Abstract
Pneumothorax is reported infrequently in marine mammals, most often secondary to physical or barometric trauma. We describe tension pneumothorax associated with verminous bronchopneumonia in a juvenile white-beaked dolphin stranded in Québec, Canada. Autopsy findings included a violent escape of air upon opening the pleural cavity, and white nematode-filled cystic nodules extending into the subpleural space and the lung parenchyma. One of these cysts had ruptured on the pleural surface. Numerous nematodes were observed in the lumen of main and secondary bronchi. Mediastinal lymph nodes were also enlarged. Helminths were collected and processed for molecular species identification. Histologically, verminous bronchopneumonia was marked by massive infiltration of the airways by polymorphonuclear and histiocytic cells surrounding adult and larval nematodes. Regional lymph nodes also contained focal infiltrates of polymorphonuclear cells associated with larvae. The nematodes were identified as Halocercus lagenorhynchi according to their morphology and molecular characterization. We attributed the death of this dolphin calf to tension pneumothorax secondary to a massive infection by the lungworm H. lagenorhynchi.
White-beaked dolphins (Lagenorhynchus albirostris) are elusive small odontocetes endemic to the North Atlantic and infrequently encountered in the Gulf of St. Lawrence (Canada). Their distribution spans from North Carolina to the western Barents Sea. Few valid population estimates exist, which explains why their true conservation status is poorly known. 12 Reports on morbidity and mortality in stranded white-beaked dolphins are scarce, and most focus on the North Sea. Bacterial pneumonia, gastritis, and emaciation are the most common postmortem findings in this species.1,29 A few cases harbored lungworms without signs of inflammation or other associated pathologic changes. 29
Lungworms of cetaceans are in the family Pseudaliidae. 22 They are a frequent yet often misunderstood cause of morbidity and mortality in stranded animals. Authors struggle to agree on the interpretation of these parasitic infections as incidental findings, contributing factors (44.8% of published studies), or primary cause of mortality (10.5% of published studies) in stranded cetaceans. 10 One of the hypotheses for this misclassification of the pathogenicity of lungworm infections is that verminous bronchopneumonia is usually complicated by bacterial infection, which is harder to document in the opportunistic collection and examination of stranded carcasses. Reported pathogenic effects of strongyles in hosts include bronchopneumonia, edema, vasculitis, thrombosis, and hemorrhage; more recent publications revealed the possible role of lungworms in the pathogenesis of air leakage.10,20
Air leak syndrome (
We describe the autopsy findings in a case of verminous bronchopneumonia that caused tension pneumothorax in a dolphin. A female white-beaked dolphin stranded near Blanc-Sablon (QC, Canada) in July 2022 and was submitted frozen (at –20°C) by the Quebec Marine Mammal Emergency Response Network to the Canadian Wildlife Health Cooperative Quebec regional laboratory (St-Hyacinthe, Québec, Canada) for postmortem examination. The autopsy was performed in September 2022 using standard techniques. 14 The relative age of the specimen was determined based on physical maturity, body length, developmental features, and gastric content. The carcass weighed 30 kg, measured 135 cm from snout to fluke notch, and was in a good state of preservation (code 2). 14 Full girth at the level of the axilla was 84 cm. Blubber thickness at midbody was 20 mm (ventral), 15 mm (lateral), and 22 mm (dorsal). The animal was classified as endomorphic (body condition score 4), per the adequate fat reserves, convexity ventrolateral to the dorsal fin, and well-developed epaxial musculature. 18 Rostral teeth were unerupted, the tip of the tongue harbored marginal papillae, and the umbilicus was fully healed and darkly pigmented.
Upon entry into the thoracic cavity from the dolphin’s right side, air hissing was immediately noticed after the puncture of the parietal pleura, followed by chest collapse, a finding characteristic of tension pneumothorax. Scattered throughout the lungs, predominantly in the subpleural space, were numerous light-colored nodules containing nematodes ( Fig. 1A ). Abundant in the caudodorsal lungs, were thin-walled, up to 4-cm, cystic nodules covering ~20% of the lungs. These often-coalescing nodules distorted the visceral pleura and contained adult nematodes surrounded by dense, yellow-white purulent material. On the cranioventral pleural surface of the right lung, a 2-cm bulla filled with pus and nematodes was ruptured. The parenchyma of both lungs was extensively affected by encysted nematodes on cut section ( Fig. 1B ). In the main and secondary bronchi of the upper and lower respiratory tract, large numbers of light-colored nematodes caused a heavy parasite burden ( Fig. 1C ). Peribronchial and mediastinal lymph nodes were also severely enlarged. All 3 gastric compartments contained large quantities of white flocculent fluid (milk curd), but no macroscopically detectable parasites. However, direct examination of the gastric contents revealed numerous comma-shaped nematode larvae. Lacteals of the small intestine were dilated and pale. No macroscopic abnormalities were detected in other organs.
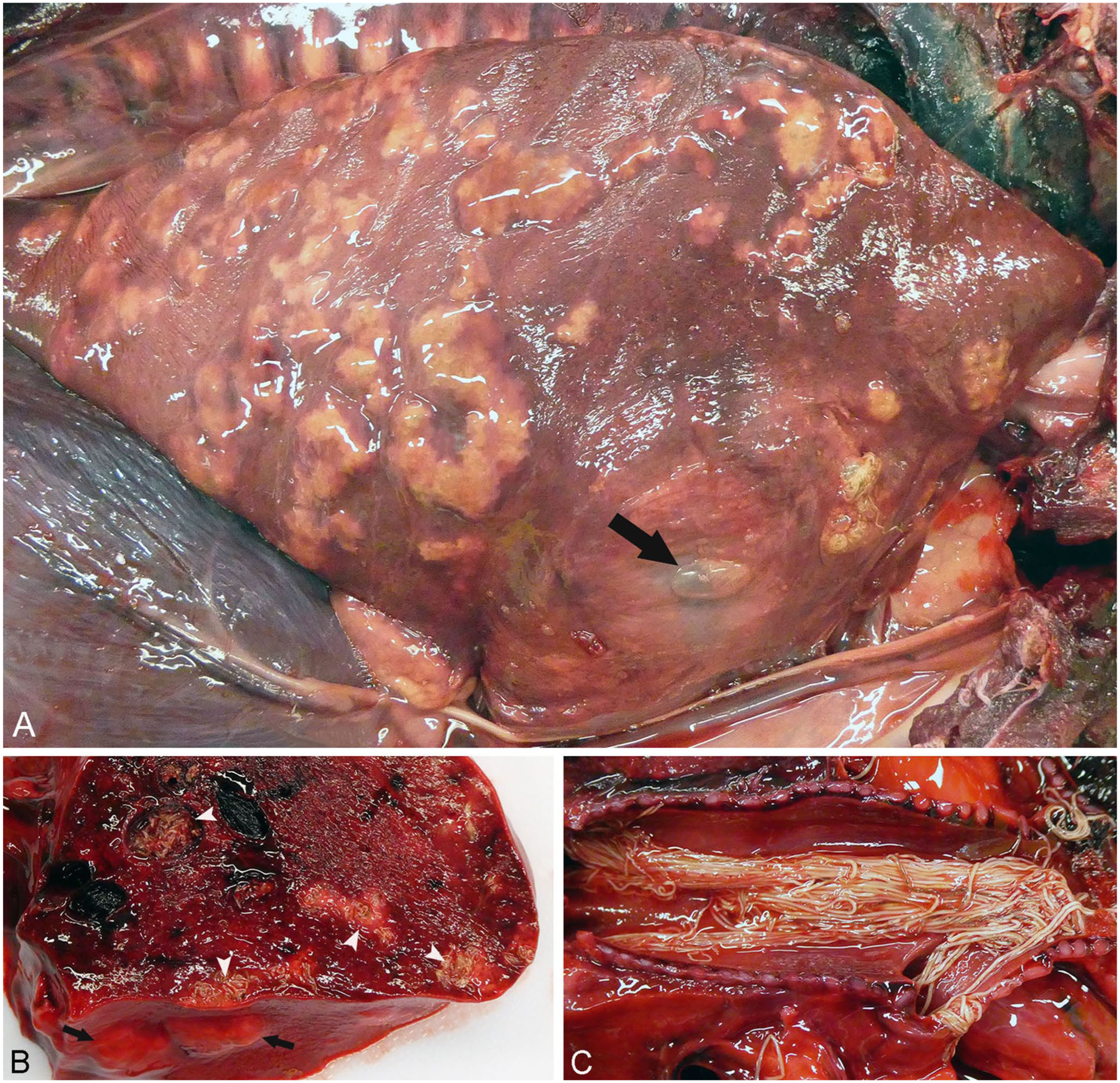
Severe verminous bronchopneumonia caused by Halocercus lagenorhynchi in a stranded white-beaked dolphin calf.
Routine samples of brain, skin, skeletal muscle, tongue, thyroid gland, trachea, lungs, heart, thymus, esophagus, forestomach, second and third gastric compartments, intestines, peribronchial, mediastinal, as well as mesenteric lymph nodes, liver, spleen, kidneys, and adrenal glands were fixed in 10% neutral-buffered formalin, processed routinely for histology, and stained with hematoxylin phloxine saffron. Lung samples were submitted for aerobic culture to investigate secondary bacterial pneumonia. Brain tissue was screened for highly pathogenic avian influenza A virus (H5N1) by RT-PCR. Whole nematodes and cystic lung lesions were frozen at –80°C and sent to the Atlantic Veterinary College (AVC; Charlottetown, Prince Edward Island, Canada) for molecular identification.
Histology of the lungs revealed marked multifocal-to-coalescent verminous bronchopneumonia, with massive infiltration by polymorphonuclear and histiocytic cells associated with numerous sections of adults and larval nematodes ( Fig. 2A ). The adult nematodes had platymyarian musculature separated by 2 symmetrical hypodermal chords, smooth cuticle, low cuboidal intestinal cells, opposed uteri, and reflexed ovaries ( Fig. 2B ).6,13 This morphology and the localization of these parasites were suggestive of the genus Halocercus (Rhabditida, Pseudaliidae). The large diameter (x̄ = 0.5 mm) and length (>30 cm) were further suggestive of H. lagenorhynchi. 2 In the regional lymph nodes, focal infiltrates of polymorphonuclear cells were associated with numerous filariform larvae.
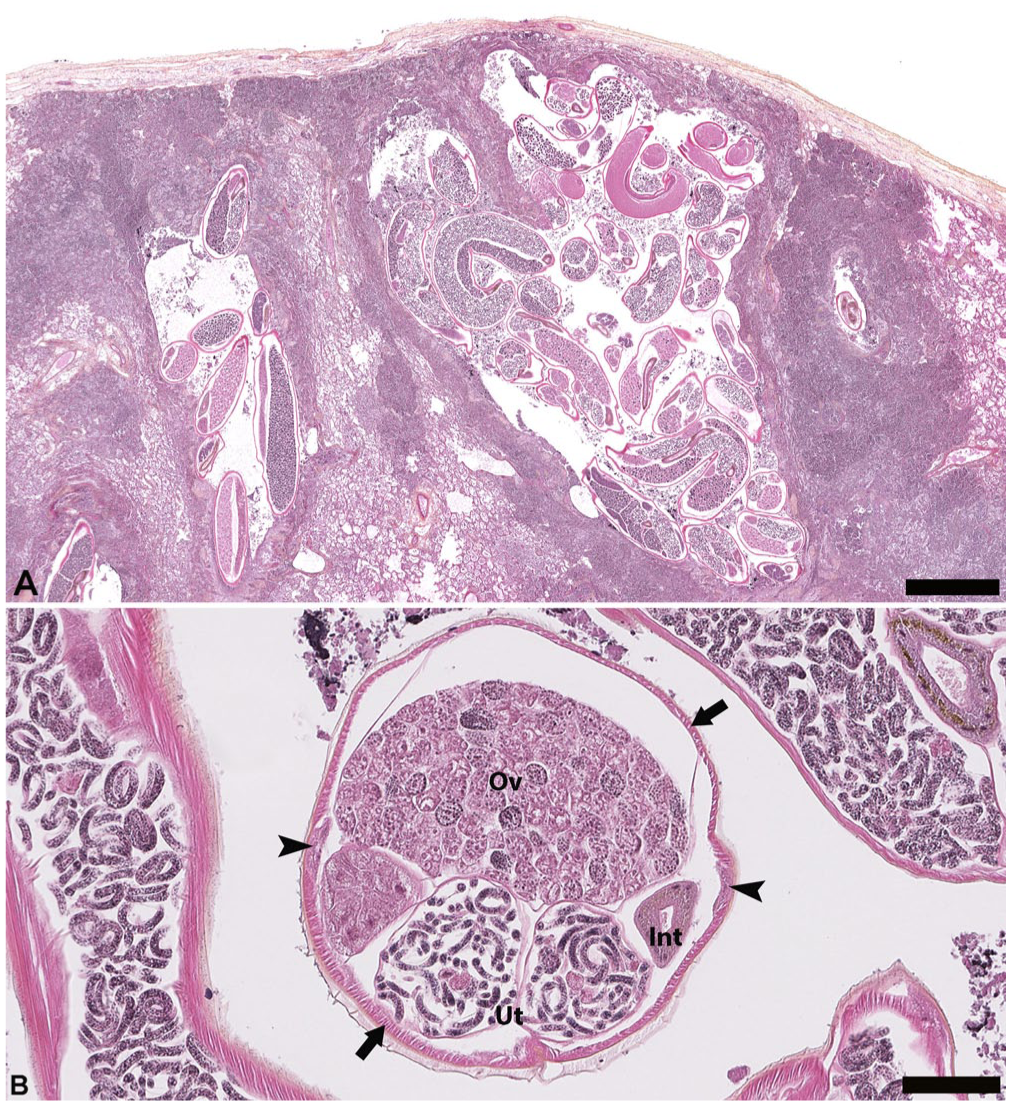
Verminous bronchopneumonia secondary to the nematode Halocercus lagenorhynchi in a stranded white-beaked dolphin calf. Hematoxylin–phloxine–saffron stain.
All bacteriology and virology results were negative. At the AVC, DNA was extracted from the unfixed frozen lung tissue (DNeasy blood and tissue kit; Qiagen) and nucleotide sequences of 2 partial regions of the large subunit (
Death was attributed to acute fatal tension pneumothorax, which was induced by a pleural tear. The tear resulted from massive cystic purulent-to-histiocytic verminous bronchopneumonia caused by an infection with H. lagenorhynchi. Regional verminous suppurative lymphadenitis was also present. Other authors have characterized similar histologic lesions in the lungs as active and subacute verminous infections, which is in line with the proposed temporality of disease in this case. 9 The death of this dolphin was therefore the result of a severe parasitic infection without bacterial complication.
Developmental features, morphometrics, and diet indicated that this white-beaked dolphin was a nursing juvenile, aged no more than 3 mo.11,12,19 Recent consumption of milk, which is very rich in fat, probably explains the dilation of intestinal lymphatic vessels. Consuming fatty foods gives a milky appearance to lymph passing through mesenteric vessels, making these lacteals unusually conspicuous. 30 The recovery of the carcass in July is in line with the summer (May to August) birth season reported for this species. 11
Little is known about the life history of cetacean lungworms, which are all in the family Pseudaliidae. Direct transmission of infective larvae, although possible, is unlikely in cetaceans due to dispersal in the vast marine environment. Transmission via paratenic hosts in the diet seems more plausible, but no intermediate hosts have been implicated in field data. However, many reports of unidentified nematode larvae from fish prey could hypothetically be pseudaliids. Large studies in odontocetes of all ages suggest that some strongyle species are acquired post-lactation, when calves begin to feed on prey, corroborating the hypothesis of horizontal transmission via the food chain. 22 The diet of white-beaked dolphins is dominated by cod (Gadidae); hence, researching the detection of infective larvae in this prey species may provide more insight into the life cycle of H. lagenorhynchi in the North Atlantic. 17 Prenatal and neonatal infections for 3 species of Halocercus spp., including H. lagenorhynchi, have been documented in bottlenose dolphins (Tursiops truncatus), orcas (Orcinus orcas), and striped dolphins (Stenella coeruleoalba).7,9,27 These reports offer convincing evidence of vertical transmission of nematodes from this genus, whether in utero from the placenta or transmammary through lactation. Because the stranded white-beaked dolphin calf in our report appeared to be milk-fed, the transmission pathway of H. lagenorhynchi was likely vertical. Suckling dolphin calves have an immature immune system, making them more susceptible to mortality from high lungworm burden.
Interestingly, despite being originally associated with the white-beaked dolphin, H. lagenorhynchi is now reported most often in other delphinids, such as the bottlenose dolphin, the striped dolphin, and the short-beaked common dolphin (Delphinus delphis).9,15,28,32,33 Lungworms with a broader host diversity can evolve to become more pathogenic, as the reliance on host–parasite coevolution becomes less important for survival. In addition to the aforementioned species, infection by H. lagenorhynchi has been documented in the Indo-Pacific bottlenose dolphin (T. aduncus), the white-sided dolphin (L. acutus), and the spotted dolphin (S. frontalis). 10 H. lagenorhynchi is particularly pathogenic as it invades the bronchi and pulmonary parenchyma and facilitates secondary bacterial infections. 22
We retrieved no cases of pneumothorax secondary to Halocercus spp. infection in a search of Google, PubMed, CAB Direct, and Web of Science, using the search terms “pneumothorax”, “odontocete”, “dolphin”, “Halocercus”, “lungworm”, or “nematode”, suggesting that this condition has not been reported previously in odontocetes. Descriptions of pneumothorax associated with lungworm infection in cetaceans are scarce, with only 3 cases reported to date. 21 This clinical presentation offers further insight into the pathogenicity of Halocercus spp. in dolphins and is complementary to the described occurrence of pulmonary atelectasis.8,9 Other lungworm species implicated in ALS in marine mammals are Otostrongylus circumlitus, Parafilaroides decorus, and Skrjabinalius guevarai.20,21
The prevalence of H. lagenorhynchi varies with geographic location and host species. 26 No data exist on the prevalence of this parasite in dolphins from the Gulf of St. Lawrence, and a detailed stranding report of this parasite for this geographic area has not been reported previously. At the time of publication, only 12 stranding events involving white-beaked dolphins had been submitted to the Canadian Wildlife Health Cooperative, which is responsible for wildlife health surveillance in this region. Our report offers useful information on a singular cause of death for this infrequently encountered species.
Footnotes
Acknowledgements
We thank and acknowledge the staff of the Centre québécois sur la santé des animaux sauvages (CQSAS), and Dr. Nina Germitsch and Haili Wang for their participation in processing this case. Our case was presented as a poster at the 71st Annual International Conference of the Wildlife Disease Association on July 29–August 4, 2023, in Athens, GA, USA.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
We received no financial support for the authorship and/or publication of this article. Our work was financially supported in part by the Government of Quebec as part of the Stratégie québécoise sur la santé des animaux sauvages.
