Abstract
After reviewing cognitive safety in healthcare, effectiveness of BrainTagger cognitive assessment games in tracking age-related cognitive changes is evaluated. One hundred eighty-three participants, aged 4 to 92, each played up to four games designed to assess cognitive speed, response inhibition, cognitive control, and working memory. Participants were recruited during two public events in Toronto: AGE-TECH Innovation Week (October 24–27, 2023) and the National Home Show (March 8–17, 2024). Piecewise linear regression analyzed game performance across three life stages: development (0–25 years); peak performance (26–50 years); age-related decline (51+ years). All four games demonstrated expected trends, with performance increasing in early years, stabilizing during middle age, and declining after age 50. BrainTagger are shown to track age-related cognitive decline. Future applications in clinical settings may enhance early identification of cognitive harm caused by medical interventions or neglect, contributing to improved cognitive safety in healthcare environments.
Introduction
Physical safety has been a major focus in preventing adverse events in healthcare. In contrast there has been relatively little interest in cognitive safety, likely because popular clinical methods of cognitive assessment are paper based and relatively insensitive. Cognitive assessment games promise to be much more sensitive and easier to administer repeatedly than traditional paper and pencil tests. Games that have been shown to be psychometrically valid in measuring targeted cognitive functions such as working memory and executive functioning now need to be applied, and thoroughly validated, in clinical settings. In this article, we review some of the cognitive harms that occur in healthcare and present novel results demonstrating that BrainTagger game performance are sensitive to age-related changes in cognitive status.
Background
Cognitive safety (i.e., preservation of the cognitive status of each person to the maximum level possible given their physiology, brain state, and age) is a significant component of overall patient safety. Cognitive safety should be a key part of conserving “the functional ability that enables well-being in older age” (Clegg et al., 2013). There are many lifestyle factors that contribute to risk of dementia and cognitive impairment in general (Livingston et al., 2020), but the role of medical interventions in contributing to risk of cognitive impairment and dementia is not well understood. Here we review some of the medical interventions that have been found to pose threats to cognitive safety and that may contribute, along with lifestyle factors, to acceleration of age-related cognitive impairment.
Cognitive impairment frequently occurs after surgery, a phenomenon referred to as postoperative cognitive dysfunction (POCD). POCD following surgery and general anesthesia is especially common in elderly patients (Li et al., 2022). Patients with POCD require longer hospitalization and have difficulty returning to their homes (van Harten et al., 2012; Wang et al., 2014), likely due to POCD-related deficits in activities of daily living (Indja et al., 2017).
Cognitive impairment is common in people with chronic kidney disease (Berger et al., 2016). Initiation of hemodialysis is associated with an accelerated decline of cognitive function and an increased incidence of cerebrovascular accidents and white matter lesions (Polinder-Bos et al., 2018). Cognitive impairment increases in prevalence with declining kidney function, being up to three times more common at the stage where kidney replacement therapy is required (Crowe et al., 2021).
Chemotherapy in cancer patients can cause a decline in short-term cognitive ability (Koppelmans et al., 2012; Wefel et al., 2010). This decline is referred to as chemotherapy induced cognitive impairment (CICI). With relatively few longitudinal studies involving long-term cognitive effects of chemotherapy, most evidence of long-term deficits involves subjective reports of cognitive impairment (Amidi et al., 2015). Schagen et al. (2022) noted that “neuropsychological monitoring is not yet standard practice in most cancer centers, clinicians frequently learn of potential cognitive dysfunction through patient self-reports in clinics or on questionnaires.”
Radiotherapy for brain cancer seems to be particularly harmful, with neurological side effects of cranial irradiation being reported to occur in 50% to 90% of adult patients (Greene-Schloesser et al., 2012).
Intensive Care Unit (ICU) stays are also hazardous for cognitive health. Pandharipande et al. (2014) examined the impact of respiratory failure or shock in the ICU and found that 6% of the 821 patients enrolled in the study had cognitive impairment at baseline, while 26% and 24% of patients had cognitive scores associated with mild dementia after 3 months, and 12 months, respectively. In a systematic review of cognitive impairment in the ICU, most of the studies reviewed suggested that critical illness and ICU treatment are associated with long-term cognitive impairment (Wolters et al., 2013).
Delirium is an acute decline of attention and cognitive function (Inouye & Ferrucci, 2006) that frequently occurs in older patients who are under care. Poulsen et al. (2021) cited a systematic review, based on 48 studies, that found a pooled prevalence of delirium in the ICU to be 31%. In a systematic review (Yan et al., 2023), the incidence of postoperative delirium was found to be 19% after elective surgery and 32% after emergency surgery.
Delirium affects 10% of older emergency department (ED) patients and is often unrecognized (Hustey & Meldon, 2002). Patients discharged with unrecognized delirium have greatly increased risk of near-term mortality compared to non-delirious patients (Kakuma et al., 2003). Patients in the emergency room typically do not receive cognitive assessments, but clinical judgment cannot be relied upon to make up for missing cognitive assessments. In their study of delirium, Lee et al. (2022) found that ED nurses missed delirium in 43/78 cases and physicians missed delirium in 10/20 cases and even considered two of those cases safe to discharge. Delirium is a major threat to cognitive safety. As Fong and Inouye (2022) noted: “people with dementia have a higher risk of developing delirium than the general population, and the occurrence of delirium is an independent risk factor for subsequent development of dementia”.
Commonly used tests in the clinical setting include the Mini-Mental State Examination (MMSE; Folstein et al., 1975), which is typically used to screen for dementia, the Montreal Cognitive Assessment (Nasreddine et al., 2005) used to screen for mild cognitive impairment, and the confusion assessment method (CAM) used to assess for delirium. Traditional clinical tests are often paper-based and require trained administrators, making them less repeatable and engaging for participants. Due to their relatively high cost in terms of effort, and limited ability to track subtle cognitive changes, these tests are unsuitable for monitoring ongoing changes in cognitive status that may indicate cognitive harm. Game-based cognitive assessments offer numerous advantages, including reduced learning effects and self-administration potential. Tong et al. (2016) developed a Whackamole game which showed good results in screening for delirium in the emergency room (Lee et al., 2019). That work demonstrated how cognitive assessment games can be used to monitor cognitive health.
Cognitive decline in older people occurs against a backdrop of age-related declines in cognitive abilities, particularly with respect to fluid intelligence (Cattell, 1963; Samu et al., 2017). Fluid intelligence involves the ability to solve novel, complex problems and is closely associated with cognitive speed, executive functions and working memory. We have extended the original Whackamole game that was used in screening for delirium (Tong et al., 2016), into a suite of cognitive assessment games referred to as BrainTagger. Each game targets an aspect of fluid intelligence that corresponds to a standard psychological task. For instance, TAG-ME Only is a BrainTagger game that has been shown to be highly correlated with the Go/No-Go task for response inhibition (Tong et al., 2019). Similarly, TAG-ME Again is a BrainTagger game that has been validated against the N-Back task for working memory (Hu et al., 2023).
Method
Participants and Recruitment
A total of 183 participants (M = 44.87 years, SD = 20.47; range = 4–92 years) were recruited at AgeTech Innovation Week (October 2023) and the National Home Show (March 2024) in Toronto. All participants consented to play at least one BrainTagger game. This study was approved by the Research Ethics Board at the University of Toronto (Protocol #46000).
Game Suite
TAG-ME quick: Measures cognitive speed via response latency to mole appearances.
TAG-ME only: Assesses response inhibition (a Go/No-Go task using moles with hats as distractors).
TAG-ME bigger: A numerical Stroop task measuring cognitive control.
TAG-ME again easy: A 1-back memory task assessing working memory through number recall.
Procedure
Games were played on a tablet with a six-button custom input device, as shown in Figure 1. Each game consisted of 30 trials. For each participant the four games were played in the order shown above and participants could stop playing the games whenever they wanted. Reaction times, accuracy, false alarms, and other response types were logged automatically. Trials used randomized visual stimuli and the character (mole) appearance time in the cognitive speed and response inhibition games was adaptive so that moles disappeared faster when participants were hitting the moles faster and disappeared more slowly when participants were having trouble hitting the moles before they disappeared.

BrainTagger button box and tablet on adjustable height table (The BrainTagger Kiosk).
Participants entered age and sex before gameplay. They could choose how many games to play and were free to withdraw at any time.
Statistical Analysis
Piecewise linear regressions were applied to assess performance across three age ranges:
Development (0–25 years).
Plateau (26–50 years).
Decline (51+ years).
Readers who are concerned about family wise alpha levels can apply Bonferroni correction across the 12 regression models by using an adjusted alpha value of .05/12, that is, .004. However, in our view this would be overly conservative and collection of more data to support the present results would be preferable.
Result
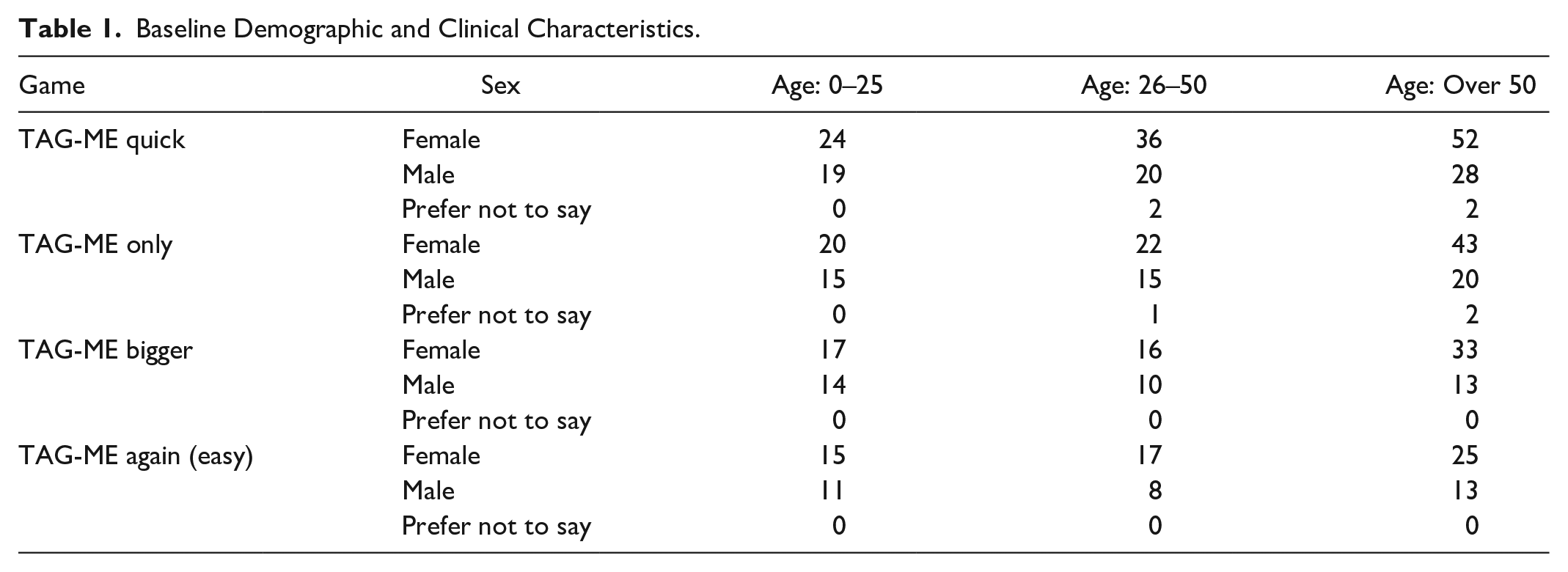
Table 1 shows baseline demographic and clinical characteristics for each game and age group. There were more female than male participants for all three age groups.
Baseline Demographic and Clinical Characteristics.
Age Effects on Performance
Performance trends followed expected U-shaped developmental curves (Cattell, 1963; Samu et al., 2017).
TAG-ME quick: As shown in Figure 2, cognitive speed improved during development (r = −.25), plateaued in midlife (r = .05), and declined significantly in older age (r = .32).
TAG-ME only: As shown in Figure 3, response inhibition, as measured by correct reaction time in the Go/No-Go task, improved during development (r = .48) and declined after age 50 (r = .39).
TAG-ME bigger: Cognitive control followed a similar trend, with a marginal effect during development (p = .055) and a near-significant decline in older age (p = .07).
TAG-ME again easy: As shown in Figure 4, working memory, measured by d’ (d-prime), showed a strong correlation with age in the younger group (r = .70). However, relationships in the older age groups were not statistically significant (p > .30 in both cases).
Most participants completed 2 to 4 games, indicating good engagement with the games.
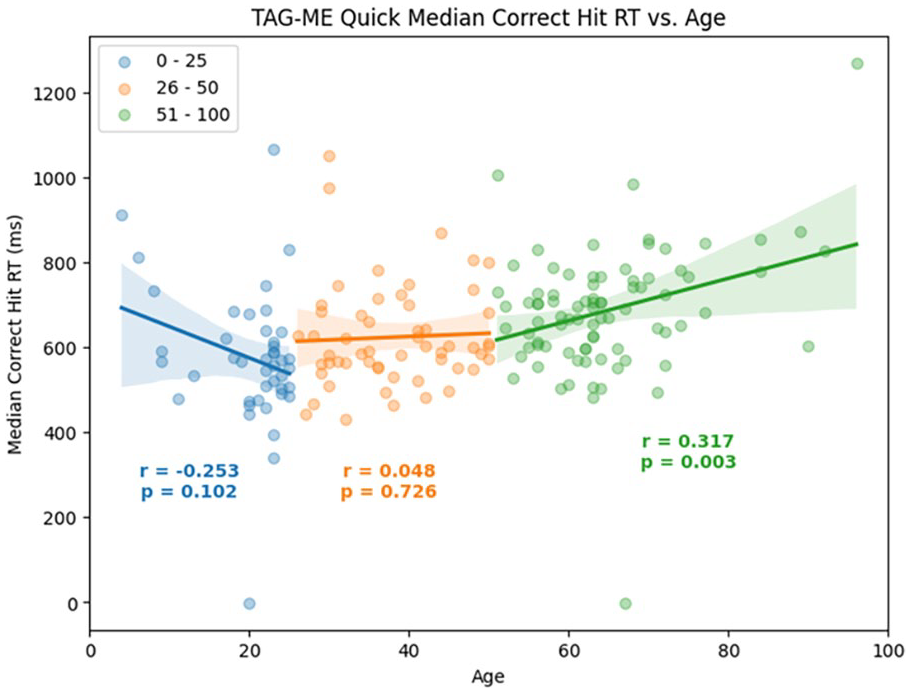
Regression analysis of age versus TAG-ME quick median correct hit response time (with Pearson correlations and p-values also shown).
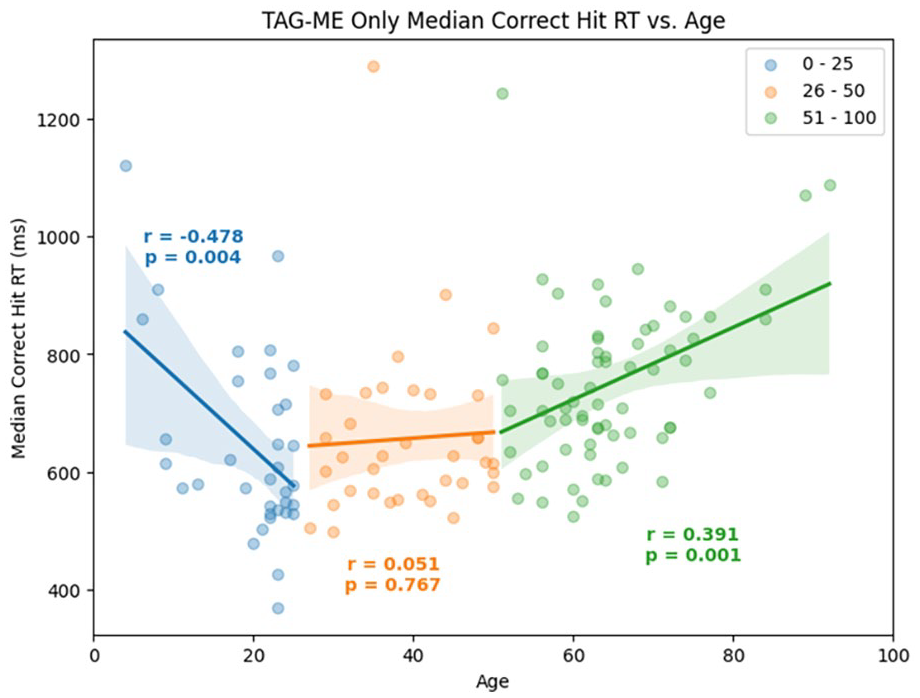
Regression analysis of age versus TAG-ME only median correct hit response time (with Pearson correlations and p-values also shown).
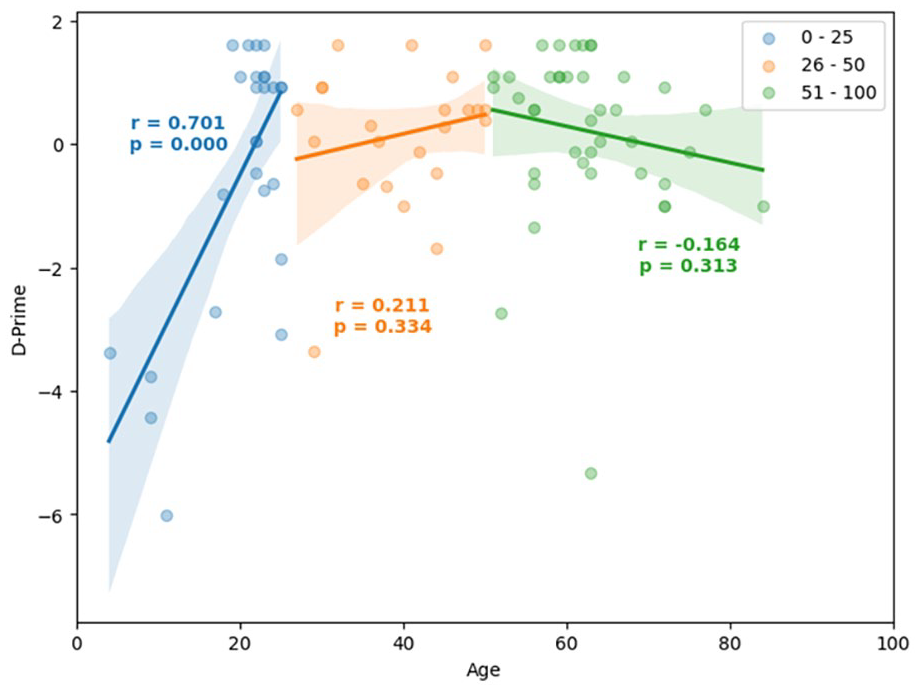
Regression analysis of age versus TAG-ME again easy d-prime values (with Pearson correlations and p-values also shown).
Discussion
The BrainTagger games demonstrated clear age-related trends consistent with the expected trajectory of fluid intelligence. The observed sensitivity to decline in cognitive performance after age 50 suggests good potential for use in hospital or long-term care settings to monitor older patients at risk of cognitive harm from interventions such as anesthesia, ICU stays, and chemotherapy.
Compared to traditional assessments, BrainTagger offers self-administration, reduced learning effects, and low cost, making it suitable for widespread use. Prior work supports its ability to detect delirium in emergency departments (Lee et al., 2019), and this study extends its relevance to cognitive aging.
Limitations
The sample was recruited from public events and may not generalize to clinical populations. Cross-sectional data were used to predict age-related trajectories and it is possible that some of the observed differences in cognitive status with age may have been due to systematic differences in the cognitive status of the difference age cohorts sampled in this study.
Conclusion
BrainTagger games show promise as tools for cognitive safety monitoring. They are sensitive to the natural aging process and could be used to detect cognitive harms associated with medical interventions. Future research should implement “before and after” cognitive tracking in hospitals and long-term care environments and explore integration into electronic medical records for continuous monitoring.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: BrainTagger is co-owned by Mark Chignell, two other off-campus researchers, and the University of Toronto. In the future it is possible that a company may be formed and that BrainTagger may earn revenues. But at present BrainTagger is freely available for research use and the main motivation of our research is to collect scientific results associated with the use of the BrainTagger Cognitive assessment games.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article:Development of the BrainTagger games was funded by grant number IA-2021-211 “Target Acquisition Games for Measurement and Evaluation (TAG-ME) of Detailed Brain Function” from the Connaught Fund. Additional support was provided by the University of Toronto work study program.
