Abstract
Canine distemper virus (CDV) is a widespread morbillivirus that causes subclinical to fatal infections in domestic and wild carnivores. Raccoons (Procyon lotor) are CDV reservoirs and suffer from associated disease. Aspects of pathogenesis may lead to difficulty in the interpretation of commonly used testing modalities, such as reverse-transcription real-time (RT-rt)PCR and immunohistochemistry (IHC). The reliance upon such tests is greater for wildlife, which are often submitted as carcasses with no clinical history. We compared CDV RT-rtPCR results to immunohistochemistry (the gold standard) in tissues from 74 raccoons. These tests had high kappa agreement (lymph node: 0.9335; lung: 0.8671) and a negative correlation between IHC score and threshold cycle (Ct) value for lymph node and lung (Spearman rank correlation coefficient [rs] = −0.8555 and −0.8179, respectively; p < 0.00001). An RT-rtPCR Ct value of 30 in lung and lymph node with sensitivity and specificity of 92.3 and 92.6% and 86.8 and 96.4%, respectively, was suitable for determining CDV involvement. Conjunctival swabs provide an alternative for distemper diagnosis, as there was a strong correlation between Ct values of conjunctival swabs and tissues (rs = −0.8498, p < 0.00001, n = 46). This information will aid in more efficient and accurate diagnoses in individuals, small-scale outbreaks, and epidemiologic investigations in wildlife.
Canine distemper, caused by canine distemper virus (CDV; order Mononegavirales, family Paramyxoviridae, genus Morbillivirus, species Canine morbillivirus), is a common and ubiquitous disease of domestic and wild carnivores. Wildlife are not only a reservoir for CDV, but many species are susceptible to disease, including members of the families Ailuridae (e.g., red panda), Canidae (e.g., red fox), Felidae (e.g., lions), Hyaenidae (e.g., hyenas), Mustelidae (e.g., mink), Procyonidae (e.g., raccoons), Suidae (e.g., peccaries), Ursidae (e.g., black bears), and Viverridae (e.g., civets).1,3 The virus is easily transmitted and may cause respiratory, gastrointestinal, and neurologic disease, the latter of which requires differentiation from rabies virus infection.2,4,12 The virus may be shed subclinically in bodily secretions during prolonged incubation periods, likely augmenting the potential for population-level effects during outbreaks. 3 Further, CDV has undergone host adaptation and exhibits cross-species transmission among an expanding mammalian host range. The importance of monitoring for CDV infections among wildlife is further highlighted by evidence that strains isolated from North American wildlife within the last 10 y are genetically distinct from vaccine strains.3,15,20
Understanding regional and large-scale epidemiologic patterns and thus, the potential impacts of CDV infection on wildlife, depends upon accurate diagnoses, which is challenging given the complex pathogenesis.4,12,20 This complexity is accompanied by a wide variation in clinical presentation, host immune response, and disease duration and outcome. 2 Further, such variation may impede the ability to diagnose distemper in wildlife involving commonly used methods, such as reverse-transcription real-time (RT-rt)PCR and immunohistochemistry (IHC).2,6,9,16 In addition, wildlife cases often lack a clinical history (e.g., clinical signs and duration of disease) that would otherwise aid in interpretation of laboratory tests. Various methods, from arbitrary to analytical, are used to determine threshold cycle (Ct) values for RT-rtPCR; however, these values may or may not be related to clinical disease or cause of death. Therefore, species- and location-specific validated thresholds for RT-rtPCR help to more accurately interpret test results. 5
Our study provides a comparison of 2 routine and widely available postmortem CDV test modalities in a common North American wildlife species, the raccoon (Procyon lotor), to improve the value of these tests. In addition, CDV detection in conjunctival swabs was evaluated as a potential rapid alternative to tissues for field sample collection in raccoons. Our objectives were to: 1) compare the results of CDV RT-rtPCR to IHC (as the gold standard) in tissues; 2) establish an RT-rtPCR Ct value relevant to the specified dataset; and 3) to assess the utility of RT-rtPCR of conjunctival swabs in comparison with commonly tested tissues (e.g., lung and lymph node) in the diagnosis of distemper in raccoons in Ontario.
Samples of caudal lung and mesenteric lymph node were collected from raccoon carcasses from January 2014 to November 2015, and conjunctival swabs from July 2015 to June 2016. Carcasses originated in southern Ontario, Canada, and were provided to the Ontario-Nunavut node of the Canadian Wildlife Health Cooperative by government agencies (e.g., the Ontario Ministry of Natural Resources and Forestry), wildlife clinics, wildlife control professionals (including roadkill), licensed fur trappers, and private citizens.
Duplicate samples of each tissue (~2 × 2 × 1 cm) were collected aseptically; one sample was placed into 2.0 mL of phosphate-buffered saline (PBS) and the other into 10% neutral-buffered formalin. Samples in PBS were vortexed for 30 s, centrifuged at 3,750 × g for 10 min, and the supernatant decanted for testing. Conjunctival swabs were collected by swabbing the inside of the lower conjunctiva of both eyes from the medial to lateral canthus with cotton-tipped applicators, which were immediately placed into 3.0 mL of universal virus transport medium for viruses, chlamydia, mycoplasmas, and ureaplasmas (BD Biosciences, Sparks, MD). Tissue supernatants and conjunctival swabs were stored at −80°C until tested.
Total nucleic acids were extracted from lung, lymph node, and conjunctival swab samples for subsequent RT-rtPCR testing. Lung and lymph node were tested either individually (n = 61) or pooled (n = 13). Nucleic acid extraction and RT-rtPCR methods, including primers and probe sequences, were used as described previously. 7
Formalin-fixed tissues were routinely processed for histopathology, and unstained slides of lung and lymph node were deparaffinized and rehydrated for IHC as described previously. 7 Briefly, the anti-CDV monoclonal primary antibody (CDV-NP-0.5, VMRD, Pullman, WA) was detected through a goat anti-rabbit horseradish peroxidase–labeled polymer detection system (EnVision+ System, Dako, Burlington, Ontario, Canada); NovaRed (Vector Laboratories, Burlington, Ontario, Canada) was used as a substrate chromogen and Harris hematoxylin as a counterstain. Positive controls consisted of lung and lymph node from a known CDV-positive raccoon. All laboratory tests were conducted by professionals at the Animal Health Laboratory at the University of Guelph (Guelph, Ontario, Canada).
IHC served as the gold standard method to determine CDV infection status. Immunoreactivity in lung and lymph node sections was subjectively scored according to the pattern of immunoreactive cells in tissue sections examined, with “0” for no immunoreactivity detected in cells, “1” for widely scattered foci involving small numbers of immunoreactive cells in a single tissue (i.e., lung or lymph node), “2” for widely scattered, variably sized foci of immunoreactive cells in both tissues (i.e., lung and lymph node), and “3” for larger foci of immunoreactive cells in both tissues.
For determination of the RT-rtPCR Ct values to be used in subsequent analyses, tissues with an IHC immunoreactivity score of 1–3 were collectively assigned a positive value of “1.” Tissues with no reactivity were considered negative and remained as “0.” As the gold standard, IHC data were compared to corresponding RT-rtPCR Ct values for tissue and swab samples from all raccoons for which IHC and RT-rtPCR were performed on both lung and lymph node (i.e., excluding pooled tissue samples tested by RT-rtPCR). These data were plotted on a scatter plot graph (Excel, Microsoft, Redmond, WA) with RT-rtPCR Ct values on the y-axis and the IHC designations of “1” and “0” on the x-axis. Samples with no amplification within 45 cycles were considered negative and assigned a Ct value of 45.01 for the purpose of the analyses. These data were used to derive a linear equation (y = −17.8x + 38.5) to determine a preliminary threshold Ct value as the Ct value on the y-axis that corresponded to an IHC score of 0.5. The value of 0.5 was considered the mid-point between positive (i.e., “1”) and negative (i.e., “0”) values, and was used to distinguish CDV-positive RT-rtPCR (i.e., ≥0.5) and CDV-negative (i.e., <0.5) samples. The resulting threshold Ct value was subsequently used as the determinant of CDV infection status.
To confirm the calculated Ct value, the sensitivity, specificity, 5 and kappa statistic (κ) for test agreement were calculated for each Ct value, represented by whole numbers from 24 to 35 for both lung and lymph node. Additionally, the correlation between the assigned IHC scores (0–3) of tissues (i.e., lung, lymph node, and pooled lung and lymph node) and the corresponding Ct values was measured using the Spearman rank correlation coefficient (rs). Coefficient rs was also used to assess the correlation between Ct values of the paired conjunctival swabs and pooled tissue samples.
Tissues from 74 raccoons were tested by both RT-rtPCR and IHC. Using the linear equations created by comparing RT-rtPCR Ct values to the binomial IHC results by tissue, we determined that the calculated threshold Ct value for CDV positivity in raccoon tissues was ≤ 29.6. Using the gold standard (i.e., IHC) and a rounded (to the nearest whole number) Ct value of ≤ 30, sensitivity and specificity for RT-rtPCR of raccoon lung were 92.3% and 92.6%, and 86.8% and 96.4% for lymph node (Fig. 1A). The κ agreement test results between IHC and RT-rtPCR were 0.9335 for lymph node and 0.8671 for lung (Fig. 1B). Additionally, there was a strong negative correlation between the assigned IHC score (0–3) and Ct value for both lymph node and lung (rs = −0.8555 and −0.8179, respectively, with p < 0.00001 for both; Fig. 1C).
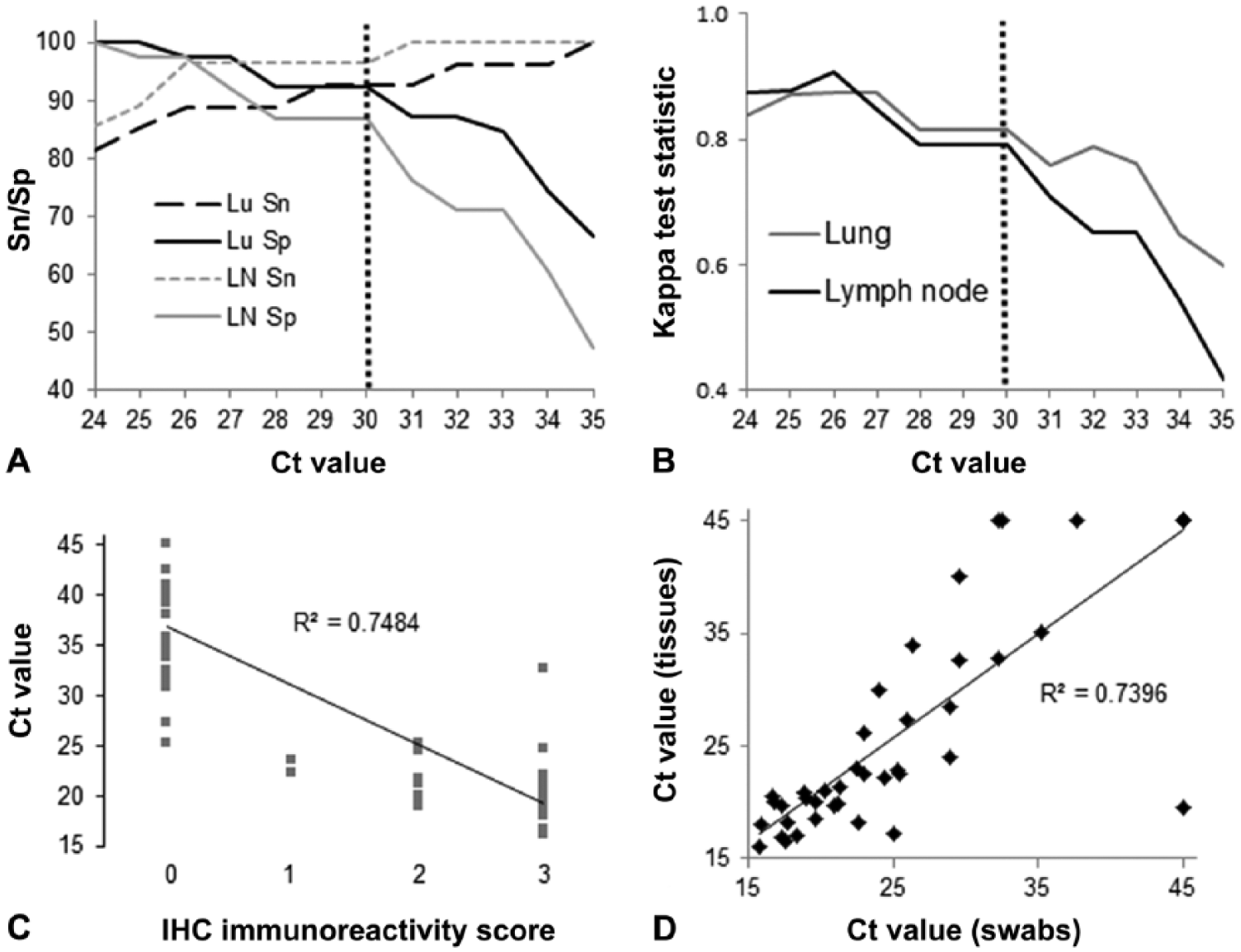
Representations of canine distemper virus (CDV) reverse-transcription real-time (RT-rt)PCR and immunohistochemistry (IHC) data for tissue (lung and lymph node) and conjunctival swab samples from raccoons in Ontario, Canada. Samples with no replication within 45 cycles were considered negative by RT-rtPCR and assigned a threshold cycle (Ct) value of 45.01 (panels C and D).
Using the calculated threshold Ct value of ≤ 30, 32 of 74 (43%) raccoons tested were CDV-positive by IHC and 35 of 74 (47%) were CDV-positive by RT-rtPCR for at least 1 sample (i.e., lung, lymph node, or pooled lung and lymph node). Among CDV-positive raccoons, lung and lymph node RT-rtPCR results were generally in agreement. Among the 61 raccoons for which both lung and lymph node were tested individually, 28 (46%) had 1 or more CDV-positive tissues. Pooled lymph node and lung were CDV-positive by RT-rtPCR in 7 of 13 (54%) raccoons.
Paired sample sets of conjunctival swab and pooled lung and lymph node were tested by RT-rtPCR for 46 additional raccoons. Among these, 29 (63%) raccoons were CDV-positive by RT-rtPCR for both sample types and 13 (28%) raccoons were CDV-negative for both sample types. Sample sets from the remaining 4 raccoons had discordant test results between sample types, 3 of which were swab-positive but tissue-negative, and 1 of which was tissue-positive but swab-negative. A strong correlation existed between Ct values of conjunctival swab and pooled tissue samples (rs = −0.8498, p < 0.00001; Fig. 1D).
Canine distemper outbreaks have been increasing in frequency on multiple continents, involve an expanding host range, and are affected by mutation-induced virulence changes.2,3,8,10,18,19 Monitoring of CDV-associated outbreaks to assess health effects and epidemiologic patterns in wildlife requires accurate interpretation of laboratory test results.6,16 This is especially true given the lack of clinical history for many wildlife cases, necessitating the use of a secondary test to confirm distemper diagnoses. Results from our study indicate that 2 commonly used postmortem CDV tests, RT-rtPCR and IHC, were in strong agreement for samples from raccoons in Ontario, Canada. Although IHC is useful to demonstrate viral antigen in situ, RT-rtPCR proved a sensitive and specific method to detect CDV infection.
RT-rtPCR for CDV is an important test modality because a variety of easily collected samples (e.g., lung and lymph node) may be used to provide rapid, high-throughput, and economical results. The results herein suggest that, for CDV epidemiologic surveys in Ontario raccoons, RT-rtPCR may be used as the sole indicator of CDV status with a threshold Ct value of 30. This designated threshold is laboratory-specific; such calculated thresholds may vary geographically and by wildlife species, diagnostic laboratory, and other factors. 5 In our study, animals were collected from the same region and time frame to minimize the potential effects of differences in regional strain evolution.
The complex pathogenesis of CDV, along with taxonomic and individual host factors and viral strain differences, contribute to the diagnostic challenge it presents in both wild and domestic animals.4,15,20 The virus may replicate, infect, and persist in tissues, manifest as neurologic disease days to months after infection, and be shed for prolonged periods.4,13,14 Viral clearance is highly dependent upon host humoral immune response, which may commonly be compromised in free-ranging wildlife. 15 This complex and variable interplay between virus and host may lead to disparaging diagnostic results; for example, only 45% of 54 mustelids positive for CDV by IHC exhibited characteristic histologic lesions (e.g., encephalitis). 19 In naturally CDV-infected raccoons and gray fox (Urocyon cinereoargenteus), mean quantitative RT-rtPCR Ct values were not consistently associated with microscopic visualization of inclusion bodies in tissues (e.g., brain, lung, kidney, lymph node), whereas pooled tissues from both clinically (86%, n = 36) and subclinically infected animals (55%, n = 22) tested CDV-positive by RT-rtPCR. 15
Species- and region-specific parameters are needed for more accurate CDV etiologic and disease diagnoses in wildlife based on the evolving virulence and host range of infections across a wide geographic range.5,12 Domestic dogs are a major reservoir, and the increase in CDV epidemics worldwide is in part attributed to continued urbanization and the concomitant rise in human and corresponding domestic dog populations. 12 Further, CDV maintenance within wildlife hosts hampers disease eradication and fuels virus spillover and spillback between wild and domestic animal hosts (e.g., raccoons and dogs).3,11,16 In 2016, a novel CDV strain (proposed America 4 lineage) that was genetically distinct from vaccine strains was isolated from raccoons and gray fox in Tennessee, and CDV-associated disease has been reported in vaccinated dogs.14,15 In addition, subclinically infected raccoons had simultaneous coinfections with multiple CDV strains, supporting the potential for within-host genetic recombination. 15 Therefore, the emergence and evolution of CDV strains originating from wildlife should be considered in vaccination protocols for domestic animals and captive wildlife.
Conjunctival swabs are considered a suitable sample choice for early CDV detection by RT-PCR in dogs with respiratory disease. 17 In our study, conjunctival swabs proved a reliable alternative to tissues for RT-rtPCR testing in raccoons based on highly correlated test results. The collection of conjunctival swabs from wild raccoon carcasses requires less time and effort, reduces personnel training and risk of contamination, and eliminates the need for carcass transport and handling. From a wildlife disease surveillance perspective, in addition to reliable and economical test results, rapid sample collection that precludes the need to open carcasses or transport them from the field is an important factor.
Footnotes
Acknowledgements
We thank the veterinarians, clinicians, pathologists, biologists, and public for the submission of carcasses, especially Wildlife Technologies Inc. Erin Harkness, David Cristo, and Lenny Shirose of the Canadian Wildlife Health Cooperative and Emma Delitala, Sarah Brisson, and Elizabeth Hartnett of the Ontario Veterinary College provided logistical support. Josepha DeLay of the Animal Health Laboratory, University of Guelph provided immunohistochemistry expertise.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This study was supported by the Canadian Wildlife Health Cooperative and the Ontario Ministry of Natural Resources and Forestry.
