Abstract
Two immunocompetent dogs from separate households were presented to a tertiary referral hospital with soft tissue wounds attributable to severe bacterial infection. A 5-y-old, castrated male Golden Retriever dog (case 1) was presented with extensive hemorrhagic cellulitis of the forelimbs and neck that developed over 48-h and was attributable to Acinetobacter lactucae infection. Additionally, an 8-y-old, spayed female Labrador Retriever-mix dog (case 2) sustained a deep penetrating wound over the left hip that progressed over 24 h to necrotizing fasciitis, from which Bacillus paramobilis was isolated. Histopathologic findings in both cases included severe acute, skeletal muscle necrosis and necrosuppurative myofasciitis, dermatitis, and panniculitis with intralesional rod-shaped bacteria. Whole-genome sequencing and phylogenetic analysis of the bacterial isolates revealed numerous cytolytic toxins and other virulence genes carried by the isolates, expanding the profile of these 2 bacteria. To our knowledge, canine cases of A. lactucae and B. paramobilis associated with necrotizing fasciitis have not been reported previously. In both cases, progression from an inciting incident to septic disease was extremely rapid, occurring within 48 h in case 1 and 24 h in case 2, underscoring the fulminant nature of necrotizing fasciitis.
Keywords
Acinetobacter species frequently acquire antimicrobial resistance genes, enabling them to survive in the hospital environment.
10
The Acinetobacter calcoaceticus-baumannii (
Here, we describe 2 cases of necrotizing fasciitis in dogs, one caused by an ACB-group organism and the other by a B. cereus-complex bacterium. We retrieved no cases of necrotizing fasciitis caused by either of these bacteria in a search of Google, PubMed, CAB Direct, Web of Science, and Scopus, using the search terms “Acinetobacter lactucae” and “Bacillus paramobilis” in the context of “necrotizing fasciitis”, separately.
A 5-y-old, castrated male Golden Retriever dog (case 1) developed lethargy, pain, axillary swelling, and right forelimb lameness 2 d after a dental procedure under general anesthesia. The dog was not noted to be immunocompromised prior to the dental procedure, and the procedure was performed uneventfully. On presentation at the referring veterinarian, a locally extensive area of pitting edema and bruising extended from the ventral neck to the axilla, bilaterally, and down the medial aspects of both forelimbs. Treatment included lactated Ringer solution (
The patient was diagnosed with sepsis, defined as infection accompanied by the systemic inflammatory response syndrome (
At autopsy, the skin, subcutis, and musculature along the entire ventrolateral cervical and thoracic regions from the mandible to the xyphoid process were edematous and necrotic (
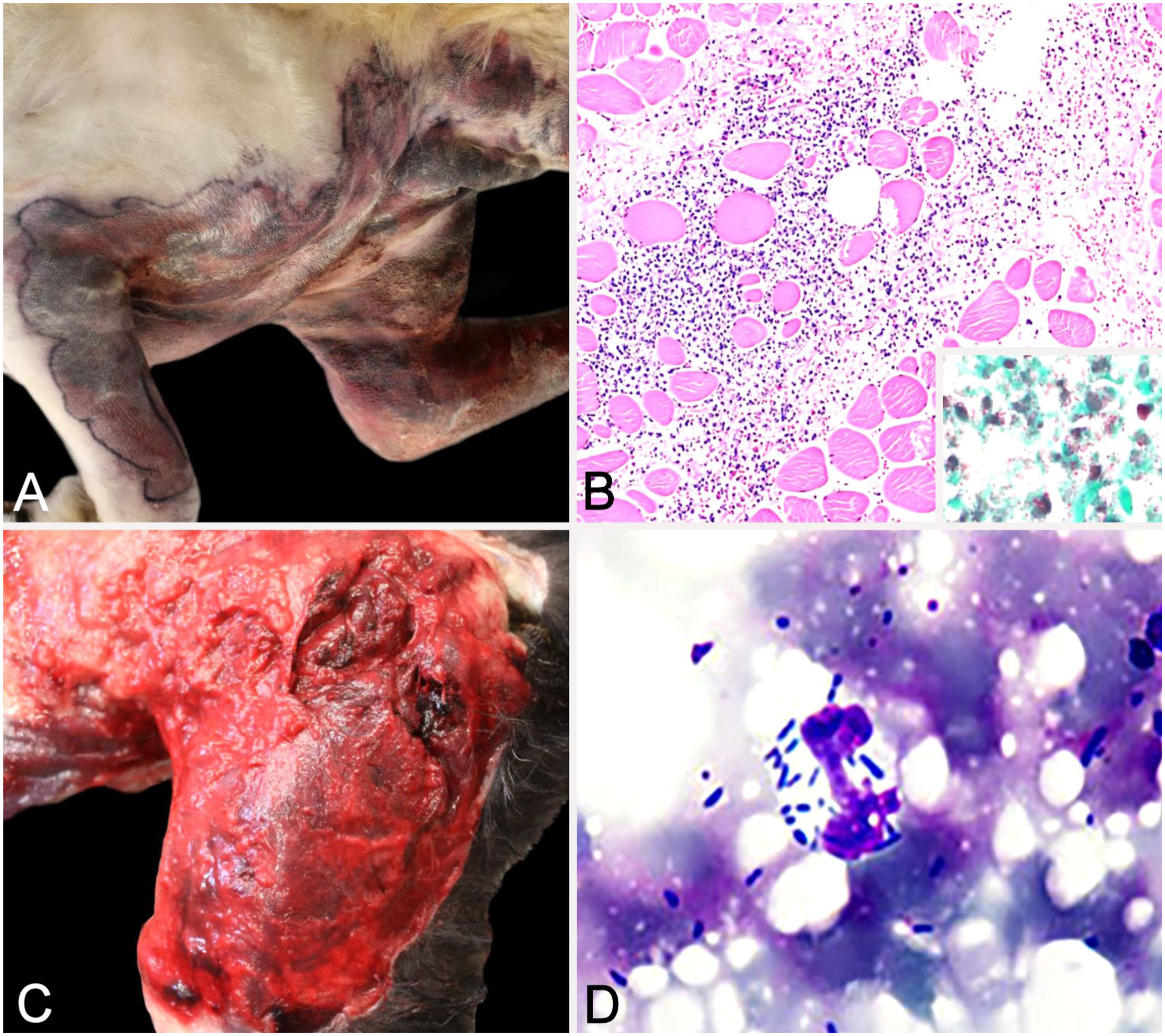
Gross, histologic, and cytologic findings of 2 canine cases of necrotizing fasciitis.
An 8-y-old, spayed female, immunocompetent Labrador Retriever-mix dog (case 2) was found with a large open wound over the left hip 24 h after escaping from the client’s yard. The patient was non-ambulatory but was responsive to stimuli. The patient was immediately taken to the Texas A&M University Small Animal Veterinary Medical Teaching Hospital (College Station, TX, USA) after discovery, but became comatose while in transit. On presentation, the patient was in hemodynamic shock with the following abnormal perfusion parameters: comatose mentation with no apparent corneal reflex and bilaterally fixed mydriatic pupils, tachycardia, prolonged capillary refill time with hypodynamic pulses, hypothermia, and hyperlactatemia (8.2 mmol/L). 9 Emphysematous wounds were noted over the left leg and at the base of the right ear.
Within 24 h of the inciting incident, the patient was diagnosed with sepsis, supported by tachycardia, tachypnea, and hypothermia consistent with SIRS. 4 A CBC and chemistry panel were not performed; hence, WBC concentrations were not available for SIRS criteria, and SOFA scoring could not be completed. A blood gas with electrolytes analysis revealed mixed acidosis, hypoglycemia (3.50 mmol/L; RI: 4.4–6.7 mmol/L), hyperkalemia (5.6 mmol/L; RI: 3.5–5.5 mmol/L), and azotemia (creatinine 194 µmol/L, RI: 44–133 µmol/L; urea nitrogen 14 mmol/L, RI: 2.5–11.4 mmol/L). Treatment consisted of supplemental flow-by oxygen, active warming, dextrose supplementation, and IV boluses of 7.2% hypertonic saline followed by LRS. The patient’s mentation improved to quiet and alert, with cranial nerve function returning during resuscitation and improvement in all vital signs. However, euthanasia was elected given the severity of the wounds and financial constraints.
At autopsy, 3 full-thickness skin lacerations were noted caudal to the base of the right ear, over the left hip joint, and along the ventral midline in the inguinal region. The subcutaneous tissues of the ventrolateral neck, chest, and bilaterally from the hips to the tarsal joints were markedly hemorrhagic and edematous. A large cavitation, interpreted as a necrotic puncture wound, was within the left quadriceps femoris and biceps femoris muscle bellies (
Aerobic culture of the skin from case 1 had 3+ growth of Acinetobacter lactucae and 1 colony of Malassezia sp.; culture of adipose tissue from case 2 had 3+ growth of a Bacillus sp., 3 colonies each of Escherichia coli and Streptococcus infantarius, and 2 colonies each of Proteus mirabilis and Ligilactobacillus sp. No Clostridium spp. were isolated from the wounds on anaerobic culture in either case. Aerobic culture consisted of inoculating plates (including trypticase soy agar supplemented with 5% sheep blood [blood agar plate], MacConkey agar, and Columbia nutrient agar with 5% sheep blood [CNA plate]); incubating them at 37 ± 2°C in a 5% CO2 atmosphere; and inspecting the culture plates daily for growth. Initial bacterial identification was performed (MALDI-TOF MS Biotyper with FlexControl v.3.4.206.94 software, MBT Compass 4.1 library; Bruker).
Whole-genome sequencing of the Bacillus sp. and A. lactucae isolates was performed to confirm identification and to evaluate virulence gene carriage. Briefly, a single colony was used to inoculate 10 mL of lysogeny broth that was then incubated at 37°C for 5 h. The cultures were pelleted, washed once with sterile PBS (pH 7.4), and incubated for 24 h at 37°C in 200 µL of TE buffer containing 5 µL of 25 mg/mL lysozyme from chicken egg white dissolved in PBS (Research Products International). DNA extraction was performed (MasterPure gram positive DNA purification kit; LGC Biosearch Technologies) at the lysis step per the manufacturer’s recommendation. Double-stranded DNA concentration in both samples was confirmed with a spectrophotometer (Qubit 4; Thermo) and conformed with recommendations for commercial long-read sequencing. Extracted bacterial genomic DNA from both cases was sent to Plasmidsaurus (SNPsaurus) for Oxford Nanopore MinION long-read sequencing per a standard protocol. 13 The bottom 5% worst fastq reads were removed via Filtlong v.0.2.1 (https://github.com/rrwick/Filtlong) using default parameters. Miniasm v.0.3 (https://github.com/lh3/miniasm) was used to generate a rough assembly to allow resampling and remove low-quality reads. Assembly was done with Flye v2.9.1 (https://github.com/mikolmogorov/Flye) with parameters selected for high quality ONT reads. Plasmid identification was done with Sourmash v.4.6.1 (https://github.com/sourmash-bio/sourmash) using GenBank. Sequence data from both agents were deposited in GenBank (BioProject PRJNA1110197): chromosome and plasmid sequences of A. lactucae (CP162643-CP162644) and the chromosome and 3 plasmid sequences of B. paramobilis (CP155622-CP155625).
The A. lactucae (128-92) isolated from case 1 was assigned multi-locus sequence typing (MLST)-Pasteur 2734 and ribosomal MLST (rMLST) 277522. The B. paramobilis (N24509) isolated from case 2 was designated ST3300, rMLST 276351, and core genome MLST 3540 using PubMLST.org databases.
12
Phylogenetic trees comparing 128-92 and N24509 with other isolates in GenBank were constructed using iTOL (
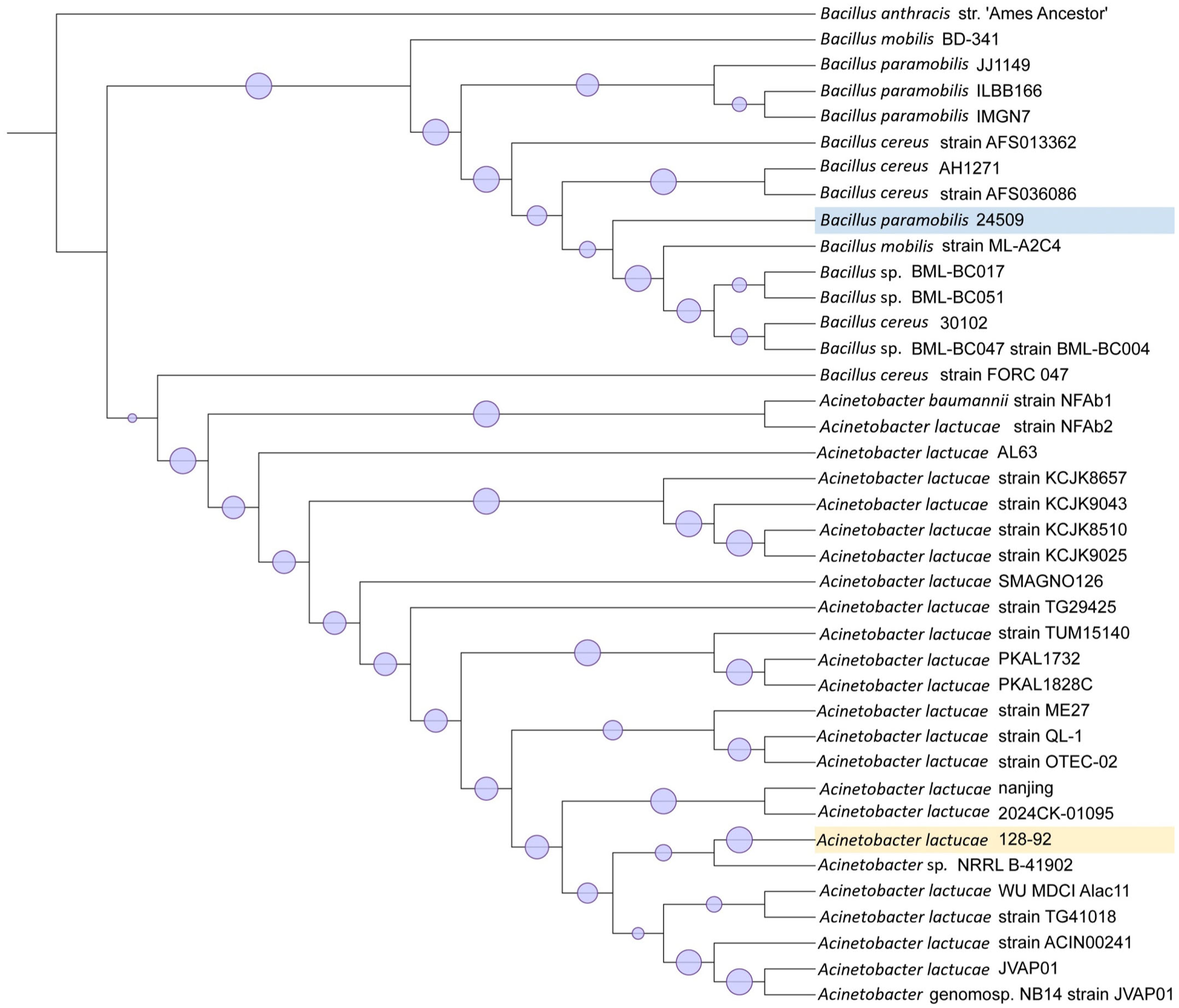
Phylogenetic tree of closest related bacteria to isolates 128-92 and N24509 in GenBank (as of 2025 Oct 24), using the Tamura–Nei model of nucleotide substitution with 1,000 bootstrap replicates.
In necrotizing fasciitis, subcutaneous tissue infection progresses rapidly, and common clinical signs include lameness, marked pain that is disproportionate to the outward appearance of the lesions, and hypoalbuminemia. 16 Typical gross findings include rapidly progressing hemorrhage, edema, and subcutaneous pocketing. 6 Deaths in 70–80% of dogs with necrotizing fasciitis are caused by multiple organ dysfunction, DIC, and septic shock. Histologic lesions typically consist of extensive myofiber necrosis with hemorrhage, edema, and bacterial colonies similar to those observed in both of our canine cases. 18
β-hemolytic Streptococcus canis is the most common bacterium underlying necrotizing fasciitis in dogs, although other bacteria, including Staphylococcus pseudintermedius, Staphylococcus aureus, Clostridium spp., Pasteurella multocida, Pseudomonas aeruginosa, and Escherichia coli, have been implicated in companion animals.16,19 In humans, ~50% of necrotizing fasciitis patients report an initial penetrating or crush injury that serves as an apparent inoculating event for infection. 5 Polymicrobial growth is commonly reported in human necrotizing fasciitis; a dominant organism may initiate disease, with subsequent secondary colonization. 2 The relative abundance of B. paramobilis in culture, together with concordant histologic findings, supports a likely contribution to wound development in case 2 despite finding additional microorganisms.
A. baumannii is associated with canine pyoderma, respiratory infections, and necrotizing fasciitis in cats. 23 In a study investigating community-acquired infections, 17 A. baumannii was found in 6 of 74 (8%) dogs with respiratory signs. A. lactucae has been recovered from the respiratory tracts of normal dogs but has also been reported to cause synovitis and nasal infection. Other ACB complex bacteria have been isolated from wounds, ear canals, and the reproductive and urinary tracts of cats, dogs, and horses. 3 Virulence factors that contribute directly to necrotizing fasciitis have not been specifically defined in A. baumannii; however, strains associated with invasive disease have been reported to cause reduced biofilm formation, increased hemolytic activity, and enhanced cellular invasion. 8
Bacillus spp. have been implicated in a wide range of infections in people, including abscesses, septicemia, wound and burn infections, osteomyelitis, and urinary tract infections. 15 B. paramobilis is a novel member of the Bacillus cereus group that was first cultured from the blood of 3 people hospitalized in Japan. B. paramobilis is a gram-positive, facultatively anaerobic, non-motile, rod-shaped bacterium with centrally formed, ellipsoidal endospores. 20 The bacterium has been isolated from the uterus of a beef cow 24 and is found in soil. 25 Cutaneous infections caused by B. cereus can closely mimic the clinical presentation of clostridial gangrenous myositis. Myositis caused by B. cereus typically occurs with sepsis in immunocompromised patients, and inflammation often spreads from the subcutaneous tissue to skeletal muscle. 15 The pathogenicity of B. cereus is associated with the production of enterotoxins and virulence factors that are involved in hemolysis and tissue necrosis. CDCs, a large family of pore-forming bacterial proteins, insert into and form pores in plasma and vesicular membranes. 22 A CDC closely resembling cereolysin O was identified in isolate N24509 and is a plausible contributor to the extensive tissue necrosis observed in case 2. Major toxin-encoding genes among B. cereus group strains are nheABC, hblABCD, and spH, with isolate N24509 carrying nheC, nheA, and hblACD. 1
In case 2, aerobic culture yielded additional bacterial species, which raises a limitation in definitively distinguishing monomicrobial from polymicrobial infection. However, the predominance of Bacillus spp. in culture, together with cytologic and histologic identification of abundant gram-positive rod-shaped bacteria, supports B. paramobilis as a likely major contributor to the necrotizing fasciitis observed in case 2.
Footnotes
Acknowledgements
We thank the staff of the Veterinary Medical Teaching Hospital and Clinical Microbiology service for their efforts in patient care and preservation of bacterial isolates.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
