Abstract
Gliomas are the most prevalent brain tumors among children and adolescents. The occurrence and development of various malignant tumors is closely related with LIN28A gene, but its relationship with glioma susceptibility has not been widely discovered. In this case-control study, we conducted four single nucleotide polymorphisms (SNPs) (rs3811464 G>A, rs3811463 T>C, rs34787247 G>A, and rs11247957 G>A) of LIN28A gene to investigate whether they increase the risk of glioma. Odds ratios (ORs) and 95% confidence intervals (CIs) were used to evaluate their relationship. There was no significant correlation between four SNPs and glioma risk in single polymorphism and conjoint analysis. However, in stratification analysis, we found that rs3811463 TC/CC may add to the risk of glioma with clinical stage III (adjusted OR = 3.16, 95% CI = 1.15-8.70, P = .026) or stage III+IV patients (adjusted OR = 2.05, 95% CI = 1.02-4.13, P = .044). Our research suggested that four SNPs of LIN28A gene have a weak relationship with the risk of glioma in Chinese children. LIN28A rs3811463 TC/CC may increase the possibility of glioma in clinical stage III or stage III+IV patients which need larger samples and further confirmation.
Introduction
Glioma, one of the nervous system tumors, is the most prevalent and occurs in primary intracranial neoplasm among children. 1 Besides, the most common secondary primary neoplasms of childhood cancer survivors were central nervous system tumors of which 30% were glioma approximately. 2 The stages of glioma were classified by WHO as I to IV according to the degree of malignancy. With the exception of stage I or II (low-grade gliomas, LGGs), the stage III or IV (high-grade gliomas, HGGs) shows significant aggressiveness. Outcomes of LGGs are usually good with 5-year overall survival up to 95%. 3 In contrast, owing to aggressive characteristic and limited therapeutic efforts, HGG patients still maintain poor prognosis. 4
Many factors that contribute to the occurrence of glioma have been explored.5-10 Radiation is the only verified risk factor for glioma, especially in childhood. 11 However, several genetic disorders have been linked to glioma in children including neurofibromatosis type 1 (NF1), Turcot syndrome, and Li-Fraumeni syndrome. 12 This indicates that genetic factors are particularly important for the pathogenesis of glioma and it is necessary to clarify the pathogenesis for treatment and observation of prognosis. Moreover, evidence shows that P53, BRAF V600E, and H3-K27M mutations are associated with malignancy of pediatric glioma.13-16 It is worth mentioning that genetic diversity and disease susceptibility show up to be connected with single nucleotide polymorphisms (SNPs). Margaret Wrensch et al. 17 confirmed that rs1412829, which is near CDKN2B in chromosome 9p21, may be a promoting element of glioma susceptibility. Marc Sanson et al. 18 exhibited that EGFR rs11979158 and rs2252586 at 7p11.2 facilitated the transformation from benign glioma to malignant glioma. However, the discovery of these risks is only a small part of the etiological mechanism; further exploration of polymorphisms is needed.
LIN28 is a highly conserved RNA-binding protein in structure which was first found to be a timing controlling gene. 19 LIN28 can inhibit the synthesis of let-7, a tumor suppressor gene, to promote the occurrence and development of tumors.20,21 In mammals, there are two LIN28 homologous, LIN28A, and LIN28B, respectively. LIN28A blocks let-7 progressing using Zcchc11 mechanism to promote tumor growth. 22 A vast number of LIN28A-related cancers have been identified in recent years including breast cancer, 23 ovarian cancer, 24 colon cancer, 25 osteocarcinoma, 26 and pancreatic cancer. 27 In addition, a result of a multicenter study demonstrated that LIN28A polymorphisms, especially rs34787247 G>A, may add the risk of neuroblastoma. 28 Jinjing Lu et al. 29 verified that LIN28A impeded tumor suppressor gene IRF6 to promote glycolysis and proliferation of glioma cells by stabilizing SNHG14.
Although abundant studies have suggested the significance of gene polymorphism in glioma, the effect of the LIN28A polymorphisms on pediatric glioma has not been discovered. In the present study, we investigated whether LIN28A gene polymorphisms are associated with the risk of glioma in Chinese children.
Material and Methods
Patients and Controls
In this study, totally 191 cases with newly diagnosed glioma and 248 controls were enrolled belonging to the Chinese population. The included 191 children were examined at the medical center and pathologically typed and staged as WHO classification. All cases with other cancers or exposure to chemotherapy in the last 6 months were excluded. The non-cancer controls only were examined and matched on sex and age from the same residing areas as patients. The controls were confirmed without nervous system diseases, congenital genetic diseases, and infectious diseases. None of the subjects had blood relationship. Approval for this study was obtained from the institutional review board of Guangzhou Women and Children’s Medical Center (2016021650). The patients provided their necessary written informed consent for the research use of their blood sample according to relevant laws and regulations. The protocol of this study was complied with ethical guidelines.
SNP Selection and Genotyping
Four LIN28A polymorphisms (rs3811464 G>A, rs3811463 T>C, rs34787247 G>A, and rs11247957 G>A) were selected through the dbSNP database (http://www.ncbi.nlm.nih.gov/SNP) and SNPinfo software (http://snpinfo.niehs.nih.gov/snpfunc.htm). Selection criteria are briefly described below: (1) the minor allele frequency (MAF) reported in HapMap was > 5% for Chinese Han subjects; (2) putative functional potential SNPs located in the 5′- flanking region, exon, 5′- untranslated region (5′ UTR), and 3′ UTR, which might affect transcription activity or binding capacity of the microRNA binding site; (3) SNPs in low linkage disequilibrium with each other (R2 < .8). rs3811464 G>A is located in transcription factor binding sites (TFBS); rs3811463 T>C and rs34787247 G>A are located in transcription factor binding sites (TFBS) and also affect binding capacity of the microRNA binding site; rs11247957 G>A is located in TFBS, affects binding capacity of the miRNA binding site, and affects splicing regulation activity. There was no significant LD (R 2 < .8) among these 4 SNPs of LIN28A (R2 = .183 between rs3811464 and rs3811463, R2 = .009 between rs3811464 and rs34787247, R2 = .054 between rs3811464 and rs11247957, R2 = .03 between rs3811463 and rs34787247, R2 = .052 between rs3811463 and rs11247957, R2 = .002 between rs34787247 and rs11247957). We used a standard commercial TaqMan real-time PCR kit to genotype the DNA samples.30,31 About 10% samples were randomly chosen and re-genotyped for accuracy of genotyping results and quality control. We acquired 100% concurrence for the quality control samples.
Statistical Analysis
We utilized χ2 test to estimate the dissimilarity of gender, subtypes, and clinical stages between cases and cancer-free controls. The goodness-of-fit test was applied to check Hardy–Weinberg equilibrium (HWE) in controls. The strength between the selected polymorphisms and risk of glioma was counted by odds ratio (ORs) and 95% confidence intervals (CIs). We adjusted age and gender as adjusted ORs, neglecting corresponding stratify factor. Besides, stratification analysis was accomplished to validate the connection of age, gender, clinical stages, and subtypes. P-value <.05 was considered as statistically significant.
Results
Association Between LIN28A Polymorphisms and Glioma Susceptibility
Association Between LIN28A Gene Polymorphisms and Glioma Susceptibility in Chinese Children.

Abbreviations: OR, odds ratio; CI, confidence interval; HWE, Hardy–Weinberg equilibrium.
aχ2 test for genotype distributions between glioma patients and cancer-free controls.
bAdjusted for age and gender.
cRisk genotypes were carriers with rs3811464 GA/AA, rs3811463 TC/CC, rs34787247 GA/GG, and rs11247957 GA genotypes.
Stratification Analysis
Stratification Analysis of Risk Genotypes with Glioma Susceptibility.
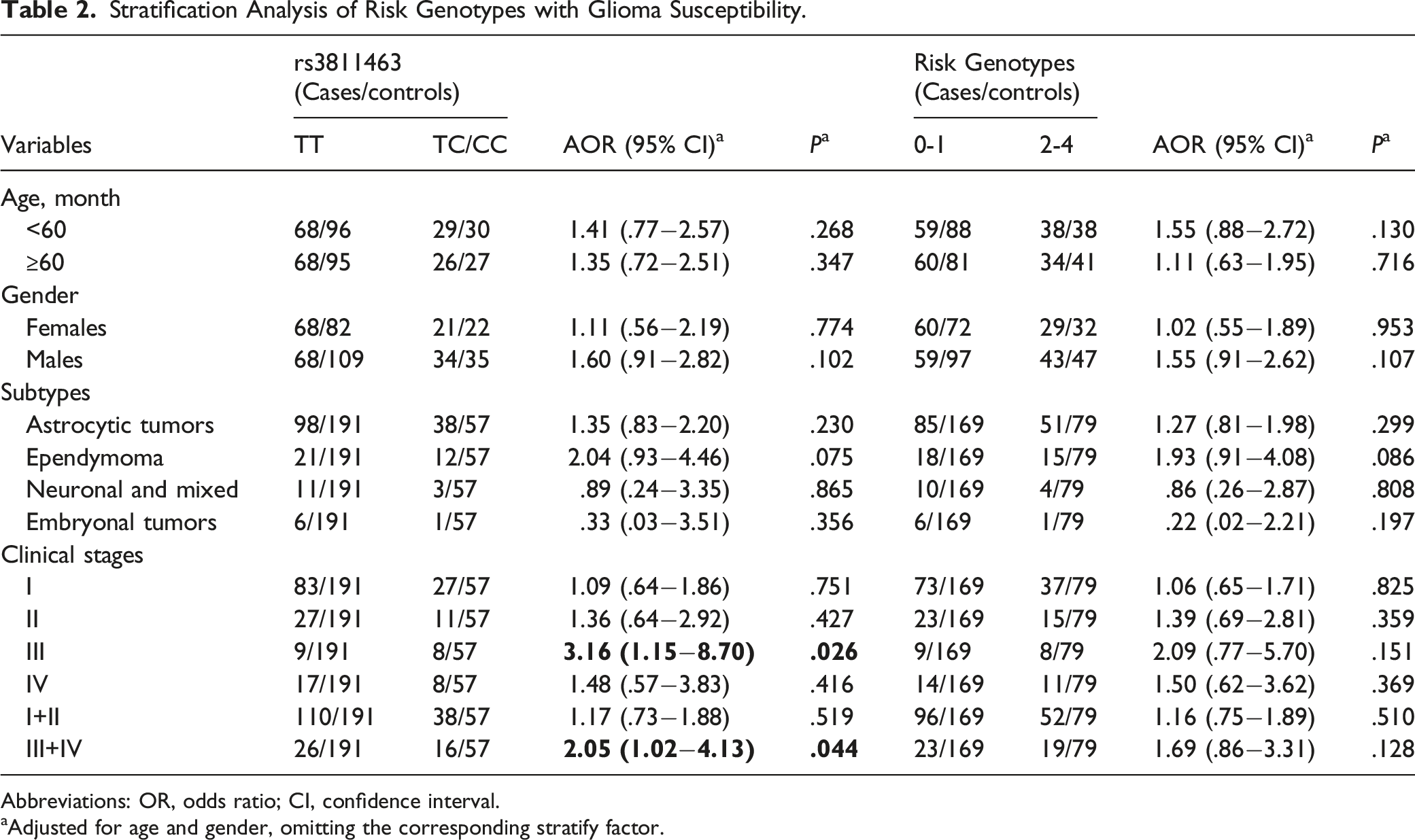
Abbreviations: OR, odds ratio; CI, confidence interval.
aAdjusted for age and gender, omitting the corresponding stratify factor.
Discussion
Gliomas are thought to originate from neuroglial stem or oligodendroglial progenitor cells; despite various therapies, the median survival of malignant glioma is still less than 15 months.32,33 Although the rate of progression from LGGs to HGGs is much lower among children than adults, HGGs can be fatal in childhood. Unlike adults, there are relatively few high-grade gliomas in children and with differences in molecular genetics. 12
Abundant explorations have been found that show variety of gene mutations could lead to glioma, and further study on the susceptibility of glioma is continuing. The most common somatic mutation in pediatric LGGs is BRAF V600E and it is often accompanied with deletion of CDKN2A/B in pleomorphic xanthoastrocytomas which shows a tendency of transformation.33,34 Carl Wibom et al. 35 confirmed that EGFR gene variations (rs17172430 and rs11979158) would lead to the homozygous loss of CDKN2A/B. Isocitrate dehydrogenase (IDH)1 mutation was generally considered to occur in children over 14 years old with HGGs, 36 yet there have been cases of mutation detected under the age of ten. 37 Interestingly, the prognosis of IDH1 mutation is better than that of the wild type. After SNP detection, Robert B. Jenkins et al. 38 found that the variant rs55705857 located on 8q24.21 was strongly correlated with IDH1 mutation glioma. In addition, gliomas with H3-K27M mutation are mostly classified as WHO grade IV, and usually occur together with p53 gene. 39 p53 gene is a suppresser gene and rs78378222 of p53 gene susceptibility was detected to be connected with glioma (OR = 2.35) in a genome-wide association study. 40 H3-K27M mutation gliomas are deadly; more related mechanisms need to be discovered to treat patients.
Currently, the main view is that LIN28A regulates the occurrence and development of tumors by controlling let-7 molecules. Let-7, a series of highly conserved miRNA sequence, is a tumor suppressor gene that acts by inhibiting proto-oncogenes such as RAS and HMGA2.41,42 LIN28A plays an inhibitory role to let-7 by blocking the transformation of pre-let-7 to let-7. 43 Meanwhile, the proliferation of tumor cells depends on glycolysis, a study demonstrated that LIN28A facilitated glycolysis in glioma cells by downregulating the suppressor gene IRF6. 29 Therefore, as a tumor-promoting factor, LIN28A abnormally expressed in numerous tumors and represents a poor prognosis. In a clinical trial, Ning Li et al. 44 found that cyclins such as CDK2 and CDKN1 could be controlled by LIN28A in tumor cells to support cell cycle progression. It has been reported that LIN28A can interact with coiled-coil containing protein kinase2 to accelerate the malignant transformation of ovarian cancer. 45 Two case-control studies demonstrated that LIN28A polymorphisms changed susceptibilities of neuroblastoma and Wilms tumor.28,46 Despite evidence has shown that LIN28A increases the incidence of numerous tumors, there has been no previous study about the relationship between LIN28A polymorphisms and glioma susceptibility.
In this study, we estimated 191 pediatric glioma patients and 248 cancer-free controls to work out whether LIN28A gene polymorphisms are associated with the risk of glioma. Although the result implied that none of these picked polymorphisms were related to glioma susceptibility, the stratification analysis suggested that rs3811463 TC/CC may increase the risk of clinical stage III or III+IV patients with glioma. From what we know, this is the earliest study of assessing the association between LIN28A polymorphisms and glioma susceptibility. However, several limitations of our study should be noticed. First of all, due to insufficient sample size, there may exist some deviations between our study and real situation. More samples needed to be included in the future to obtain more accurate conclusions. Second, because specimens from other ethnic groups were not available, our findings do not apply to ethnic groups other than the Han. Third, the large differences exist in genetic variation between children and adults with glioma, thus our conclusions may not be applicable to the entire population. Moreover, our patients were recruited from hospital; many factors including environmental influence, diet background, and personality preference were not taken into account which may be led to bias eventually.
In conclusion, this study suggested that LIN28A polymorphisms have a weak relationship with glioma susceptibility and further investigation with lager samples is needed to proceed.
Supplemental Material
Supplemental Material - Association Between LIN28A Gene Polymorphisms and Glioma Susceptibility in Chinese Children
Supplemental Material for Association Between LIN28A Gene Polymorphisms and Glioma Susceptibility in Chinese Children by Huiqin Guo, BA, Yuxiang Liao,MD, Ao Lin, MS, Huiran Lin, MS, Xiaokai Huang, MS, Jichen Ruan, MD, Li Yuan, PhD, and Zhenjian Zhuo, PhD in Cancer Control
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by grants from the National Natural Science Foundation of China (No. 81802346, 82002636) and China Postdoctoral Science Foundation (No. 2020T130132).
Ethics Approval
Approval for this study was obtained from the institutional review board of Guangzhou Women and Children’s Medical Center (2016021650).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
