Abstract
The early clinical manifestations of pyoderma gangrenosum are not characteristic, it resembles other infectious skin lesions and is difficult to identify in the early stages. The exploratory surgery for direct observation of the superficial fascia of the suspected infection site for signs of infection, together with the collection of tissue samples for bacterial culture, combined with histopathological biopsy and clinical manifestations, was of significant value in the differential diagnosis of this disease and infectious skin lesions with similar manifestations. We introduce a patient who was admitted to hospital with redness and swelling of the left calf and foot for 7 days. The local appearance of the lesion resembled a carbuncle or early-stage necrotizing fasciitis. We performed surgical exploration for a definitive diagnosis but found no signs of infection, therefore, infection could be preliminarily ruled out. The disease was finally diagnosed as pyoderma gangrenosum, which was treated with hormone therapy and recovered after dressing change.
Introduction
Pyoderma gangrenosum does not show characteristic skin lesions during early stages of the disease. Epidemiological studies indicate the average age of PG onset is in mid-40s, with an incidence of a few cases per million person-years.1,2 We present a case of pyoderma gangrenous, focusing on the experience of early diagnosis.
Case presentation
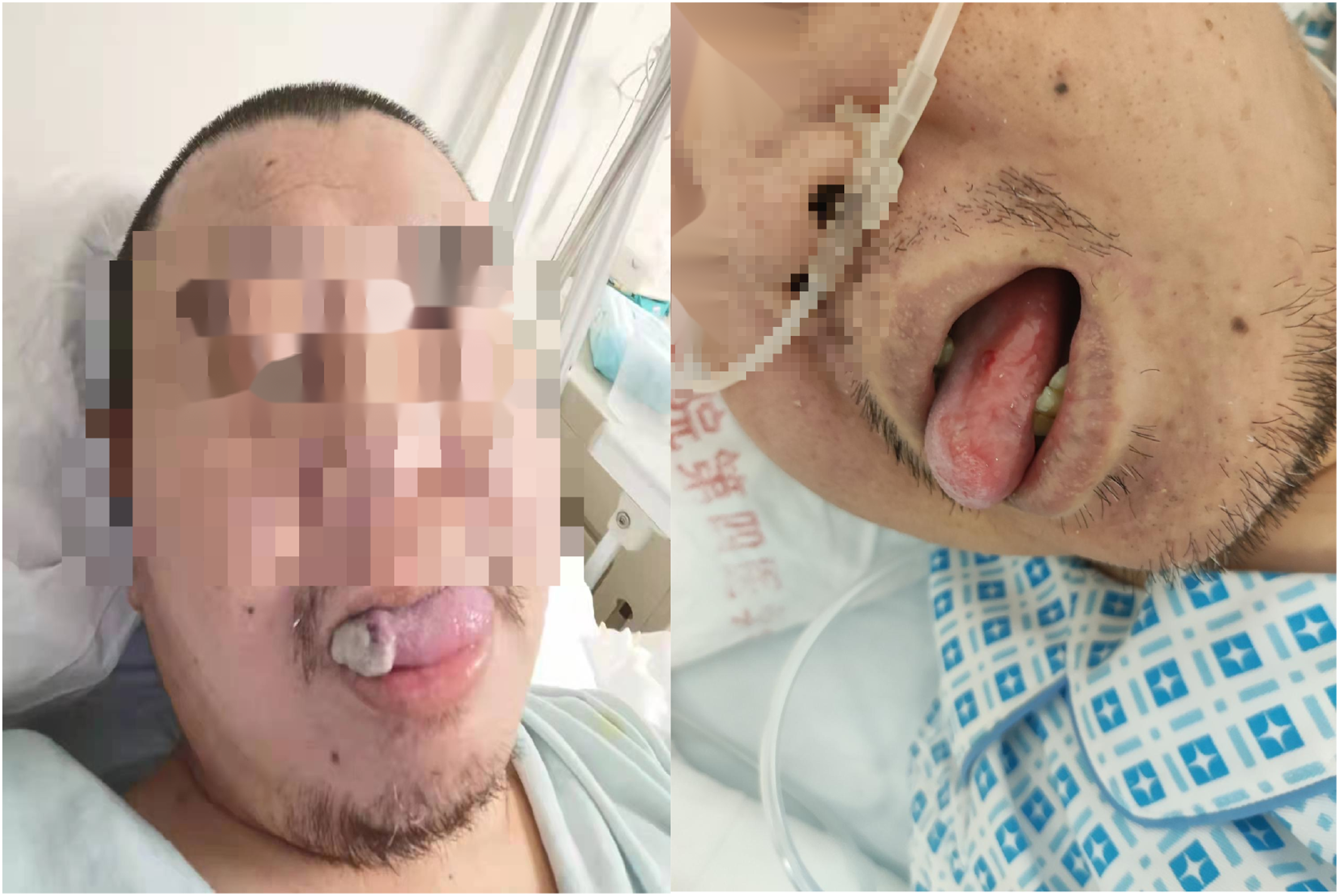
A 41-year-old male was admitted to hospital with a red and swollen area on dorsal area of the left foot and calf. The area became larger, redder, and harder, with severe pain (Figure 1). The patient had suffered from diabetes for 4 years, and his fasting blood glucose was controlled at 6 to 8 mmol/l by insulin and oral medications. He had no family history of genetic disease. The vital signs were stable, with a temperature of 36.7°C, pulse of 87 beats/min, respiration of 20 beats/min, and blood pressure of 133/83 mmHg. The back of his left foot and lower limb were red and swollen, and part of the skin was purplish and extremely tender with a blister in the center, which was 3 cm × 2 cm in size and contained turbid bloody fluid (Figure 2). Skin temperature of the left foot and leg was 39°C. The patient showed recurrent oral ulcers (Figure 3). Redness and swelling on the left calf and left foot dorsum. There were blisters in the red and swollen areas of the left calf and left dorsal foot with purplish-red skin borders on admission. Oral ulcers.

The main laboratory tests on admission were as follows: White blood cell count 6.65 × 10^9/L; Percentage of neutral cells, 87.5%; Hemoglobin, 104 g/L;Platelet count, 195 × 10^9/L.
Ultrasound examination showed there was no obvious abnormal liquid dark areas under the dermis around the skin blisters on the left foot and inner calf.
The patient was initially treated for skin-soft tissue infection with an intravenous infusion of cefuroxime sodium 1.5 g tid. On the day of admission, he developed chills and fever, with a temperature of 39.5°C. It did not return to normal untill administration of 10 mg dexamethasone sodium phosphate. Physical cooling and ibuprofen were ineffective. We changed the antibiotic to meropenem 2g tid and linezolid tablets 600 mg bid. An exploratory surgery on left foot and calf were performed with local anesthesia on the day following admission. The fluid in blisters of both lesions was clear and light yellow. After removal of the rotting skin, dark red edematous tissue was visible at base of the wound together with additional serous exudation. No purulent discharge was found in both superficial fascia and deep fascia (Figure 4). 2 mm × 2 mm skin tissue was removed and sent for pathological examination of tissue block and bacterial culture. Negative pressure wound therapy was used to cover the wound. The bacterial culture of the wound tissue was sterile and cultures of secretions from two wounds were negative. The pathological results showed the presence of severe suppurative inflammation with necrosis and bullae with abscess formation on the skin surface (Figure 5). Oral methylprednisolone was added at a dose of 36 mg/d. Meanwhile, considering the patient’s diabetes and high inflammatory index (PCT peaked at 0.568 ng/mL), antibiotics were not withdrawn until the PCT decreased to less than 0.1 ng/mL. Further tests were negative for systemic lupus erythematosus, mixed connective-tissue disease, and scleroderma, so other immune system diseases were excluded. Pyoderma gangrenosum was considered. He has received regular clinical follow-up for 6 months after diagnosis, and the wound healed 2 months after onset (Figure 6). There was no local recurrence. Local exploration of the lesions on the left calf and left dorsal foot showed dark red edematous tissue at the base, no purulent secretions in the superficial fascia, intact deep fascia, and no purulent secretions in the superficial and deep surfaces. Histological examination of the skin tissue of the left calf and left foot showed severe suppurative inflammation with necrosis, and bullae with abscess formation were seen on the surface. The left calf and left foot dorsum healed completely 2 months after onset.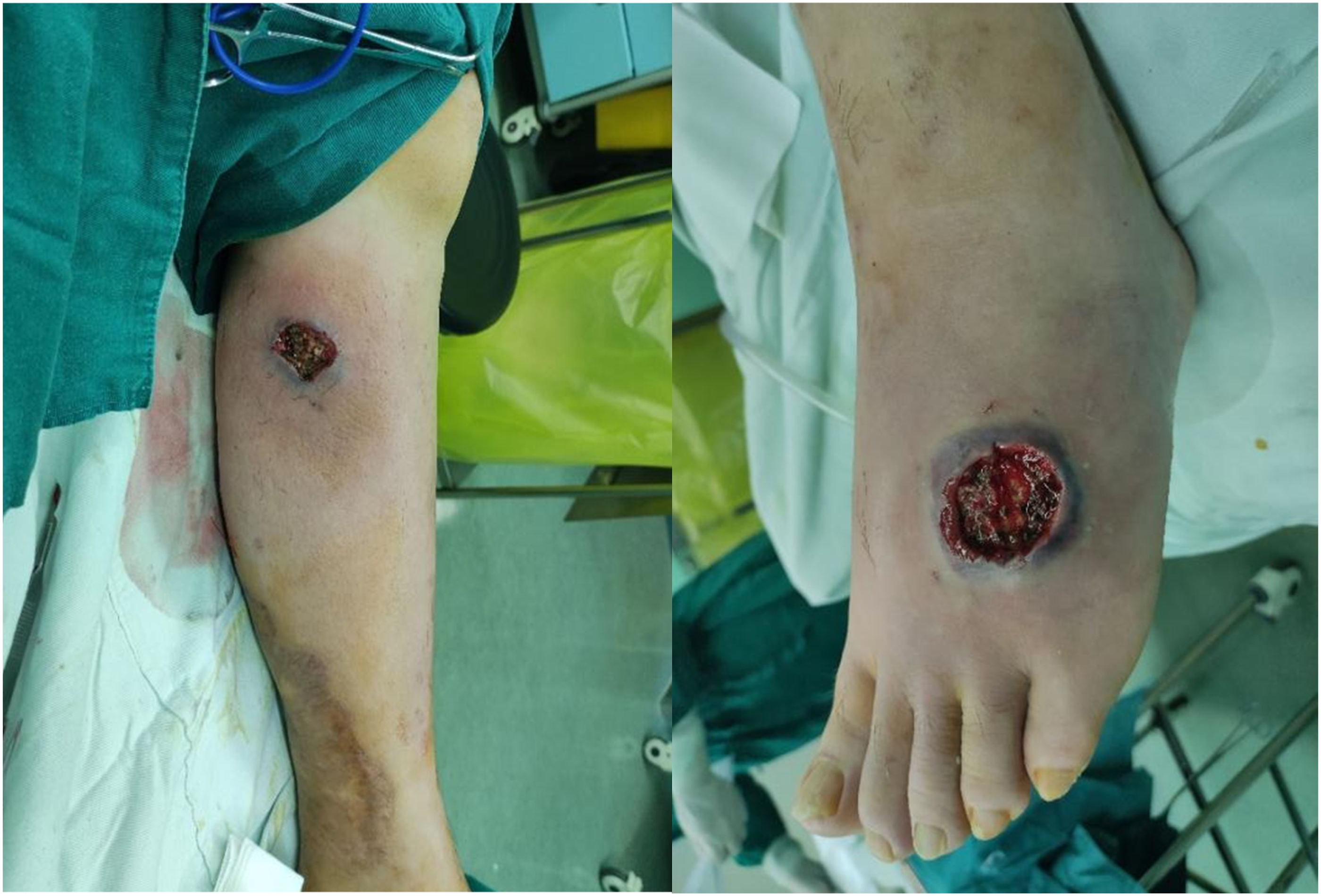


Discussion
The typical presentation of pyoderma gangrenosum is generally described as a rapidly progressive and painful lesion, irregularly shaped and with a mauve margin, together with a latent destructive necrotizing ulcer.
3
However, these typical manifestations were only observed when skin lesions were actively progressive. The main pathological manifestations include infiltration of lymphocytes or mixed inflammatory cells mainly neutrophils, at the edge of the ulcer.4–8 The patient came to our hospital at an early stage before skin lesions had progressed to the stage of typical one. The skin manifestations of redness, swelling, heat, and pain were very similar to those of a carbuncle or an early presentation of necrotizing fasciitis.
9
We performed surgical exploration to investigate the possibility of infection in the subcutaneous tissue where infection was likely to occur. No signs of infection were observed and thus bacterial infection was preliminarily ruled out. According to Paracelsus scoring system developed by Jockenhofer et al., it can be considered as gangrenous pyoderma.10,11 NPWT combined with systemic immunosuppression is a safe option for adjuvant treatment of wounds caused by pyoderma gangrenous. To cover the wound with skin grafts could be used to speed up healing.12–16 However, the use of debridement remains controversial and complete debridement may interfere with wound healing and requires great caution. Otherwise, this patient presents with mouth ulcers, skin erythema, and positive pathergy tests (Figure 7). Behcet's disease may also not be completely excluded.
17
Positive pathergy tests.
The author believes that appropriate local exploration is valuable for early differential diagnosis of infectious skin lesions such as carbuncles and necrotizing fasciitis. 18 The surgical exploration allowed a direct assessment of the presence or absence of infection in the superficial fascia. It provides a new diagnostic idea for diseases with refractory skin ulcers as main clinical manifestations.19,20 Of course, attention should be paid to the retention of healthy tissue to prevent the wound from continuing to expand and thorough debridement is not necessary to avoid damaging the capacity for tissue repair.
Conclusion
Surgical exploration may provide direct evidence of purulent infection of the superficial fascia. To provide a new diagnostic idea for diseases with skin ulcers due to necrotizing fasciitis or pyoderma gangrenosum as the main clinical manifestations.
Footnotes
Acknowledgments
The author appreciates all the staff of the burn and plastic surgery for their efforts in treating patients, and thanks the patient's families for their care at the same time.
Author contributions
Shen cong mo is the primary author and was responsible for data acquisition, analysis, interpretation and manuscript preparation. Li feng participated in data interpretation and manuscript editing. All authors have read and agreed to the published version of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical statement
Informed consent
Written informed consent was obtained from the individual included in this study. Written informed consent was obtained from all subjects before the study.
