Abstract
Acute hemorrhagic diarrhea syndrome (AHDS), formerly named canine hemorrhagic gastroenteritis, is one of the most common causes of acute hemorrhagic diarrhea in dogs, and is characterized by acute onset of diarrhea, vomiting, and hemoconcentration. To date, histologic examinations have been limited to postmortem specimens of only a few dogs with AHDS. Thus, the aim of our study was to describe in detail the distribution, character, and grade of microscopic lesions, and to investigate the etiology of AHDS. Our study comprised 10 dogs with AHDS and 9 control dogs of various breeds, age, and sex. Endoscopic biopsies of the gastrointestinal tract were taken and examined histologically (H&E, Giemsa), immunohistochemically (
Introduction
Acute hemorrhagic diarrhea syndrome (AHDS) is a common cause of hemorrhagic diarrhea in dogs.20,27 There are numerous terms used for this syndrome, such as canine hemorrhagic gastroenteritis, canine intestinal hemorrhage syndrome, acute intestinal hemorrhage syndrome in dogs, acute hemorrhagic diarrheal syndrome, and acute hemorrhagic enteropathy.6,8,11,13,27 The syndrome is characterized by acute onset of hemorrhagic diarrhea and is frequently associated with vomiting and hemoconcentration.6,20 A predilection for small breeds has been observed.6,19
To date, the etiology of AHDS is unconfirmed. Bacterial endotoxins, hypersensitivity to food components, or autoimmune reactions could play a role in the pathogenesis.6,11,13 The involvement of
In previous studies, histologic examinations have been limited to only a small number of dogs with AHDS, in which samples were collected postmortem.13,21,23,24 The information in these studies is limited by: 1) the difficulty of interpreting intestinal changes in specimens collected postmortem given postmortem autolysis; 2) the fact that these dogs did not have the usual course of disease that typically is associated with recovery; and 3) the low number of cases. We describe herein the pathology of AHDS using endoscopic gastrointestinal biopsies, and the characterization of the
Material and methods
Cases
Our study population partly overlaps with the patient populations of a previously published study.
31
Ten dogs of various breeds, sex, and age that had been presented with acute onset of bloody diarrhea (<3 d) were included (Table 1). Exclusion criteria included: pretreatment with antibiotics, hemorrhagic diarrhea caused by non-steroidal anti-inflammatory or corticosteroid drugs, anticoagulant toxicosis, hypoadrenocorticism, hepatic failure, portal hypertension, acute or chronic renal failure, pancreatitis, inflammatory bowel disease, gastrointestinal neoplasia or foreign bodies, and/or enteric infection with parvovirus,
Signalment of the 10 dogs diagnosed with acute hemorrhagic diarrhea syndrome.
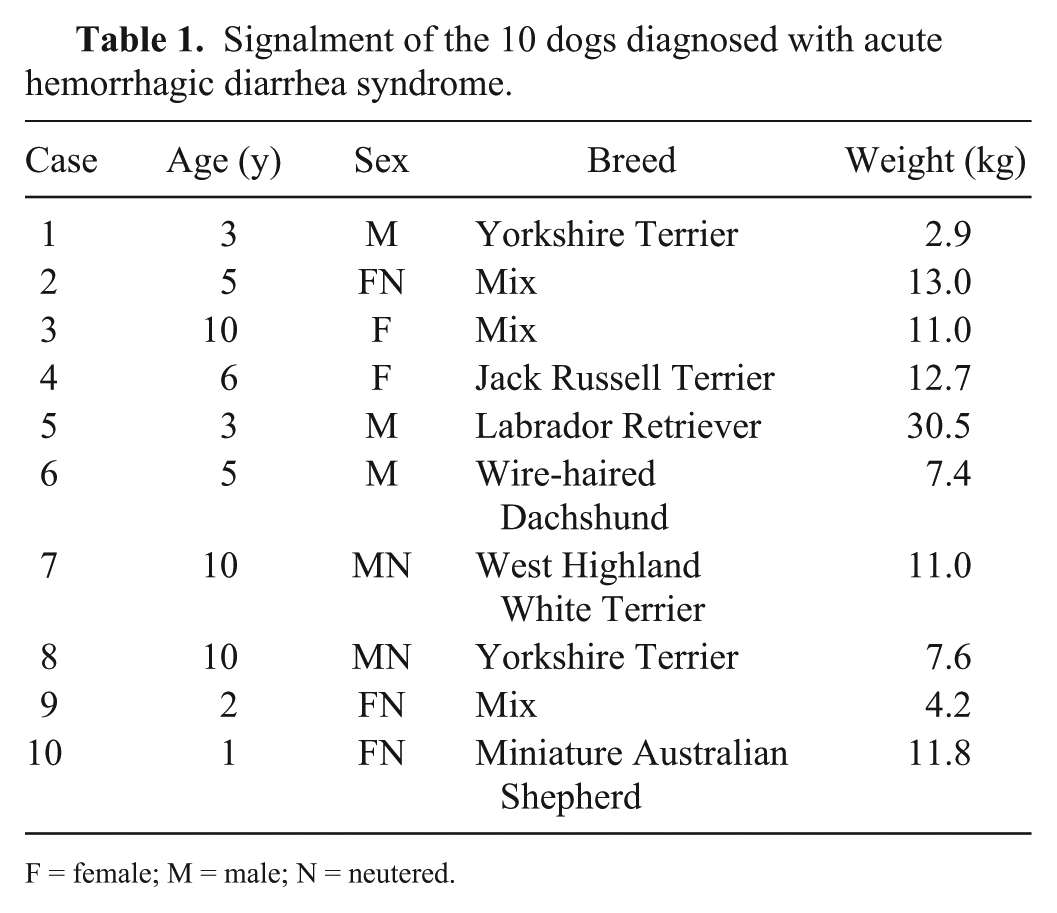
F = female; M = male; N = neutered.
Nine dogs with chronic non-hemorrhagic gastrointestinal disorders that had been presented to the Clinic of Small Animal Medicine of Ludwig-Maximilians-University Munich were used as controls for histologic, immunohistochemical, and microbiologic investigations of the gastrointestinal mucosa (Table 2). In 2 additional dogs, in which endoscopy was performed for causes unrelated to acute hemorrhagic diarrhea,
Signalment and diagnoses of the 9 dogs in the control group.
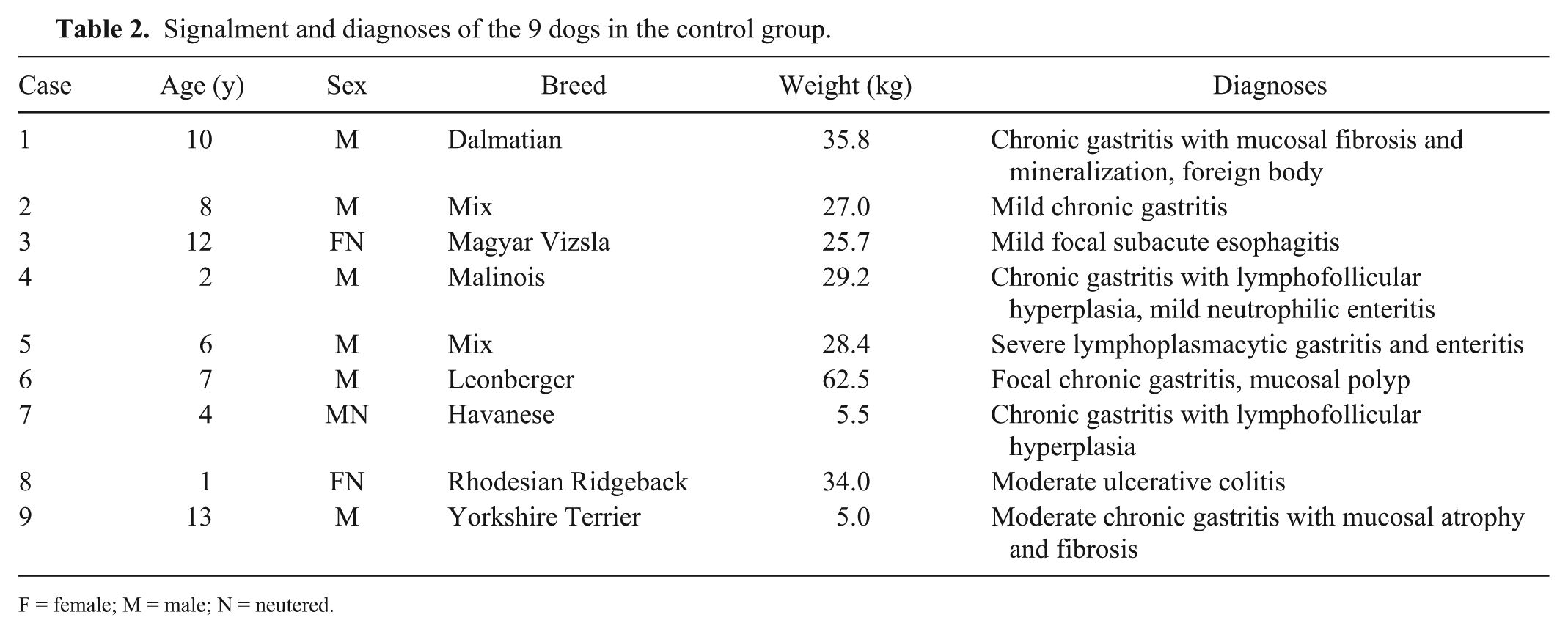
F = female; M = male; N = neutered.
Biopsy and histologic examination
Biopsy samples of stomach, duodenum, ileum, and colon were taken endoscopically. All of the biopsies included mucosa and submucosa. Biopsy samples were immersed in 10% neutral-buffered formalin immediately after collection and embedded in paraffin and plastic 12 using standard histologic techniques. For histologic evaluation, the sections were stained with hematoxylin and eosin. Additional sections were stained with Giemsa and Gram. Biopsy samples were evaluated histologically according to the standard criteria of the World Small Animal Veterinary Association Gastrointestinal Standardization Group. 9
Immunohistochemistry
Sections of the intestine were evaluated immunohistochemically using antibodies against
b. Parvovirus: mouse anti-canine/feline parvovirus (1:100 dilution, AbD Serotec) as primary antibody; peroxidase-labeled rabbit anti-mouse immunoglobulins (1:100 dilution, Dako) as secondary antibody; rabbit normal serum (1:80 dilution, MP Biomedicals).
All incubations were completed at room temperature. Following manual deparaffinization and rehydration, 4-μm tissue sections, mounted on positive-charged glass slides, were treated with 1% hydrogen peroxide to quench endogenous peroxidase activity and washed in a bath of Tris-buffered saline (TBS, 0.5 M, pH 7.6). Slides for parvovirus IHC were pretreated with proteinase K (Dako) for 10 min, followed by a TBS bath for 10 min. After blocking with normal serum for 30 min, slides were incubated with the primary antibody for 60 min. Subsequently, slides were washed in baths of TBS, incubated with the secondary antibody for 1 h, and washed again with TBS. Binding of peroxidase coupled to the secondary antibodies was visualized by the reaction with H2O2 and 3’3’-diaminobenzidine-tetrahydrochloride-dihydrate (Biotrend Chemikalien, Köln, Germany) as chromogen. Slides were counterstained with Mayer hematoxylin.
For clostridial IHC, positive controls included a clostridial suspension (
The positive control for parvovirus infection originated from routine autopsy material (dogs with histologic and virologic confirmed parvovirus infection). For negative controls, the primary antibodies were replaced with homologous non-immune sera.
Bacterial culture
Bacteriologic investigation was performed using duodenal biopsies of 9 dogs. Within 30 min of biopsy collection, samples were plated onto agar plates using sterile tweezers, followed by 3-phase streaking using an inoculation loop for semi-quantification. Nutrient agar (with 5% defibrinated sheep blood for aerobic incubation) and Schaedler agar (with 5% sheep blood for anaerobic incubation) were used for cultivation. Plates were incubated at 38 ± 1°C. Anaerobic incubation was performed using gas generators (GENbox anaer, bioMérieux, Marcy l’Etoile, France) in anaerobic jars. Colony growth was monitored for 3 d. Every colony type was quantified (from (+) = total <10 colonies up to +++ = dense colony growth in the third streak) and differentiated by mass spectrometry using MALDI-TOF (Microflex LT, Flex analysis software 3.0, Bruker-Daltonics, Bremen, Germany). Biotyper score values of ≥2.3 were accepted as identification on species level. From every instance of clostridial growth on culture plates, 5–7 colonies were subcultured and preserved at –20°C. One isolate per dog was subsequently used for further investigation.
Genotyping of C. perfringens isolates and determination of genes encoding for toxin netF
In
Statistical analysis
To identify statistical differences in the severity of mucosal lesions between different locations (duodenum, ileum, colon) of the intestinal tract, histologic scores were compared with a repeated measures ANOVA and Tukey multiple comparison post-test. This analysis was conducted twice, both as intention-to-treat analysis with the last value carried forward and as a per-protocol analysis excluding the animals that did not have histologic scores from every intestinal region.
Results
Histopathology
Gastric mucosa
Gastric biopsies were available from 10 dogs (4–9 biopsies per animal,
Duodenal mucosa
Duodenal biopsies were available from 10 dogs (8–15 biopsies per animal,
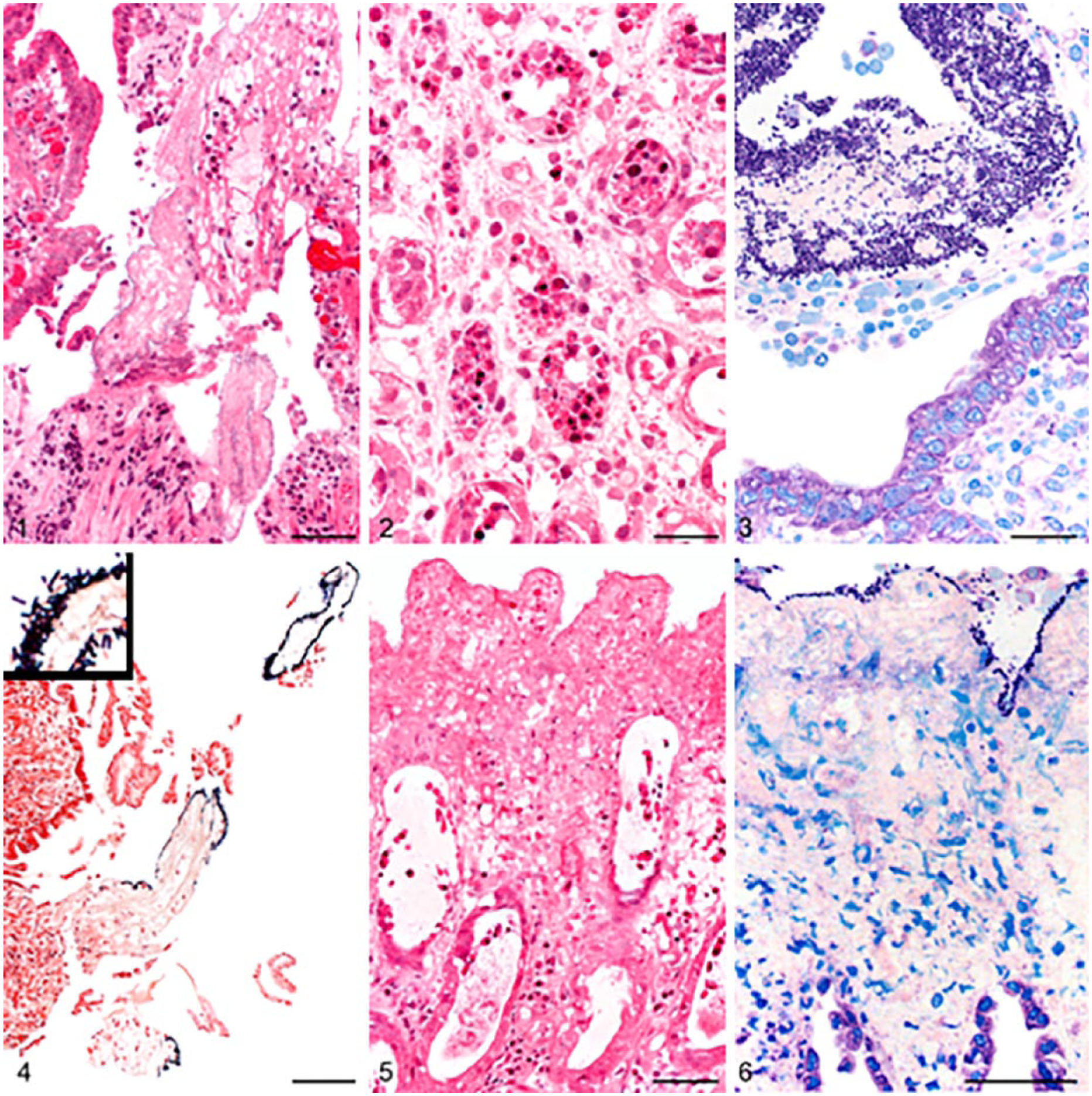
Intestinal biopsies of dogs with acute hemorrhagic diarrhea syndrome.
Ileum
Ileal biopsies were available from 8 dogs (2–9 biopsies per animal,
Colon
Biopsies of the colon were available from 9 dogs (2–10 biopsies per animal,
There was no significant difference in the necrosis scores of the duodenum, ileum, and colon with either per-protocol or intention-to-treat analysis (
Distribution of acute histologic lesions in the intestine of 10 dogs diagnosed with acute hemorrhagic diarrhea syndrome.
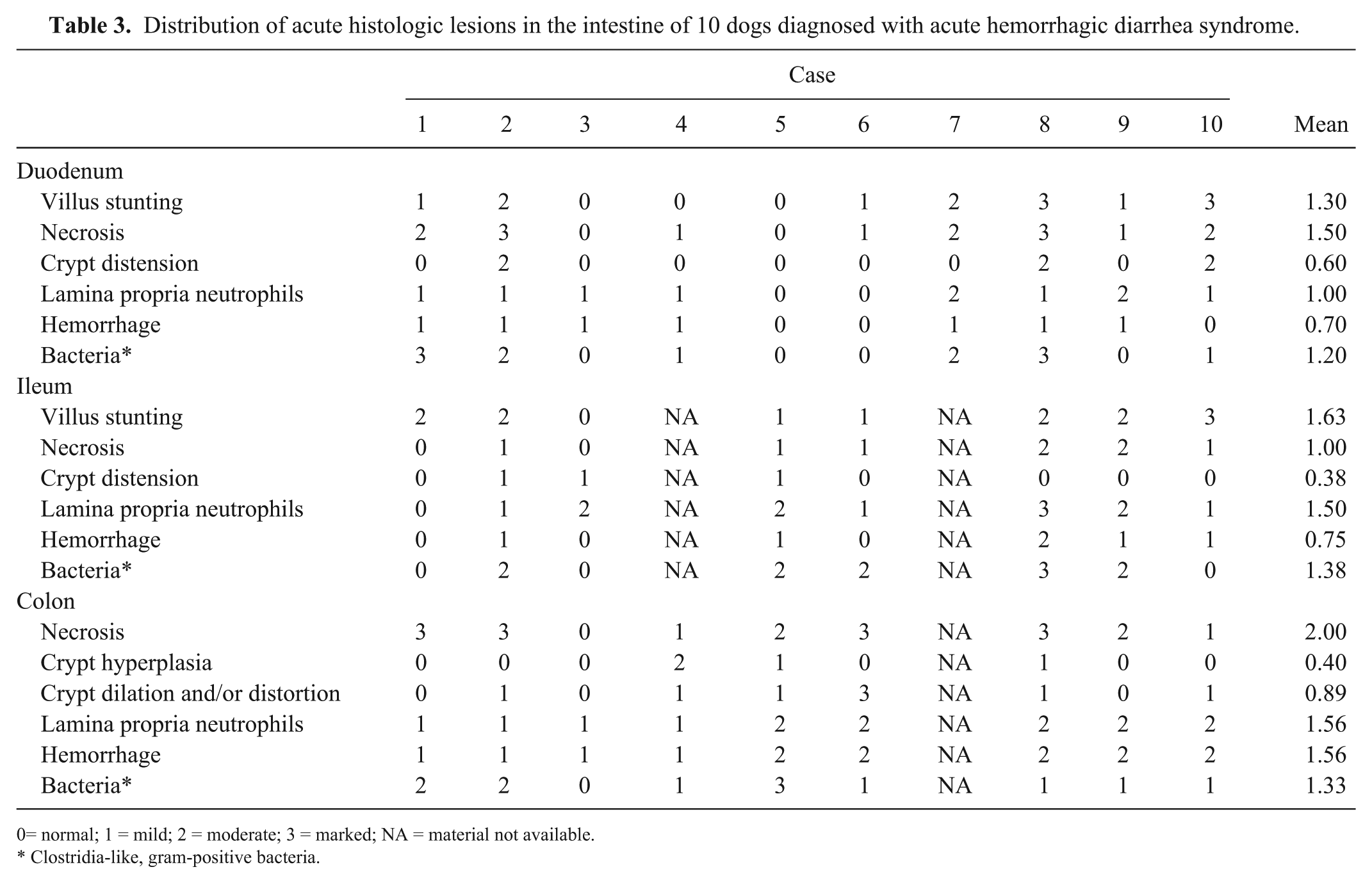
= normal; 1 = mild; 2 = moderate; 3 = marked; NA = material not available.
Clostridia-like, gram-positive bacteria.
Immunohistochemistry
Clostridium spp
Results of immunohistochemistry (IHC) using antibodies against
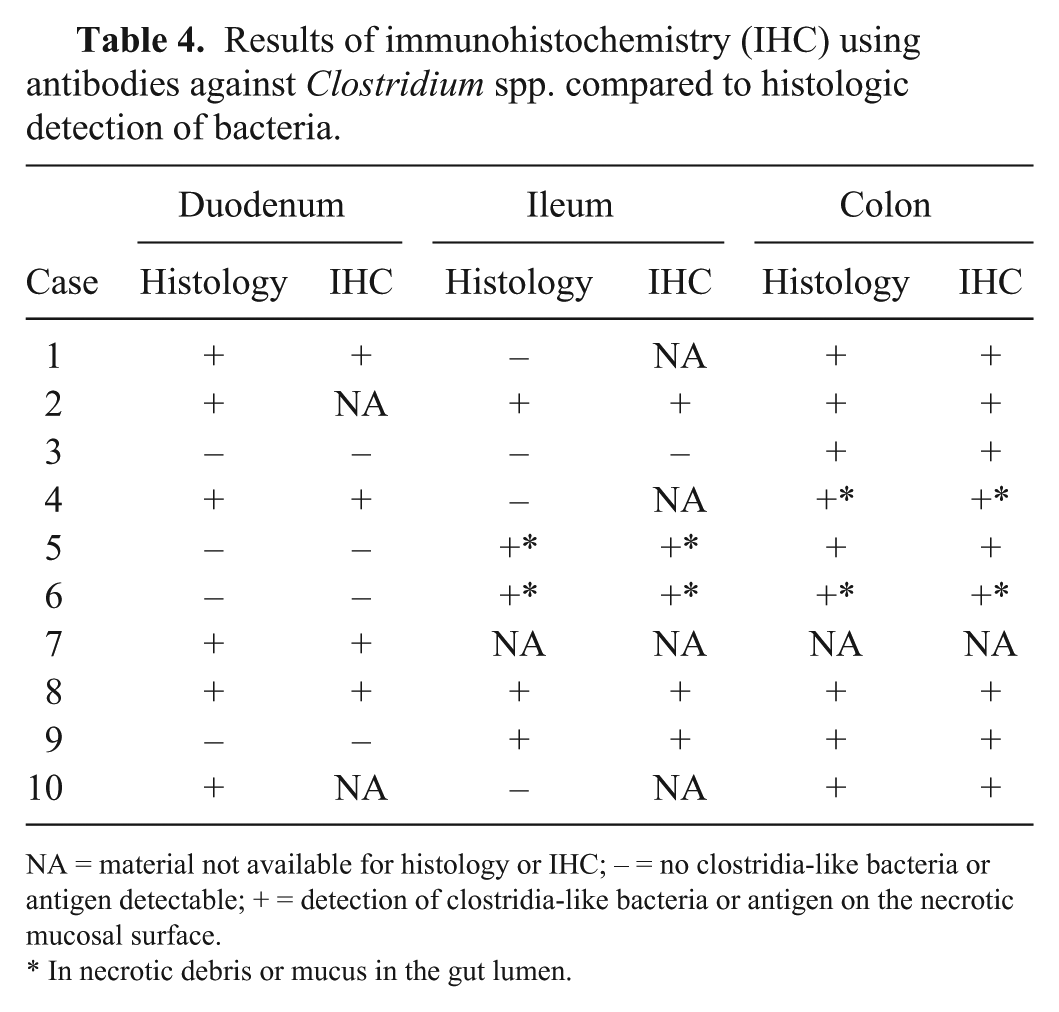
NA = material not available for histology or IHC; – = no clostridia-like bacteria or antigen detectable; + = detection of clostridia-like bacteria or antigen on the necrotic mucosal surface.
In necrotic debris or mucus in the gut lumen.
Parvovirus
Parvoviral antigen could not be demonstrated in the small intestine in any of the cases.
Bacterial culture, genotyping of C. perfringens isolates, and determination of genes encoding for toxin netF
Bacterial culture of duodenal biopsies revealed growth of bacteria in 8 of 9 dogs with AHDS and no bacterial growth in 1 dog with AHDS. MALDI-TOF typing of representative colonies resulted in
PCR results for the toxin-encoding genes in clostridial strains isolated from 5 dogs with acute hemorrhagic diarrhea syndrome and 2 dogs from the control group (C).

– = negative; + = positive.
Growth of bacteria was observed in 8 of 9 biopsies from control dogs. The following bacteria were identified:
In 2 dogs, a 10-y-old Beagle, in which endoscopy was performed to remove a foreign body, and in a 4-y-old Havanese, in which lymphangiectasia was diagnosed,
Discussion
Although AHDS is common and a clinical entity well-known to veterinarians, little is known about the pathology and pathogenesis of the syndrome. 6 Although it has been reported that untreated dogs can die from hypovolemic and endotoxic shock soon after onset of clinical signs, 6 dogs usually recover completely after supportive treatment.27,32 Histologic studies of AHDS have described lesions in single cases.8,21,23,24 Moreover, in these studies, all dogs died because of the disease, which usually only occurs in the most severe case or results from additional complicating factors. The low mortality rate and rapid autolysis of the intestine after death has prevented acquisition of sufficient material for detailed histologic characterization of the lesions.6,11
Our study extends previously published histopathologic findings 31 based on rapidly processed endoscopic biopsies of a larger number of acutely diseased dogs with AHDS that had a typical course of disease with recovery and convalescence within a few days. The bacteria associated with the lesions in these dogs are also characterized.
The major histopathologic feature was necrotizing and neutrophilic inflammation of the mucosa of the small and large intestine. Lesions were detectable in the entire intestinal tract with variable individual distribution. The colonic mucosa was affected in each case, together with at least one part of the small intestine. In previous reports, distribution of lesions has been variable: small and large intestine except very proximal and distal portions 13 ; whole small intestine to colon 6 ; isolated segment of jejunum, terminal jejunum 6 ; ileum, cecum, and proximal colon 6 ; whole intestine with highest degree in the colon. 21 In our study, we detected no significant differences in the necrosis scores of the duodenum, ileum, and colon (Table 3). Because samples were obtained through endoscopy, it was not possible to obtain biopsies from the jejunum, but it is likely from our study that all sections of the small and large intestines are affected.
Interestingly, significant differences in lesion severity were observed between animals. This might be explained by the different time points of presentation after onset of clinical signs and by the different times required for individual dogs to be stabilized before anesthesia for endoscopy was performed. These differences in times together with the ability for rapid regeneration of the intestinal mucosa could be responsible for the observed difference in severity of necrosis and neutrophilic infiltration found in the histologic examination. In previous AHDS studies using postmortem specimens,21,24 the gastric mucosa was occasionally involved. In our study, only chronic lesions, likely not related to AHDS, were found. Therefore, the most commonly used name “hemorrhagic gastroenteritis (HGE)” in dogs is not an adequate term for the syndrome. The term “acute hemorrhagic diarrhea syndrome”8,31 seems to be more appropriate.
Morphologic findings in the intestine resemble previously reported histologic findings in postmortem examinations of dogs with AHDS.13,21,24 In previous reports, villus necrosis was described without evidence of inflammation 13 and cryptal involvement,13,23 whereas in our study at least a mild acute inflammatory infiltration was present in all cases, and the cryptal epithelium was affected in most cases including cryptal necrosis and regeneration.
As also found in our study, other case reports have described bacteria on the necrotic mucosal surface of dogs with AHDS.21,23,24 In previous reports, lesions were suggested to be associated with clostridial infection.21,23,24 Given the close association of bacteria and epithelial lesions, a toxic insult is considered to be the most likely explanation for the mucosal damage. In contrast to a previous study,
24
we did not detect clostridial organisms in deeper parts of the lamina propria. Clostridia-like, gram-positive bacilli, and clostridial antigen were only demonstrated on the necrotic mucosal surface and in necrotic debris in the intestinal lumen in our study population. This difference between the dogs presented in our study and the fatal case of the disease might be related to the severity of clostridial overgrowth with isolation of
The etiology of AHDS has remained unclear for decades.
27
Over time, the suspicion of involvement of clostridia in the pathogenesis of AHDS has grown.8,11,21,23,24 Currently, a pathogenic role of clostridia is assumed, given that the demonstration of clostridia-like bacteria histologically and the detection of
Despite an extensive search, a common triggering event or predisposing factor could not be found in the 10 dogs examined. 31 As found in other studies, 18 most of the dogs were small breed (9 of 10 dogs ≤13 kg body weight) in our study. Previously, the high incidence of AHDS in dogs of small breed was associated with the common occurrence of pancreatitis in these breeds. 18 However, pancreatitis was not confirmed in the cases examined in our study.
Apart from
Given that specific virulence factors of the cultured
Some of the other bacteria (e.g.,
To identify the specific
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
