Abstract
Objective
To evaluate patients’ and caregivers’ perceived benefit of using a custom 3-dimensional (3D)-printed transparent model of a skull with unilateral congenital aural atresia as a tool for surgical counseling.
Design
Prospective study.
Setting
Tertiary academic children's hospital.
Patients/Participants
Twenty-three families with children with congenital aural atresia.
Interventions
A custom 3D-printed skull model was created from a computed tomography scan of a 5-year-old male patient with right unilateral congenital aural atresia, using transparent VEROCLEAR for bone and contrasting colors for the facial nerve, auditory ossicles, and bony labyrinth. This 3D model was used during standardized counseling about congenital aural atresia. Families were surveyed regarding their understanding of congenital aural atresia and their impressions of the model.
Main Outcome Measures
Scale of 1 to 5 (strongly disagree to strongly agree) regarding utility of the 3D model.
Results
Resources used most by families to learn about congenital aural atresia were online (57%) and doctor's visits (83%). Families strongly agreed that having a 3D model made it easier to understand the anatomy of congenital aural atresia (4.7/5, SD 0.5), the goals of surgical repair (4.7/5, SD 0.7), and the risks of atresiaplasty surgery (4.6/5, SD 0.6). Model use also increased reported comfort level with treatment decision-making (4.7/5, SD 0.6).
Conclusion
A novel, transparent 3D-printed model of congenital aural atresia that allows patients and families to visualize and physically interact with complex temporal bone anatomy could be a valuable, effective, and readily available tool to improve patient surgical counseling.
Introduction
Congenital aural atresia (CAA) is the absence of a patent ear canal evident at birth and encompasses various degrees of failed external ear canal development. This condition affects 10 000 to 20 000 live births per year. 1 It is also associated with microtia, or a small or malformed ear diagnosed at birth.2,3 Congenital aural atresia causes severe conductive hearing loss, which, if untreated, can delay speech development and contribute to social integration challenges and feelings of inferiority due to its physical manifestations.4–7 Management options for CAA include bone conduction hearing devices (BCHD) and surgical reconstruction of the ear canal (atresiaplasty).8,9 However, treatment decisions require careful consideration of factors such as surgical risks, aesthetic outcomes, and variability in hearing improvement.10–12
Congenital aural atresia presents with a complex and multifaceted pathology, making both its understanding and the navigation of treatment options challenging. Unfortunately, past studies have shown that most parents of children with CAA describe their initial consultation experience as “terrible” or “bad” due to the lack of clear and transparent information, especially regarding treatment options and due to lingering questions between discharge and follow-up appointments. 13 Many families reported not being given any specific information and found online resources to be “unclear” and “overwhelming.” 14
Understanding treatment options for CAA within the context of complex temporal bone anatomy is challenging for patients and families. We explored the utility of a custom 3-dimensional (3D)-printed transparent model of a skull with unilateral CAA as a tool for surgical counseling. We hypothesized that patients and families would find the opportunity for anatomic visualization and hands-on interaction useful in their understanding of CAA and its treatment options.
Methods
Study Participants
This prospective study enrolled patients with CAA seen in an outpatient clinic at a tertiary academic children's hospital between October and November 2024. Prior to this study, typical counseling for CAA utilized printed graphical visual, review of patient's computed tomography (CT) imaging, and standard office anatomical ear models. Demographic and clinical data were collected prospectively. The study was approved by the Institutional Review Board. All patient records/data were anonymized and deidentified.
Patients and families were approached in Otolaryngology clinic or in the preoperative holding area and asked whether they would like to participate in the study. This captured participants with a wide range of experiences with CAA, from new diagnoses to several years into follow-up. Families were notified that the decision to participate in the study would not impact the clinical care received during their outpatient visit, and no financial compensation was offered in exchange for participation. Interpreter services were utilized to interact with non-English-speaking families.
Three-Dimensional Printing Process
A custom 3D-printed model skull was created using a CT scan of a 5-year-old male with right unilateral CAA. Using a representative CAA patient with a high-resolution CT scan, a Digital Image and Communications in Medicine image file removing all identifiable patient information was exported using Materialise Mimics inPrint (Figure 1A). Key anatomical landmarks were manually segmented and highlighted in different colors. The bone layer was then printed in transparent VEROCLEAR material. This enabled an easy view of the facial nerve, auditory ossicles, and the bony labyrinth which were each printed in contrasting colors within the model (green, orange, and blue, respectively) (Figure 1B). The left side of the model had an open ear canal (Figure 1C) while the right side of the model had an atretic ear canal (Figure 1D). The final product was sanded and polished to provide the best resolution of all printed structures.
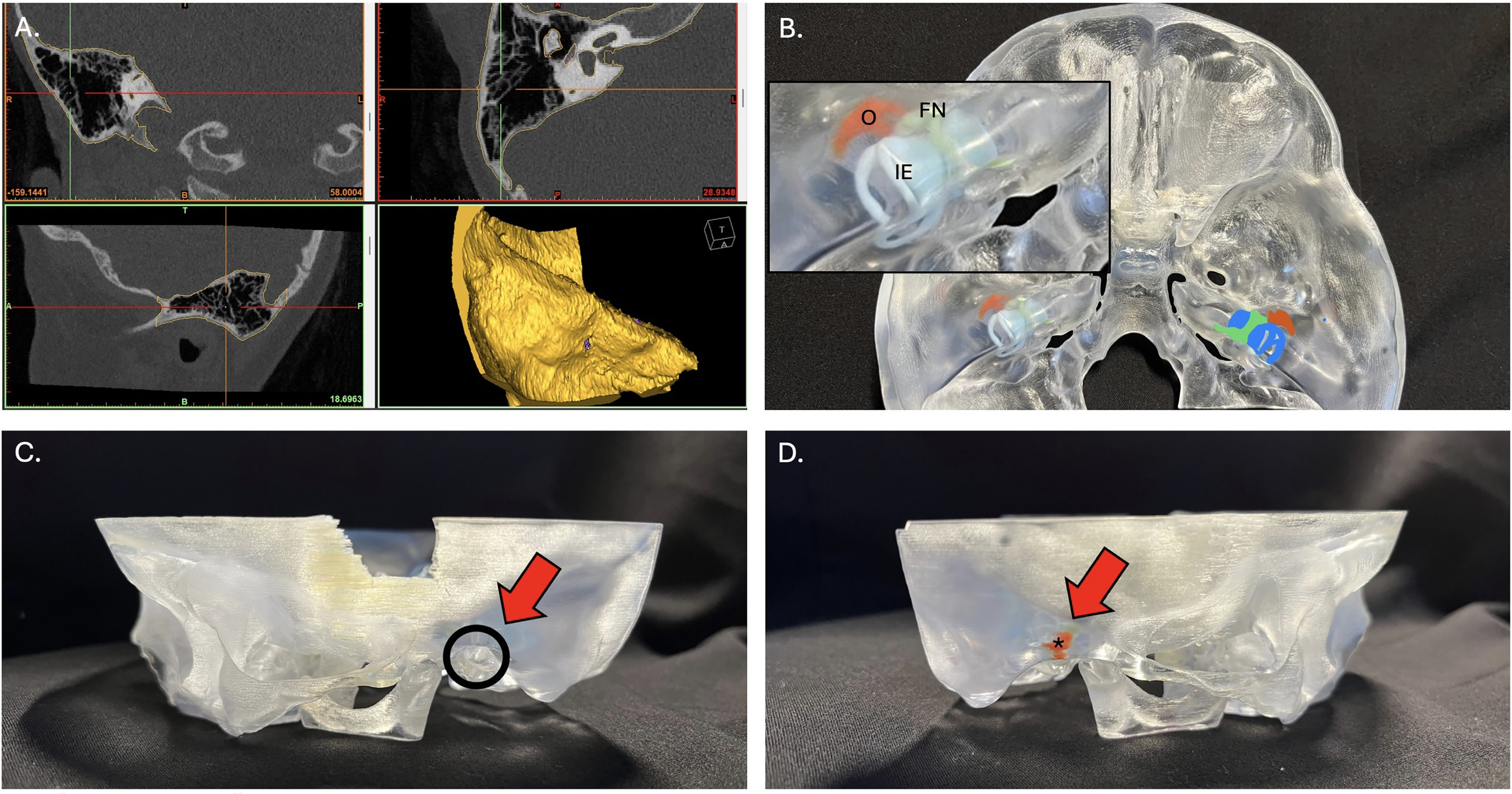
(A) A computed tomography (CT) scan was used was used to create the 3-dimensional (3D) model. (B) Because the bone layer was printed in transparent material, there was a view of the facial nerve (FN, green), auditory ossicles (O, orange), and bony labyrinth (IE, blue). (C) The left side of the model had an open ear canal (circle). (D) The right side of the model had an atretic ear canal (*).
Study Methods
Consent to participate in the study was obtained while parents were waiting in the clinic exam room or in the preoperative area. They then were given a presurvey to gather information about their child's clinical history and to assess their self-perceived understanding of CAA (Table 1). The presurvey also asked questions about sources of information the family had used to learn more about CAA. The 3D model was then introduced during standardized counseling about CAA, which took approximately 5 min (see Supplemental Material). The same 3D model was used with all study participants. Finally, immediately after the standardized counseling, families were given a postsurvey regarding the use of the model to understand CAA using a 5-point scale correlating with answer choices (Table 2). Outcome measures included the following: improved understanding of CAA anatomy, surgical repair, surgical risks, and BCHD, as well as comfort in the treatment options that the participants chose to pursue.
Presurvey Used to Gather the Child's History With CAA and Parental's Understanding and Resource Use.
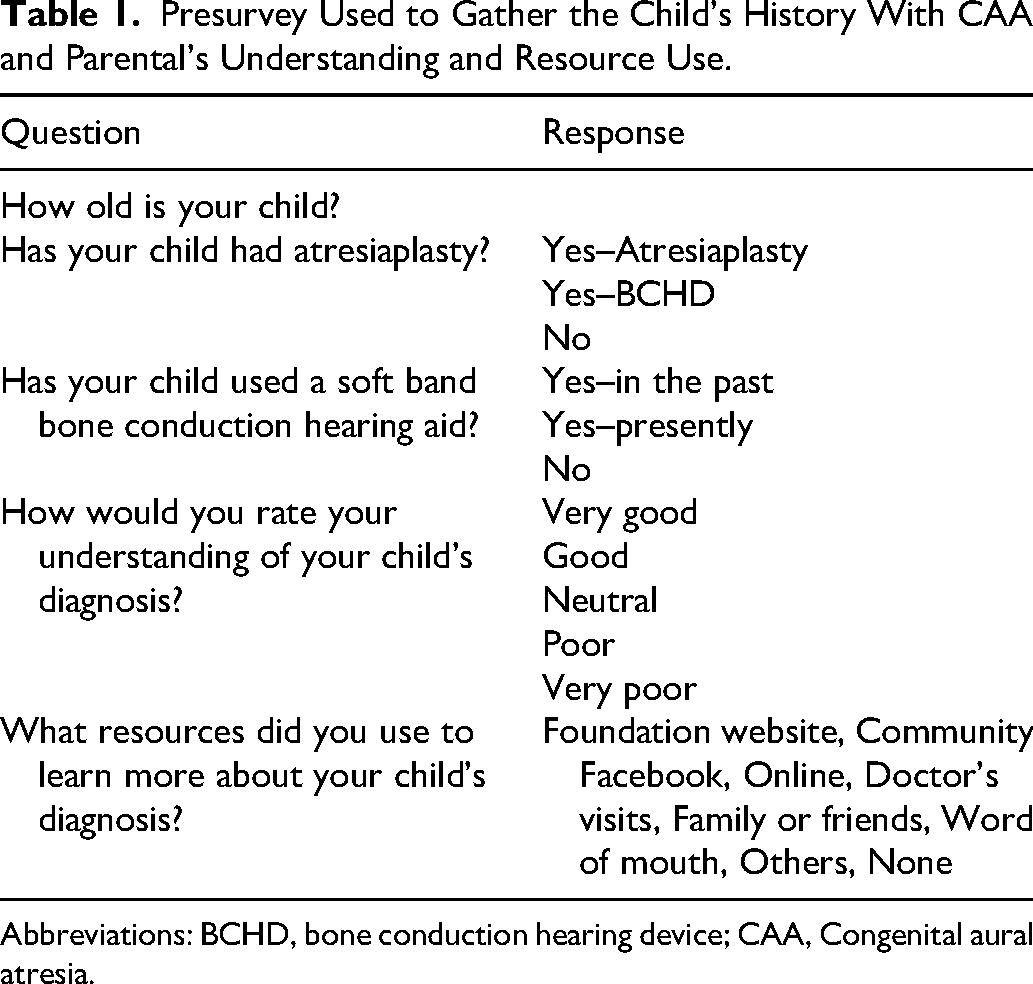
Abbreviations: BCHD, bone conduction hearing device; CAA, Congenital aural atresia.
Postsurvey to Assess Parents’ Feedback of the Model.

Abbreviation: CAA, Congenital aural atresia.
Statistical Analyses
All statistical analyses and graphing were performed using GraphPad Prism 10 and within REDCap. The participants’ responses were analyzed on a Likert scale—1 represented strongly disagree, 2 represented disagree, 3 represented neutral, 4 represented agree, and 5 represented strongly agree. Participants’ responses to the question “How would you rate your understanding of your child's diagnosis?” were grouped into low understanding (poor, very poor), neutral, and high understanding (good, very good). Descriptive statistics were reported. The mean of the participants’ responses to the model was compared between the child's age, prior surgical history, use of soft-band BCHD, and the parent's understanding of CAA. Kruskal-Wallis nonparametric testing was used to compare results between groups, and a predetermined threshold P value of .05 used for determination of statistical significance.
Results
Twenty-three children with CAA were enrolled in this study, and their parents completed the surveys with the child's involvement at the parent's discretion. The mean age was 7.74 (SD 5.63) years old. 6 (26%) had atresiaplasty 5 (22%) had BCHD surgery, and 12 (52%) had no prior surgery to treat their CAA (2 A). Most children had used a soft bone BCHD either in the past (9, 39%) or presently (6, 26%) (Figure 2B). Most parents rated their understanding of CAA as very good (10, 44%) or good (7, 30%). Fewer parents rated their understanding of CAA as poor (1, 4%) or very poor (1, 4%). Resources utilized included the foundation website, community Facebook, family or friends, and word of mouth. Online websites (13, 57%) or doctor's visits (19, 83%) were the most used resources (Figure 3).
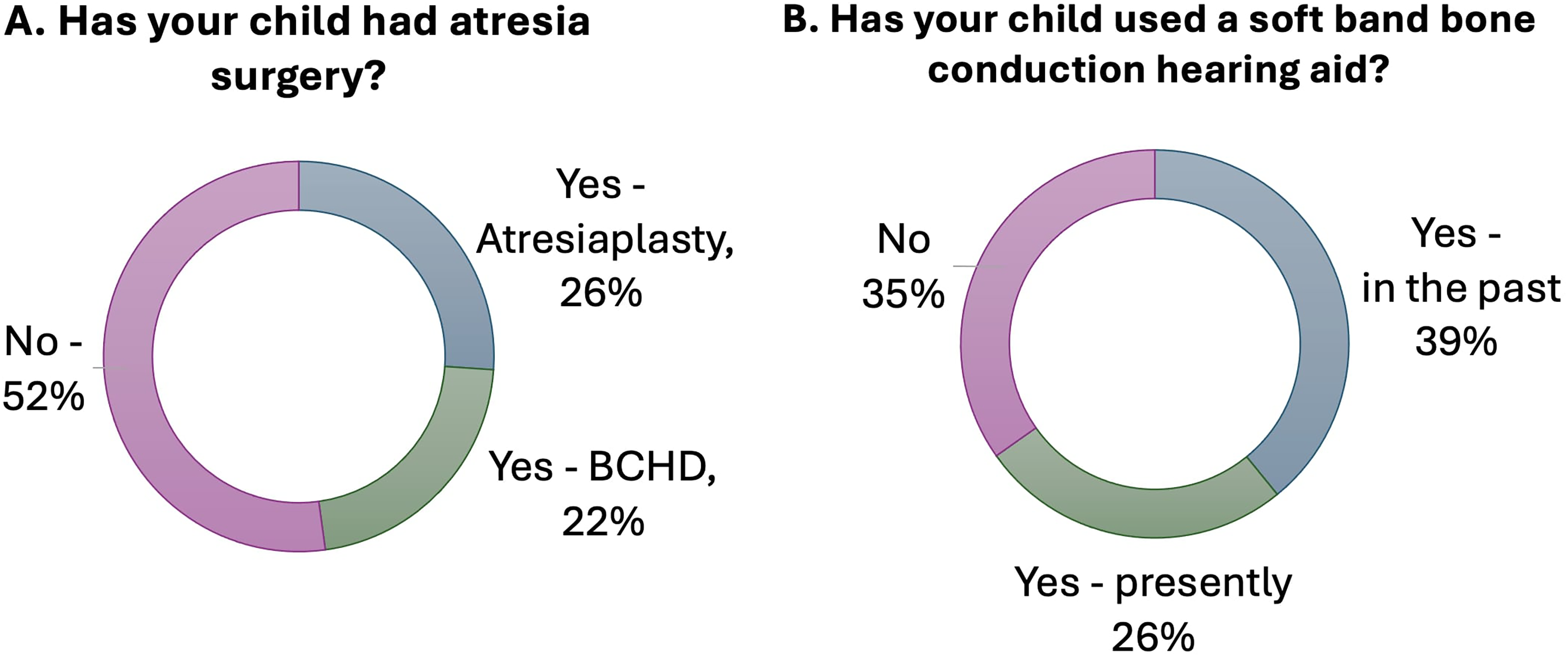
Families were asked questions about (A) their child's history of atresiaplasty and (B) their child's use of soft band bone conduction hearing aids.
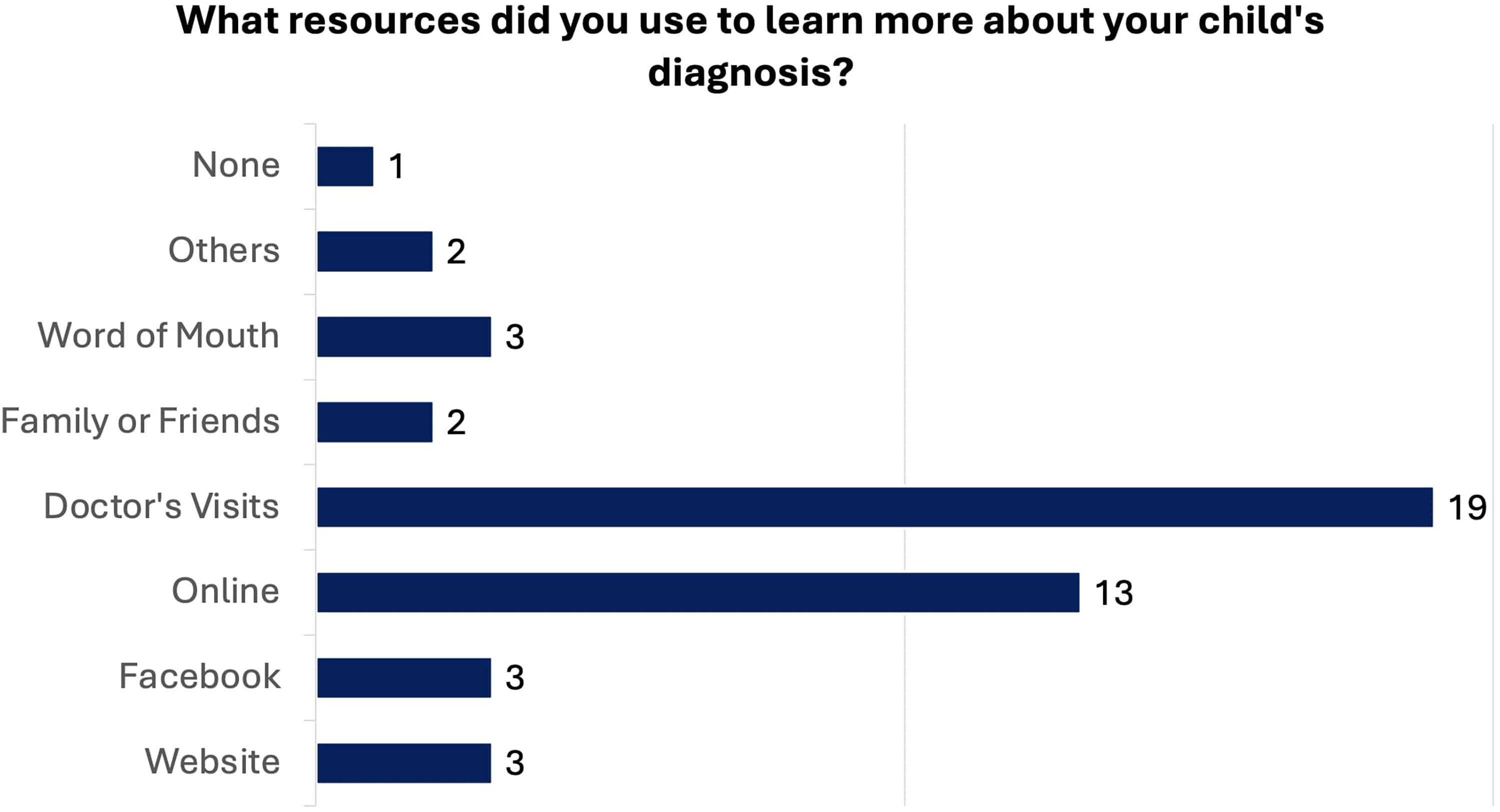
Types of resources utilized by parents to learn more about congenital aural atresia (CAA). Online websites (13, 57%) and doctor's visits (19, 83%) were the most used resources.
On a scale of 1 to 5 (strongly disagree to strongly agree), families strongly agreed that having a 3D model made it easier to understand the anatomy of CAA (4.74/5, SD 0.45), goals of surgical repair (4.70/5, SD 0.70), and risks of atresiaplasty (4.61/5, SD 0.58) (Figure 4A-C). Using the model also increased understanding of BCHD (4.74/5, SD 0.450) (Figure 4D) and increased comfort level with treatment decision-making (4.65/5, SD 0.57).
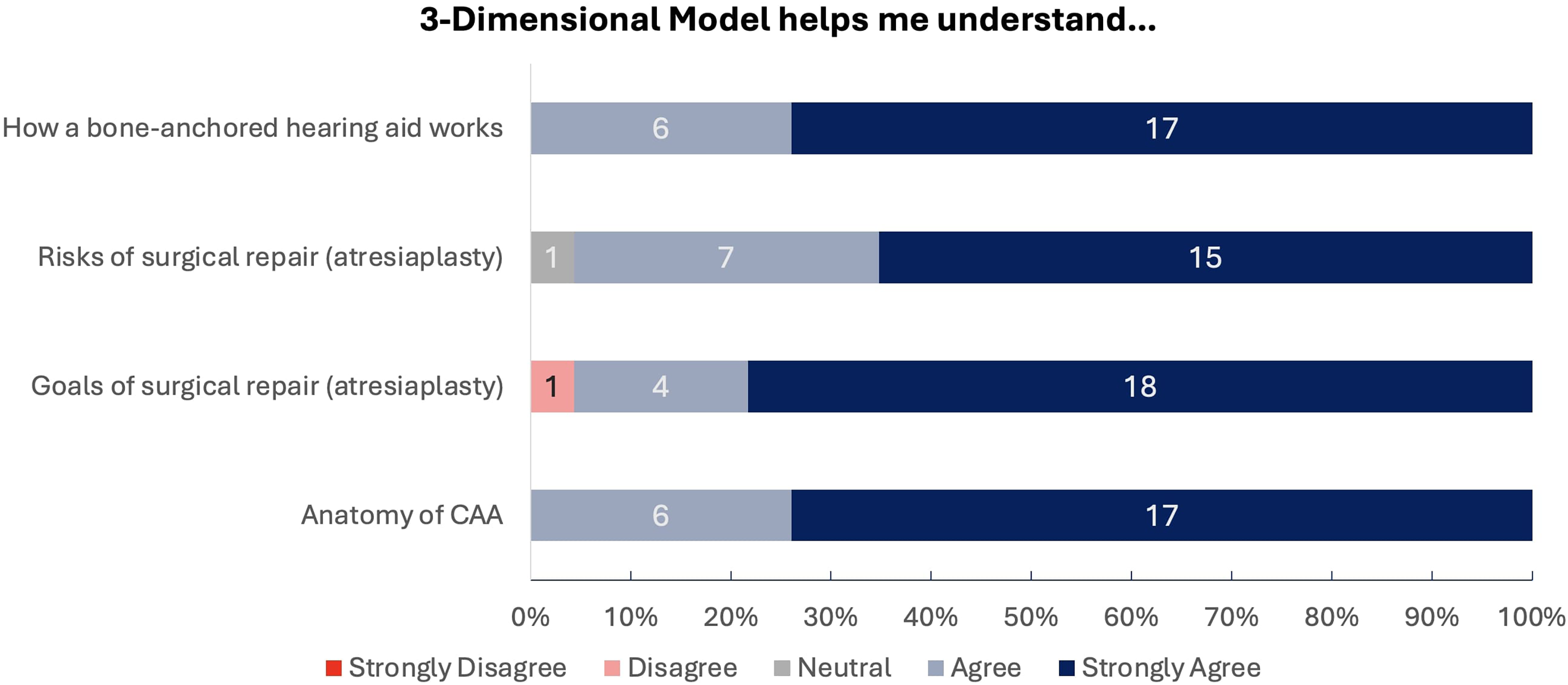
Parents’ responses to the usefulness of a 3-dimensional model in understanding how bone anchored hearing aids work, risks of atresiaplasty, goals of atresiaplasty, and anatomy of congenital aural atresia (CAA).
Differences in surgical history or use of soft band BCHD did not result in significantly different answers to the questions about the model in the postsurvey. Having a high self-perceived understanding of CAA was correlated with perceived usefulness of the 3D model for understanding the anatomy of CAA (P = .042) and understanding atresiaplasty (P = .0079). Having a high or low self-perceived understanding of CAA was not correlated with the perceived usefulness of the 3D model for understanding the risks of atresiaplasty, the mechanism of BCHD, or comfort with the treatment option chosen.
Discussion
This study investigated how parents and patients perceived the use of a 3D-printed model for parents and patient education in the field of CAA. Our study shows that most families and patients felt that a CAA 3D-model helped them understand the complex anatomy of CAA, the goals and risks of surgical repair, and the mechanism of BCHDs. Overall, the model increased family comfort with their choices of treatment, and many participants shared positive feedback about the model. In addition, we found no statistically significant differences in the perception of the model by age, surgical history, and prior use of BCHD, indicating the model's potential utility in a variety of settings and with families of different experience levels. These results suggest that transparent 3D-printed CAA models are a useful tool for counseling of patients with CAA and their families.
Improving parental literacy about CAA is crucial to a child's treatment plan, particularly due to the complex care that involves coordination with multiple specialists over many years. The functional and aesthetic impacts of CAA also affect children's psychological and social well-being.15,16 How providers communicate information is important given the implications on the parents’ and patients’ perspective of illness, their long-term relationships with clinicians, and both patient and provider satisfaction.17–20 However, the literature shows that many families of children with CAA and microtia report feeling dissatisfied and stressed after visits with physicians.13,14 Surveys indicate that parents often desire more information, particularly about treatment options. 13 Integrating CAA-specific 3D models into clinical practice may be an easy and helpful step for improving family understanding and support for CAA. Participants in this study expressed that they wished they had seen the CAA model during the initial consultation, as it would have provided a clear framework for the physician to discuss next steps and management options.
Otolaryngology and other medical specialties such as hepatology, neurosurgery, and cardiology have utilized 3D models to improve patient education.21–23 In Louisiana, a 3D vestibular model enhanced patients’ understanding of symptom causes, increased confidence in prevention strategies, reduced anxiety, and improved satisfaction with educational sessions. 24 Similarly, 3D sinonasal models have been employed to create patient-specific irrigation strategies, optimizing drug delivery and treatment outcomes while fostering greater physician and patient engagement. 25 Like dizziness-related disabilities and sinonasal diseases, CAA involves complex pathology and treatment options that can be challenging for patients to comprehend through verbal explanations or 2-dimensional images alone. 26 This study demonstrated the effectiveness of a tactile, interactive visual model in enhancing patient education and understanding of CAA and its treatment options. These models can be reused with different patients, enhancing their value and cost-effectiveness in a clinical setting.
This study had several limitations. First, this pilot study only included 23 families at one institution, and small heterogeneous sample sizes may introduce bias, limit statistical power, and the ability to detect significant effects. The study did not include a control group receiving counseling without the use of the CAA 3D model, limiting the ability to assess whether the model was better than standard counseling without it. Instead, the study focused on participant attitudes toward counseling with the 3D model itself. The model was based on a patient with right-sided complete aural atresia and not specific for every patient's anatomy. This evaluation was model-centric and did not include a control group using conventional 2D imaging or existing physical models, limiting our ability to quantify the added benefit of the 3D model. This study lacked long-term follow-up, making it difficult to assess if the use of 3D models led to long-term improvements in patient care. We also did not collect patient sex/gender as a variable in our questionnaire. Future studies could involve a larger patient sample and include a control group to compare education sessions that incorporate 3D CAA models with those that do not. Next steps include studies that also incorporate a comparative arm using traditional 2D imaging or standard physical models to more rigorously assess the added value of 3D modeling in patient and trainee education. Additionally, it could be valuable to study patients and families at various stages of their medical journey with CAA—such as during the initial consultation, preoperatively, and after years of follow-up—to determine the most beneficial timing for using the model.
Future initiatives could also focus on creating reading materials that cater to patients from diverse educational backgrounds or exploring augmented reality tools that use patient CT scans to visualize anatomy and treatment options. The model can continue to evolve, including adding insets into the model to label anatomy. In addition, while the model used in this study was based on a single patient's CT scan, future advancements could involve creating patient-specific 3D models derived from individual CT scans for personalized counseling and education—albeit at increased cost. Models could also be used by otolaryngologists for surgical planning.
Conclusions
In conclusion, patients reported that use of a transparent 3D-printed CAA model skull during counseling was helpful in improving their understanding of anatomy and treatment options. This simple and reusable tool allows patients and families to visualize and physically interact with complex anatomy. Future studies with larger sample sizes and control groups would be valuable in assessing the benefit of this type of model as compared to existing visual aids.
Supplemental Material
sj-docx-1-cpc-10.1177_10556656251363440 - Supplemental material for Use of a 3-Dimensional Printed Model of Congenital Aural Atresia for Family Surgical Counseling
Supplemental material, sj-docx-1-cpc-10.1177_10556656251363440 for Use of a 3-Dimensional Printed Model of Congenital Aural Atresia for Family Surgical Counseling by Ina Lee, MS, Shreyas Krishnapura, MD, Brenton Griffith, BS, Marc Bennett, MD, FACS, MMHC, Sumit Pruthi, MBBS, and Jason Park, MD, PhD in The Cleft Palate Craniofacial Journal
Footnotes
Ethical Approval and Informed Consent
This study received ethical approval (as an exempt study) from the IRB # 222122 on September 11, 2023. This is an IRB-approved retrospective study, all patient information was deidentified and patient consent was not required.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
