Abstract
Abnormal sudden mortality of 2 goats and 10 sheep occurred in a 27-head flock in Verona province (northeastern Italy). In the 24 h before death, animals had acute ataxia, lateral recumbency, spastic convulsions, and dyspnea. Autopsy of 4 animals was performed at the Verona Veterinary Diagnostic Laboratory of the Istituto Zooprofilattico Sperimentale delle Venezie (IZSVe; Italy). In the forestomachs, abundant fibrous material and numerous seeds were observed. Bacteriologic, parasitologic, and histologic investigations were carried out; samples of rumen contents and liver were analyzed by direct analysis real-time high-resolution mass spectrometry (DART-HRMS), which allows rapid screening of toxic substances. The combination of DART-HRMS and liquid chromatography high-resolution tandem MS (LC-HRMS/MS) confirmed the acute intoxication and provided insights into the clinicopathologic findings due to the ingestion of Chimonanthus praecox (wintersweet), a plant species that contains alkaloids including calycanthine, which is known to be toxic in several domestic species, including small ruminants.
Chimonanthus praecox, commonly known as wintersweet, is a bush native to China, popular worldwide as an ornamental plant due to its distinctive winter flowering. 12 This plant belongs to the Calycanthaceae family, whose members contain various alkaloids, including calycanthine, described as a powerful convulsant. 2 Calycanthine acts centrally, interfering with the release of, and receptors for, gamma-aminobutyric acid (GABA), a major inhibitory neurotransmitter.2–4 The toxicity of calycanthine is known in ruminant pathology, but few descriptions of accidental intoxication by wintersweet in ruminants exist in the literature. Clinically, limb rigidity, hyperesthesia, dyspnea, and death were described in a case involving 3 cattle; ataxia, tetanic seizures, dilated pupils, and hyperesthesia, with elevated heart and respiratory rates, were described in another intoxication involving 4 goats; tonic-clonic convulsions and dyspnea before death were described in a case involving 2 sheep.1,6,10 In all available studies, the suspicion of intoxication was based on identification of the plant within the digestive tract, and the diagnosis was based on clues provided by the anamnestic information, not by detecting calycanthine in biological materials. In addition, we retrieved no cases of a precise description of pathologic findings.
In our case, an episode of suspected intoxication occurred in June 2022 in a backyard farm of 27 goats and sheep of different ages, living together. These animals were confined to a fenced olive grove where they normally ate hay and had water available. Suddenly, 12 of 27 animals developed ataxia, lateral recumbency, spastic convulsions, dyspnea, and eventual death within 24 h from the onset of clinical signs; no clinical signs were recorded in the remaining animals. The investigation could not exclude a non-accidental event, and the owner reported the animals had not received pharmacologic treatment. He also added that the animals had accessed branches of several plant species that he had pruned but could not say which ones they had possibly eaten or in what quantity.
Of 12 dead animals, 1 goat and 3 sheep were brought directly to the Verona Veterinary Diagnostic Laboratory of IZSVe (northeastern Italy) and underwent autopsy between the afternoon and the next morning. Postmortem examination of the goat, a 2-y-old Saanen, revealed diffuse severe pulmonary edema with abundant serous effusion in the pericardium, hemorrhagic content in the duodenum, foamy content in the jejunum and ileum, and moderate segmental chronic catarrhal typhlitis with adult nematodes within the cecum. In addition, 3 live dipteran larvae, which were identified as Oestrus ovis, were found in the frontal and ethmoidal sinuses. The sheep examined were one 5-y-old and two 1-y-old crossbreds. Autopsy of the oldest sheep revealed marked diffuse congestion of the abomasal mucosa with foamy content (Fig. 1), mild diffuse acute catarrhal enteritis with moderate mesenteric lymphadenomegaly, and numerous mineralized parasitic cysts in the liver and lung. One of the 1-y-old sheep had moderate diffuse congestion of the abomasal mucosa and moderate segmental acute hemorrhagic enteritis of the small intestine and cecum. In the other young sheep, postmortem autolysis of the thoracic and abdominal organs had occurred, which allowed only a marked mesenteric lymphadenomegaly to be observed. In all of the examined animals, the forestomachs, particularly the rumen, contained abundant fibrous plant material and numerous seeds, oblong, ~2-cm long, varying in color between dark- and light-brown and resembling those of Chimonanthus praecox (Figs. 2, 3).
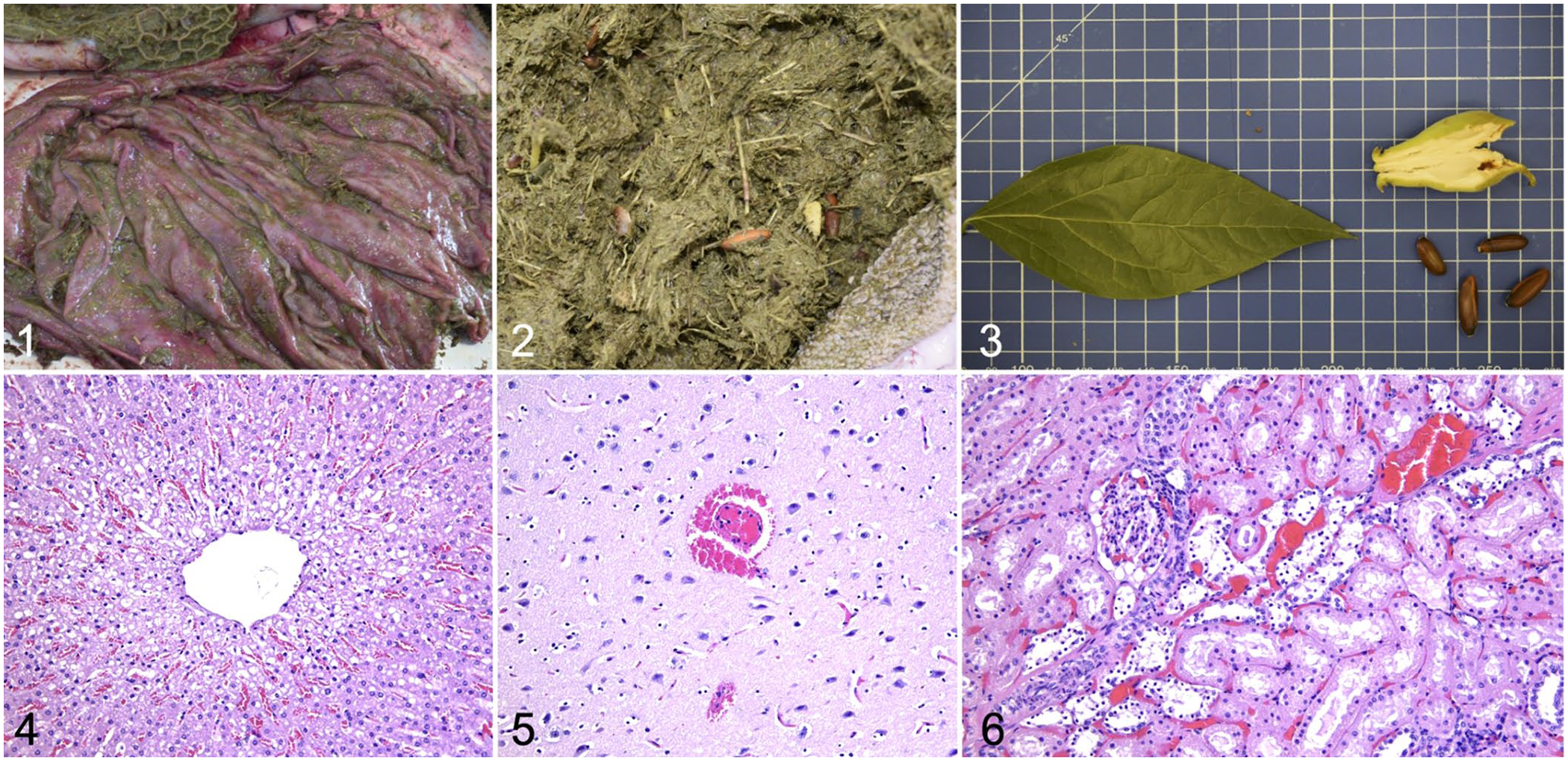
Macroscopic and histologic findings in small ruminants with acute Chimonanthus praecox (wintersweet) intoxication.
From each animal, a sample of brain, liver, and intestine was collected for routine bacteriologic investigations, including the microbial load of Clostridium perfringens in the intestine. Normal intestinal microbiota was detected in all the sheep; in the goat, a marked growth of α-toxin–producing C. perfringens was found. Negative bacteriologic cultures resulted from brain and liver of all animals. On intestinal flotation parasitology, gastrointestinal strongyles were found in all animals, and coccidial oocysts in one subject.
Liver, brain, and kidney from each subject were submitted for histologic examination. In all the examined liver sections, moderate diffuse congestion with mild-to-moderate hydropic degeneration of centrilobular hepatocytes was observed (Fig. 4). Severe diffuse vascular congestion, edema of the meninges, and rare parenchymal perivascular microhemorrhages were observed in the brain. In the goat, occasional meningeal hemorrhages were detected (Fig. 5). The kidneys from all 4 animals had severe, diffuse congestion and multifocal, mild-to-moderate necrosis of the tubular epithelium within the cortex (Fig. 6). Detailed gross and histopathological features are described in Supplemental Table 1.
Autopsy features and collateral laboratory results did not allow attribution of the animals’ deaths to a specific infectious agent, except for the suspicion of C. perfringens in the goat. Aliquots of rumen content and liver samples were collected and sent to the Experimental Chemistry Laboratory of the IZSVe. Aliquots of rumen content and liver samples from animals not involved in this event were used as negative controls. Samples of C. praecox plants were also found on the property where the animals became ill, and were collected, identified, and sent to the laboratory. All of the samples were analyzed by direct analysis real-time high-resolution mass spectrometry (DART-HRMS) using a fast and reliable screening method for toxicants. 14 The DART-HRMS spectra obtained from the seeds in the rumen were compared with those obtained from C. praecox from the property. The DART-HRMS spectra of C. praecox extract showed intense signals that were assigned to 3 alkaloids (i.e., calycanthine, calycanthidine, folicanthidine). The seeds collected from the rumen contents had the same spectrometric profile, as did the spectra obtained from liver and rumen content samples, with intense signals that were assigned to the aforementioned alkaloids in all 4 animals. The absence of C. praecox–related ions in control liver and rumen content was also verified.
The detection of alkaloids by DART-HRMS supported the hypothesis that C. praecox was the cause of the animal intoxication. Calycanthine was confirmed in the liver and rumen content samples, and it was quantified by liquid chromatography–high-resolution tandem mass spectrometry (LC-HRMS/MS). Screening and confirmatory analytical chemistry procedures have been described thoroughly. 14 The diagnosis of C. praecox poisoning by LC-HRMS/MS allowed us to attribute the clinical signs, such as ataxia, lateral recumbency, and spastic convulsions, to the strychnine-like toxicity caused by calycanthine, as described previously. 8 On the other hand, we found no specific gross or histologic findings; the alkaloids and other toxins contained in C. praecox might have been responsible for findings of edema, congestion, and hemorrhage, which could be consequences of increased cell permeability and circulatory or coagulation disorders. Given the incomplete understanding of the toxicity mechanisms of the detected alkaloids, both direct and indirect toxic effects may be considered.
The previously reported GABA interference induced by calycanthine may explain the severe muscle spasms, with respiratory distress and eventual death due to pulmonary and cardiac failure. The ensuing acute systemic hypoxia may have contributed to renal tubular injury, centrilobular hepatic degeneration, and multiorgan vascular congestion, macroscopically remarkable in the abomasal mucosa of all the examined animals. In addition, the renal damage, observed in all of the animals studied and previously hypothesized in a human case of intoxication with C. praecox, may also be related to other active molecules from the plant or their metabolites that may have caused a direct toxic effect on the renal tubules.5,7 Beyond calycanthine and derivatives, Calycanthaceae contain other compounds that might have biological activity that could contribute to the onset of clinical and pathologic findings in small ruminants, such as other tryptamine-related alkaloids (e.g., chimonanthine, chimonanthidine, tryptamine, Na,Nb–dimethyltryptamine), coumarins, flavonoids, terpenoids, lignans, and essential oils. 13 Among these, it cannot be excluded that coumarins could have produced circulatory changes consequent to their vasodilatory effect in addition to their anticoagulant capacity, as well as their hepatotoxicity (cytochrome P-450 enzyme–related), causing decay of ATP levels, loss of ionic gradients, cell swelling, and damage.9,11 Nonetheless, evidence to support a direct relationship of specific molecules to effects in small ruminants and, more generally, in mammals is scarce.
Multiorgan petechiation, previously described in a sheep intoxicated with C. praecox, was rare in our cases. 8 The case in the goat was complicated by an intestinal isolate of C. perfringens, possibly because of paralytic ileus. Clostridial α-toxin is known to alter vessel permeability and might have aggravated the clinical course, accelerating the fatal outcome. A few scientific articles have described clinical signs in small ruminants associated with intoxication by C. praecox.8,10 Unfortunately, neither clinical nor pathologic features are pathognomonic for such intoxication, and previous reports were based on anamnestic suspicion, neurologic signs, or morphologic identification of the plant in the gastrointestinal tract. We retrieved no cases of laboratory confirmation of C. praecox intoxication in a search of Google, PubMed, Web of Science, and Scopus, using the search terms “Chimonanthus praecox in animals”, “diagnosis of Chimonanthus praecox poisoning in animals”, suggesting that this condition has not been confirmed previously in small ruminants. We confirmed our case of C. praecox intoxication by detecting plant-produced alkaloids by fast DART-HRMS screening and LC-HRMS/MS, namely calycanthine, calycanthidine, and folicanthidine.
The toxicity of C. praecox and other ornamental plants is largely unknown to farmers who, despite knowing the potential toxicity of some cultivated plants, are often unaware of the dangers of ornamental plants to which their animals may have accidental or non-accidental access. In small ruminants, the ingestion of toxic plants must be considered among the causes of sudden death and uncertain medical history, even if identification may be difficult due to the mastication and digestion of plant parts. Our report highlights new aspects that veterinarians should consider in relation to the toxicity of C. praecox, increases the knowledge of visible macroscopic and microscopic signs of Chimonanthus alkaloid poisoning in small ruminants, and overcomes the difficulty of morphologic identification of the plant thanks to the innovative use of analytical chemistry tools.
Supplemental Material
sj-pdf-1-vdi-10.1177_10406387251382610 – Supplemental material for Clinical and pathologic changes in small ruminants with acute Chimonanthus praecox (wintersweet) intoxication confirmed by analytical chemistry
Supplemental material, sj-pdf-1-vdi-10.1177_10406387251382610 for Clinical and pathologic changes in small ruminants with acute Chimonanthus praecox (wintersweet) intoxication confirmed by analytical chemistry by Stefania Leone, Ivana Pallante, Claudia Zanardello, Carmela Zacometti, Tommaso Patregnani, Greta Foiani, Alessandra Tata, Roberto Stella, Marianna Merenda and Nicola Pozzato in Journal of Veterinary Diagnostic Investigation
Footnotes
Acknowledgements
We thank Dr. Roberto Bianconi, the veterinary practitioner who pragmatically approached the case, and Giancarlo Alessandri for technical assistance regarding bacterial cultures.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
ORCID iDs
Supplemental material
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
