Abstract
Background
Ten percent of patients with melanoma develop in-transit metastases (ITM). Isolated limb infusion (ILI) is a well-established therapy for unresectable ITM on the extremities, but the ideal sequencing/line of therapy of ILI has not been defined. This study evaluates ILI as first-line, second-line, or third or later-line therapy.
Methods
A retrospective review included all patients with unresectable ITM who underwent ILI from 2006-2023.
Results
A total of 130 patients were identified, 61% female, median age of 71 (31-89) years. Median follow-up was 37.5 months. ILI was first-line therapy in 80% (n = 104), second-line in 15% (n = 19), and third or later-line in 5.4% (n = 7). Overall response rate (ORR) and complete response (CR) rates for ILI as any line of therapy were 74% and 41%, respectively. ORR for ILI as first, second, or third or later-line therapy were 78%, 63%, and 57%, respectively. CR rates for ILI as first, second, or third or later-line therapy were 42%, 37%, and 43%, respectively. There were no significant differences in ORR, progression-free survival (PFS), overall survival, or disease-free survival between therapy lines. Median PFS for ILI as first, second, or third or later-line therapy were 6.9, 5.4, and 18 months, respectively.
Conclusion
Patients responded well to ILI, whether or not they received previous therapies for unresectable ITM. First-line ILI appears to have a better ORR than later lines of ILI. Although sample size limited statistical significance, there was a 21% improvement in ORR when ILI was used as first-line vs third-line therapy, which is clinically meaningful. ILI is effective for unresectable melanoma ITM and can be used as salvage therapy.
Plain language summary
Isolated limb infusion (ILI) is an excellent treatment option for patients with in-transit melanoma. In this study, patients responded notably well when ILI was used as first-line therapy, and also responded well when ILI was used as a salvage therapy.
Keywords
Introduction
Melanoma is a type of skin cancer formed by malignant transformation of melanocytes, the melanin-producing cells that reside in the basal layer of the epidermis. Melanoma is the fifth most common cancer in the United States. Although melanoma represents only 1% of skin cancers, it accounts for the majority of skin cancer related deaths. 1 Up to 10% of patients diagnosed with high-risk early-stage melanoma may develop in-transit metastases (ITM), which are characterized by tumor nodules that spread along lymphatics in the subcutaneous or dermal layers between the primary lesion site and the regional nodal basin. 2 The development of ITM portends a poor prognosis, with 5-year survival rates ranging 24%–54%.3,4
Management of melanoma patients with unresectable ITM is not standardized, as literature is limited, and almost all randomized control trials of systemic therapy either exclude patients with ITM or it is difficult to understand (and extract any data) if any patients with ITM were treated in these trials.5,6 Current treatment modalities for unresectable melanoma ITM include systemic, intralesional, and regional approaches.
Systemic therapy options for unresectable melanoma ITM include immune checkpoint inhibitors such as anti-PD1 agents such as nivolumab, and ani-CTLA4 agents such as ipilimumab or targeted therapy options including BRAF inhibitors such as dabrafenib, and MEK inhibitors such as trametinib. Systemic therapies, either used in combination or as single-agent therapy, have shown complete response (CR) rates between 5%–19% and overall response rates (ORR) between 19%–69%.7-9 Intralesional therapy options for unresectable melanoma ITM include agents commercially available or on clinical trial such as interleukin-2 (IL-2), PV-10, and talimogene laherparepvec (TVEC), which have shown variable rates of success in several studies, with reported CR rates between 26%–78%.7,10-12
Regional therapy with isolated limb infusion (ILI) or isolated limb perfusion (ILP) for unresectable melanoma ITM have both shown excellent results. 13 ILP, first described by Creech and Krementz in 1958, consists of surgical isolation of the extremity for delivery of localized heated chemotherapy (typically melphalan), at doses that would be toxic systemically.13,14 ILI, first described by Thompson et al, in 1996, is a minimally invasive percutaneous alternative to ILP, with less technical complexity and lower procedure-related morbidity.13,15,16 One large study found an ORR of 64.1%, with CR 28.9% and median overall survival (OS) of 38.2 months for first-line ILI therapy for stage IIIB and IIIC melanoma. 15 Since ILI is associated with lower morbidity, it is considered to be the first-line regional therapy for unresectable in-transit melanoma at several institutions.17-19
In the absence of a universally accepted protocol for the treatment of unresectable melanoma ITM, there is a critical need to elucidate the optimal sequencing of therapeutic modalities. The purpose of this study is to evaluate the response rates and survival outcomes associated with ILI when used as either first-line, second-line, or third or later-line therapy for unresectable melanoma ITM. Our hypothesis is that ILI would result in better response rates and survival outcomes when used as first-line therapy for patients with unresectable melanoma ITM.
Methods
Patient Selection
After receiving an exempt determination from the Institutional Review Board (MCC #22585/IRB #Pro00072457), a retrospective review study at this single-institution cancer center was conducted. Patients included in this study were 18 years or older, with American Joint Committee on Cancer (AJCC) eighth edition stage IIIB or IIIC melanoma with unresectable in-transit metastasis confined to an extremity, who underwent ILI between 2006 and 2023. 20 Patients with distant metastatic disease were excluded. Patients meeting these criteria were selected consecutively, eliminating potential bias. The reporting of this study conforms to STROBE guidelines. 21
Description of Procedure
The ILI procedure at this institution has been described previously by Carr et al. 22 Preoperatively, the infusion dosing is determined based on limb volume, assessed through sequential circumference measurements along the length of the extremity. The melphalan doses used were 7.5 mg/L (with a maximum of 100 mg) for lower extremities and 10 mg/L (with a maximum of 50 mg) for upper extremities. The melphalan dose was adjusted based on ideal body weight. Actinomycin-D was administered at 100 mcg/L. 22
Postoperatively, patients are placed in a monitored setting with hourly neurovascular exams for 24 hours after ILI. After serum creatine phosphokinase levels peak and then start to return to baseline, patients are discharged on 3 months of aspirin 325 mg and pantoprazole 40 mg daily. This usually occured around day 2 for upper extremity ILIs and day 4-5 for lower extremity ILIs. Restaging full body positron emission tomography and computed tomography scans are typically performed at 3-4 month intervals for 3 years, then every 6 months for years 4-5, and yearly thereafter. 22
Data Collection
Data was collected through retrospective chart review. Outside institution patient record documentation was concurrently reviewed, when available. A diagnosis of ITM was confirmed on clinical documentation as well as dermatopathology reports. All patients presented with unresectable ITM, and therefore, surgery was not feasible in this population. Best response was defined by clinical documentation and lesion measurements; as the provider notes generally denote partial response (PR) or CR vs stable disease (SD) or progressive disease (PD). Any progression was defined as an increase in the size of any ITM lesion, development of a new ITM lesion, development of nodal disease, or development of distant metastatic disease. In-field progression was defined as an increase in size of an ITM lesion or development of a new ITM lesion on the ipsilateral extremity, within the treated portion of the extremity (ie: distal to the tourniquet used during ILI). Out-of-field progression was defined as development of a new metastatic lesion at a site distant to the affected extremity (proximal to the tourniquet used during ILI) and ipsilateral lymph node basin. All patient data were de-identified.
Statistical Methods
Categorical variables were compared using Fisher’s exact test. Progression-free survival (PFS) was measured from the date of ILI to the date of first progression, which was sub-divided into in-field and out-of-field progression. Disease specific survival (DSS) was measured from the date of ILI to the date of death secondary to melanoma. OS by ILI was measured from the date of ILI to the date of death of any cause. PFS, DSS, and OS probability estimates were calculated using Kaplan-Meier analysis method. The log-rank test was used to compare PFS, DSS, and OS between two or more independent groups of patients. Statistical tests were two-sided, and significance was defined as P-value of <.05. All statistical analyses were conducted using the R version 4.2.
Results
Patient Demographics.
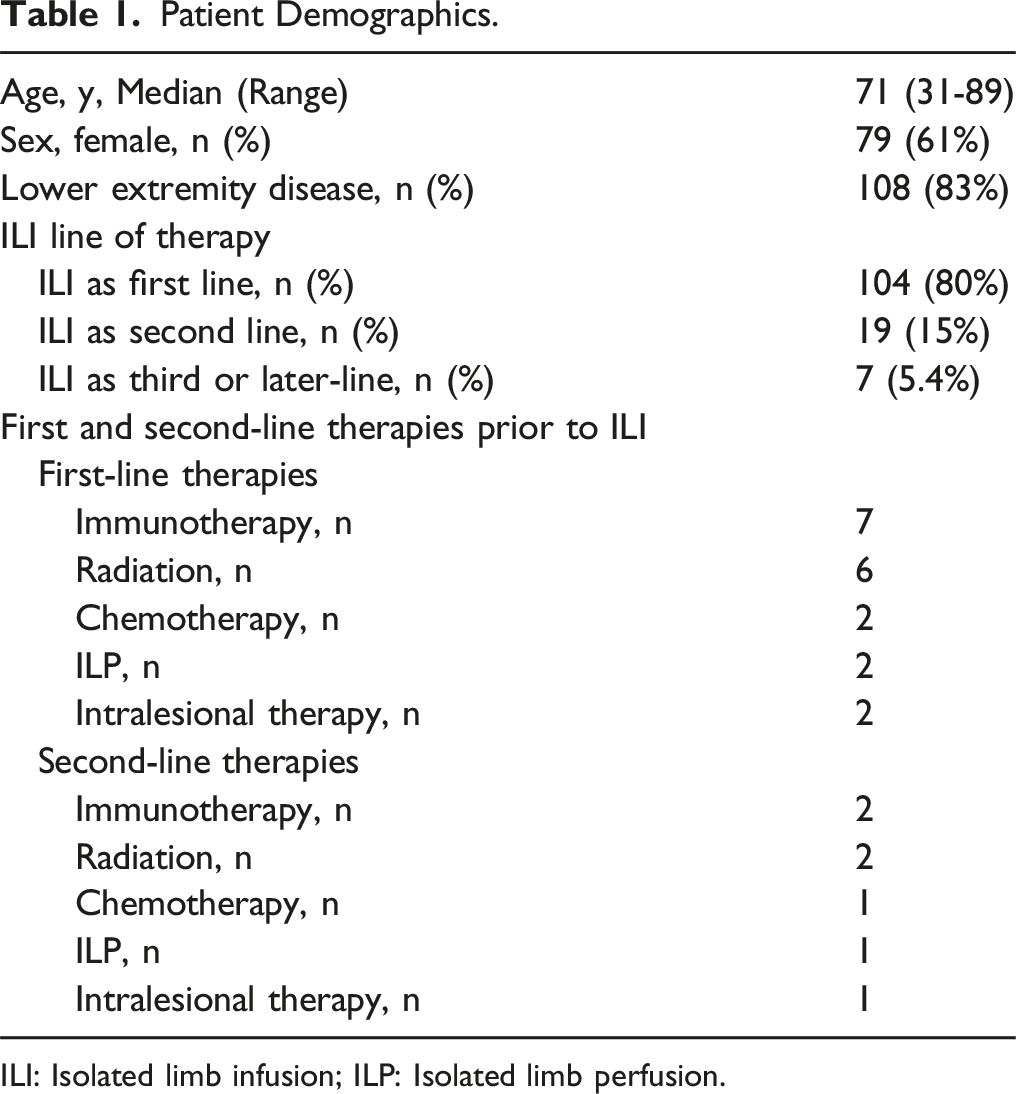
ILI: Isolated limb infusion; ILP: Isolated limb perfusion.
ILI was given as first-line therapy in 80% of patients (n = 104), as second-line therapy in 15% of patients (n = 19), and as third or later-line therapy in 5.4% of patients (n = 7). Of the patients who received ILI as second-line or later therapy, the prior first-line treatments they progressed on included immunotherapy (n = 7), radiation (n = 6), chemotherapy (n = 2), ILP (n = 2), and intralesional therapy (n = (2) were reported. Of the patients who received ILI as third or later-line therapy, the prior second-line treatments they progressed on included immunotherapy (n = 2), chemotherapy (n = 2), ILP (n = 1), radiation (n = 1), and intralesional therapy (n = (1) (Table 1).
Intraoperative Outcomes
Intraoperative Parameters and Postoperative Outcomes After Undergoing ILI for Advanced Extremity Melanoma.

ILI: Isolated limb infusion.
The median length of hospital stay after ILI was 6 days. Two patients (1.5%) experienced longer term side effects in the form of lymphedema 3 months after the ILI, which eventually resolved. The median peak postoperative CPK level was 341 U/L (range 3 to 12 808 U/L) (Table 2).
Disease Response
Best Response After ILI for Extremity Melanoma With In-Transit Metastases.
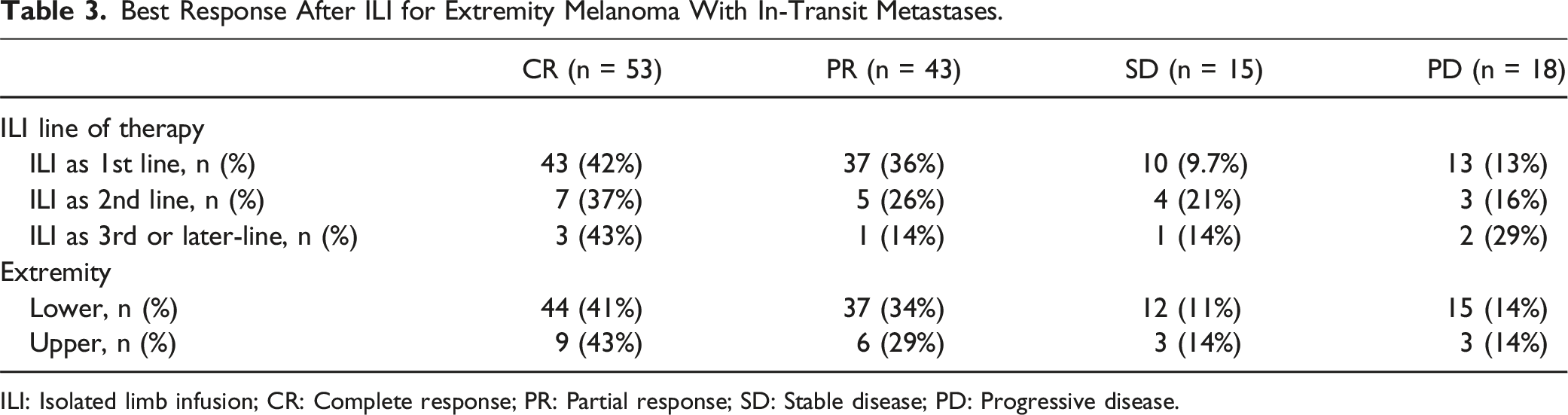
ILI: Isolated limb infusion; CR: Complete response; PR: Partial response; SD: Stable disease; PD: Progressive disease.
Progression and Survival Outcomes
A total of 98 patients (75%) ultimately experienced disease progression (defined as any progression whether in-field or out-of-field) at some point after ILI treatment. One patient required an amputation after ILI due to regional disease progression and development of multiple, symptomatic fungating tumors. At a median follow up of 37.9 months, 37% of patients died of the disease, 18% of patients died of unknown causes, 19% of patients were alive with disease, and 26% of patients had no evidence of disease.
Survival Outcomes After ILI for Extremity Melanoma With In-Transit Metastases.

ILI: Isolated limb infusion; OS: Overall survival; DSS: Disease-specific survival; PFS: Progression-free survival; NR: Not reached.

Survival analyses for in-field progression-free survival (A) and out-of-field progression-free survival (B), based on ILI line of therapy. ILI: Isolated limb infusion.
Median OS for ILI as first-line therapy was 59.6 months, as second-line therapy was 39.3 months, and as third or later-line therapy was 113.5 months. Median DSS for ILI as first-line therapy was not reached, as second-line therapy was 39.3 months, as third or later-line therapy was not reached (Table 4). No statistically significant differences were found in OS or DSS based on ILI line of therapy (although DSS between therapies approached significance) (P = 0.35 and P = 0.07, respectively) (Figure 2). Survival analyses for overall survival (A) and disease-specific survival (B), based on ILI line of therapy. ILI: Isolated limb infusion.
Survival Outcomes by Disease Response
Patients who responded well to ILI, with CR or PR were found to have a significantly longer DSS than those who did not respond well to ILI, with SD or PD (HR 0.48 (95% CI 0.27-0.87); P = 0.015). There were no significant differences between responders (CR or PR) and non-responders (SD or PD) with respect to OS, overall PFS, in-field PFS, or out-of-field PFS.
Discussion
Patients with melanoma who develop ITM have a poor prognosis with low 5-year survival rates. Although several therapy options exist for unresectable ITM, including systemic, intralesional, and locoregional approaches, there is a critical need to determine which line of therapy is optimal for each treatment modality to achieve the best oncologic outcomes. This is the first study to analyze patient outcomes after receiving ILI as either first, second, or third or later-line therapy for unresectable melanoma ITM, to determine which line of therapy may be optimal for ILI.
Although statistical significance was not illustrated, likely due to the low sample size of patients in second-line and third or later-line ILI therapy groups, a clinically meaningful 21% improvement in ORR was apparent in patients who received ILI as a first-line therapy compared those who received ILI as third or later-line therapy, and a 15% improvement was seen between first-line and second-line ILI for ITM. This large percentage difference based on line of therapy (78% ORR in treatment-naiive compared to 57% ORR as third or later-line therapy) is something for patients and physicians to consider in shared decision-making and multidisciplinary discussions to determine first-line treatment strategy for unresectable melanoma ITM. For those who received ILI as third or later-line therapy, the most common therapy modalities that patients progressed on prior to ILI were immunotherapy and radiation therapy. It is possible that these therapies, most notably radiation, lead to alterations in the local vascular environment of the tumor sites, potentially affecting regional chemotherapy delivery to the superficial ITM lesions. At the same time, there is also evidence that concomitant systemic therapy with ILI therapy may show synergistic effects. 23
The survival analyses in this study indicate that ILI appears to be efficacious as first-line, second-line, or third or later-line therapy. Patients with ITM who failed prior therapy lines had favorable DSS and PFS outcomes that were not statistically different compared to those who received upfront ILI. Although there were no statistically significant differences, the absolute differences (numerically and percentage wise) are clinically meaningful, with a greater than 20% ORR difference between first and third or later-line ILI.
This study builds upon our group’s earlier findings, as well as multi-institutional reports, and serves as an updated account of our previous experience. 24 The ORR of 78% and CR rate of 42% for ILI as first-line therapy in this study are generally higher than those found in the literature. Currently, the largest study (n = 687) on long-term outcomes after first-line ILI for stage IIIB and IIIC melanoma found an ORR of 64.1% (CR 28.9%, partial response (PR) 35.2%, stable disease (SD) 14.5%, progressive disease (PD) 19.8%). 15 The difference between this multi-institutional study and the current study could be attributable to the extensive experience with ILI at this institution for almost 20 years and potential bias in choosing the best candidates for the procedure, and/or offering it early in the course/ diagnosis of ITM. Our higher response rates correlated, as expected, with slightly longer survival than previous reports, with a median OS for ILI as first-line therapy of 59.6 months in this study. This is consistent with reports in the literature that patients who respond well to ILI (CR + PR) tend to have longer OS, PFS, in-field PFS, and distant metastatic-free survival and DSS than those who are non-responders (SD + PD).15,25 Although our study did not find any significant differences between responders and non-responders with respect to OS, overall PFS, in-field PFS, or out-of-field PFS, we did find a significantly longer DSS in patients with CR + PR to ILI vs SD + PD. Miura et al found, with a median follow up of 47 months and a median OS of 38.2 months, that patients with CR or PR had a significantly longer median in-field PFS (21.9 vs 3 months, P < .0001), distant PFS (53.6 vs 12.7 months, P < .0001), and OS (46.5 vs 24.4 months, P < .0001) than non-responders (SD + PD). 15
Limitations
Limitations of this study include its retrospective nature. Furthermore, statistical analyses were somewhat limited by low sample size in the second-line and third or later-line therapy groups. Although every effort was made to retrieve updated patient information, some patients were ultimately lost to follow up. Lastly, sometimes the best response was not clearly denoted in the clinical documentation. Despite these limitations, this study contributes valuable insights into the use of ILI in patients with unresectable melanoma ITM.
Conclusion
The majority of patients with melanoma ITM responded to ILI therapy regardless of line of therapy. Although the sample size may have constrained the statistical significance, a clinically meaningful 15%–21% improvement in ORR was noted for patients who received ILI as a first-line therapy compared to second- or third or later-line therapy.
Survival outcomes were not different based on ILI therapy line, indicating the utility of ILI across all treatment lines, as either first-line or salvage therapy after failure of prior therapy modalities can still positively impact response and survival. DSS differences approached significance at P = 0.07. Responders after ILI do much better than non-responders, with significantly longer PFS and OSS. Overall, these results support the consideration of ILI in multidisciplinary discussion and management of melanoma patients with unresectable ITM whether treatment naïve or after failure of other local, regional or systemic therapies, as ILI can salvage some patients and lead to durable repsonses.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Disclosures
Aleena Boby, Dr Michelle M. Dugan, Helana Ghali, Shaliz Aflatooni, Dr Danielle K. DePalo, and Wenyi Fan have no conflicts of interest or financial ties to disclose. Dr Jonathan S. Zager has received payments from Delcath Systems – Medical Advisory Board. He has consulting agreements with Philogen, Merit Medical, Castle Biosciences, and Merck and Replimune. His department receives research funding from Philogen, SWOG, Delcath Systems, and Provectus. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript; or in the decision to publish the results. Dr. Zager holds an US patents for high-flow ILI - US patent numbers -10,583,246 and 11,779,698.
