Abstract
Cervical cancer is a common female cancer. It is strongly associated with human papillomavirus (HPV) infection. However, HPV infection alone is not sufficient to induce cervical cancer because its development is dependent on the coexistence of several factors that enable the virus to overcome the host immune system. These include individual genetic background, environmental factors, or diet, including dietary selenium intake. Selenium is an essential trace element with antiviral properties and has been shown to exert antitumor effects. Surprisingly, the role of selenium in cervical cancer has not been studied as intensively as in other cancers. Here, we have summarized the existing experimental data on selenium and cervical cancer. It may be helpful in evaluating the role of this nutrient in treatment of the mentioned malignancy as well as in planning further studies in this area.
Introduction
Cervical cancer is the fourth most common female cancer worldwide, with an estimated 570,000 new cases and 310,000 deaths in 2018 (Globocan 2018). The main risk factor associated with this disease is infection with human papillomavirus (HPV; mostly types 16 and 18), which is found in the majority of cases. 1 However, HPV infection alone is not sufficient to induce cervical cancer. Certain factors, such as individual genetic variation and environmental factors or diet, may influence the risk of this disease. 2 Recent retrospective epidemiological studies suggest that there may be a relationship between selenium (Se) status and cervical cancer. 3 Se is an essential trace element that plays some important functions in the human organism through selenoproteins. These functions are related to the antioxidant system, redox signaling, thyroid hormone metabolism, immune system, and many others. 4 Notably, studies have shown that Se possesses antiviral activity. 5 At the same time, Se has been implicated in human cancers. 6 Scientific research has focused on the association between Se and cancer at different sites, including the head and neck, esophagus, colon, liver, skin, lung, breast, bladder, and prostate. 7 However, little attention has been paid to the role of Se in cervical cancer. Considering the antiviral activity of Se, 5 it is important to investigate its possible therapeutic effect in cervical cancer. Since this type of cancer is a challenging clinical problem due to its high metastatic potential and resistance to therapy, effective treatment methods are highly required. Altough the possible preventive role of Se in cervical cancer is still under question (no association was shown in one prospective study 8 and there was no possibility of concluding the causative relationship based on case-control studies), 3,9 -11 data from laboratory experiments conducted in cells and animals show that Se may be used in cervical cancer therapy. However these observations need to be further confirmed in clinical studies. In this short review we summarized the existing experimental data on Se and cervical cancer. This may be helpful in evaluating the therapeutic potential of this nutrient in the mentioned malignancy as well as in planning further research.
Methods
We searched literature in electronic databases including PubMed and Scopus until the 15th of September 2020. Search terms included a combination of terms “selenium” with “cervical cancer” or “HPV” or “CIN1” (cervical intraepithelial neoplasia grade 1). Titles and abstracts were further screened in order to select experimental studies that aimed to assess the cancer therapeutic effects of Se or Se compounds in patients with cervical cancer or CIN1, or in different laboratory cervical cancer models (
Laboratory Studies on Se and Cervical Cancer
In Vitro Studies on Se Compounds in Cervical Cancer.
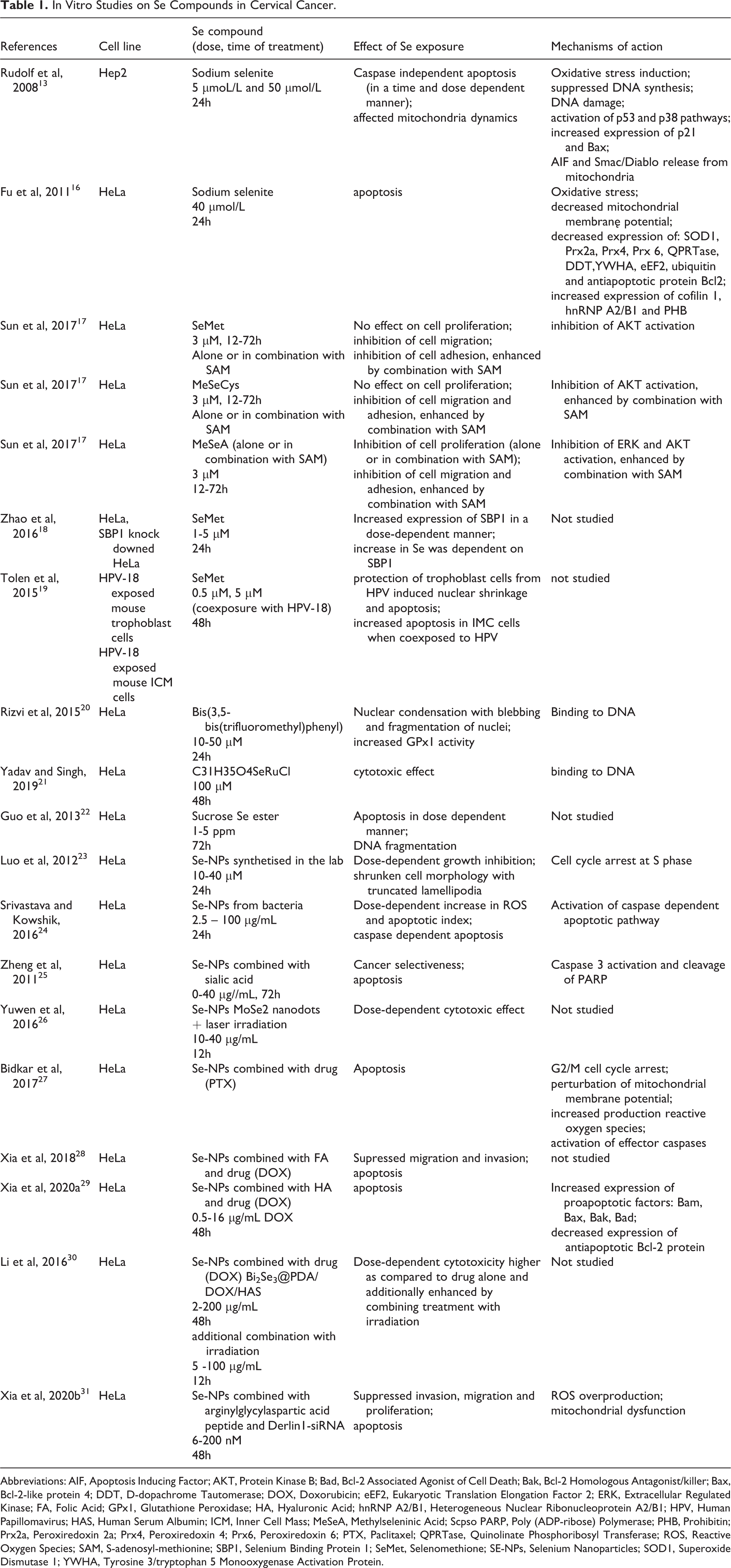
Abbreviations: AIF, Apoptosis Inducing Factor; AKT, Protein Kinase B; Bad, Bcl-2 Associated Agonist of Cell Death; Bak, Bcl-2 Homologous Antagonist/killer; Bax, Bcl-2-like protein 4; DDT, D-dopachrome Tautomerase; DOX, Doxorubicin; eEF2, Eukaryotic Translation Elongation Factor 2; ERK, Extracellular Regulated Kinase; FA, Folic Acid; GPx1, Glutathione Peroxidase; HA, Hyaluronic Acid; hnRNP A2/B1, Heterogeneous Nuclear Ribonucleoprotein A2/B1; HPV, Human Papillomavirus; HAS, Human Serum Albumin; ICM, Inner Cell Mass; MeSeA, Methylseleninic Acid; Scpso PARP, Poly (ADP-ribose) Polymerase; PHB, Prohibitin; Prx2a, Peroxiredoxin 2a; Prx4, Peroxiredoxin 4; Prx6, Peroxiredoxin 6; PTX, Paclitaxel; QPRTase, Quinolinate Phosphoribosyl Transferase; ROS, Reactive Oxygen Species; SAM, S-adenosyl-methionine; SBP1, Selenium Binding Protein 1; SeMet, Selenomethione; SE-NPs, Selenium Nanoparticles; SOD1, Superoxide Dismutase 1; YWHA, Tyrosine 3/tryptophan 5 Monooxygenase Activation Protein.
In Vivo Studies on Se Compounds Used in the Experimental Studies on Cervical Cancer.
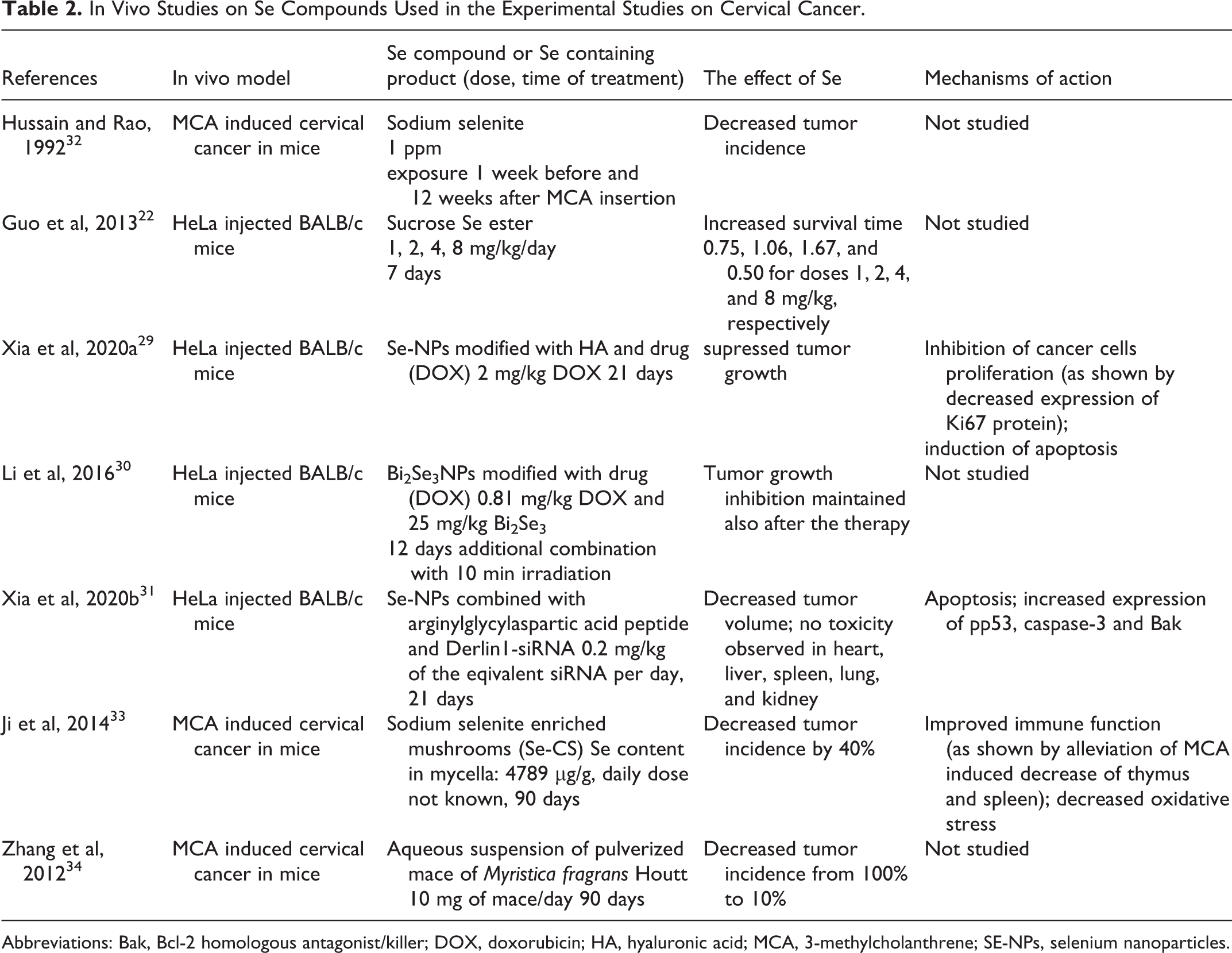
Abbreviations: Bak, Bcl-2 homologous antagonist/killer; DOX, doxorubicin; HA, hyaluronic acid; MCA, 3-methylcholanthrene; SE-NPs, selenium nanoparticles.
Sodium Selenite
Among the inorganic forms of Se, sodium selenite (Na2SeO3) was the only one tested in studies on cervical cancer (Tables 1–3). It is an excellent example of Se compounds with redox activities. Its ability to generate reactive oxygen species underlies its toxicity in cancer cells. 35
Randomized Placebo Controlled Trials Investigating the Effect of Se Supplementation on Clinical Outcomes in Cervical Intraepithelial Neoplasia or Cervical and Uterine Cancer.

Abbreviations: CIN1, cervical intraepithelial neoplasia 1; RT, radiotherapy.
Rudolf et al exposed Hep2 cells to sodium selenite at 5 and 50 µmol/L for 24h. 13 The authors observed that Se induced oxidative stress in the cells, which resulted in suppressed DNA synthesis, DNA damage, and subsequent activation of the p53 pathway. Along with these changes, the Se activated p38 pathway led to the accumulation of proapoptotic Bax protein and release of AIF and Smac/Diablo proteins from mitochondria, which together led to caspase-independent apoptosis. 13 Proteomic changes upon sodium selenite treatment were investigated by Fu et al in HeLa cells treated with the compound at a concentration of 40 µmol/L for 24h. 16 It was shown that selenite altered the expression of 13 proteins, including proteins involved in redox balance, apoptosis, signal transduction, mRNA transcription, protein translation, degradation, and translocation. Increased generation of ROS and decreased mitochondrial membrane observed along with downregulation of antioxidant proteins led the authors to conclude that sodium selenite induced apoptosis in HeLa cells via a ROS-mediated mitochondrial pathway. 16
Hussain and Rao conducted a study in mice to assess the chemopreventive properties of sodium selenite
Organic Se Compounds
Organic selenium compounds may be of natural and synthetic origin and include compounds largely differing in chemical structure, cellular uptake, metabolic pathways and biological effects. Some of them were shown to exert antitumor activity
The potential antitumor activity of a combination of organic Se compounds with a methyl group donor, S-adenosyl-methionine (SAM), was investigated in HeLa cells by Sun et al. 17 The authors tested three organic Se compounds, two of which, selenomethionine (SeMet) and methylselenocysteine (MeSeCys), are found naturally in the food, and the third one, methylseleninic acid (MeSeA), is of synthetic origin. The anticancer effect was studied at the level of cell proliferation, migration and adhesion, with further assessment of the levels of activated AKT and ERK proteins, which are both related to cell proliferation and migration. MeSeA alone (3 µM) inhibited both ERK and AKT activation and suppressed proliferation, migration and adhesion in HeLa cells, and all these effects were enhanced by combination with SAM (3 µM). Two other compounds, SeMet (3 µM) and MeSeCys (3 µM), significantly inhibited AKT alone and this inhibition was enhanced by SAM in the case of MeSeCys. No effect on cell proliferation was shown in the case of MeSeCys and SeMet either alone or in combination with SAM. However, both MeSeCys and SeMet inhibited cell migration alone (with the enhancing effect of combination with SAM observed for MeSeCys) and adhesion (with the enhancing effect of combination with SAM observed for the two compounds). 17 Altogether, this study shows that among the three tested organic Se compounds, MeSeA exerted the most promising tumor-suppressive effects in HeLa cells.
Zhao et al observed that exposure of HeLa cells to SeMet at concentrations of 1-5 µM for 24h increased the expression of selenium-binding protein 1 (SBP1). 18 SBP1 is a protein that binds covalently to Se (in contrast to selenoproteins to which Se is incorporated co-translationally as a consequence of decoding selenocysteine UGA codon). Human studies have shown that SBP1 is often underexpressed in cancer. This suggests a tumor-suppressive function of this protein. 39 Zhao et al showed in their study that the knockdown of SBP1 in HeLa affected intracellular Se levels in these cells in response to SeMet treatment (the dose-dependent increase was lower compared to that in the control cells). 18
Interesting observations regarding SeMet and HPV coexposure were made by Tolen et al. 19 These authors did not investigate the cervical cancer model. They used HeLa cell lysates to isolate HPV-18 DNA, which they further used to infect mouse embryos. The embryos were exposed to HPV in the presence of SeMet at 0,5 µM and 5 µM, and it was observed that SeMet at both concentrations prevented nuclear shrinkage caused by the virus and decreased the number of apoptotic cells as compared to cells exposed only to HPV. On the other hand, co-exposure to HPV18 and SeMet increased apoptosis in the second type of cells associated with the trophoblast (inner cell mass), showing that the effect of SeMet on HPV-exposed cells depends on the specific cell type. 19
Apart from methylseleninic acid, there are few other synthetic organoselenium compounds studied, including bis(3,5-bis(trifluoromethyl)phenyl)selane, synthesized as an example of an organometallic candidate for cervical cancer chemotherapeutic drugs.
20
HeLa cells exposed to this compound at 10-50 µM were shown to undergo dose-dependent changes typical for late apoptosis, including nuclear condensation with blebbing and nuclei fragmentation. Yadav and Singh investigated the anticancer properties of different organometallic ruthenium complexes with Se and compared them to those of organometallic ruthenium complexes with sulfur.
21
Within the studied complexes, the complex containing Se (C31H35O4SeRuCl) seemed to inhibit the proliferation of HeLa cells most efficiently. Sucrose selenium ester, synthesized from sucrose and selenium oxychloride, was shown to inhibit proliferation of HeLa cells as well as three other cancer cell lines (bladder carcinoma cell line 5637, human malignant melanoma cell line A375, and gastric carcinoma cell line MGC-803).
22
The inhibitory effect was dose-dependent and associated with the induction of apoptosis. Importantly sucrose-Se was not toxic in the normal human liver cell line HL-7702. Furthermore, in the
Se Nanoparticles
Special interest in the therapeutic potential of Se in cancer has been paid in recent years to Se nanoparticles (Se-NPs). Nanoparticles (particles with at least one dimension less than 100 nm) are generally attractive because of their unique biomedical features, allowing them to function as very effective drug carriers. Thus combining nanotechnology with the anticancer effects of Se offers very promising possibilities for cancer treatment.
40
Most importantly, Se-NPs have been shown to exert lower toxicity
Several
Different modifications of Se-NPs by adding additional factors to their structure improved their therapeutic utility specifically in cervical cancer treatment. For example, Zheng et al added sialic acid (SA) to Se-NPs to enhance their cancer-targeting and cell-penetrating abilities. The combination with SA significantly increased the cellular uptake of Se-NPs in HeLa cells as compared to SeNPs alone. 25 Apart from dose-dependent cytotoxicity in HeLa cells, Se-NPs decorated with SA (SA-Se-NPs) were shown to be non-toxic in normal human kidney HK-2 cells. The mechanism underlying the cytotoxic effect of SA-Se-NPs in HeLa was apoptosis induced by activation of caspase-3 and subsequent cleavage of PARP. 25
Se-NPs have also been shown to be effective photothermal therapy (PTT) agents. PTT agents are factors that induce high temperature in the cell upon irradiation and consequently lead to cell death. The combination of PTT with molybdenum diselenide (MoSe2) nanodots was shown to exert cytotoxic effects in HeLa cells, suggesting that MoSe2 nanodots are a novel effective PTT agent. This study also shows a broad spectrum of general anticancer potential in the case of Se-NPs, which results not only from its biological activity but also from its physical properties. 26
Nevertheless, the most promising property of Se-NPs relates to their drug-carrying ability. Effective combinations of Se-NPs with drugs in cervical cancer treatment were shown in HeLa cells and a xenograft mouse model. For example, paclitaxel (PTX)-loaded Se-NPs induced apoptosis in HeLa cells (but also in various other cancer cell lines). The authors indicated that the combination of Se-NPs with the drug led to G2/M cell cycle arrest, perturbation of mitochondrial membrane potential, increased production of reactive oxygen species, and activation of effector caspases.
27
More complex Se-NP-based drug delivery systems include additional decoration, for example, with folic acid (FA) or hyaluronic acid (HA). These molecules were added to the system in order to enhance the tumor-targeting ability of the drug since tumor cells were shown to express high levels of cell surface receptors for FA and HA.
46,47
Se-NPs modified with FA increased, for example, the anticancer effects of doxorubicin (DOX) in HeLa cells.
28
Xia et al tested the antitumor activity of HA-Se@DOX - DOX-loaded Se-NPs decorated with HA. HA-Se@DOX was shown in this study to exert very good cytotoxic effects both
Apart from drugs, nanoparticles have been shown to be good delivery platforms for small interfering RNAs (siRNAs). These are sequence-specific molecules with the ability to silence genes, which are very promising candidates for novel anticancer drugs. Using siRNA technology for therapeutic purposes is particularly attractive due to the ability to target specific genes and have less toxic effects than traditional chemotherapy.
48
Combining siRNA with nanotechnology is expected to become the gold standard in cancer therapy in the near future. An example of using Se-NPs combined with siRNA in cervical cancer treatment was presented recently by Xia et al.
31
In their study a tumor-targeting gene delivery vehicle, RGDfC-Se@siRNA was constructed. For this purpose, Se-NPs were loaded with RGDfC peptide and Derlin1-siRNA. RGDfC peptide (arginylglycylaspartic acid peptide) was used here to enhance the tumor-targeting ability of the compound since it binds to membrane protein, integrin alpha V beta 3, shown to be overexpressed in cancer cells. Similarly, the product of the siRNA target gene, Derlin1, is a protein that is overexpressed in cancer. This combination resulted in a specific cellular uptake of the compound in HeLa and effective downregulation of Derlin1 at both the mRNA and protein levels. Furthermore, RGDfC-Se@siRNA suppressed the invasion, migration, and proliferation of HeLa cells by inducing apoptosis, probably by ROS-mediated mitochondrial dysfunction.
Mushrooms and Plants Rich in Selenium
Ji et al analyzed the
Zhang et al investigated the effect of seeds from the herb
On the other hand, Se fortification had no effect on the cytotoxic activity of kale and kohlrabi sprouts in SiHa cells as similar cytotoxic effect was observed in both fortified and unfortified sprouts. 14
Human Studies on Se and Cervical Cancer—Randomized Controlled Trials
As far as we are concerned, no clinical trials on Se treatment efficacy in cervical cancer have been conducted so far. Considering that some mechanisms of Se (such as the antiviral activity) may overlap both in treatment and prevention, it is important to examine the outcomes of supplementation trials conducted for preventive purposes (Table 3). Such a chemopreventive potential of Se was suggested by observational retrospective studies showing decreased Se status in cervical cancer patients or subjects infected with HPV as compared to healthy controls.
3,9
-11
Although these studies do not provide evidence on the causative relationship between low Se status and increased risk of cervical cancer, and the only observational cohort study of relevant interest did not indicate any association between Se status and cervical cancer risk,
8
some promising data were revealed in the supplementation trial conducted by Karamali et al.
36
This randomized, double-blind placebo-controlled trial aimed to assess the possible preventive activity of Se against the progression of cervical intraepithelial neoplasia 1 (CIN1), which is a premalignant lesion, usually associated with HPV infection.
50
In this study, 26 women with CIN1 were supplemented with 200 µg for six months and notably, Se supplementation was associated with higher CIN1 regression (88% vs. 56% in the placebo group,
Little attention has been paid to the effects of Se supplementation on clinical outcomes in cervical cancer patients (Table 3). Muecke et al seemed to be the only authors who investigated this issue. 37,51,52 They conducted a study of 81 patients with cervical cancer (n = 11) and uterine cancer (n = 70) described as Se deficient (blood Se <84 µg/L) who were randomly assigned to receive either Se or placebo during radiotherapy. The subjects were supplemented with Se at a dose of 500 µg on the days of radiotherapy and 300 µg on the days without radiotherapy. Adjuvant Se supplementation increased Se status in patients and was associated with a lower incidence of radiation-induced diarrhea. 51,52 However, Se supplementation had no effect on disease-free survival and overall survival in this group. 37 It should be noted that the majority of patients in the above study were diagnosed with uterine cancer (endometrial cancer), which, unlike cervical cancer, appears not to be associated with HPV infection. 53 The important issue to be taken into account in terms of analyzing the Se effects in humans, especially when comparing different studies, is the chemical form of the used supplement. In the study by Karamali et al, Se yeast was used, 36 whereas Muecke et al used sodium selenite. 51 This fact could have an impact on the final outcome of the study because different Se compounds generate different biological effects. 54,55
Summary, Concluding Remarks, and Research Gaps
Experimental studies conducted in cells and mouse models of cervical cancer indicate that different Se compounds exert anticancer effects in cervical cancer induced by HPV or chemical carcinogen. Of all the tested Se compounds, the most promising anticancer effects in cervical cancer seem to be associated with Se-NPs, which when combined with antineoplastic drugs and factors enhancing their tumor-targeting ability, were highly selective towards cancer cells and effective at relatively low doses. An additional combination of Se-NPs with thermal therapy or with siRNA technology further expanded the therapeutic possibilities of Se in cervical cancer. The most recently investigated combinations of Se-NP platforms with immunotherapy based on the ability of Se-NPs to deliver therapeutic mRNAs have not been tested in cervical cancer yet and await evaluation in this type of malignancy. 56 -58 Importantly, future studies should be conducted on different cervical cancer cell lines since most of the existing evidence is concerned only with HeLa cells. Most importantly, Se-NP drug delivery platforms were shown to be non-toxic to normal cells or tissues. This is a critical issue because depending on its dose and chemical form, Se can be extremely toxic, 35 as clearly indicated in humans by cases of intoxication from misformulated dietary supplements. 59 Moreover, long-term Se exposure at dose 200 µg (considered to be non-toxic) may be associated with adverse health effects in humans, including diabetes 60,61 and cancer. 62 Altogether, these facts should be taken into account when using Se for therapeutic purposes.
Laboratory evidence on specific mechanisms of action of Se in cervical cancer is scarce (Figure 1). According to the
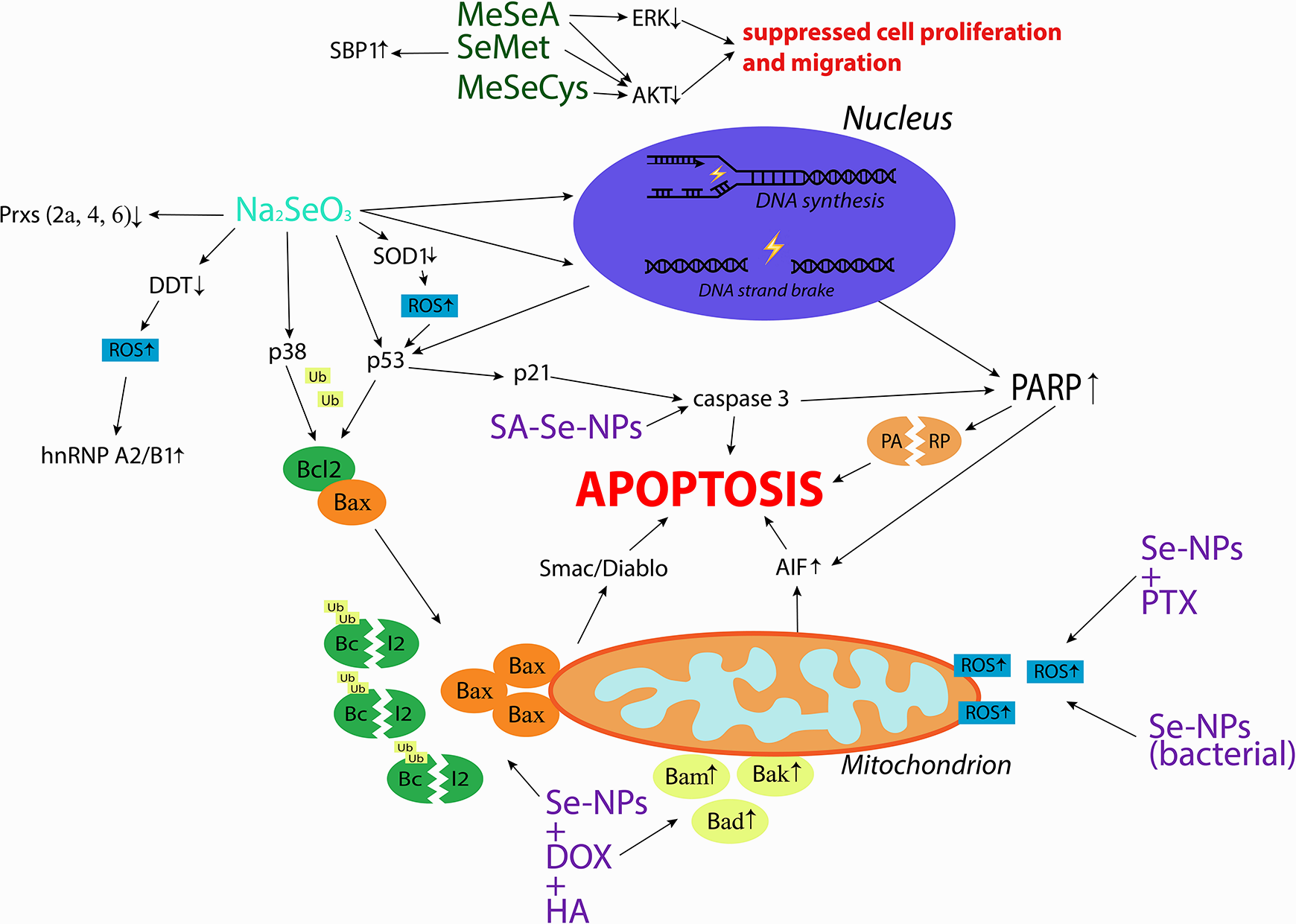
Mechanisms of action investigated in studies on Se compounds effects in cervical cancer cells. Organic Se compounds are indicated in green, inorganic Se compound is indicated in blue, Se nanoparticles are indicated in purple. AIF, Apoptosis Inducing Factor; AKT, Protein Kinase B; Bad, Bcl-2 Associated Agonist of Cell Death; Bak, Bcl-2 Homologous Antagonist/killer; Bax, Bcl-2-like protein 4; Bcl-2, B-cell lymphoma 2; DDT, D-dopachrome Tautomerase; DOX, Doxorubicin; ERK, Extracellular Regulated Kinase; HA, Hyaluronic Acid; hnRNP A2/B1, Heterogeneous Nuclear Ribonucleoprotein A2/B1; MeSeA, Methylseleninic Acid; MeSeCys, Methylselenocysteine; PARP, Poly (ADP-ribose) Polymerase; Prx2a, Peroxiredoxin 2a; Prx4, Peroxiredoxin 4; Prx6, Peroxiredoxin 6; PTX, Paclitaxel; ROS, Reactive Oxygen Species; SA, Sialic Acid; SBP1, Selenium Binding Protein 1; SeMet, Selenomethione; SE-NPs, Selenium Nanoparticles; SOD1, Superoxide Dismutase 1; Ub, Ubiquitin. 13,17,20-21,34-35,37, 41.
Analysis of human cervical cancer tissues as well as human cervical cancer cell lines (all with HPV) indicated the downregulation of selenium-dependent glutathione peroxidase 3 gene, which may further suggest possible role of selenoproteins in cervical cancer and HPV infection. 68 This issue has not been discussed in this review because most studies concerning Se and cervical cancer have focused on Se compounds in terms of their cytotoxic properties. Nevertheless, the activity and functions of selenoproteins are also considered to be responsible for the anticancer effect of Se. 6 Hence, more interest should be gained in the role of selenoproteins in cervical cancer.
Overall, the existing experimental evidence on possible therapeutic potential of Se in cervical cancer is limited. However, because the available data is promising, it should be further explored, especially with respect to the specific mechanisms of action exerted by this element in HPV infection. A more in-depth investigation of the antiviral activity of Se is also of high interest in terms of its possible chemopreventive effect in cervical cancer, a topic that was not addressed in this review. The fact that cervical cancer is a preventable malignancy makes it important to identify potential nutritional chemopreventive strategies to reduce its incidence and mortality rates. In this context, the hypothesis that high or adequate Se status may decrease the risk of cervical cancer by protecting against HPV infection is very attractive. However, it currently lacks solid scientific evidence. Epidemiological studies indicate a decreased Se status in cervical cancer patients, but the causative nature of this relationship is still not clear. Therefore, more data from human studies (prospective studies and supplementation trials) are needed to verify the hypothesis that higher Se status may prevent HPV infection and/or the development of cervical cancer. Certainly, the role of Se in cervical cancer treatment (and prevention) requires further research addressing the issues discussed in this review It would also be important in the light of the scientific discussion about the general role of Se in cancer and the many still unresolved issues in this area. 69
Footnotes
Authors' Note
Our study did not require an ethical board approval because it did not contain human or animal trials.
Acknowledgment
The authors would like to thank Nataliia Pavlenko for graphical support in preparing this manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by the Nofer Institute of Occupational Medicine—grant number IMP 14.5/2020; Postdoctoral Scientific Development Fund of Heilongjiang Province—grant number LBH-Q17096; China Postdoctoral Science Foundation—grant number 2015M571440; National Natural Science Foundation of China—grant number 81773368.
