Abstract
We tested whether a low-literacy-friendly, multimedia information and assessment system used in daily clinical practice enhanced patient-centered care and improved patient outcomes. This was a prospective, parallel-group, randomized controlled trial with 2 arms, CancerHelp-Talking Touchscreen (CancerHelp-TT) versus control, among adults with Stage I–III breast or colorectal cancer receiving chemotherapy and/or radiation therapy in safety net settings. Each patient was assessed for outcomes at 4 timepoints: after starting treatment (baseline), during treatment, immediately after treatment, and at follow-up assessment. The primary outcomes were health beliefs, cancer knowledge, self-efficacy, and satisfaction with communication about cancer and its treatments. Health-related quality of life (HRQOL) was a secondary outcome. A total of 129 patients participated in the study (65 intervention and 64 control), and approximately 50% of these completed the study. Patients randomized to receive the CancerHelp-TT program had a significantly larger increase in their cancer knowledge in comparison to those randomized to the control arm (effect size = .48, P = .05). While effect sizes for differences between randomized groups in self-efficacy, health beliefs, HRQOL, and satisfaction with communication were small (.10–.48), there was a consistent trend that participants in the intervention group showed larger increases over time in all outcomes compared to the control group. The CancerHelp-TT software was favorably rated by intervention participants. The CancerHelp-TT program showed promise to increase vulnerable cancer patients’ cancer knowledge and adaptive health beliefs and attitudes. However, vulnerable patients may need additional interventional support in settings outside cancer clinics.
Introduction
Improving the delivery of patient-centered health information is especially important for vulnerable patient populations.1–3 Patients with limited economic resources, low literacy skills, or racial/ethnic minority status are particularly vulnerable to receiving suboptimal care over time. These patients experience a greater burden of disease, are less informed about diagnosis and treatment, and are less likely to be satisfied with communication with their health-care providers.4,5 Improving communication in health-care settings may lead to higher quality of care, higher satisfaction with care, and better health outcomes.6–8 To facilitate these efforts, appropriate communication tools are needed.
Cancer patients are an appropriate population in which to test health education interventions because of cancer’s multifactorial symptomatology and treatment, and its large prevalence, with 15.5 million Americans with a history of cancer currently and 1.5 million new cancer cases diagnosed annually. 9 In addition, there are significant, persistent racial, and socio-economic disparities in likelihood of developing cancers, access to cancer care, and cancer outcomes.10,11 These disparities are not uniform across all cancer sites or metrics. For example, in comparison to non-Hispanic whites, black women have a lower lifetime probability of developing breast cancer (11.1% vs. 13.1%), but they have a higher likelihood of dying from breast cancer (3.3% vs. 2.7%). 12 For colon and rectal cancer, the racial disparities are worse 12 and are intertwined with socioeconomic characteristics. 13 For instance, a recent study found that whites had the highest rate of colorectal cancer screening (62%), in comparison to 59% among blacks and 31% among Spanish-speaking Hispanics; however, when controlling for socioeconomic characteristics, these differences were diminished for black but not Spanish-speaking Hispanics. 13 A systematic review of health literacy studies in adult cancer patients also found that non-white patients tended to have lower health literacy than white patients. 14 Health literacy also represents an important barrier to cancer care access, and is associated with timing of key events like cancer diagnosis and screening, and ultimately less coordinated care.14,15 Therefore, health literacy and related constructs (e.g., cancer knowledge, self-efficacy) are promising targets for intervention.
Tailored interactive multimedia programs usable among patients with low health literacy have the potential to optimize patient health behaviors and reduce health disparities, 16 but, with a couple exceptions focused mostly on increasing cancer screening,17-19 few interventional studies to date have been conducted. 20 Here, we describe the results of a randomized, controlled trial testing whether a low literacy, multimedia information and assessment system used in daily clinical practice—the CancerHelp-Talking Touchscreen (TT)—enhances patient-centered care and improves patient outcomes for underserved cancer patients. The theoretical framework guiding this study was the Behavioral Model for Vulnerable Populations.21-23 This model was designed to include domains that are especially relevant to understanding the health and health-seeking behavior of vulnerable populations. Specifically, in comparison to National Cancer Institute (NCI)-created educational paper brochures, it was hypothesized that the CancerHelp-TT would increase cancer patients’ health beliefs, knowledge of cancer, and treatment, self-efficacy, HRQOL, and satisfaction with health-care communication.
Materials and Methods
Study Design
This was a 2-arm, prospective, parallel group randomized controlled trial. Each participant was randomized with equal allocation (1:1) to receive the CancerHelp-TT intervention or control cancer education (standard verbal and written patient education). A block-permuted randomization sequence, stratified by clinic and diagnosis, was generated by the study statistician. Patients’ random assignment was revealed at the time of enrollment by the study coordinator. Blinding was not possible, but assignment was concealed in a computer program until requested by the coordinator. Each patient was followed through the end of treatment, and for the first follow-up visit. Assessments were made after starting treatment (baseline), during treatment (on average, 40 days post-baseline), immediately after treatment (on average, 8 months post-baseline), and a follow-up assessment (on average, 1 year post-baseline).
Setting and Sample
Ambulatory cancer patients were enrolled at the clinic where they were receiving their primary treatment course (chemotherapy and/or radiation therapy). Eligibility criteria included: 18 years or older, English-speaking, recent diagnosis (within 6 months) of Stage I-III breast or colorectal cancer, starting adjuvant or neo-adjuvant therapy (within 1 month of starting infusion chemotherapy or within 2 weeks of starting radiation therapy), and having sufficient sensory and cognitive ability to interact with a multimedia computer touchscreen. Prior computer experience was not required. The only exclusion criterion was that patients could not have been concurrently involved in any disease management, performance improvement or psychosocial intervention initiative or study. Patients were recruited from 3 medical centers in Chicago, IL: the John H. Stroger Hospital of Cook County, the Advocate Illinois Masonic Medical Center, and Mt. Sinai Hospital. These centers were chosen because they serve significant proportions of racial/ethnic minorities, and socioeconomically disadvantaged and low literacy populations, and they provide safety net services.
Control Arm
Patients randomized to the control arm received NCI diagnosis- and treatment-specific paper brochures: What You Need to Know Series/Patient Summary and Chemotherapy and You and/or Radiation Therapy and You. 24 They used the multimedia (text plus audio) Talking Touchscreen25,26 to complete assessments for each of the study endpoints, but did not have access to the CancerHelp-TT educational program. All aspects of care were driven by usual clinical practice. Each of the study sites provided reasonably standard patient education to all patients, using combined verbal and written materials.
Intervention Arm
Patients randomized to the intervention arm received the same NCI diagnosis- and treatment-specific paper brochures and used the multimedia Talking Touchscreen to complete assessments for each of the study endpoints. In addition, they were given access to the CancerHelp-TT patient education software which adapted the NCI brochures for multimedia access (see screen images in Figure 1). They were shown how to log in on their own so that they could access the software at any time and as often as they wished, and they were also given a CD-ROM to take home. Patients were able to personalize the intervention by selecting modules that were most relevant/important to them at each session. The program included the option to generate and print tailored, personally relevant checklists of issues they may wish to discuss during medical visits (“Topics for Today”). The software also included a short video introduction by a clinician from each site, and a video module based on the NCI Facing Forward booklet (https://pubs.cancer.gov/ncipl/detail.aspx?prodid=P119). The CancerHelp-TT software and the data collection platform both employ an interactive design with audio and visual tools and user-friendly features to enhance accessibility for patients with low literacy (see screen images in Figure 1). CancerHelp-TT was available on a kiosk in a private room at any time during clinic hours. CancerHelp-Talking Touchscreen (CancerHelp-TT) screen images.
Measurements
Outcomes
Participants in both arms used the multimedia TT data capture system25,26 to complete knowledge, satisfaction, HRQOL and other study measures up to 4 times, including baseline, during treatment, end-of-treatment treatment, and follow-up. A research assistant introduced the computer equipment and sat with the participant during a brief practice session. When the patient was ready to begin, the research assistant stepped away but remained available for assistance. At each subsequent study visit, the research assistant met the patient in the waiting room to assist in completing an assessment prior to the patient’s clinical encounter. The primary outcomes of this trial were health beliefs, cancer knowledge, self-efficacy, and satisfaction with providers’ communication about cancer and its treatments. Health-related quality of life was a secondary outcome.
Health beliefs were measured with 10 items representing beliefs, attitudes, and values about how cancer is developed and progresses and how cancer should be treated. 27 Each item has 4 response options from “Strongly Disagree” to “Strongly Agree.” Responses were summed to create a scale ranging from 10 to 40 with higher scores indicating more adaptive health beliefs. The coefficient alpha for this measure fell below the threshold for adequate reliability (<.70). Knowledge of cancer and treatment was measured with a combined scale featuring breast cancer questions or colon cancer questions, plus general cancer questions. 28 Each of the 11 questions had a true/false/do not know format; scores were created by summing correct answers, resulting in a possible range of 0–11 with higher scores indicating higher cancer knowledge. The coefficient alpha for this measure exceeded the threshold for adequate reliability (>.70).
Self-efficacy was measured with the Communication and Attitudinal Self-Efficacy scale for cancer (CASE-cancer). 29 The CASE-cancer has 3 scales: understand and participate in care (UPC; 4 items), maintain a positive attitude (MPA; 4 items), and seek and obtain information (SOI; 4 items). Each item has 4 response options ranging from “Strongly Disagree” to “Strongly Agree.” For each scale, responses are summed for a possible range of 3–12 with higher scores indicating higher self-efficacy. The CASE scales have demonstrated adequate reliability (>.70), convergent validity, and differential item functioning. 29
To measure patient satisfaction with providers’ communication about cancer and its treatment, a 6-item composite index was created using items from the Commonwealth 2006 Quality of Health Care Survey (www.commonwealthfund.org) and the Functional Assessment of Chronic Illness Therapy Treatment Satisfaction (FACIT-TS). 30 For example, one item asked “Do you have an opportunity to ask questions?” Each item has 5 response options ranging from “No, not at all” to “Yes, as much I want.” Responses were summed to create a scale ranging from 0 to 24 with higher scores indicating greater satisfaction. The coefficient alpha (internal consistency reliability) for these 6 items was .93.
Health-related quality of life was measured with the Functional Assessment of Cancer Therapy-General (FACT-G). 31 The FACT-G is a multidimensional, 28-item scale of HRQOL targeted for cancer patients. Each item has 5 response options ranging from “Not at all” to “Very much” and is scored so that higher scores indicate better HRQOL (scores range from 0 to 108). The FACT-G has demonstrated high reliability (>.90) 32 and has evidenced construct validity (e.g., convergent, divergent, known groups), 31 and sensitivity to change over time. 31
Intervention evaluation measures
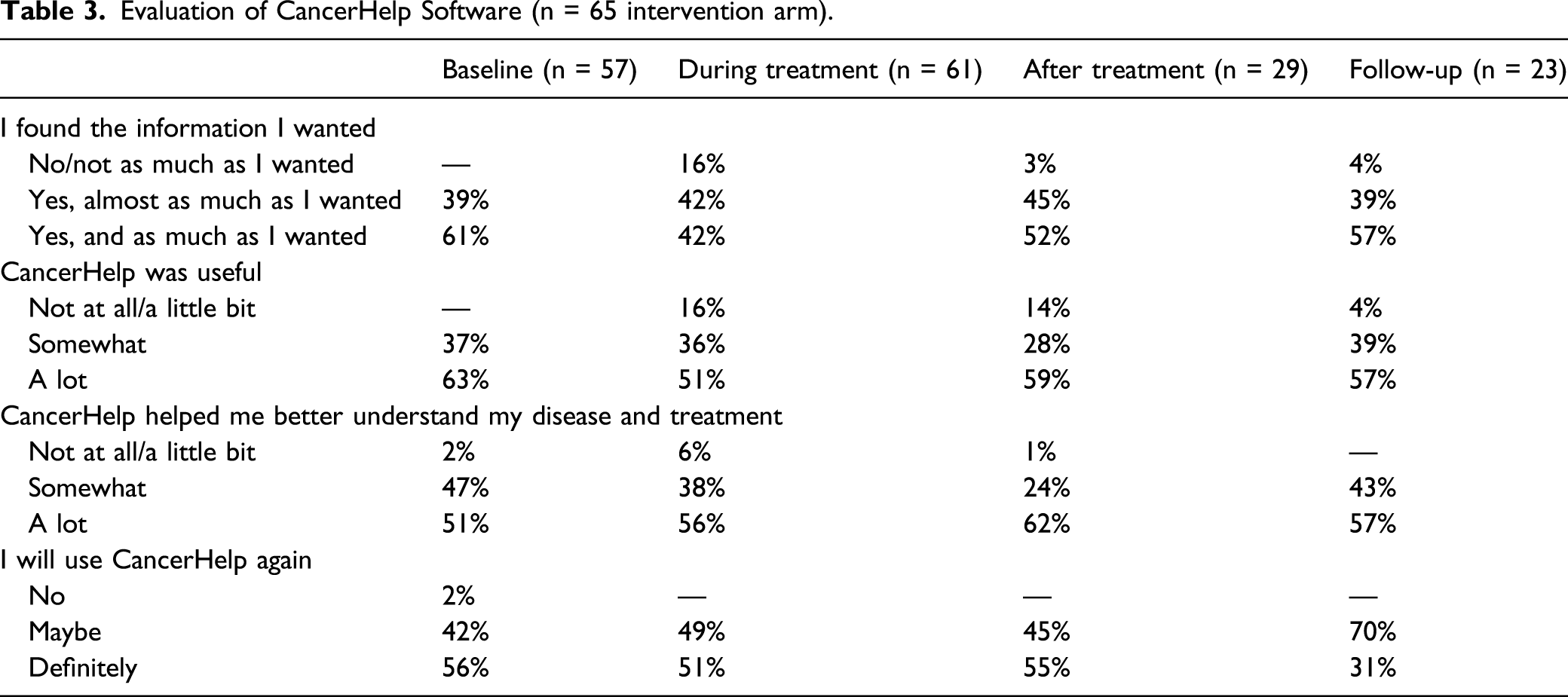
Participants in the CancerHelp-TT arm provided an evaluation of the program. This included questions about whether patients found the information they were looking for, whether they found CancerHelp-TT useful, whether the program helped them better understand their disease and treatment, and whether they would use CancerHelp-TT again.
Other measures
In addition to the outcomes, patients’ demographic and clinical characteristics were collected, including race/ethnicity, age, sex, cancer type, education level, self-reported health literacy, type of health insurance, financial strain, and performance status rating using the Eastern Cooperative Oncology Group Performance Status Rating (ECOG PSR). 33 Patients also answered one item on preferences for making decisions about health care 34 and questions about whether they had previously looked at cancer booklets/pamphlets, looked on the internet for information about cancer, talked to a doctor about cancer, or talked to a nurse about cancer.
Health literacy was measured with the Health Literacy Assessment Using Talking Touchscreen Technology (Health LiTT). 35 Health LiTT has 3 item types: prose, document, and quantitative. Participants in this study completed a 10-item short form. The Health LiTT is scored on a T-score metric with a mean of 50 and standard deviation of 10, with higher scores indicating better health literacy. Scores of ≥55 indicate adequate health literacy and <55 indicate low health literacy. 36
Statistical Analyses
Sample Size and Power Considerations
Power calculations were performed using formulas for sample size estimation for normal data in longitudinal models with a two-sided significance level of .05 and power of 80%. A correlation of .50–.60 between repeated measures was considered appropriate, as determined from previous longitudinal studies of HRQOL and satisfaction in cancer patients.37,38 A sample size of 80–90 patients per group measured over 2–3 timepoints would achieve 80% power to detect an effect size of .40 between study arms. Assuming 10% dropout, the enrollment sample size was set at 200 (100 per arm).
Efficacy and Intervention Evaluation Analyses
All statistical tests used a nominal P-value of <.05 to indicate statistical significance and were conducted in SAS version 9.4. 39 Since this was an exploratory trial (ie, the first test of the effect of this intervention on outcomes) instead of confirmatory trial, we did not adjust the P-value for multiplicity. 40
Efficacy analyses on health beliefs, cancer knowledge, HRQOL, and satisfaction with communication were conducted with random intercepts models to estimate the difference-in-differences in score change from baseline to after treatment (2 measurement timepoints). The independent variable in these models was study arm of randomization. Effect sizes were calculated as the difference-in-differences estimate divided by the pooled standard deviation for the change score (difference in means) from the appropriate post-baseline assessment and baseline. Cohen’s conventions for magnitude of effect were used: small = .20 < d < .50, medium = .50 < d < .80, and large = d > .80. 41
In addition, for satisfaction with communication and HRQOL, the differences between study arms in outcomes trajectories over 3 (HRQOL) or 4 (satisfaction with communication) measurement timepoints were analyzed with individual growth curve models. These models use both fixed and random effects parameters added sequentially from model to model (4 models for each outcome) and examine growth (change in outcomes over assessment in timepoints) for patients in the study. The first model (Model 1) includes a random intercept and the assessment timepoint as a continuous fixed effect. The random intercept parameter indicates the amount of variation in outcomes between patients at baseline, and the assessment timepoint parameter indicates the growth trajectory in outcomes for all patients. The second model (Model 2) adds a random slope for the assessment timepoint to Model 1, and this parameter indicates whether growth trajectories vary between patients. A third model (Model 3) adds an interaction term between study arm of randomization and assessment timepoint to Model 1, which determines whether outcome trajectories vary between patients randomized to the CancerHelp-TT intervention arm and the control arm. Finally, a fourth model adds the interaction term between study arm of randomization and assessment timepoint to Model 2, making it similar to Model 3, but with a random slope for assessment timepoint. Model fit was compared with −2 log likelihood, Akaike’s information criterion (AIC), the Bayesian information criterion. For each, lower values indicate better model fit.
Finally, evaluations of the CancerHelp educational software were summarized using proportions for patients randomized to the intervention arm (n = 65).
Results
In total, from January 2010 to October 2011, 178 patients were approached to participate in the study, with 152 of these eligible. Of these, 129 consented to participate (85%; Figure 2). Half were randomized to receive the CancerHelp-TT intervention and half were randomized to receive control cancer education; all participants completed baseline assessments. Sixty-three (33 intervention, 30 control) completed the end-of-treatment assessment and 49 (25 intervention, 24 control) completed the follow-up assessment. Recruitment and retention flow diagram.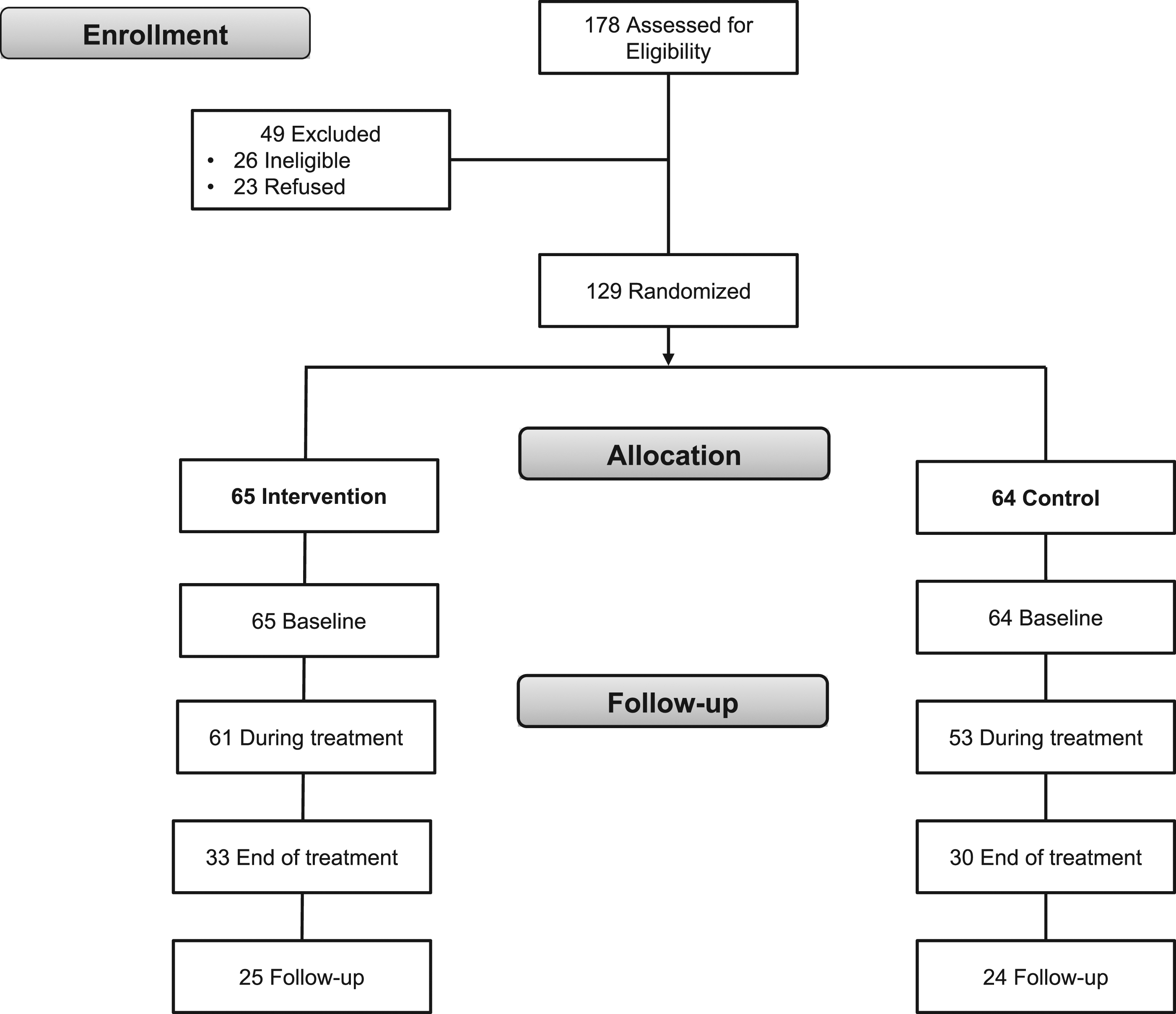
Baseline Patient Characteristics by Study Arm (n = 129).

Entries in the table represent number (percentage) of participants, or mean (standard deviation).
aThe Health LiTT score is a t-score calculated from 10 items with scale mean of 50, SD of 10.
bThe Health Beliefs scale is the sum of 10 items with a theoretical range of 10–40.
cThe Cancer Knowledge scale is the sum of 11 items with a theoretical range of 0–11.
dThe Understand and Participate in Care subscale is the sum of 4 items with a theoretical range of 4–16.
eThe Maintain Positive Attitude subscale is the sum of 4 items with a theoretical range of 4–16.
fThe Seek and Obtain Information subscale is the sum of 4 items with a theoretical range of 4–16.
gThe Satisfaction with Communication scale is the sum of 6 items with a theoretical range of 0–18.
hThe FACT-G is the sum of 27 items with a theoretical range of 0–108.
Differences in Health Beliefs, Cancer Knowledge, HRQOL, and Satisfaction with Communication Across Assessment Timepoints.
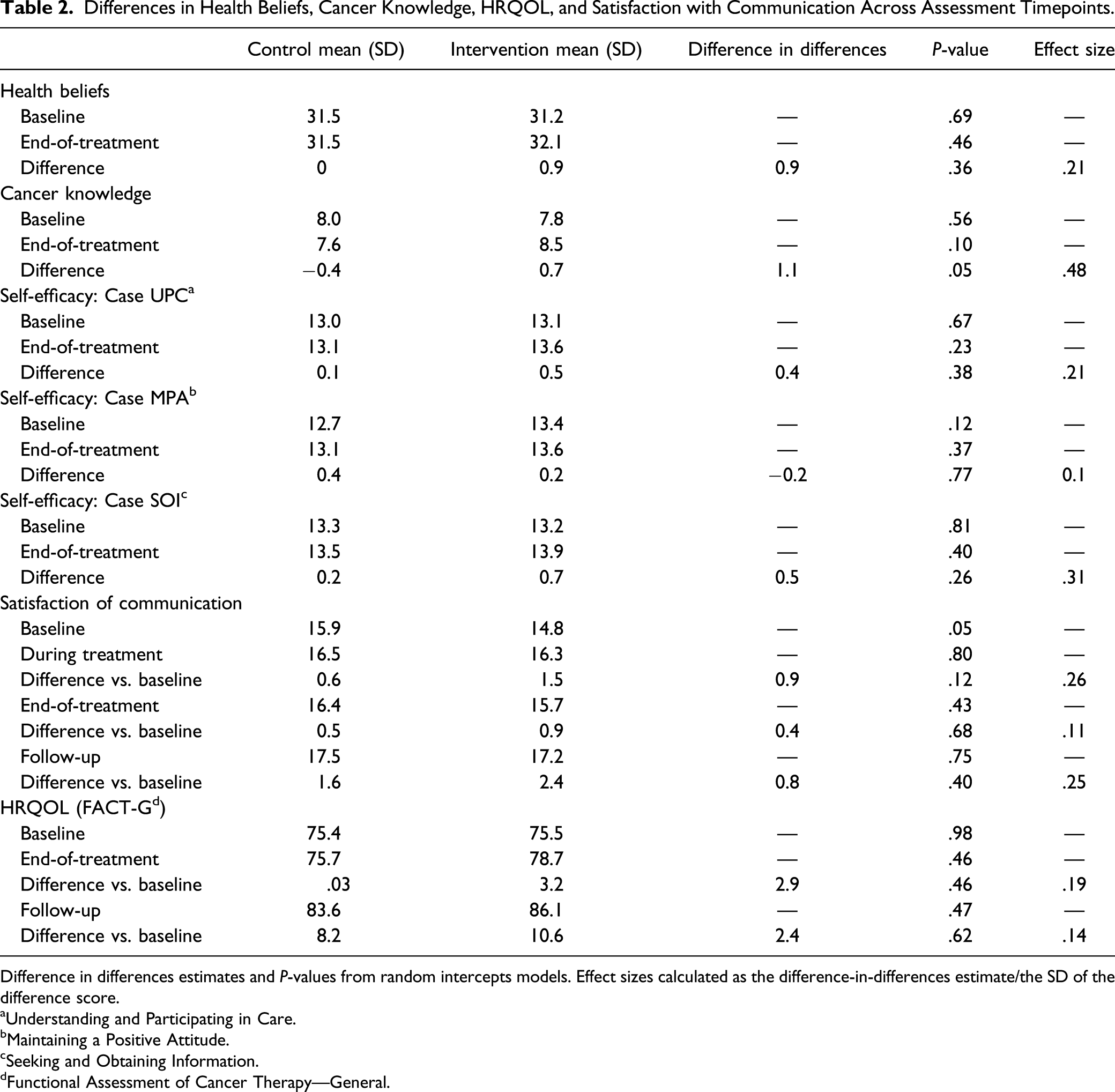
Difference in differences estimates and P-values from random intercepts models. Effect sizes calculated as the difference-in-differences estimate/the SD of the difference score.
aUnderstanding and Participating in Care.
bMaintaining a Positive Attitude.
cSeeking and Obtaining Information.
dFunctional Assessment of Cancer Therapy—General.
No significant differences between study arms were observed in the individual growth models for comparison of change across multiple post-baseline assessments on HRQOL and satisfaction with communication (Table S1). For HRQOL, in all 4 models, the random intercept was significant, indicating that HRQOL varied significantly between patients at baseline. In addition for all 4 Models, the fixed effect for assessment timepoint was positive and statistically significant, indicating that, overall, patients increased in HRQOL over from baseline to end-of-treatment, and through follow-up. Comparing models 3 and 4, −2 log likelihood and AIC indicated better fit for Model 4 (model includes random effect for the assessment timepoint). In this model, the fixed effect for study arm and the interaction term for study arm x assessment timepoint were not statistically significant. Very similar patterns were observed in the models for satisfaction with communication. For both outcomes, though differences in trajectories across timepoints were not significant, trends favored the intervention arm, with positive fixed effects for the study arm x assessment timepoint interaction term.
Evaluation of CancerHelp Software (n = 65 intervention arm).
Discussion
Innovative interventions to improve education about cancer and its treatments are needed for patients in low-resource communities, and racial/ethnic minority and low literacy patients. These interventions must help cancer patients overcome barriers to accessing information about cancer, which are often cultural- or socioeconomic-related. Leveraging health information technology to make cancer education more easily access for vulnerable populations has been suggested as a promising approach interventions, which in turn is hypothesized to improve health outcomes and experiences with care. In this randomized control trial testing a targeted, health information technology-based intervention for cancer patients in community settings, we found improvements in cancer knowledge were associated with using the intervention.
Though effect sizes for differences in outcomes between the CancerHelp-TT intervention arm and the control arm were of small magnitude, differences tended to trend in the expected direction. Most notably, the observed cancer knowledge difference between arms exceeded the hypothesized effect size (hypothesized = .40; observed = .48). These outcomes were observed despite potentially significant barriers faced by these very vulnerable patients to participate in the study. Participant retention in cancer research, especially breast cancer research, is a known challenge, and low socioeconomic status has been identified as a risk factor for breast cancer trial attrition in the past. 43 Although previously-administered, community-based cancer education programs in clinics have been effective, 44 the most vulnerable patients may need more intensive education interventions to overcome barriers related to stigma and socioeconomic challenges. For example, recent efforts in the field of kidney transplant education have been successful at overcoming racial disparities in access to gold standard treatments by providing home-based education, where families can be involved in educational discussion in a setting comfortable for patients and their support systems.45,46 Similar approaches should be considered for application with cancer patients.
Despite challenges faced by vulnerable cancer patients that may have posed barriers to participation, there were aspects of the intervention that appeared to work well. Positive ratings of the CancerHelp-TT likely reflect, in part, efforts that were made to make the program accommodating and easy to use for participants, especially those with low health literacy. For example, study activities were integrated into the clinical workflow of the sites where the study was conducted. This allowed the receipt of cancer education from CancerHelp-TT to fit into the activities patients were already involved in at the clinic without experiencing too much inconvenience for the sake of the study. In addition, the research assistants established personal relationships with providers and staff in each clinic and contributed in assisting staff and patients in basic, nonmedical clinical activities. The CancerHelp-TT program was available on multiple laptops and kiosks in clinic to make it easily accessible and provide options so the patients could use the program where they are most comfortable. Noting these positive responses, application of the CancerHelp-TT program in settings outside the clinic, such as patient’s homes (via the web or in person) or locations favored by patients such as beauty salons and barber shops, may impart a better opportunity for patients who struggle to come to the clinic to benefit from the CancerHelp-TT program.47,48
Like any study, this study has important limitations to consider when considering its results. First, since this study was a randomized, controlled trial, the emphasis in study design was on its internal validity, aiming to ensure valid comparisons between intervention and control groups. However, the generalizability of this study’s results may be limited. Replication of this trial in other geographical areas and clinical settings is warranted. Second, this trial did not include Spanish-speaking patients. Spanish-speaking patients in community oncology clinics may be among the most vulnerable patient population, and they would likely benefit from a Spanish-language version of the CancerHelp-TT program. Third, though this was an exploratory trial, and hence we elected not to adjust for multiple outcomes, the presence of multiple comparisons may have increased the probability of a false positive result. Finally, the planned sample size for this study was not achieved due to lower than expected recruitment and significant attrition. In addition to the resulting reduced statistical power to detect expected effect sizes, the patients who dropped out of the study were likely to have experienced the most severe, negative impact from their cancer, and who also started most vulnerable due to lower socioeconomic status. These patients are also hypothesized to have derived the most benefit from the CancerHelp-TT program. Additional trials testing the CancerHelp-TT program must make additional efforts to retain these patients through the end of the study.
In conclusion, this prospective, randomized, controlled trial comparing the impact of CancerHelp-TT to NCI education brochures on cancer patients’ health beliefs, cancer knowledge, HRQOL, and satisfaction with communication with cancer and cancer treatment found few statistically significant differences in study arms to achieve benefit in these outcomes. This trial’s capacity to evaluate the CancerHelp-TT program was diminished by significant patient drop-out over the course of follow-up. Despite this, encouraging trends were found, indicating that future studies may indeed find beneficial effects from CancerHelp-TT. Given persistent disparities in access to appropriate cancer treatments and cancer outcomes, there remains significant need for CancerHelp-TT and programs like it to help ensure all cancer patients have the information they need to manage their condition to maximize their quality of life.
Footnotes
Acknowledgments
The authors thank the research assistants who recruited and interviewed study participants (Beatriz Menendez, Niraj Patel, Evie Sprague, Veronica Valenzuela), the CancerHelp Institute (Ed. Miller, Celia Muench), the programming and analysis staff (Michael Bass, MS, Jennifer Beaumont, MS, Mark Monforti, Monica Prudencio, Katy Wortman, MSW), co-investigators (David Cella, PhD, Jin-Shei Lai, PhD) and physicians and clinical staff at John H. Stroger Hospital of Cook County (Elizabeth Marcus, MD, Michael Mullane, MD), Advocate Illinois Masonic Medical Center (Maritess Caamic, RN, Ann Mauer, MD, Samir Undevia, MD), and Mt. Sinai Hospital (Grace Golden, Ervin Hire, MD, Gina Menyah, Anna Liza Rodriguez, RN.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by AHRQ Grant # R18-HS017300 (awarded to E. A. Hahn).
Ethical Approval
The study and intervention protocol was approved by the Institutional Review Board (#CR5_STU00013521), and all participants provided informed consent.
