Abstract
The number of patients with cancer being admitted to intensive care units (ICUs) is increasing worldwide, and these patients are vulnerable to infection. This study aimed to address the long-term impact of positive cultures during admission on 1-year mortality among patients with cancer who received perioperative intensive care. This retrospective cohort study enrolled adult patients with cancer who were admitted to ICUs and received surgery during 2011 to 2016 at a tertiary hospital in central Taiwan. Cancer-related data were retrieved from the cancer registry, and data during ICU admissions were obtained from the electronic medical records. We compared the survival curves between patients with and without positive clinical cultures using log-rank test and used a multivariable Cox proportional hazards regression model to evaluate the influence of positive clinical cultures on 1-year mortality. A total of 638 patients were included for analyses, and 37.9% of them had positive cultures during the index admission. In-hospital mortality was 9.1%, while 1-year mortality was 21.0%. Compared with patients who survived, patients who died were significantly more likely to have positive cultures (59.7% vs 32.1%), to have a higher Acute Physiology and Chronic Health Evaluation II scores (median 21.8 vs 19.0), and to receive mechanical ventilation (86.6% vs 77.4%). Survival analysis found that positive cultures of blood, the respiratory tract, the urinary tract, or the skin and soft tissue were associated with an increased 1-year mortality. Multivariable Cox proportional hazards regression analysis found that positive cultures of blood, the respiratory tract, the urinary tract, or the skin and soft tissue (hazard ratio: 1.621; 95% confidence interval: 1.087-2.419) were significantly associated with 62.1% increased hazards of death within 1 year after the ICU admission. A positive culture during admission was associated with a worsened long-term survival among patients with cancer who received perioperative intensive care. Further studies are needed to confirm this association.
Introduction
Advances in cancer treatments in recent years have led to a steady improvement in survival rates of patients with cancer. 1,2 Physicians in intensive care units (ICUs) are more inclined to consider admitting critically ill patients with cancer worldwide. 3,4 One recent population-based study that used a cancer registry database in the United Kingdom reported that approximately 5% of patients with solid tumors required an ICU admission within 2 years of cancer diagnosis. 5 Another multicenter study in Europe found that nearly 1 in 7 patients admitted to ICUs had a malignant neoplasm. 6
The outcome, including short-term and long-term outcome, of perioperative intensive care is currently a crucial issue in critical care medicine. 7 One 7-day cohort study, investigating 3599 patients receiving perioperative intensive care in 28 European nations, showed the in-hospital mortality was approximately 14%. 8 However, the long-term outcome of patients receiving perioperative intensive care, particularly those having cancer, remains unclear.
Recent studies of long-term outcomes in patients with septic shock, sepsis, bloodstream infection, or pneumonia have found that the infection may substantially worsen patients’ long-term survival 9 -13 or quality of life 9,10 and increase the frequency of cardiovascular events. 11 In patients with cancer requiring the intensive care, few studies, using data of cancer registry, have shown that outcomes, mainly short-term outcomes, were correlated with older age, advanced cancer status, the severity of illness, and the need for mechanical ventilation. 14 -17 Given that the microbiology culture data are often not recorded in cancer registry, studies on patients with cancer rarely evaluated the effect of positive clinical cultures on long-term outcome among critically ill patients with cancer. Therefore, we aimed to address the long-term impact of having positive cultures during admission on 1-year mortality among patients with cancer who received perioperative intensive care by using both data in cancer registry and electronic medical records.
Methods
Ethical Approval
This study was approved by the institutional review board of the Taichung Veterans General Hospital (TCVGH; CE16250A). Informed consent was waived given that this study utilized data that were previously collected in the course of normal care and the patients were not identifiable.
Study Design and Study Population
We conducted this retrospective cohort study at TCVGH, a tertiary care referral hospital with 1514 beds and 6 ICUs in central Taiwan. We included all adult patients who were registered in the TCVGH cancer registry and had at least one ICU admission to an ICU at TCVGH from 2011 through 2016 following their cancer diagnosis. The first ICU admission following cancer diagnosis was defined as the index ICU admission and included in the analysis. We excluded patients whose discharge status for the index admission was unknown and those without surgery. After applying the inclusion and exclusion criteria, we included 638 patients as our study population (Figure 1).

Selection process of the study population.
Data Source
We used 2 data sources: the clinical data warehouse and the cancer registry. Data regarding demographic information, ICU admission, discharge diagnoses, metastasis, surgical history, Acute Physiology and Chronic Health Evaluation (APACHE) II score, clinical culture results, mechanical ventilation usage, renal replacement therapy, antibiotic use, and length of hospital stay were retrieved from the TCVGH clinical data warehouse. Data related to cancer diagnosis, cancer type, and cancer treatment were pulled from the cancer registry of TCVGH. The TCVGH is one of the participating hospitals of the Taiwan Cancer Registry, a population-based cancer registry that is organized and funded by the Ministry of Health and Welfare in Taiwan since 1979. 18
Microbiology Clinical Cultures
The main exposure of interest was at least one culture of samples obtained during the index hospital admission that was positive. The results of all clinical cultures of blood, the respiratory tract (eg, tracheal aspirate, bronchoalveolar lavage fluid, pleural effusion, or expectorated sputum), the urinary tract (ie, midstream urine, urinary catheter, or percutaneous nephrostomy), skin and soft tissue (ie, wound, pus, or discharge), or the abdomen (ie, ascites, bile, or peritoneal drainage) during the index admission were retrieved. We performed subgroup analysis of patients who had positive clinical cultures for gram-positive cocci (GPC), gram-negative bacilli (GNB), or Candida.
Covariates
Comorbidities (Charlson comorbidity index), the presence of metastasis, acute respiratory failure, acute cardiac conditions, acute neurological conditions, pulmonary embolism, and acute renal conditions were defined by the International Classification of Diseases, Ninth Revision, Clinical Modification (ICD-9-CM) or ICD-10-CM discharge codes for the index admission. Mechanical ventilation was defined by the insurance claims for mechanical ventilation during ICU admission. Renal replacement therapy was defined by the physician’s order for hemodialysis during hospital admission. Antibiotic usage was defined as any oral or intravenous administration of antibiotics during the index hospital admission.
Mortality
The main outcome of interest was the mortality within 1 year following the index ICU admission. The date of death was obtained from the cancer registry. If the patient did not have a date of death, he/she was censored at the last contact date.
Statistical Analysis
Data for categorical variables were shown as frequencies and percentages and data for continuous variables were presented as median and interquartile range (IQR). For univariable analyses, we used the Wilcoxon rank sum test for continuous variables and the χ2 test for categorical variables to compare the difference between patients with cancer who survived for at least 1 year after ICU admission (survivors) and those who died within 1 year of ICU admission (nonsurvivors). We used survival analysis to compare the 1-year survival between patients who had any positive clinical cultures during the index ICU admissions and patients who did not have any positive clinical cultures during their index ICU admissions. Patients whose follow-up times were shorter than 1 year were treated as censored observations. We assessed the Kaplan-Meier survival curves and compared survival between patients with positive clinical cultures and patients without positive clinical cultures using the log-rank test. We obtained the unadjusted hazard ratio (HR) for each specific culture site and specific pathogens from separate univariable Cox proportional hazards regression models. We included variables that were clinically relevant or statistically significant in a multivariable Cox proportional hazards regression model to evaluate the influence of positive clinical cultures on 1-year mortality while adjusting for other covariates. We confirmed that the Cox proportional hazards model met the proportional hazards assumption. The HR and the corresponding 95% confidence interval (CI) for each variable were presented. All statistical analyses were 2 sided, and the level of significance was .05. Data cleaning and analysis were performed using SPSS software version 22.0 (SPSS Inc, Chicago, Illinois) and SAS program version 9.4 (SAS Institute Inc, Cary, North Carolina).
Results
Demographic and Baseline Characteristics
Of the 638 patients included in our study, 77.3% were male, and the median age was 60.4 years (IQR: 52.1-71.8 years; Table 1). Major comorbidities included type II diabetes mellitus (21.6%), chronic obstructive pulmonary disease (16.0%), and cerebrovascular disease (14.6%). The most common cancer type was gastroesophageal cancer (28.4%), followed by head and neck cancer (19.6%), brain cancer (14.6%), and colon cancer (11.8%). Given that TCVGH is a referral hospital that often admits patients with advanced conditions, up to 54.9% of the patients had metastatic cancer, 87.1% had undergone surgical interventions to treat cancer, and 62.5% had undergone chemotherapy. Notably, the in-hospital mortality rate was 9.1 (58/638) and the mortality within 1 year after the index ICU admission was 36.8% (134/638). The post-acute 1-year mortality rate among patients who survived the index ICU admission was 13.1% (76/580).
Characteristics of the 638 Patients With Cancer Receiving a Surgery With ICU Admission Categorized by 1-Year Survival.
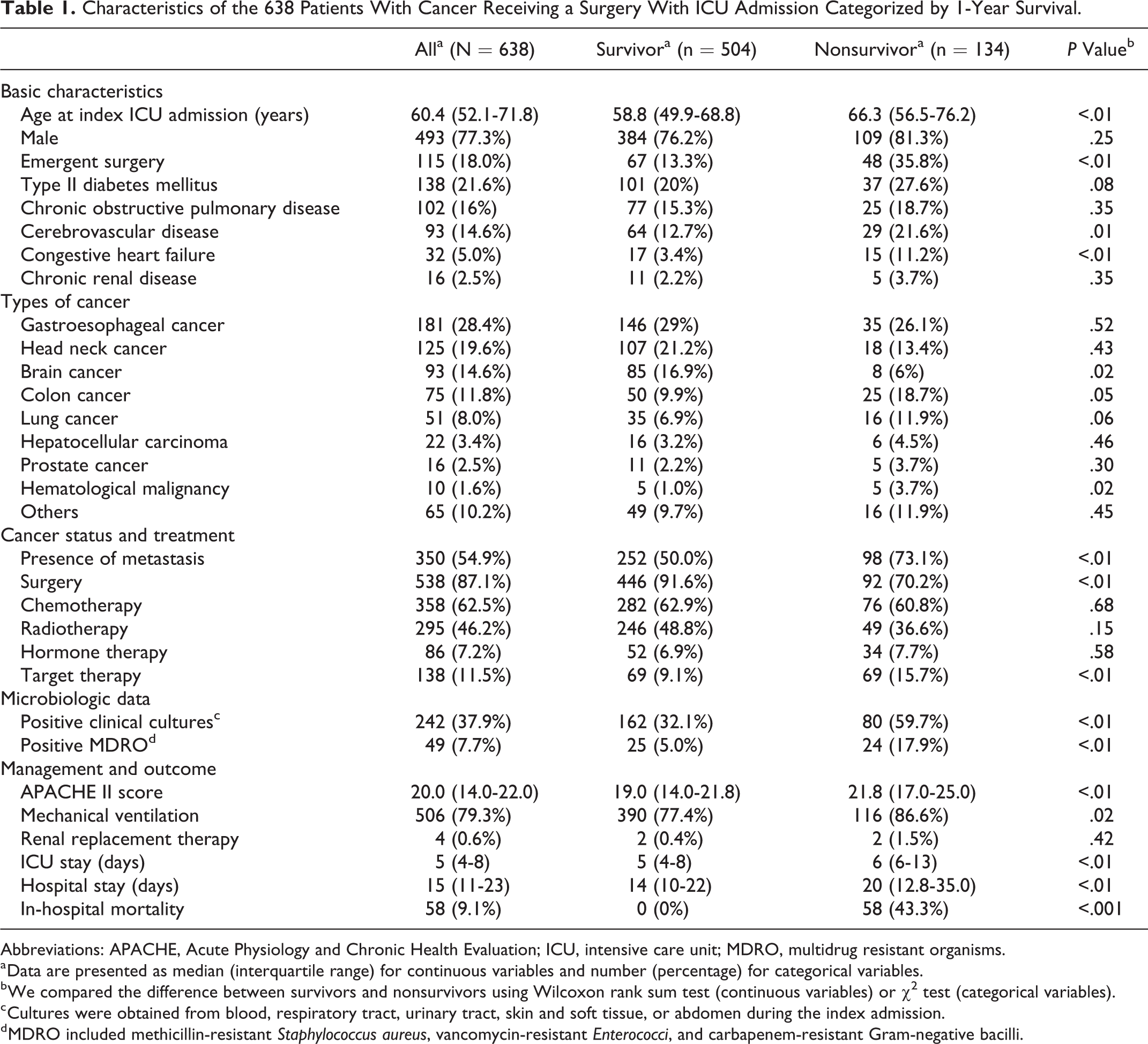
Abbreviations: APACHE, Acute Physiology and Chronic Health Evaluation; ICU, intensive care unit; MDRO, multidrug resistant organisms.
a Data are presented as median (interquartile range) for continuous variables and number (percentage) for categorical variables.
b We compared the difference between survivors and nonsurvivors using Wilcoxon rank sum test (continuous variables) or χ2 test (categorical variables).
c Cultures were obtained from blood, respiratory tract, urinary tract, skin and soft tissue, or abdomen during the index admission.
d MDRO included methicillin-resistant Staphylococcus aureus, vancomycin-resistant Enterococci, and carbapenem-resistant Gram-negative bacilli.
Of the study population, 504 patients survived (survivors) and 134 patients died (nonsurvivors) within 1 year following the index ICU admission. Compared with survivors, nonsurvivors were significantly more likely to have positive cultures during the index admission (59.7% vs 32.1%), to be older (median 66.3 vs 58.8), to have metastasis (73.1% vs 50.0%), to have a higher APACHE II score (median 21.8 vs 19.0), to receive mechanical ventilation (86.6% vs 77.4%), and to have a positive culture for multidrug resistant organisms (MDROs) including methicillin-resistant Staphylococcus aureus (MRSA), vancomycin-resistant Enterococci, and carbapenem-resistant GNB (17.9% vs 5.0%; Table 1).
Types of Common Pathogens
Of 638 included patients, 242 (37.9%) had at least 1 positive culture during the index admission, which compromised 36 (5.6%) blood cultures, 153 (24.0%) respiratory tract cultures, 56 (8.8%) urinary tract cultures, 114 (17.9%) skin and soft tissue cultures, and 33 (5.2%) cultures of tissue or fluid from the abdomen. Gram-negative bacilli were the most common organism found in blood cultures (69.4%), respiratory tract cultures (96.1%), urinary tract cultures (66.1%), and skin and soft tissue cultures (88.6%; Table 2).
Pathogens Identified in the Cultures of 242 Patients With Cancer During Their Index Admission.

Abbreviation: GNB, gram-negative bacilli.
Positive Clinical Cultures and 1-Year Survival
We found that positive clinical cultures from blood (unadjusted HR: 4.113; 95% CI: 2.519-6.716; P < .01), the respiratory tract (unadjusted HR: 2.802; 95% CI: 1.986-3.953; P < .01), the urinary tract (unadjusted HR: 3.453; 95% CI: 2.259-5.277; P < .01), and the skin and soft tissue (unadjusted HR: 1.584; 95% CI: 1.069-2.347; P = .02) were associated with worse 1-year mortality, whereas positive clinical cultures from the abdomen (unadjusted HR: 1.391; 95% CI: 0.681-2.844; P = .37) were not associated with decreased survival (Figure 2).
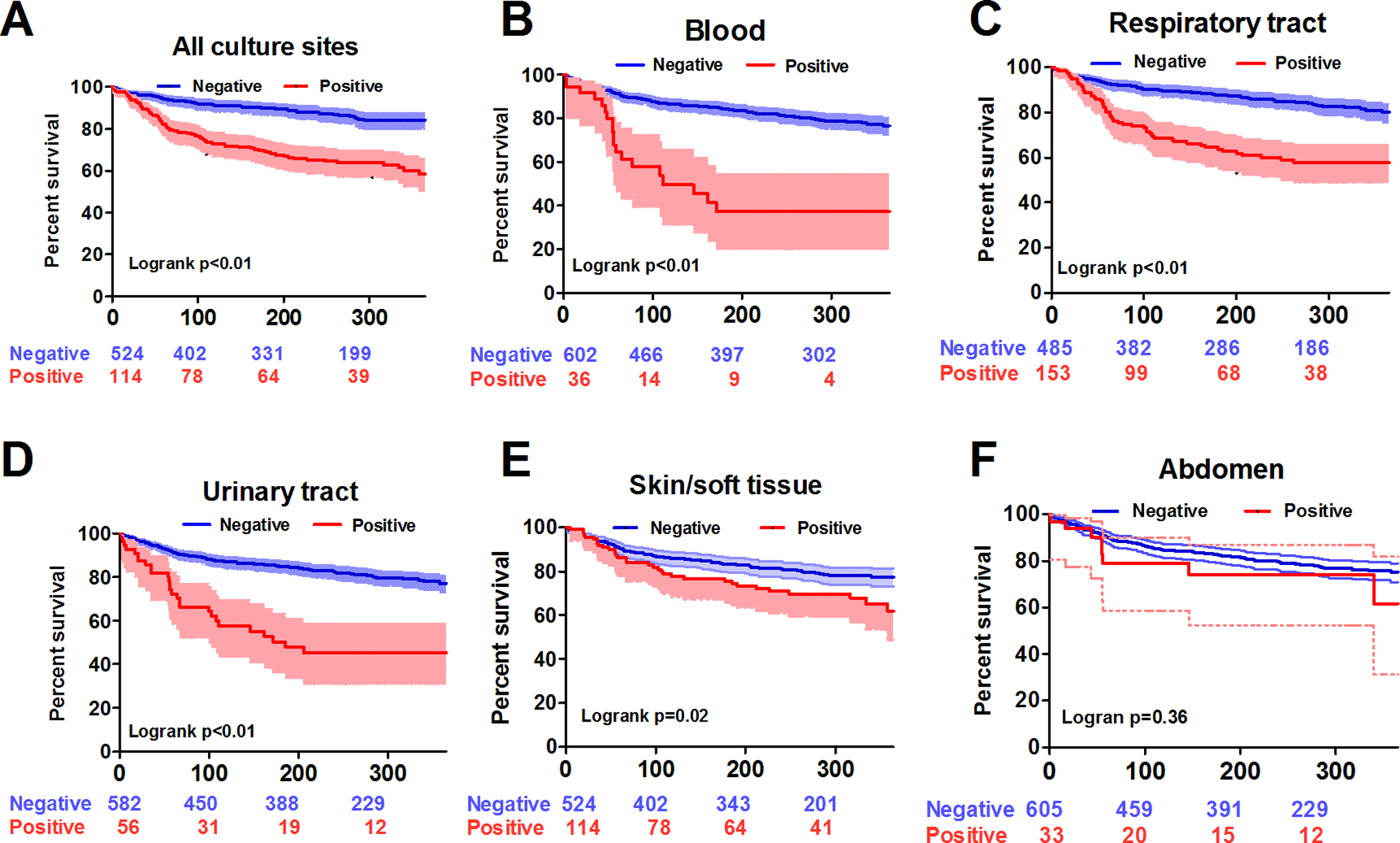
Kaplan-Meier survival curves for patients with and without positive cultures categorized by culture sites. All culture sites (A), blood (B), respiratory tract (C), urinary tract (D), skin or soft tissue (E), abdomen (F).
To further assess the association of positive clinical cultures with 1-year mortality, we included results of cultures from blood, the respiratory tract, the urinary tract, or the skin and soft tissue as one of the predictors in a multivariable Cox proportional hazards model (Table 3). We found that positive clinical cultures from blood, the respiratory tract, the urinary tract, or the skin and soft tissue (adjusted HR [aHR]: 1.621; 95% CI: 1.087-2.419) were significantly associated with a 62.1% increased hazard of 1-year mortality, after adjusting for age (aHR: 1.017; 95% CI: 1.001-1.032), male gender (aHR: 1.622; 95% CI: 1.038-2.535), having congestive heart failure (aHR: 2.074; 95% CI: 1.161-3.704), presence of metastases (aHR: 2.293; 95% CI: 1.552-3.387), emergent surgery (aHR: 1.992; 95% CI: 1.305-3.041), and APACHE II score (aHR: 1.047; 95% CI: 1.015-1.079).
Cox Proportional Hazards Regression for 1-Year Mortality.

Abbreviations: APACHE, acute physiology and chronic health evaluation; CI, confidence interval; HR, hazard ratio; MDRO, multidrug resistant organisms.
Types of Common Pathogens and 1-Year Survival
Patients who had positive clinical cultures for GPC (unadjusted HR: 2.130; 95% CI: 1.279-3.549; P < .01), GNB (unadjusted HR: 2.562; 95% CI: 1.822-3.603; P < .01), or Candida or yeast (unadjusted HR: 4.491; 95% CI: 2.981-6.766; P < .01) had significantly worse 1-year survival, compared with patients without positive clinical cultures (Figure 3). The presence of an MDRO was associated with 1-year mortality in the univariate analysis (unadjusted HR: 3.282; 95% CI: 2.451-5.979). However, after adjusting for other factors in the multivariable Cox proportional hazards analysis, the presence of an MDRO was no longer significantly associated with 1-year mortality (aHR: 1.407; 95% CI: 0.846-2.338). Furthermore, the magnitude of difference appeared to be the strongest for Candida or yeast, which was independently associated with 1-year mortality among the 242 patients with positive cultures (aHR: 1.828; 95% CI: 1.070-3.124; Supplemental Table 1).

Kaplan-Meier survival curves for patients with and without 1 of the 3 major pathogens. Gram-positive cocci (A), gram-negative bacilli (B), and candida/yeast (C).
Discussion
This study investigated the association between positive cultures during the admission and the 1-year mortality following an ICU admission among patients with cancer receiving perioperative intensive care. We found that having positive cultures in blood, respiratory tract, urinary tract, or skin and soft tissue (aHR: 1.621) was independently associated with increased hazards of 1-year mortality following ICU admission. Our results demonstrate the potential impact of positive cultures and indicate the need for vigilance for the long-term impact of positive culture among patients with cancer who were admitted to the ICU for perioperative care.
Understanding both the short-term and long-term outcomes is crucial in critically ill patients. For example, studies of sepsis survivors revealed that sepsis has a long-term influence on cognitive and functional impairments, inflammation and immune dysfunction, worsened quality of life, and increased mortality. 19 Shankar-Hari et al conducted a systematic review of 43 studies that evaluated the 1-year post-acute mortality in adult patients who survived their index sepsis admission. The authors found that the pooled 1-year post-acute mortality was 16.1% (95% CI: 14.1%-18.1%). 20 Currently, the biological evidence for a causal link between sepsis and long-term mortality is not well understood. 20 Sepsis is currently defined as life-threatening organ dysfunction caused by a dysregulated host immune response to infection. 21 It is likely that post-acute immunosuppressive responses and frailty resulting from the residual effects of sepsis could lead to new infection or recurrent infection in sepsis survivors and thus result in an additional long-term risk of death, readmissions, cardiovascular disease, cognitive impairment, and poor quality of life. 22,23
Increasing number of critically ill patients with cancer are now being admitted to ICUs because the new treatments for cancer and advances in intensive care of critically ill patients with cancer have improved their prognosis and survival. 4 One cohort study conducted in 24 European countries found that ICU mortality (20% vs 18%) and in-hospital mortality (27% vs 23%) rates were similar between patients with a solid tumor and those without cancer. 6 Similarly, the in-hospital mortality was 9.1% in this study and was similar to those reported in studies regarding in-hospital mortality of perioperative intensive care in the general population. 8,24 Furthermore, we also investigated the issue of post-acute mortality in this study. Currently, only a few studies reported the post-acute mortality in critically ill patients with cancer. Fisher et al found that the 180-day post-acute mortality was 21% in 300 patients with solid tumors. 17 Auclin et al reported that the 90-day post-acute mortality was 18.3% in 262 elderly patients with solid tumors. 25 In our study, the post-acute 1-year mortality (13.1%, 76/580) was relatively lower than those in other studies, possibly because that our study population was younger (median age: 60.4 years) than those in Fisher et al’s study (median age: 66.5 years) 17 or Auclin et al’s study (mean age: 75.2 years). 25
Our study is unique because we illustrated that having positive clinical cultures was associated with worse 1-year survival in critically ill patients with cancer receiving surgery. Although we could not directly distinguish colonization from infections, it is reasonable to postulate that the positive culture among the 242 patients in this study likely resulted from infection given that 99.6% (241/242) of the patients received antimicrobial agents, and the durations of using antibacterial and antifungal agents use were 21.9 (14.5-41.0) days and 13.9 (3.8-25.5) days, respectively. Furthermore, in addition to the positive association between positive culture of all sites with 1-year mortality, the positive culture of all sites was also highly correlated with ICU stay (β: 3.913; 95% CI: 2.439-5.387) and hospital stay (β: 12.924; 95% CI: 10.595-15.253) after adjusting for relevant variables. Taken together, the aforementioned evidence indicates that the presence of a positive culture is most likely due to an infection, rather than colonization. Additionally, several studies have shown that antimicrobial-resistant organisms were associated with an increased risk of subsequent infection and worse outcomes. One retrospective matched cohort study of ICU patients found that vancomycin-resistant Enterococcus colonization was associated with a 2.35-fold increase in in-hospital mortality, a 1.22-fold increase in ICU costs, and a 1.25-fold longer hospitalization. 26 In patients with cancer, Hess et al found that prior positive culture with any antibiotic-resistant GNB was associated with antibiotic-resistant GNB bacteremia. 27 Another prospective study of ICU patients showed that colonization with either methicillin-susceptible Staphylococcus aureus (aHR: 2.47; 95% CI: 1.52-4.01) or MRSA (aHR: 4.70; 95% CI: 3.07-7.21) had an increased risk of subsequent ICU-acquired Staphylococcus aureus infections. 28 Herein, the presence of MDRO was not significantly associated with 1-year mortality (aHR: 1.407; 95% CI: 0.846-2.338) after adjusting for relevant variables, and more studies are hence warranted to clarify the long-term impact of drug-resistant organisms among patients with cancer receiving perioperative intensive care in the future.
It is interesting that culture sites have distinct impacts on long-term outcomes. Prior research has indicated that having positive cultures from certain culture sites could be correlated with worse long-term outcomes in patient with sepsis. 29 Therefore, we evaluated the culture site-specific impacts on 1-year survival in this study. We found that 1-year mortality was highly associated with positive cultures from blood, respiratory tract, or urinary tract, slightly associated with positive cultures from the skin and soft tissue, but not associated with positive cultures from the abdomen. Recently, the European Centre for Disease Control investigated the attributable mortality of health-care-associated infections in ICUs, mainly medical ICUs (62.7%), in Europe and found that the attributable mortality rate of pneumonia and bloodstream infection was 3.5% and 5.0%, respectively, whereas urinary tract infection (UTI) had no impact on mortality in matched cohort analysis. 30 In the present study, we found a positive culture of urinary tract was associated with a higher 1-year mortality rate in patients receiving the surgery, but not in those without the surgery (Figure 2D and Supplemental Figure 1D). As shown in Supplemental Figure 1D, critically ill patients with cancer admitted for medical reasons had apparently high mortality regardless of their culture status in the urinary tracts; therefore, the impact of positive culture in the urinary tract on mortality only existed in participants receiving the surgery because there was a relatively low mortality rate in participants without a positive culture (Figure 2D). Thus, we postulated that the distinct patient populations might account for the varying impacts of UTI on mortality in critically ill patients. Interestingly, one recent systematic review included 19 studies to address the mortality correlated with infection sites among adult patients with sepsis. 29 The infection sites that have been studied the most were respiratory, abdominal, genitourinary, and skin/soft tissue infections. The authors summarized the findings from prior studies and showed that respiratory infection appeared to have the highest risk of hospital mortality (range of the adjusted odds ratio [aOR] from included studies: 0.78-2.23), followed by abdominal infections (0.60-3.92), genitourinary infections (0.05-1.17), and skin or soft tissue infections (0.42-1.38). 29 Furthermore, He et al found that sepsis survivors who had pulmonary sepsis had significantly worse 1-year mortality (aOR: 2.547; 95% CI: 1.513-4.288; P < .01) and quality of life (aOR: 2.846; 95% CI: 1.530-5.294; P = .004), compared with those who had abdominal sepsis. 31 In this study, positive cultures of the abdomen were mainly (81.8%, 27/33) from fluid obtained during the surgery, which wound have prompted early control of the infectious source. We hence postulated that the early control of infection might explain the lack of association between the positive culture in the abdomen and 1-year mortality rate among patients with cancer who were admitted to the ICU for perioperative care.
Our study has few limitations. First, this is a retrospective study using secondary data that have been already collected for clinical purposes. We combined 2 databases, the cancer registry and the electronic medical records data, to complement each other. The cancer registry in TCVGH routinely collected cancer-related information and outcome using the standardized National Cancer Registry System in Taiwan. 18 In addition, patients admitted to ICUs at the TCVGH were routinely sampled for microbiology culture. Therefore, the information bias of the cancer-related data and the sampling bias of the clinical culture results should have been minimized. Second, the generalizability of our findings to critically ill patients with cancer without a surgery could be limited; however, we found a similar pattern among the 558 patients without a surgery (Supplemental Figure 1). Third, we could not evaluate cause of death, the changes in patients’ performance status, and quality of life before and after ICU admission because the Cancer Registry and medical records did not contain these information. It will be interesting to evaluate the influence of positive clinical cultures in patients’ functional status and activity of daily living. Lastly, the study results from this single-center study may not be generalizable to other health-care settings.
Conclusions
The short-term mortality of critically ill patients with cancer who were admitted to ICUs has been largely improved due to the advances in cancer management and critical care, whereas the long-term mortality of these patients with cancer, particularly those who received perioperative intensive care, remains obscure. Our study found that having positive clinical cultures from blood, respiratory tract, urinary tract, or skin and soft tissue during admission was associated with 62.1% increased hazard of 1-year mortality. Our findings point out the impact of microbiology in the long-term outcome that has been previously omitted among patients with cancer who were admitted to ICU for perioperative management. Robust prospective studies that collect clinical samples from patients at ICU admissions, assess their quality of life, and follow them in a long term after discharge are warranted to confirm the association between positive clinical cultures and long-term outcomes, as well as to explore the mechanisms underlying the post-acute mortality in critically ill patients with cancer.
Supplemental Material
Supplemental_figure_1 - Association Between Positive Cultures During Admission and 1-Year Mortality in Patients With Cancer Receiving Perioperative Intensive Care
Supplemental_figure_1 for Association Between Positive Cultures During Admission and 1-Year Mortality in Patients With Cancer Receiving Perioperative Intensive Care by Hsiu-Yin Chiang, Tung-Han Wu, Chiann-Yi Hsu, and Wen-Cheng Chao in Cancer Control
Supplemental Material
Supplemental_table_1 - Association Between Positive Cultures During Admission and 1-Year Mortality in Patients With Cancer Receiving Perioperative Intensive Care
Supplemental_table_1 for Association Between Positive Cultures During Admission and 1-Year Mortality in Patients With Cancer Receiving Perioperative Intensive Care by Hsiu-Yin Chiang, Tung-Han Wu, Chiann-Yi Hsu, and Wen-Cheng Chao in Cancer Control
Supplemental Material
Supplemental_information - Association Between Positive Cultures During Admission and 1-Year Mortality in Patients With Cancer Receiving Perioperative Intensive Care
Supplemental_information for Association Between Positive Cultures During Admission and 1-Year Mortality in Patients With Cancer Receiving Perioperative Intensive Care by Hsiu-Yin Chiang, Tung-Han Wu, Chiann-Yi Hsu, and Wen-Cheng Chao in Cancer Control
Footnotes
Authors’ Note
H-.Y.C., T-.H.W., and W-.C.C. contributed to study concept and design. H-.Y.C., C-.Y.H., and W-.C.C. contributed to acquisition of data. H-.Y.C. and W-.C.C. contributed to analysis and interpretation of data. H-.Y.C. and W-.C.C. drafted the manuscript. Ethics approval was obtained from Taichung Veterans General Hospital CE16093A.
Acknowledgments
The authors thank the Clinical Informatics Research & Development Center and the Cancer Registry database of Taichung Veterans General Hospital for the support of clinical data acquisition (Registered number F16317). The authors also thank Dr Loreen Herwaldt in the Department of Internal Medicine, University of Iowa Carver College of Medicine for reviewing this manuscript and providing valuable suggestions. The interpretation and conclusions contained herein do not represent those of Taichung Veterans General Hospital.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
