantly over time (Figure 1C), while in multiple infections, HPV-16 also prevailed from 2006 to 2019, reaching its maximum peak in 2015 (Figure 1D). Prevalence of HPV grouped by year in the study population. A) Prevalence of HPV groups. B) Prevalence of coinfections with 2 or 3 HPV genotypes. C) Prevalence of HR-HPV in patients with single infections. D) Prevalence of HR-HPV in patients with multiple infections. Human papillomavirus (HPV) types according to International Agency for Research on Cancer (IARC), Muñoz et al and Burk et al. High-risk HPVs (Group 1); Probable high-risk HPVs (Group 2); Low-risk HPVs (Group 3); Undefined-risk HPV. Abbreviations: HPV: Human papillomavirus; HR-HPV: high-risk HPV; pHR-HPV: probable high-risk HPV; LR-HPV: low-risk HPV; UR-HPV: Undefined-risk HPV; M-HPV: multiple HPV types.
Prevalence and Distribution of HPV Genotypes by Lesion Grade
HPV type distribution according to cervical lesion grades-.

Data are expressed as n (%) unless indicated otherwise. No-IL: non intraepithelial lesion; LSIL: Low-grade squamous intraepithelial lesion; HSIL: High-grade squamous intraepithelial lesion; CC: Cervical cancer. Human papillomavirus (HPV) types according to International Agency for Research on Cancer (IARC), Muñoz et al and Burk et al. HPV classification system. High-risk HPV (Group 1); Probable high-risk HPV (Group 2); Low-risk HPVs (Group 3); Undefined-risk HPV. Abbreviations: HPV: Human papillomavirus; HR-HPV: high-risk HPV; pHR-HPV: probable high-risk HPV; LR-HPV: low-risk HPV; UR-HPV: Undefined-risk HPV; M-HPV: multiple HPV types; No-IL: women without intraepithelial lesion; LSIL: low-grade squamous intraepithelial lesion; HSIL: high-grade squamous intraepithelial lesion; CC: Cervical cancer.
The infection with a single HPV genotype in women with No-IL, LSIL, HSIL, and CC was observed in the 91.02% (95% CI 89.92–92.60, n = 1054), 76.40% (95% CI 74.65–78.08, n = 1845), 65.94% (95% CI 57.39–73.79, n = 91), and 84.40% (95% CI 79.30–88.66, n = 211) of the patients, respectively. While the most common group in women with No-IL, LSIL, HSIL, and CC were high-risk HPVs (HR-HPVs or group 1), accounting for 49.83% (95% CI 46.91–52.75, n = 577) 40.75% (95% CI 38.78–42.74, n = 984), 56.52% (95% CI 47.82–64.93, n = 78), and 81.60% (95% CI 76.23–86.20, n = 204) of the infections, respectively (Figure 2). Likewise, multiple HPV infections in women with No-IL, LSIL, HSIL, and CC were detected in the 8.98% (95% CI 7.40–10.78, n = 104), 23.60% (95% CI 21.92–25.35, n = 570), 34.06% (95% CI 26.21–42.60, n = 47), and 15.60% (95% CI 11.33–20.70, n = 39) of the samples, respectively (Table 2 and Figure 2). On the other hand, it was observed that independently of the method used, the HPV group most found was HR-HPV (Table S3). Likewise, it was observed that with the introduction of the INNO-LiPA assay, a greater number of cases with multiple infections could be identified (Table S3) Prevalence of HPV groups among patients with different cervical lesion grades. A) Prevalence of HPV groups in women No-IL. B) Prevalence of HPV groups in patients with LSIL. C) Prevalence of HPV groups in patients with HSIL. D) Prevalence of HPV groups in patients with CC. Human papillomavirus (HPV) types according to International Agency for Research on Cancer (IARC), Muñoz et al and Burk et al. High-risk HPVs (Group 1); Probable high-risk HPVs (Group 2); Low-risk HPVs (Group 3); Undefined-risk HPV. Abbreviations: HPV: Human papillomavirus; HR-HPV: high-risk HPVs; pHR-HPV: probable high-risk HPV; LR-HPV: low-risk HPV; UR-HPV: Undefined-risk HPV; M-HPV: multiple HPV types; No-IL: non intraepithelial lesion; LSIL: Low-grade squamous intraepithelial lesion; HSIL: High-grade squamous intraepithelial lesion; CC: Cervical cancer.
Prevalence and Distribution of HPV Types in Single Infections
The two most prevalent HR-HPV genotypes (group 1) in women with No-ILs, LSILs, and HSIL were HPV-16 and HPV-33, (Table 2 and Figure 3A to 3C). In women with CC, the two most prevalent HR-HPV genotypes (group 1) were HPV-16, and HPV-18, which were identified in 56.40% (95% CI 50.00–62.64, n = 141) and 9.20% (95% CI 5.92–13.48, n = 23), of the patients, respectively (Table 2 and Figure 3D). Prevalence of HPVs among patients with different cervical lesion grades and with single infections. A) Prevalence of HPV genotypes in women No-IL. B) Prevalence of HPV genotypes in patients with LSIL. C) Prevalence of HPV genotypes in patients with HSIL. D) Prevalence of HPV genotypes in patients with CC. Human papillomavirus (HPV) types according to International Agency for Research on Cancer (IARC), Muñoz et al and Burk et al. High-risk HPVs (Group 1: HPV-16, 18, 31, 33, 35, 39, 45, 51, 52, 56, 58, and, 59); Probable high-risk HPVs (Group 2: HPV-26, 34, 53, 66, 67, 68, 69, 70, 73, and 82); Low-risk HPVs (Group 3: HPV-6, 11, 13, 40, 42, 43, 44, 54, 61, 72, 81, and 89; Undefined-risk HPVs (HPV-7, 32, 55, 57, 62, 71, 74, 83, 84, 86, 87, 90, and 102). Abbreviations: HPV: Human papillomavirus; HR-HPV: high-risk HPV; pHR-HPV: probable high-risk HPV; LR-HPV: low-risk HPV; UR-HPV: Undefined-risk HPV; M-HPV: multiple HPV types; No-IL: non intraepithelial lesion; LSIL: Low-grade squamous intraepithelial lesion; HSIL: High-grade squamous intraepithelial lesion; CC: Cervical cancer.
The participants that were positive for one HPV genotype were divided into five groups ranging from ≤24 years, 25–34 years, 35–44 years, 45–54 years, and ≥55 years. The prevalence of HPV-16 and HPV-33 by age group shows an increased prevalence for the youngest women with No-IL (HPV-16: 5.27% and HPV-33: 4.49%) and LSIL (HPV-16: 6.62% and HPV-33: 1.82%) in the population aged 25–34 years. In women with HSIL the highest prevalence of HPV-16 (9.42%) and HPV-33 (5.07%) was observed in the age range of 35–44 years. While the prevalence of HPV-16 (20.40%) was higher in the age group of 45 to 54 years in CC patients (Table S4 and Figure 4). On the other hand, it was observed that independently of the method used, the HPV genotype most found was the HVP-16 (Table S5). Prevalence of HPV grouped by age in patients with single infections. A) prevalence of HR-HPVs infection in No-IL women in each age group. B) prevalence of HR-HPVs infection in LSIL patients in each age group. C) prevalence of HR-HPVs infection in HSIL patients in each age group. D) Prevalence of HR-HPVs infection in CC patients in each age group. Abbreviations: HPV: Human papillomavirus; HR-HPV: high-risk HPV; pHR-HPV: probable high-risk HPV; LR-HPV: low-risk HPV; UR-HPV: Undefined-risk HPV; M-HPV: multiple HPV types; No-IL: non intraepithelial lesion; LSIL: Low-grade squamous intraepithelial lesion; HSIL: High-grade squamous intraepithelial lesion; CC: Cervical cancer.
Prevalence and Distribution of HPV Genotypes in Patients With Multiple Infections
The presence of multiple HPV genotypes in one individual was common. The prevalence of multiple HPV infections in this study was 8.98% (95% CI 7.40–10.78, n = 104) in women with No-IL, 23.60% (95% CI 21.92–25.35, n = 570) in LSIL patients, and 34.06% (95% CI 26.21–42.60, n = 47) in HSIL patients, whereas in CC patients, it was 15.60% (95% CI 11.33–20.70, n = 39) (Table 2 and Figure 2).
The two most prevalent HR-HPV genotypes (group 1) in women with No-IL were HPV-16 and, HPV-58, accounting for 3.37% (95% CI 2.40–4.58, n = 39), and .78% (95% CI 0.36–1.37, n = 9), respectively (Table 2 and Figure 5A). In patients with LSIL, the two most prevalent HR-HPV genotypes (group 1) were HPV-16, and HPV-31, accounting for 7.87% (95% CI 6.82–9.01, n = 190), and 3.81% (95% CI 3.08–4.65, n = 92), of the infections, respectively (Table 2 and Figure 5B). Among the women with HSIL, the most prevalent HR-HPV genotypes (group 1) were HPV-16, and HPV-31, which were detected in 11.59% (95% CI 6.77–18.14, n = 16), and 8.70% (95% CI 4.57–14.70, n = 12), of the patients, respectively (Table 2 and Figure 5C). In women with CC, the most prevalent HR-HPV genotypes (group 1) were HPV-16, and HPV-33/HPV-45/HPV-52, which were detected in 9.20% (95% CI 5.92–13.48, n = 23), and 2.40% (95% CI 0.89–5.15, n = 6) of the patients, respectively (Table 2 and Figure 5D). Prevalence of HPVs among patients with different cervical lesion grades and with multiple infections. A) Prevalence of HPV genotypes in women No-IL. B) Prevalence of HPV genotypes in patients with LSIL. C) Prevalence of HPV genotypes in patients with HSIL. D) Prevalence of HPV genotypes in patients with CC. Human papillomavirus (HPV) types according to International Agency for Research on Cancer (IARC), Muñoz et al and Burk et al. High-risk HPVs (Group 1: HPV-16, 18, 31, 33, 35, 39, 45, 51, 52, 56, 58 and, 59); Probable high-risk HPVs (Group 2: HPV-26, 34, 53, 66, 67, 68, 69, 70, 73, and 82); Low-risk HPVs (Group 3: HPV-6, 11, 13, 40, 42, 43, 44, 54, 61, 72, 81, and 89; Undefined-risk HPVs (HPV-7, 32, 55, 57, 62, 71, 74, 83, 84, 86, 87, 90, and 102). Abbreviations: HPV: Human papillomavirus; HR-HPV: high-risk HPV; pHR-HPV: probable high-risk HPV; LR-HPV: low-risk HPV; UR-HPV: Undefined-risk HPV; M-HPV: multiple HPV types; No-IL: non intraepithelial lesion; LSIL: Low-grade squamous intraepithelial lesion; HSIL: High-grade squamous intraepithelial lesion; CC: Cervical cancer.
The prevalence of HPV-16 in multiple infection by age group shows an increased prevalence for young women with No-IL (.86%), LSIL (1.28%) and HSIL (2.90%) in the population aged 25–34 years. While the prevalence of HPV-16 (1.20%) was higher in the age group of 45 to 54 years in CC patients (Table S6 and Figure 6). Prevalence of HPV grouped by age in patients with multiple infections. A) Prevalence of HR-HPVs infection in No-IL women in each age group. B) prevalence of HR-HPVs infection in LSIL patients in each age group. C) prevalence of HR-HPVs infection in HSIL patients in each age group. D) Prevalence of HR-HPVs infection in CC patients in each age group. Abbreviations: HPV: Human papillomavirus; HR-HPV: high-risk HPV; pHR-HPV: probable high-risk HPV; LR-HPV: low-risk HPV; UR-HPV: Undefined-risk HPV; M-HPV: multiple HPV types; No-IL: non intraepithelial lesion; LSIL: Low-grade squamous intraepithelial lesion; HSIL: High-grade squamous intraepithelial lesion; CC: Cervical cancer.
Risk of Lesions Associated With Single HPV Infection
Association of HPV groups with LSIL, HSIL, and CC.

Data are expressed as n (%) unless indicated otherwise; Odds ratio (OR); 95% confidence interval (CI).
aRisk to developing LSIL (values are calculated taken Negative group as reference).
bRisk to developing HSIL (values are calculated taken Negative group as reference).
cRisk to developing CC (values are calculated taken Negative group as reference).
Significant P<.05. * Odd ratio non-adjusted. ** OR adjusted by age (14–24, 25–34, 35–44, 45–54, >55), smoking (No, and Yes), and giving birth (0, 1 to 2, >3). Human papillomavirus (HPV) types according to International Agency for Research on Cancer (IARC), Muñoz et al and Burk et al. High-risk HPVs (Group 1); Probable high-risk HPVs (Group 2); Low-risk HPVs (Group 3); Undefined-risk HPVs. Abbreviations: HPV: Human papillomavirus; HR-HPV: high-risk HPV; pHR-HPV: probable high-risk HPV; LR-HPV: low-risk HPV; UR-HPV: Undefined-risk HPV; M-HPV: multiple HPV types; No-IL: non intraepithelial lesion; LSIL: Low-grade squamous intraepithelial lesion; HSIL: High-grade squamous intraepithelial lesion; CC: Cervical cancer.
Association of HPV genotypes with LSIL, HSIL, and CC in single infections.
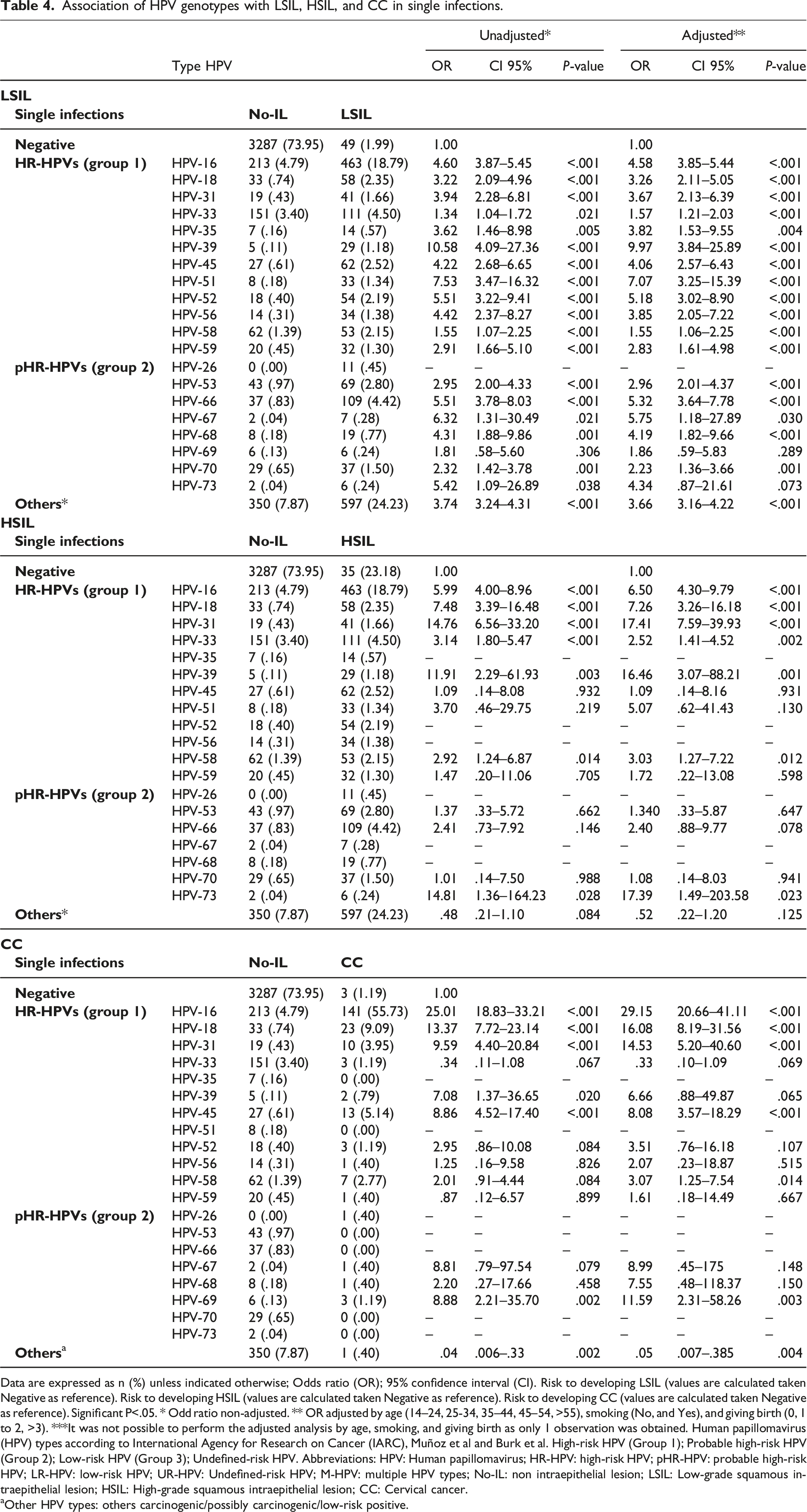
Data are expressed as n (%) unless indicated otherwise; Odds ratio (OR); 95% confidence interval (CI). Risk to developing LSIL (values are calculated taken Negative as reference). Risk to developing HSIL (values are calculated taken Negative as reference). Risk to developing CC (values are calculated taken Negative as reference). Significant P<.05. * Odd ratio non-adjusted. ** OR adjusted by age (14–24, 25-34, 35–44, 45–54, >55), smoking (No, and Yes), and giving birth (0, 1 to 2, >3). ***It was not possible to perform the adjusted analysis by age, smoking, and giving birth as only 1 observation was obtained. Human papillomavirus (HPV) types according to International Agency for Research on Cancer (IARC), Muñoz et al and Burk et al. High-risk HPV (Group 1); Probable high-risk HPV (Group 2); Low-risk HPV (Group 3); Undefined-risk HPV. Abbreviations: HPV: Human papillomavirus; HR-HPV: high-risk HPV; pHR-HPV: probable high-risk HPV; LR-HPV: low-risk HPV; UR-HPV: Undefined-risk HPV; M-HPV: multiple HPV types; No-IL: non intraepithelial lesion; LSIL: Low-grade squamous intraepithelial lesion; HSIL: High-grade squamous intraepithelial lesion; CC: Cervical cancer.
aOther HPV types: others carcinogenic/possibly carcinogenic/low-risk positive.
Risk of Lesions Associated With Multiple HPV Infections
Association of HPV genotypes with LSIL, HSIL, and CC in multiple infections.

Data are expressed as n (%) unless indicated otherwise; Odds ratio (OR); 95% confidence interval (CI). Risk to developing LSIL (values are calculated taken Negative as reference). Risk to developing HSIL (values are calculated taken Negative as reference). Risk to developing CC (values are calculated taken Negative as reference). Significant P<.05. * Odd ratio non-adjusted. ** OR adjusted by age (14-24, 25-34, 35-44, 45-54, >55), smoking (No, and Yes), and giving birth (0, 1 to 2, >3). ***It was not possible to perform the adjusted analysis by age, smoking, and giving birth as only 1 observation was obtained. Human papillomavirus (HPV) types according to International Agency for Research on Cancer (IARC), Muñoz et al and Burk et al. High-risk HPV (Group 1); Probable high-risk HPV (Group 2); Low-risk HPV (Group 3); Undefined-risk HPV. Abbreviations: HPV: Human papillomavirus; HR-HPV: high-risk HPV; pHR-HPV: probable high-risk HPV; LR-HPV: low-risk HPV; UR-HPV: Undefined-risk HPV; M-HPV: multiple HPV types; No-IL: non intraepithelial lesion; LSIL: Low-grade squamous intraepithelial lesion; HSIL: High-grade squamous intraepithelial lesion; CC: Cervical cancer.
aIncludes positive women to 2 or more types (HPV-16, HPV-18, HPV-31, HPV-33; HPV-39, HPV-45, HPV-52, HPV-56, HPV-58, HPV-68, HPV-53, HPV-66 and hpv-69) and other HPV (HR-HPV/pHR-HPV/LR-HPV).
bOther HPV types: Others HR-HPV/pHR-HPV/LR-HPV-positive.
Association of multiple HPVs infections with LSIL, HSIL, and CC.

Data are expressed as n (%) unless indicated otherwise; Odds ratio (OR); 95% confidence interval (CI).
aRisk to developing LSIL, HSIL and CC (values are calculated taken patients with hpv-16 or hpv-18 single infections).
Significant P < .05. * Odd ratio non-adjusted. ** OR adjusted by age (14–24, 25–34, 35–44, 45–54, >55), smoking (No, and Yes), and giving birth (0, 1 to 2, >3). Abbreviations: HPV: Human papillomavirus; M-HPV: multiple HPV types; No-IL: non intraepithelial lesion; LSIL: Low-grade squamous intraepithelial lesion; HSIL: High-grade squamous intraepithelial lesion; CC: Cervical cancer.
Discussion
HPV infection is one of the most common sexually transmitted infections and is related to various clinical conditions, ranging from asymptomatic infection to malignant cervical disease. 28 Our study is one of the largest studies performed in southern Mexico to detect HPV, to determine the distribution genotypes and to investigate the relation of infection with specific genotypes to the development of precancerous lesions and CC.
The HPV prevalence among women with No-IL worldwide is 11.7%. Sub-Saharan Africa (24.0%), Latin America and the Caribbean (16.1%), Eastern Europe (14.2%), and Southeast Asia (14.0%) had the highest prevalence. 29 These data differ from what was found in this study, where we observed a high positivity for HPV infection in No-IL patients (26.05%), one of the highest rates reported in the world.
Several investigations have studied the relationship between changes in the distribution of HPVs with age where some investigations report that with advanced age there is a decrease in the distribution of HR-HPV.30,31 While others report an increased frequency of HR-HPV in women older than 45 years of age.32–34 In this study, we observed that the frequency of hpv-16 increases with age, reaching its maximum peak in women >45 years of age. One of the reasons that could influence this increase is the woman’s immunological status, 35 which could affect the acquisition and reactivation of HPV infections.
The distribution of HR-HPV genotypes (group 1) differs across regions. It has also been reported that the most common genotypes worldwide are HPV-16, HPV-18, and HPV-45. 13 Our data showed that the three most prevalent HR-HPV genotypes (group 1) in CC patients were HPV-16, HPV-18, and HPV-45. Our results show agreement with the observation that HPV-16 and HPV-18 were the two most frequent genotypes in other regions of Mexico. 36 Additionally, it has been reported in populations in Africa, Asia, North America, and Oceania that HPV-16, HPV-18, and HPV-45 are the three predominant genotypes in CC patients.13,36–39 However, these results differ from those reported by Li et al, who found that the 3 most prevalent HR-HPV genotypes (group 1) in the Chinese population were HPV-16, HPV-52, and HPV-58. 40 One of the explanations for this discrepancy in the prevalence of HPV-16 in women with No-IL may be attributed to variables related to the acquisition of the virus, such as age, age at the initiation of sexual activity, number of lifetime sexual partners, socioeconomic status, level of education, number of deliveries, use of oral contraceptives, smoking, and ethnicity, which have been shown to influence the prevalence of HPV.7,16,17,19,28,41
In this study, the risk of lesions associated with single infections with HR-HPV genotypes (group 1) in women in southern Mexico was investigated. We observed substantial variation in the risk of LSIL, HSIL and CC associated with different HR-HPV genotypes. These findings were similar to the results reported in other populations, in which researchers observed that HPV-16, HPV-18, and HPV-39 were associated with a high risk of developing CC.19,38,42
Multiple HPV infections have been reported to be very common.16,43 An interesting observation in this study was the presence of a low proportion of women with multiple HPV infections: the prevalence ranged between 2.34 and 31.13%. Similar observations were reported in studies from Cozumel, México (No-ILs: 2.19%) 44 ; San Luis Potosí/Guanajuato, Mexico (LSIL: 21.10%; HSIL: 5.00%; CC: 3.40%) 45 ; Latin America and the Caribbean (HSIL: 16.80% and CC: 12.60%) 46 ; Brazil (CC: 16.43%) 47 ; Costa Rica (18.22%) 48 ; and Ecuador (CIN1: 7.01%; CIN2/3: 5.26% and CC: 14.28%), 49 but differs from that reported in other studies that have reported relatively higher proportions of participants with multiple HPV infections: China (CSCC: 72.40%), 40 Romania (HSIL: 68.20%), 50 Brazil (LSIL: 58.00%; HSIL: 55.00%), 51 and Nigeria (CC: 61.36%). 52 The prevalence of HPV coinfections has been attributed to having multiple sexual partners.48,52,53 In countries such as Nigeria, a high rate of polygamous marriages is reported, and a man can marry up to four wives. 52 In the state of Guerrero, México, polygamy is not common. 19 It has also been observed that women with multiple lifetime sexual partners may be exposed to different HPV genotypes. 54 Therefore, the low prevalence of multiple HPV infections in our study may be attributed to the notion that most marriages in this region are monogamous, and widows usually do not remarry; the cultural practices may differ in countries in which a high percentage of women have multiple HPV infections.
Few studies have described the association of HPV coinfections and the risk of LSIL, HSIL, and CC. In this study, we determined that patients infected with more than 2 HR-HPV genotypes, had significantly increased risks of LSIL, HSIL, and CC. Our result agrees with the finding in another study that showed that women with multiple HPV infections had a higher risk of developing CIN2.
55
Likewise, Lee
Our study has important limitations to consider. First, it is well known that the primers used from 1997 to 2010 do not amplify all HPV genotypes with the same sensitivity, and therefore such differences are observed in the frequency of multiple infections. However, the INNO-LiPA assay has been used extensively and allows easy detection of multiple infections due to its high sensitivity. Second, as it is a retrospective study, it cannot be verified whether there is a low proportion of multiple infections in the 1997–2010 samples. Third, we cannot assess the influence of the HPV vaccine. The HPV vaccine was introduced in Mexico in 2007 and the target population were women under 15 years of age, this means that the frequency and distribution of HPVs is not yet affected by vaccination. Moreover, the impact of multiple infections on the molecular mechanisms and epidemiology of cervical cancer needs to be fully elucidated, and additional studies should be performed.
In conclusion, this study focused on the epidemiology of HPV infections in women in southern Mexico. The most prevalent HR-HPV genotype (group 1) was HPV-16, in women with No-IL, HSIL, and CC. Patients with HPV-39 single infections had a higher risk of developing LSIL; those with HPV-31 single infections had a higher risk of developing HSILs; and those with HPV-16 single infections had a higher risk of developing CC. Patients with multiple infections with HR-HPV genotypes (group 1: HPV-16, HPV-18, HPV-39, HPV-52, HPV-53, HPV-66, and HPV-69) and other HPV genotypes had significantly higher risks of developing LSIL, HSIL and CC. These findings reaffirm that HPV infection is an independent factor for the development of LSIL, HSIL, and CC. The results of this study confirm that high-risk HPV infections are valuable markers to identify patients at high risk of developing precancerous lesions and CC, as well as useful evidence that can be used for future HPV and cancer screening programs in Mexico. In addition, that with these results they could be used for the development of vaccines against HPV more prevalent in the female population.
Supplemental Material
sj-pdf-1-ccx-10.1177_10732748221103331 – Supplemental Material for Prevalence and Distribution of Human Papillomavirus Genotypes (1997–2019) and Their Association With Cervical Cancer and Precursor Lesions in Women From Southern Mexico
Supplemental Material, sj-pdf-1-ccx-10.1177_10732748221103331 for Prevalence and Distribution of Human Papillomavirus Genotypes (1997–2019) and Their Association With Cervical Cancer and Precursor Lesions in Women From Southern Mexico by Luz del Carmen Alarcón-Romero, Jorge Organista-Nava, Yazmín Gómez-Gómez, Julio Ortiz-Ortiz, Daniel Hernández-Sotelo, Oscar del Moral-Hernández, Miguel Angel Mendoza-Catalán, Ramón Antaño-Arias, Marco Antonio Leyva-Vázquez, Natividad Sales-Linares, Verónica Antonio-Véjar and Berenice Illades-Aguiar in Cancer Control
Footnotes
Acknowledgments
We thank all of the Instituto Estatal de Cancerología ‘‘Dr Arturo Beltrán Ortega’’ and Secretaría de Salud personnel who helped with this study at the clinic sites. We also thank technicians of Laboratorio de Biomedicina Molecular for their excellent laboratory assistance.
Authors’ Contributions
LCAR: Investigation, Methodology, histological evaluation of all biopsy material. JON and YGG: Conceptualization, Data curation, Formal analysis, Investigation, Writing - original draft, Writing - review & editing. JOO, DHS, OMH, MAMC, RAA, NSL and VAV: Investigation and Methodology. MALV and BIA: Conceptualization, Investigation, Methodology, Funding acquisition, Project administration, Review and approved the final version of the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The present study was supported by grant from CONACYT, México (Investigación Científica Básica 2016; grant no. 288612).
Ethical Approval and Consent to Participate
This study was reviewed and approved by the Institutional Review Board “Comité de Bioética de la Dirección de Investigación de la Universidad Autónoma de Guerrero” (approval no. CB-005/2016). Informed consent was obtained from all participants. Data confidentiality was maintained throughout the study. All procedures were in accordance with the Helsinki Declaration.
ORCID iDs
Supplemental Material
Supplemental material for this article is available online.
Appendix
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
