Abstract
Radial fractures are relatively uncommon (0–1%) injuries in postmortem studies of racehorses, and few cases have been published. We describe here a series of 42 cases of radial fractures observed in California racehorses. Autopsy findings from all racehorses submitted for postmortem examination between 2006 July 1 and 2022 June 30 that had a recorded radial fracture were evaluated. We included 42 horses (30 Thoroughbreds, 12 Quarter Horses), most of which were 3–5-y-old. Catastrophic radial fracture was observed in 30 (71%) horses; in the remaining 12 (29%) horses, the fracture of the radius was considered an incidental finding. All of the catastrophic radial fractures were complete, displaced, and comminuted; 70% were articular and 30% were non-articular; 61% were open and 39% were closed; 83% were diaphyseal to distal epiphyseal and 17% were proximal epiphyseal. Nine of the 30 horses with catastrophic fracture had a preexisting periosteal callus associated with the main line of fracture. Among Quarter Horse racehorses, the frequency of callus in radial fractures tended to be higher (p = 0.083) in 3-y-olds compared with other ages. In 6 of these 9 cases, the callus was located along the cranial aspect of the distal third of the diaphysis. All incidental cases consisted of osteochondral fragmentations, which occurred most frequently on the cranial border of the intermediate facet of the distal articular surface of the radius. Knowledge of the common sites of stress fractures or osteochondral stress remodeling in the radius can help to focus autopsies and to improve the characterization and interpretation of those lesions.
Musculoskeletal injuries represent ~80% of the total racehorse fatalities in California and other racing jurisdictions.27,30 Most catastrophic skeletal injuries are associated with preexisting lesions (e.g., stress fractures, subchondral stress remodeling) that occur at consistent bone-specific locations related to sites of high stresses associated with repetitive racing speed events.31,32 Knowledge of bone-specific sites allows detection of premonitory lesions and thus provides an opportunity for intervention to prevent catastrophic fractures. Discovery of the skeletal sites and pathologic and imaging abnormalities associated with stress fractures and subchondral stress remodeling that predispose to catastrophic fractures enable industry participants and regulatory bodies to detect and rehabilitate affected horses before injuries became catastrophic.
The enhanced culture of safety in California has resulted in a marked reduction of annual racehorse fatalities from 317 (of 58,446 starts) in FY 2005/2006 to 66 (of 29,136 starts) in FY 2022/2023.2,3 Despite the lower number of starts in 2022/2023, the ratio of starts:year (2:1) compared with the ratio of injuries:year (5:1) indicates a net reduction of injuries over the years. The study of the fracture patterns, exercise history,4,28 and other associated risk factors6,14,38 is essential to understanding and preventing many catastrophic fractures in racehorses. Whereas sites of preexisting stress fractures and subchondral stress remodeling have been documented in many long and flat bones of the fore- and hindlimbs, 31 preexisting lesions associated with catastrophic fractures of the radius have not been reported in racehorses, to our knowledge.
Radial fractures, a cause of lameness in horses, may result from high-impact trauma, such as a kick or a fall, or being predisposed by a stress fracture.1,5,12,19,26 Traumatic events, the most common causes of radial fractures, usually result in incomplete and minimally displaced fractures; however, complete, displaced fractures can also occur.1,5,10,26,29,34 In a study of 26 racehorses with stress fractures of the upper-limb bones (i.e., humerus, radius, and tibia), 3 live horses had evidence of stress fracture in the radius. 19 In 2 retrospective studies of arthroscopic surgery of the antebrachiocarpal joint, osteochondral fragmentation of the radius was observed mainly in the craniolateral and/or craniomedial border of the distal articular surface.11,21 A pathologic fracture of the radius was associated with a bone cyst in a 9-y-old Thoroughbred horse. 22 In 3 postmortem studies of musculoskeletal injuries, catastrophic radial fractures had a low prevalence (0.4–1.3%) in Quarter Horses and Thoroughbreds.13,23,27
However, there is scant information published about radial fracture in racehorses particularly regarding the type and location of preexisting lesions. Here, we describe the postmortem findings in a series of cases of radial fractures of racehorses in California.
Material and methods
We retrieved and analyzed autopsy reports of all racehorses submitted for postmortem examination to the California Animal Health and Food Safety laboratory (CA, USA), through the California Horse Racing Board postmortem program, between 2006 July 1 and 2022 June 30, that had a documented radial fracture. In 2 cases included in our retrospective study, postmortem radiograph images of cadaveric specimens were also available and reviewed. During this period, a total of 3,505 racehorses were submitted for autopsy, of which 2,453 (70%) were submitted because of musculoskeletal injuries.
Data collected from the autopsy records included breed, age, circumstances of the fracture (non-exercise, racing, training), fracture classification, limb distribution, and preexisting lesions and/or other fractures. Fractures were classified as complete or incomplete; displaced or non-displaced; simple or comminuted; open or closed; articular or non-articular; epiphyseal (proximal or distal), diaphyseal, physeal, or metaphyseal; and oblique, transverse, axial/longitudinal, and/or segmental.
The preexisting lesions included in our study were classified as A) stress fracture or remodeling comprised of: 1) hyperemia or hemorrhage and thickening of the periosteum, 2) petechiation of the subperiosteal cortex circumscribing the periosteal reaction, 3) patchy, slightly raised, thin, red, rough, plaques of poorly mineralized woven bone (Fig. 1), 4) raised red or pink or gray, rough or smooth, palpable, nodular, periosteal callus immediately adjacent to the fracture line (Fig. 1), and/or 5) endosteal callus; or B) subchondral stress remodeling (Fig. 1) comprised of: 1) focal brown discoloration (subchondral bone necrosis [coauthor unpublished observation]), 2) focal red discoloration (subchondral osteopenia), 28 and/or 3) high compacted subchondral bone (subchondral sclerosis).9,32 The presence or absence of stress fracture consistent with periosteal callus, the anatomic site (location) of the periosteal callus on the radius, and the distribution of the periosteal callus between limbs were recorded.
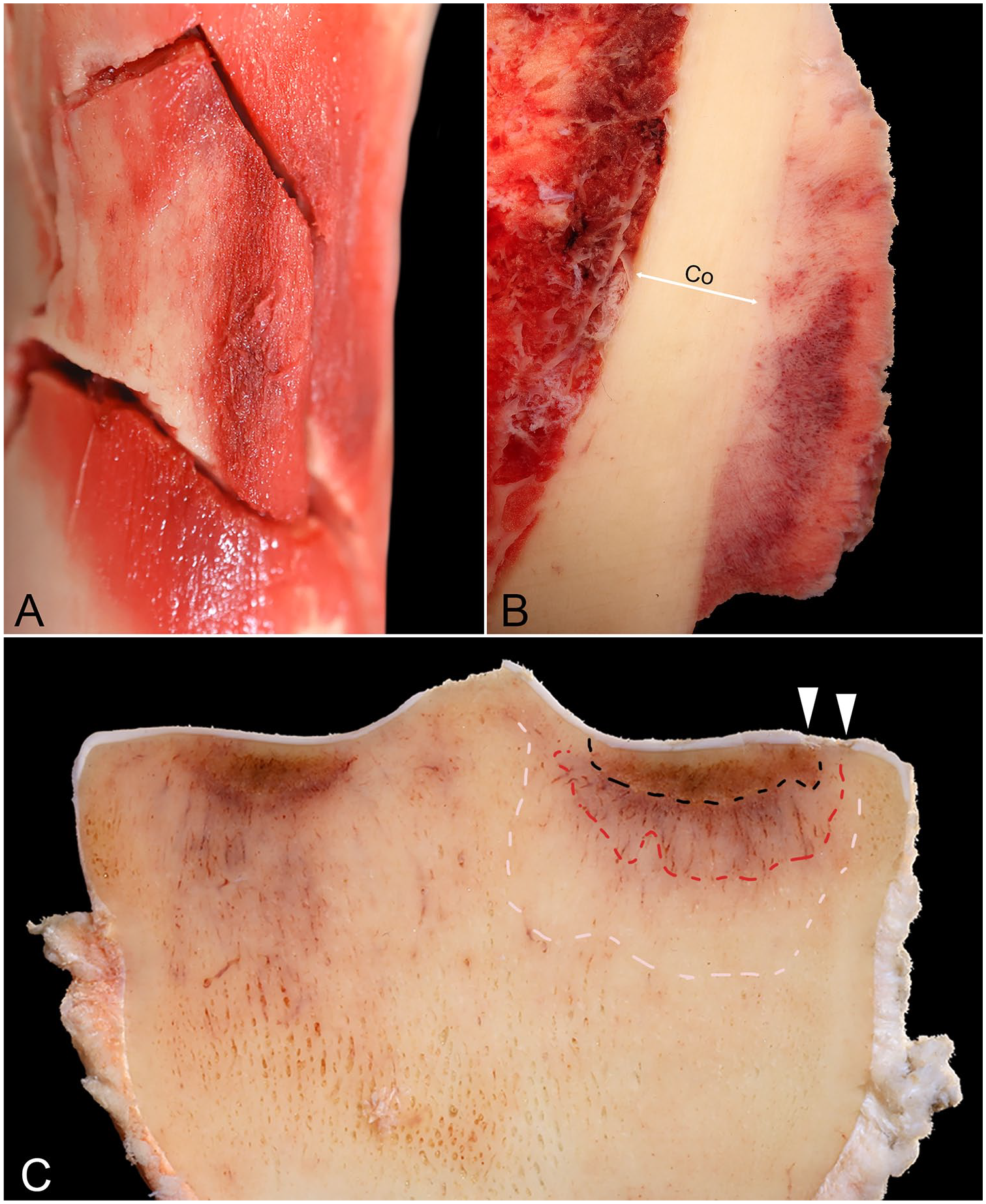
Preexisting lesions at different stages of healing. Images are from different racehorses.
Each fracture was described, and the absolute frequency was determined. A multiple correspondence analysis (MCA) was performed among the 30 horses that sustained a complete radial facture and among the 12 horses that sustained osteochondral fragmentation of the radius to evaluate the relationships between categorical variables of interest [i.e., periosteal callus (assessed in horses with complete radial fracture), breed, age, circumstance, and limb distribution] on a 2-dimensional plot, allowing spatial clusters to be identified. 7 Variables that clustered spatially were considered similar (associated or dependent) to each other, whereas those plotted farthest apart were considered rarely associated or independent of each other. 7
The periosteal callus among horses that sustained a radial fracture was compared by breed, age, circumstance, and limb distribution using a Fisher exact test. Alpha levels for significance and tendency were 0.05 and 0.1, respectively. The data were analyzed using Stata v.14.0 (StataCorp) and InfoStat v.2020 (https://www.infostat.com.ar/).
Results
Between 2006 July 1 and 2022 June 30, 42 racehorses were submitted that had sustained radial fractures, including catastrophic fractures and/or marginal osteochondral fragmentation. Catastrophic fractures were defined as those that either prompted the euthanasia of the horse or caused the death of the animal. Osteochondral fragmentation was considered an associated finding in horses that had died spontaneously or had been euthanized due to injuries other than radial fractures.
The 42 racehorses with radial fractures represented 1.2% of the total number of racehorses submitted during the study period and 1.8% of the horses submitted because of musculoskeletal injuries during that period. These 42 horses were mostly 3–5-y-old (median = 4; min = 2; max = 9), and they were Thoroughbred (32) or Quarter Horse (10). Complete, catastrophic radial fractures occurred in 30 of 42 (71%) horses with documented radial fractures; 28 horses were euthanized, and 2 died spontaneously. Osteochondral fragmentation occurred in 12 of 42 (29%) horses with documented radial fractures. Of these, 10 horses were euthanized due to fractures other than radial, and 2 died spontaneously (Table 1).
Frequency of catastrophic radial fractures and osteochondral fragmentation of the radius in Thoroughbred and Quarter Horse racehorses of California by age of the horses.
The 30 complete, catastrophic radial fractures were unilateral, with 19 occurring in the left leg and 11 in the right leg. Radial fractures occurred during racing (18 of 30) or training (12 of 30) that included official workouts (11 of 12), and non-official exercise (1 of 12; Table 2).
Characteristics of catastrophic radial fractures in Thoroughbred and Quarter Horse racehorses of California.
The fracture involves >1 location in 16 horses.
Salter-Harris type I (complete separation of the physis without fracture through the bone) and type III (complete fracture that extends longitudinally from the distal articular surface, through epiphysis, into deep zone of the grow plate and then transversally along the physis to its periphery).
The fracture involves >1 configuration in 17 horses (12 Thoroughbreds, 5 Quarter Horses).
Eight of 30 horses with a complete catastrophic radial fracture had a fracture only in the radius; the remaining 22 had at least one other bone fractured in addition to a radial fracture. In 13 of these 22 horses, there was only 1 other fractured bone, but in the remaining 9 horses, there were ≥2 other fractured bones. The fracture in other bones included the carpus (6 of 22), ulna (4 of 22), vertebrae (4 of 22), proximal sesamoid bones (3 of 22), humerus (2 of 22), third metacarpal bone (2 of 22), proximal phalanx (2 of 22), and rib, tibia or ilium (1 of each). Two of the 4 horses with concomitant radial and vertebral fractures died spontaneously after a fall due to acute fracture of the C2 vertebra with complete transection of the spinal cord.
The catastrophic radial fractures were, in all 30 cases, complete, displaced, and comminuted (Fig. 2). Twenty-one (70%) of these fractures were articular, affecting the distal (20 of 21; Fig. 2) and proximal (1 of 21) articular surface of the radius; 9 were non-articular. Seventeen fractures were open (Fig. 2), and 13 were closed. In most cases, the fractures included more than one configuration (e.g., oblique and transverse) and had oblique (23 of 30; Fig. 2), transverse (19 of 30), longitudinal/axial (5 of 30), and/or spiral (1 of 30) components. The fracture location was diaphyseal to distal epiphyseal (25 of 30), or proximal epiphyseal (5 of 30). Two of the distal epiphyseal fractures affected the physis and were classified as Salter–Harris type I (one 2-y-old male Quarter Horse) and type III (one 2-y-old female Quarter Horse).
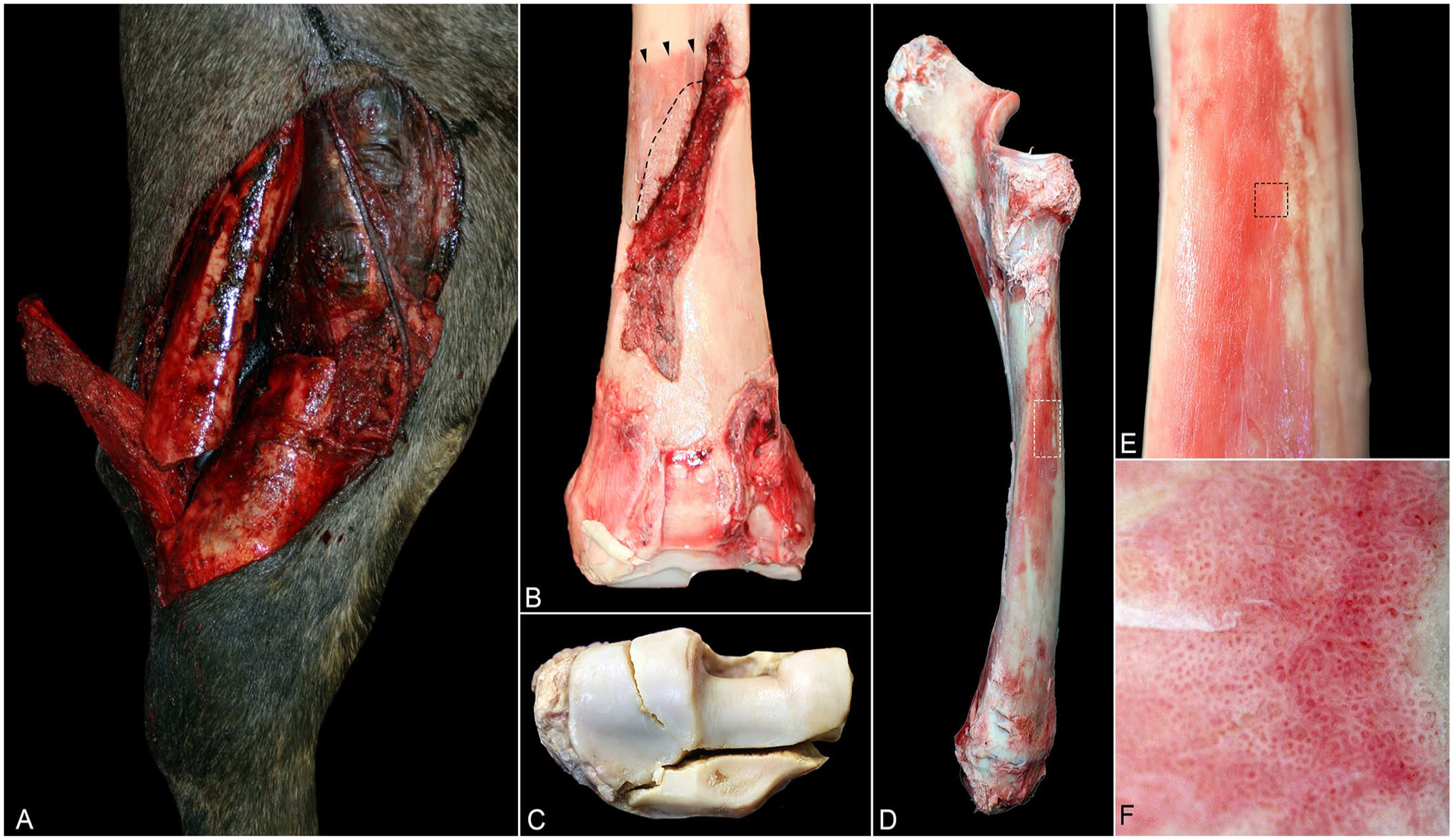
Catastrophic fracture of the radius. Images are from different racehorses.
Thirteen of 30 horses with catastrophic radial fractures had evidence of at least 1 preexisting lesion, including stress fracture consisting of: 1) slightly raised, thin, red, rough, plaques of woven bone (Fig. 2), 2) raised red or pink or gray, rough or smooth, nodular, periosteal callus immediately adjacent to the fracture line (Fig. 2), and/or 3) endosteal callus (Fig. 3). Of these 13, 7 horses had a stress fracture only in the radius, and 2 horses had a stress fracture in the radius and stress remodeling in the subchondral bone of the proximal sesamoid bones and lateral condyle of the third metacarpal bone. Four horses had a preexisting lesion (i.e., stress fracture or subchondral stress remodeling) within the other bone (i.e., not in radius) including the tibia (1; Suppl. Fig. 1), ulna (1), proximal sesamoid bone (1; Suppl. Fig. 1), and 6th lumbar vertebra (1).
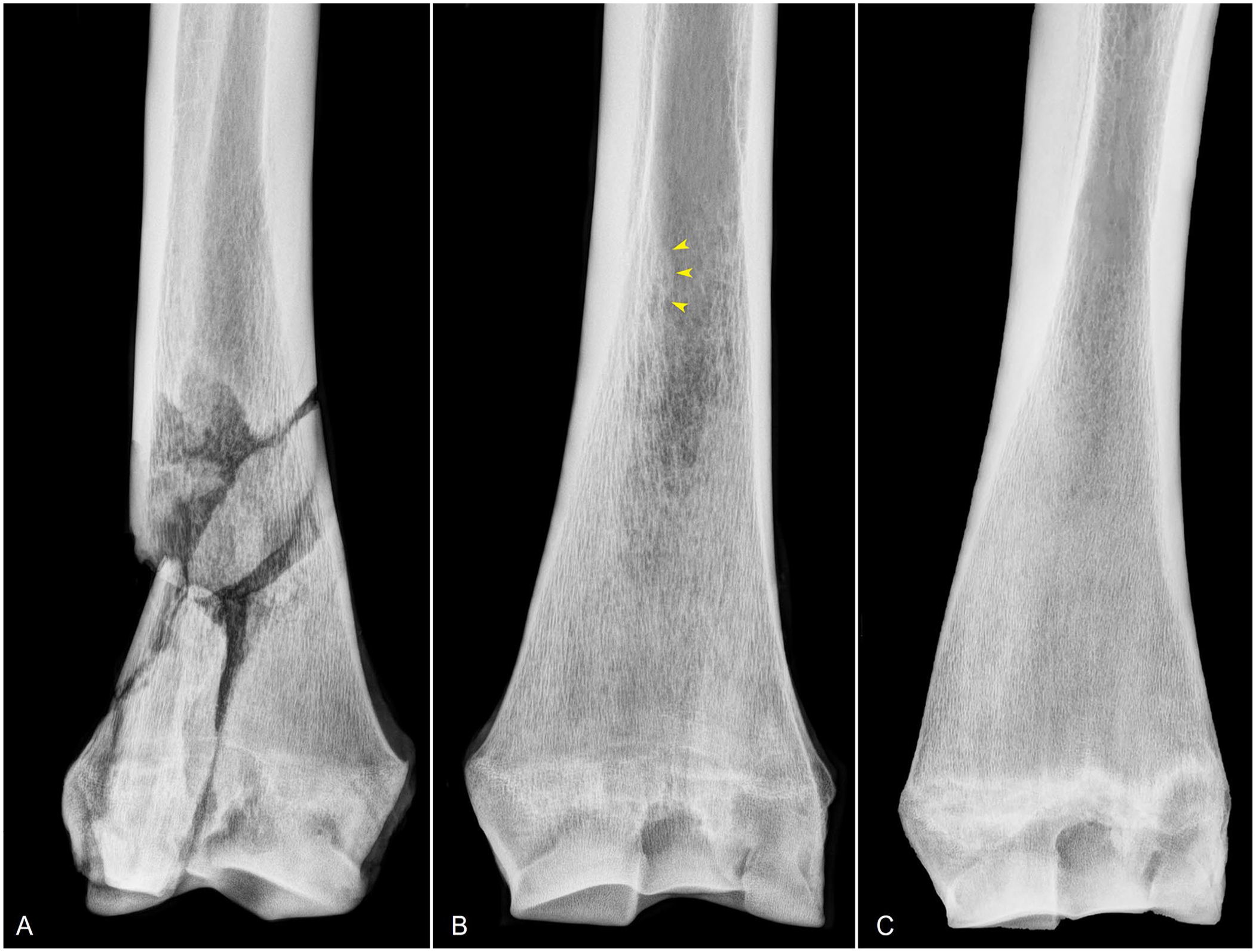
Radiographic images of a horse with a catastrophic radial fracture, an endosteal callus in the contralateral non-fractured radius, and a normal radius of a racehorse.
Nine of the 30 horses with catastrophic radial fractures had a periosteal callus associated with the main fracture line (Fig. 4), 11 did not have a periosteal callus, and in the remaining 10 no information was available regarding the presence or absence of a periosteal callus. In all cases, the callus was in one leg; 5 in the left and 4 in the right leg. The callus was more frequent in 3-y-old horses (4 of 9), followed by 2-y-old horses (2 of 9) and 4-, 5-, and 8-y-old horses (1 of each). The callus was located in the distal third of the diaphysis in 7 of 9 cases and in the mid-proximal third of the diaphysis in the remaining 2 cases. The callus found in the distal third of the diaphysis (n = 7) was in the cranial (6), caudal (3), and/or medial (2) side of the bones (Fig. 4).
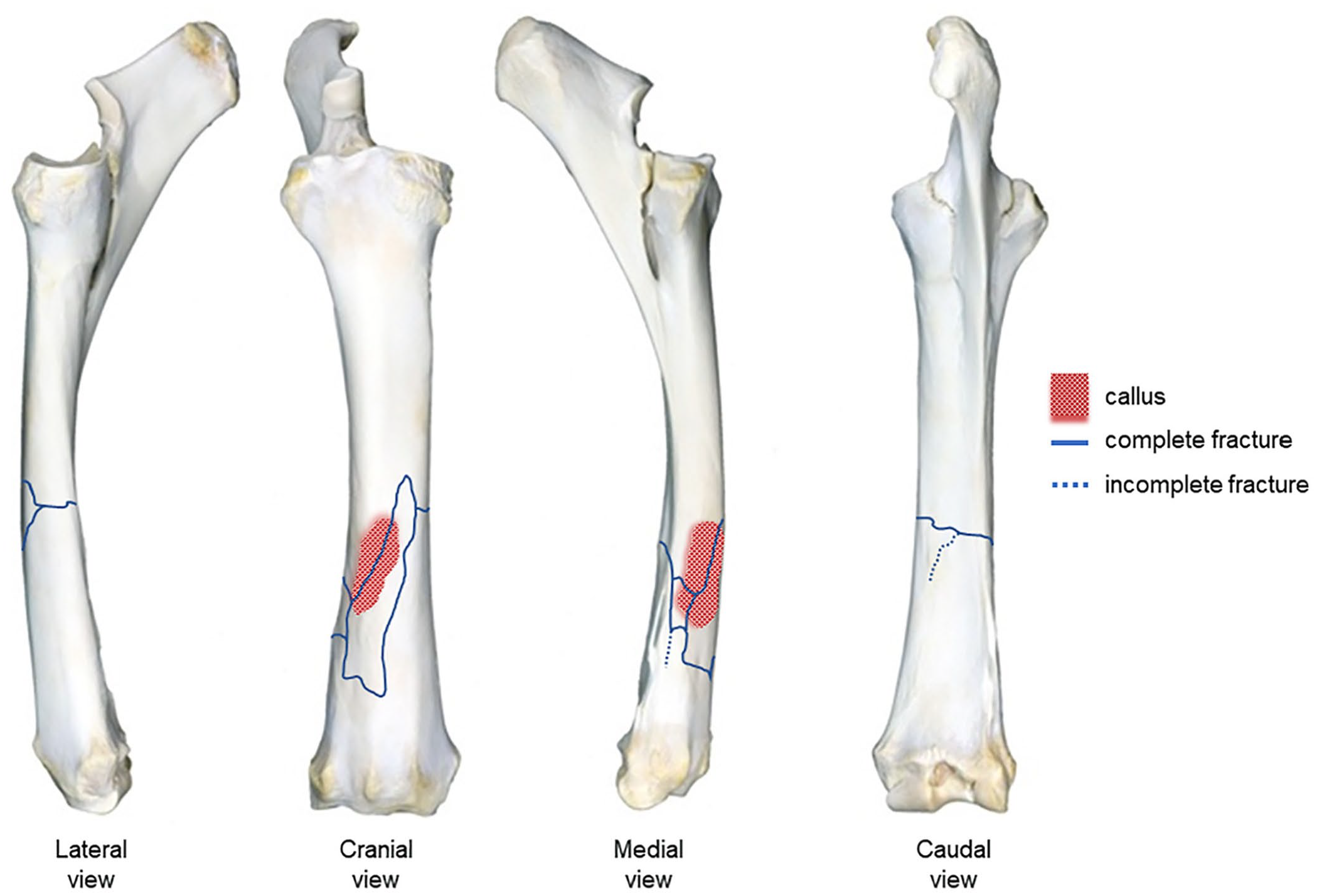
Anatomic localization and postmortem diagrammatic documentation of the stress fracture of the radius shown in Fig. 1A, 1B. Preexisting stress fractures (i.e., periosteal callus, red highlighted regions) in our series were observed most commonly on the distal third of the diaphysis on the cranial and medial side of the radius.
For the 30 horses that sustained complete catastrophic radial fracture, 63% of the total inertia (variance) in the 2 dimensions of the MCA was explained by the presence of stress fracture (callus), breed, age, circumstance, and limb distribution (Suppl. Fig. 2). There was evidence of relationship between age (3-y-old) and the presence of callus. According to the Fisher exact test, the frequency of callus in fractured radii tended to be higher (p = 0.083) in 3-y-old Quarter Horses (2; 67%) compared with Quarter Horses of other ages (1; 33%).
Osteochondral fragmentation occurred bilaterally in 4 horses and unilaterally in 8 horses (Fig. 5), equally distributed between the right and left leg (Table 3). Ten of 12 horses had, in addition to radial osteochondral fragmentation, one or more concomitant complete fracture in ≥1 other bone, including the carpus (5 [4 in the intermediate; 3 in the radial; 2 in the third and second; and 1 in the ulnar and fourth carpal bones]), proximal sesamoid bone (4), and scapula, humerus, third metacarpal, or proximal phalanx (1). Euthanasia of these 10 horses was not because of their distal radial osteochondral fragmentation, but due to the poor prognosis associated with the other aforementioned fractures that were not amenable to surgical repair. The remaining 2 horses with only osteochondral fragmentation of the radius reportedly died in association with general anesthesia for surgical repair or removal of the osteochondral fragment.
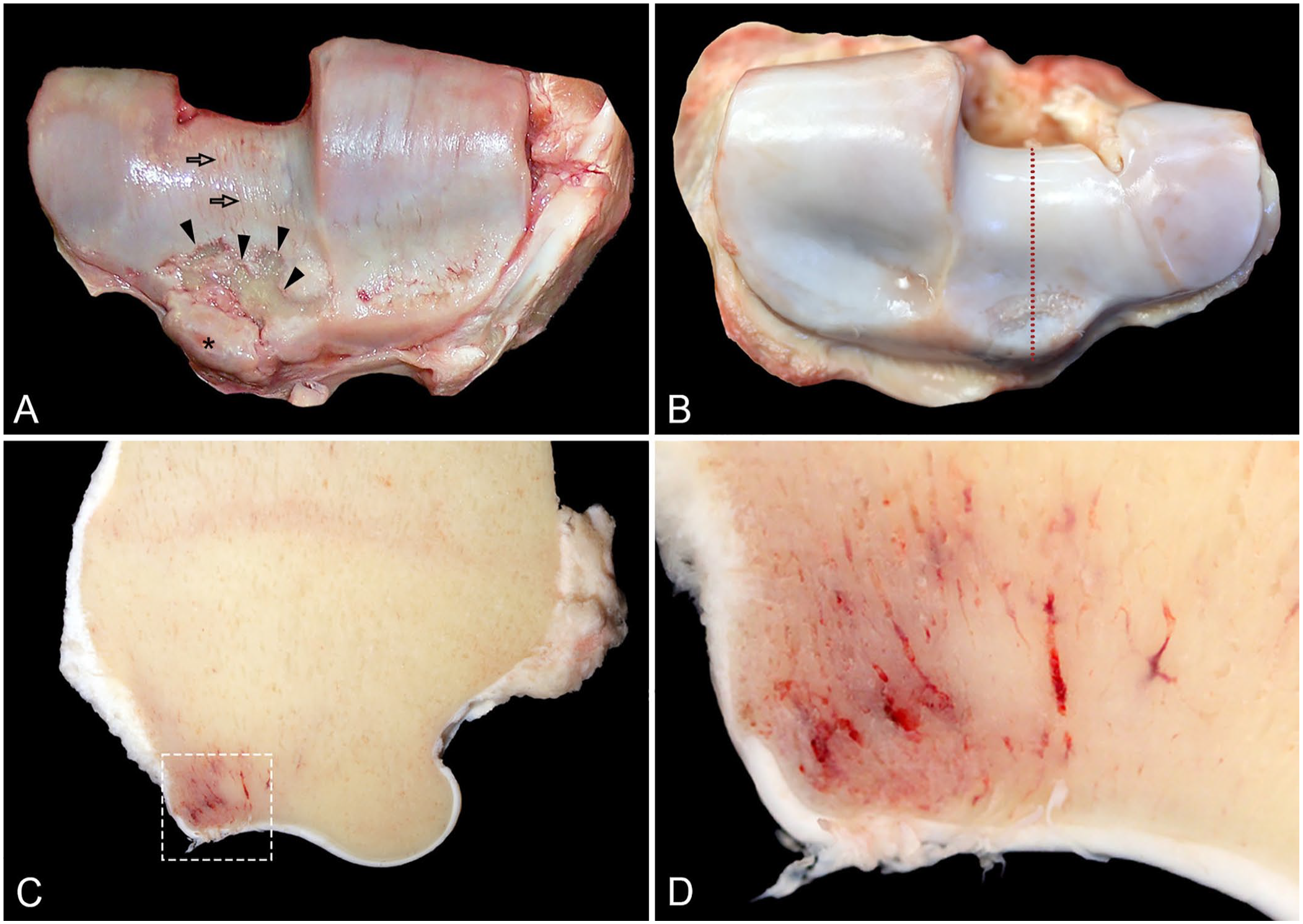
Osteochondral fragmentation of the radius. Images are from different racehorses.
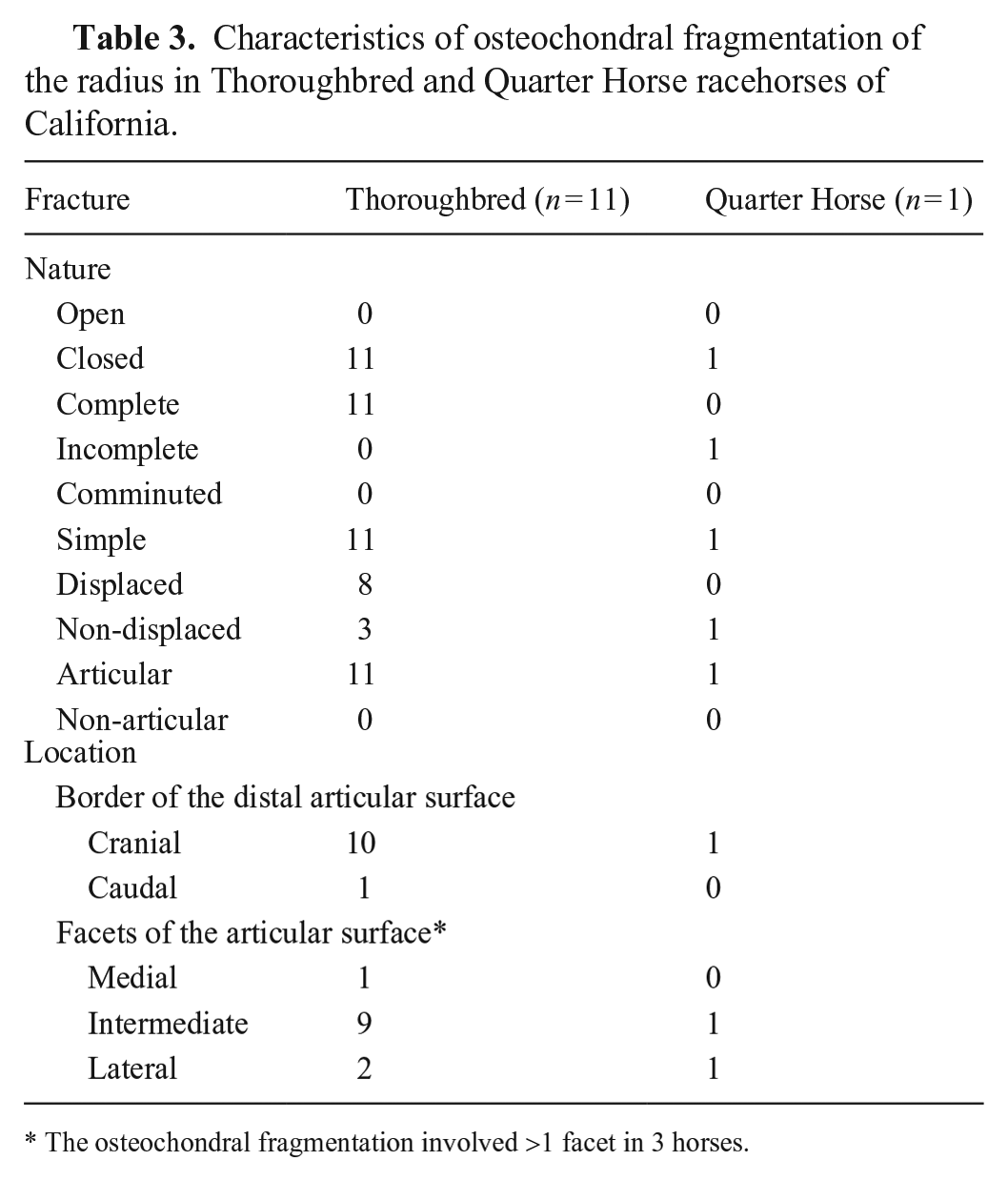
Characteristics of osteochondral fragmentation of the radius in Thoroughbred and Quarter Horse racehorses of California.
The osteochondral fragmentation involved >1 facet in 3 horses.
The osteochondral fragmentations were complete in 10 horses, and incomplete in 2 horses; 9 fractures were displaced (Fig. 5); 3 were non-displaced (Fig. 5). In all 12 cases, osteochondral fragmentations were simple, articular, and closed. Osteochondral fragmentations involved the cranial (11) and caudal (1) border of the distal articular surface of the radius; on the cranial border, the osteochondral fragmentations affected ≥1 facet, including the intermediate facet (Fig. 6) in 10 cases, the medial facet in 4 cases, and the lateral face in 1 case.
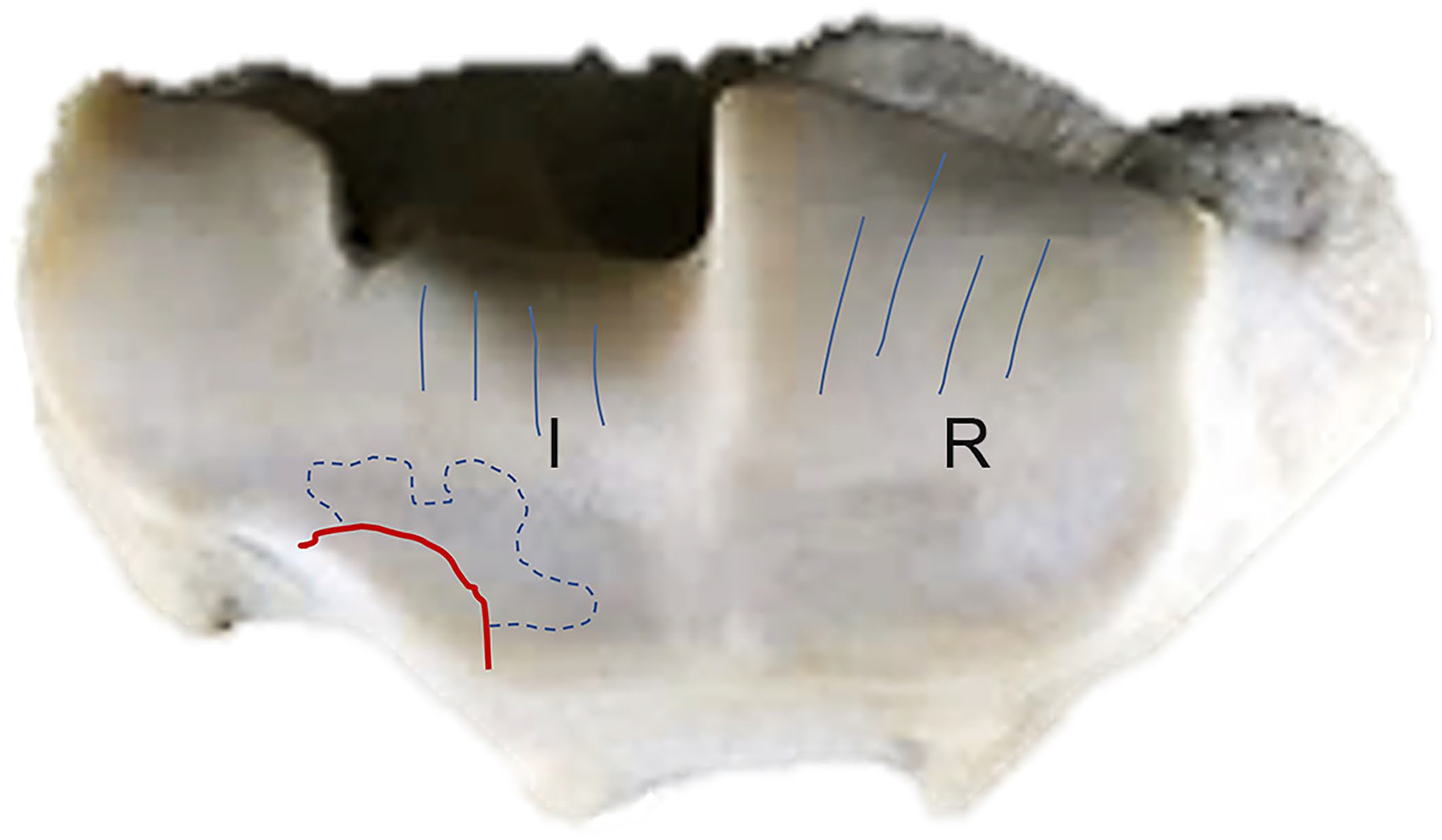
Anatomic localization and postmortem diagrammatic documentation of the osteochondral fragmentation of the radius (red line) shown in Fig. 5A. The osteochondral fragmentation was observed most commonly on the intermediate (I) and radial (R) facets of the distal articular surface of the radius. The blue dotted line shows thinning of cartilage. The blue lines indicate the location of wear lines in the cartilage.
Six of 12 horses with osteochondral fragmentation of the radius had preexisting subchondral stress remodeling or stress fracture in ≥1 other fractured bone, including: radial and intermediate carpal bones (3), proximal sesamoid bones (1), third metacarpal bone (1), scapula (1), and humerus (1). In a subset of 3 cases with radial osteochondral fragmentation of the intermediate facet and complete slab fracture of the intermediate carpal bone, there was evidence of chronic degenerative change in the radius including focal red discoloration that indicated preexisting subchondral stress remodeling (subchondral osteopenia, Fig. 5), and highly compacted subchondral bone (subchondral sclerosis); also, there were rounded margins (lipping) of the osteochondral fragment (Fig. 5) and multifocal-to-coalescing deep ulcerations of the articular cartilage near the fragment (Fig. 5).
For the 12 horses that sustained osteochondral fragmentation, 70% of the total inertia in the 2 dimensions of the MCA was explained by the breed, age, circumstance, and limb distribution of the fracture (Suppl. Fig. 3). There was a lack of evidence of significant relationship between the breed, age, limb distribution, and circumstance among horses that sustained osteochondral fragmentation.
Discussion
Our classification and location of fractures were similar to previous studies on radial fractures.1,19,26 We documented several characteristics of radial fractures in racehorses that were not described previously, to the best of our knowledge. We retrieved no studies of case-series of the pathology of radial fracture in racehorses in a search of Google, PubMed, CAB Direct, Web of Science, and Scopus, using the search terms “radial fracture” plus “racehorses” plus “pathology”, suggesting that this condition has not been reported previously in racehorses. An important finding in our study was that the catastrophic complete radial fractures were associated with preexisting stress remodeling (i.e., stress fracture in various stages of healing). The preexisting stress remodeling (observed in 9 horses) was located mainly in the external cortical surface of the cranial side of the distal third of the diaphysis, similar to the location of the stress fracture (detected by scintigraphy) observed in 3 live racehorses with a history of lameness. 19 Preexisting stress fractures usually have a bone-specific pattern of distribution 31 (e.g., the most common place to find stress fractures in the scapula and humerus is the neck of the bone25,31,33). Knowing this pattern helps the clinician detect the earliest stages of the disease and prevent catastrophic scapular or humeral fracture, respectively. Information about identification, classification, and distribution of the preexisting lesions is an important tool for pathologists because it allows them to perform focused autopsies and make accurate interpretations about their relation to etiopathogenesis of catastrophic musculoskeletal injuries. This information allows multi-institutional collaborations, mainly with catastrophic lesions with low prevalence but which end in the destruction of the animal, such as complete rupture of the distal sesamoid ligaments.
Stress fracture or subchondral stress remodeling occurs subsequent to mechanical damage that occurs due to repeated high-magnitude load on bones during increases in intensity of training and racing. 30 Bone microdamage leads to focal cortical and/or trabecular osteopenia. 32 This, in turn, causes microarchitectural changes and weakening of the bone structure, which increases the risk of a complete fracture occurring at the site of stress fracture or subchondral stress remodeling.19,32 In studies of other bones (e.g., scapula, humerus, and/or proximal sesamoid bones) during autopsy, the most common findings associated with stress lesions included: 1) periosteal/endosteal callus in the cortical region, with bone proliferation, adjacent or bridging the main acute fracture lines (in the earliest stages, the callus consists of a thin [<1-mm thick], red, highly vascular, rough, poorly mineralized, woven bone tissue; in advanced stages, the callus consists of large, usually multinodular, firm, pale, smooth, lamellar bone tissue 32 ); and 2) subchondral focal osteopenia in the earlier stages, accompanied by subchondral bone compaction and sclerosis in advanced stages. 32 In our study, information about the presence or absence of preexisting stress fractures or subchondral stress remodeling was not available in autopsy reports for 10 of 30 horses. Several reasons could explain the lack of attention to examination or detection of preexisting changes, including missing bone fragments in comminuted open fractures (6 of 10 cases) and lack of knowledge about preexisting lesions in repetitive overuse bone injury in racehorses. Postmortem computed tomography or radiography of the affected and non-affected contralateral bones could be a useful tool for the detection of preexisting lesions in postmortem examinations.
Studying the exercise history of affected horses, and detecting and estimating the associated risk factors, may provide evidence that would help to prevent stress fractures. 30 Previous studies in racehorses have reported associations between the sex, age, and/or limb distribution of the lesion, (e.g., 2- or ≥5-y-old males have higher frequency of scapular fracture, 37 3-y-old horses more commonly sustain humeral fractures, 4 and osteochondral fragmentation is more common in the right forelimb 11 ). In our study, we found a weak (p = 0.083) association with a preexisting lesion (i.e., periosteal callus), breed, and age; however, more studies (e.g., case-control study) of the history of exercise in horses that sustained a radial fracture are necessary to validate and detect associated risk factors. The fact that 4 horses had radial fractures without callus, yet sustained concomitant fractures in another bone having evidence of stress fracture or subchondral stress remodeling (i.e., tibia, 6th lumbar vertebra, proximal sesamoid bone, or intermediate carpal bone), suggests that the radial fracture in these cases occurred secondary to the high-impact trauma associated with the catastrophic failure of preexisting repetitive stress injuries within the other bones.
Two of 30 racehorses with a complete fracture of the radius died suddenly due to an acute complete fracture of C2 with complete transverse section of the spinal cord. The lack of a preexisting lesion on the C2, but presence on the radius, suggest that primary fractures of the appendicular bones that result in a fall may lead to secondary axial skeletal fracture, which, in our 2 cases, caused sudden death associated with transection of the cranial spinal cord that compromised CNS respiratory centers. 25
In previous studies, the most frequent Salter–Harris fractures of the radius involved the proximal physeal growth plates.16,26 In our study, only 2 Quarter Horse racehorses had distal Salter–Harris fractures of the radius, 1 type I and 1 type III, both in 2-y-old (24 mo) horses; however, the evidence of open physes was not mentioned in the autopsy reports of these 2 horses. According to the literature, closure of the distal ossification center of the radius occurs at age 21–32 mo.18,24 Therefore, if radial fractures are classified as Salter–Harris fractures, and the age of the horse falls into the literature range of ossification center closure, it is recommended that the presence of an open physis be confirmed and documented in the postmortem report. Otherwise, in radii with closed physes, fractures involving metaphyseal and epiphyseal regions should be documented using regular terminology.
Osteochondral fragmentation has been reported in several bones, including carpal bones (radial, intermediate, third), proximal or distal phalanx, the articular process of cervical vertebrae, the patella, and the radius.11,15,17,20,36,39,40 In the carpus or fetlock, the most common cause of osteochondral fragmentation is traumatic injury resulting from compressive forces created by carpal or fetlock hyperextension, leading to fatigue failure and fragmentation.8,35,40 Other associated causes of osteochondral fragmentation include osteochondrosis dissecans, or injuries in the soft tissue of the joint.17,39 Similar to reports on osteochondral fragmentations of the radius,11,21 osteochondral fragmentation typically occurred in our study on the cranial border of the distal articular surface of the radius, mainly on the cranial border of the intermediate facet. Osteochondral fragmentations were also observed with a concomitant carpal fracture in 5 horses, in which 3 had a complete slab fracture of the intermediate carpal bone, the opposing bone to the intermediate facet fracture. Additionally, these 3 horses had evidence of subchondral stress remodeling and other chronic lesions, including lipping and ulceration of the articular cartilage in the osteochondral fragment or in proximity to the fragment. These changes can be considered preexisting lesions and may suggest that osteochondral fragmentation of the intermediate facet of the radius may cause instability of the antebrachiocarpal joint that predisposes to triggering catastrophic intermediate carpal bone fracture (proximal row of the carpus); slab fractures of the distal row of carpal bones are usually associated with preexisting lesions in the third carpal bone. 9
Supplemental Material
sj-pdf-1-vdi-10.1177_10406387251336267 – Supplemental material for A retrospective study of radial fractures in racehorses in California, 2006–2022
Supplemental material, sj-pdf-1-vdi-10.1177_10406387251336267 for A retrospective study of radial fractures in racehorses in California, 2006–2022 by Carlos O. Schild, Akinyi Nyaoke, Javier Asin, Eileen E. Henderson, Jeff A. Blea, Susan M. Stover and Francisco A. Uzal in Journal of Veterinary Diagnostic Investigation
Footnotes
Acknowledgements
We thank the staff, pathologists, residents, and students at CAHFS San Bernardino and Davis who performed or assisted with the autopsies during the period of the study. We also thank CHRB veterinarians for their contributions.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors declared that they received no financial support for their research and/or authorship of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
