Abstract
Hematological findings in a female killer whale (Orcinus orca) undergoing rehabilitation after sudden severe anorexia revealed continuing increases in serum lactate dehydrogenase and aspartate aminotransferase activities as well as fibrinogen concentration. Serologic evidence of herpesvirus infection and skin vesicles were detected 2 weeks into the treatment regimen of antibiotics and corticosteroids. The whale showed signs of improvement after treatment with anti-herpesvirus drugs, but sudden severe anorexia reappeared, along with marked elevation of fibrinogen concentration that continued until the death. Postmortem examination revealed multiple light tan foci of necrosis in the skeletal and cardiac muscles, and lung consolidation. Microscopic findings indicated disseminated fungal granulomas in the skeletal and cardiac muscles, as well as myocarditis, mycotic embolic thromboarteritis of cardiac blood vessels, and bronchopneumonia with numerous typical Aspergillus-like fungi. Mucor-like structures in granulomas in the heart and skeletal muscle and Aspergillus-like fungi in the lungs were identified using periodic acid–Schiff, Gomori methenamine silver stain, and immunohistochemistry. The present case involves dual infection with Mucor and Aspergillus species in a killer whale with concurrent herpesvirus.
Mycoses in marine mammals have been diagnosed and reported previously but are rare.1,9,19 Mycoses may be primary or secondary during any chronic infectious process in cetaceans. 25 A review of 143 captive and wild marine mammals found 24 species of animals with mycotic infections, including 19 different fungal species. Sixty-nine (48%) of these animals were found stranded and 24 of those had underlying mycotic disease. The other 74 animals were from various oceanaria, and 43 (58%) of these also had some underlying mycotic illness. Pulmonary mycotic infections are common in cetaceans. 23 Aspergillus fumigatus and, less frequently, Aspergillus niger or Aspergillus terreus are the best-known causes of mycotic pneumonia in marine mammals. 18 Although mycosis caused by members of the class Zygomycetes is rarely observed in marine mammals, it has been attributed to a variety of species including Apophysomyces elegans, Rhizomucor pusillus, Saksenaea vasiformis, 31 Entomophthora coronata, 30 and Rhizopus spp.19,35 The current report describes a case of a killer whale (Orcinus orca) with disseminated mucormycosis and pulmonary aspergillosis associated with herpesvirus infection.
A female adult killer whale, an odontocete belonging to the oceanic dolphin family, was maintained in Port of Nagoya Public aquarium for 16 years after arriving from Taiji Whale Museum on a lease basis. The animal suddenly became dull and lethargic, and subsequently developed anorexia. Complete blood cell count and biochemical profile were performed, and the results were compared with reference intervals cited in the literature (Table 1).6,21 The white blood cell count (WBC; 6,700 cells/µl) was within reference intervals. Increased lactic dehydrogenase (LDH; 285 IU/l), aspartate aminotransferase (AST; 42 IU/l), and alkaline phosphatase (ALP; 446 IU/l) activities were present. Approximately 2.5 weeks later, the anorexia intensified and the animal was seen lying on the bottom of the pool. Follow-up blood samples evaluated one week later revealed continued marked elevation of LDH (379 IU/liter) and AST (57 IU/liter) activities, as well as elevated fibrinogen concentration (545 mg/dl). An increased serum antibody titer to Simplex virus from a baseline value of 4-32 was also detected at this time. Five days later, the WBC was 8,800 cells/µl, and administration of corticosteroids (hydrocortisone and dexamethasone), an antifibrinolytic (tranexamic acid), and antibiotics (enrofloxacin and ampicillin hydrate) was begun. Five days later, additional antibiotics (ofloxacin and benzylpenicillin procaine) were administered. The animal’s appetite improved for 7 days but sudden severe anorexia was observed and administration of the antibiotics was discontinued. Despite treatment, LDH and AST activities and fibrinogen concentration continued to increase. The animal was diagnosed with herpesvirus infection based on an elevated serum antibody titer and the appearance of vesicles on the skin. Approximately 1 week later, the WBC increased markedly to 34,000 cells/µl. On the following day, an anti-herpesvirus drug (valacyclovir hydrochloride) was administered and continued for approximately two weeks. The blisters disappeared following treatment with the anti-herpesvirus drug, followed by a decrease in the serum herpesviral antibody titer to 8 with an associated marked improvement in appetite. Shortly thereafter, the WBC decreased to 1,500 cells/µl, at which point the clinicians began to suspect an opportunistic fungal infection. Treatment was changed to fluconazole, procaine benzylpenicillin, and cephalosporin. On further laboratory testing, the whale had increased serum potassium (6.5 mEq/l) concentration; increased creatine kinase (CK; 1,217 IU/l), LDH (2,020 IU/l), AST (399 IU/l), and alanine aminotransferase (ALT; 47 IU/l) activities; and decreased ALP (61 IU/l) activity. The whale died early the following morning, 10 weeks after initial illness.
Hematologic and biochemical test results of the killer whale necropsied in the present study (July, August, and September) and reference ranges.*
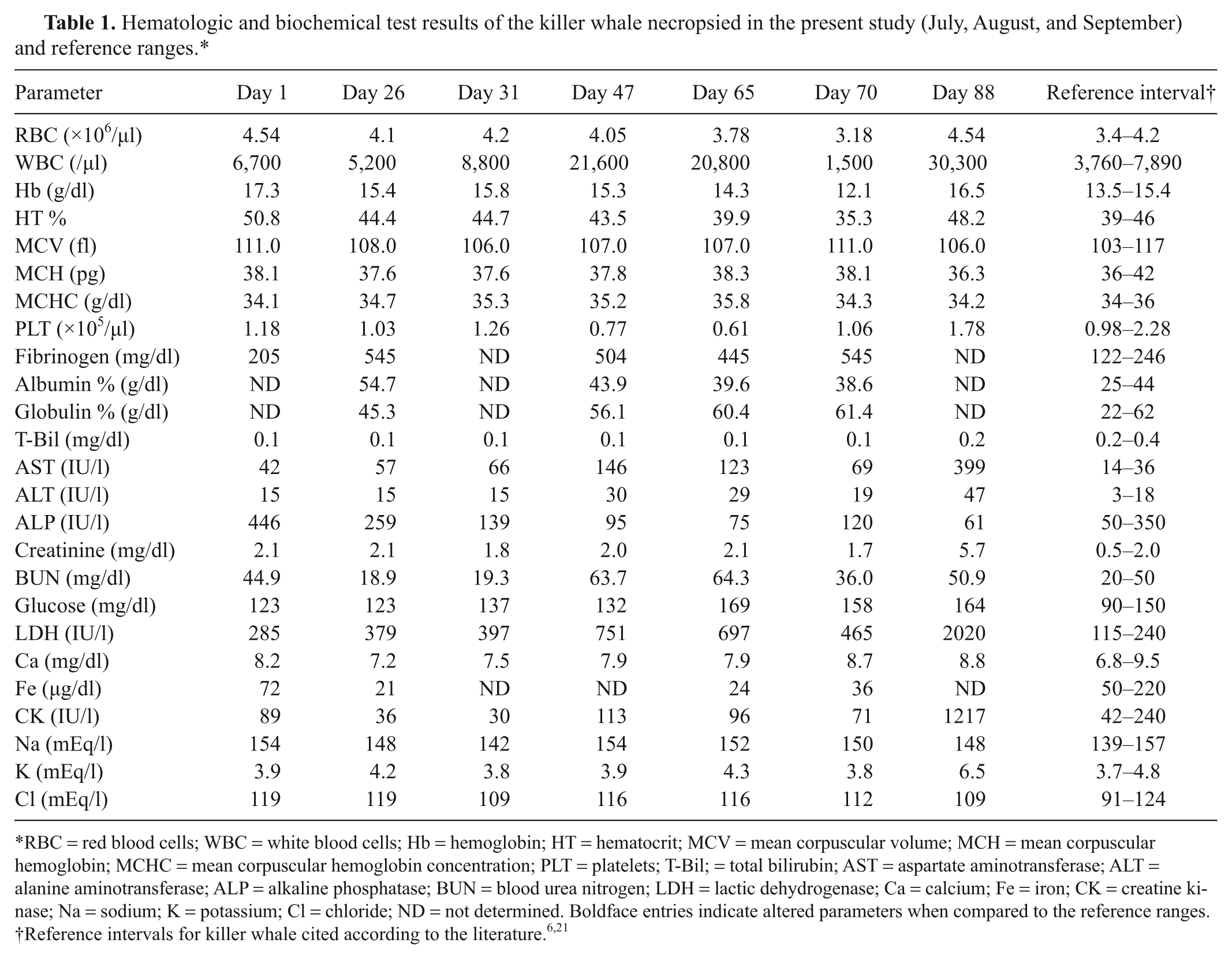
RBC = red blood cells; WBC = white blood cells; Hb = hemoglobin; HT = hematocrit; MCV = mean corpuscular volume; MCH = mean corpuscular hemoglobin; MCHC = mean corpuscular hemoglobin concentration; PLT = platelets; T-Bil; = total bilirubin; AST = aspartate aminotransferase; ALT = alanine aminotransferase; ALP = alkaline phosphatase; BUN = blood urea nitrogen; LDH = lactic dehydrogenase; Ca = calcium; Fe = iron; CK = creatine kinase; Na = sodium; K = potassium; Cl = chloride; ND = not determined. Boldface entries indicate altered parameters when compared to the reference ranges.
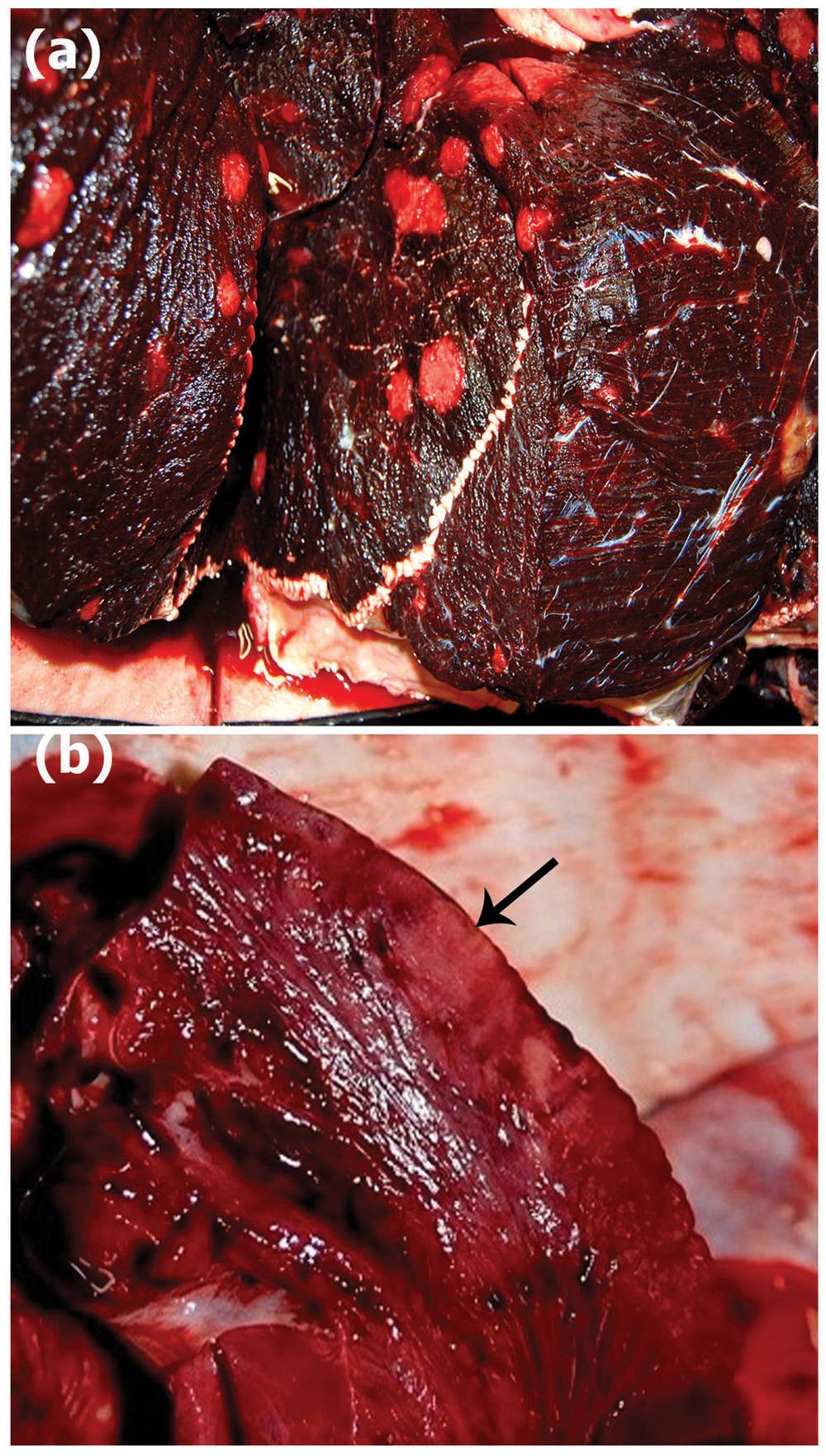
At necropsy, the animal was in slightly poor nutritional condition, with a body weight of 3.2 tons. There was a scar measuring 5 cm in length on the dorsal fin. There were multiple 3–5-cm, well-demarcated, soft, light tan foci in the various muscles of the back and abdomen (Fig. 1a). Generalized lymphadenopathy was found in the subcutaneous lymph nodes, as well as in the thoracic, mesenteric, pancreatic, hepatic, gastric, and lumbar nodes. The subcutis, including fatty tissues in the caudal thorax, was severely edematous. Lymphatic ducts and veins in the pleura around the heart showed marked dilation. The hepatic lymph node was enlarged, with central liquefactive areas. The pericardium contained approximately 0.6 liters of reddish-clear fluid. The epicardial surface was rough and light reddish-tan. There were 2 dark-red nodules, 1 cm × 4 cm, which bulged slightly from the surface of the left ventricle 7 cm from the apex (Fig. 1b). The right and left atria and ventricles were markedly dilated with a large amount of noncoagulated blood. The right and left ventricle walls were moderately thinned and discolored, and were soft. The pulmonary artery was also markedly dilated. In the lungs, there were multifocal consolidations in the caudal lobes. The cut surface was atelectatic, with a large amount of foamy exudate from the bronchi. There were a number of abscesses measuring 1–3 cm in size in the lungs. The liver showed a slight amount of swelling, and there was moderate bulging of the cut surface. The liver parenchyma was dark reddish-brown. The spleen was moderately enlarged, with a wavy thickened capsule. There were multifocal hemorrhages under the capsule. The cut surface was reddish-tan, and trabeculae and lymphoid follicles were not prominent.
Killer whale (Orcinus orca).
Tissue samples were taken from the cardiac lesion in addition to the skeletal muscle, liver, spleen, kidney, heart, lung, stomach, small intestine, uterus, mediastinal and peritoneal lymph nodes, and bladder. The samples were fixed in 10% neutral buffered formalin and embedded in paraffin wax. Sections prepared from these tissues (8 µm) were stained with hematoxylin and eosin. Selected sections were stained with periodic acid–Schiff and Grocott methenamine silver (GMS). Immunohistochemistry was performed on serial sections to identify the fungal organisms. Serial sections were dewaxed in xylene, rehydrated in graded ethanol, and quenched with 0.3% hydrogen peroxide in methanol for 20 min at room temperature. The sections were treated with 3% horse serum a for 30 min, and then incubated overnight at 4°C with either a mouse monoclonal primary antibody specific to fungi of the order Mucorales (Mab-WSSA-RA-1 b ; 1:100 dilution) in Tris buffered saline (TBS; 0.1 M, pH 7.6), or a mouse monoclonal anti–Aspergillus spp. antibody (Mab-WF-AF-1 b ; 1:200). The slides were then treated with biotinylated secondary horse anti-mouse antibody a for 30 min and avidin–biotin complex a for 30 min. The sections were rinsed 3 times with PBS between each step. After treatment, the sections were visualized with diaminobenzidine b for 5 min, washed with distilled water, and finally counterstained with Mayer hematoxylin. As a negative control, TBS was substituted for the primary antibodies, and no reactivity was seen.
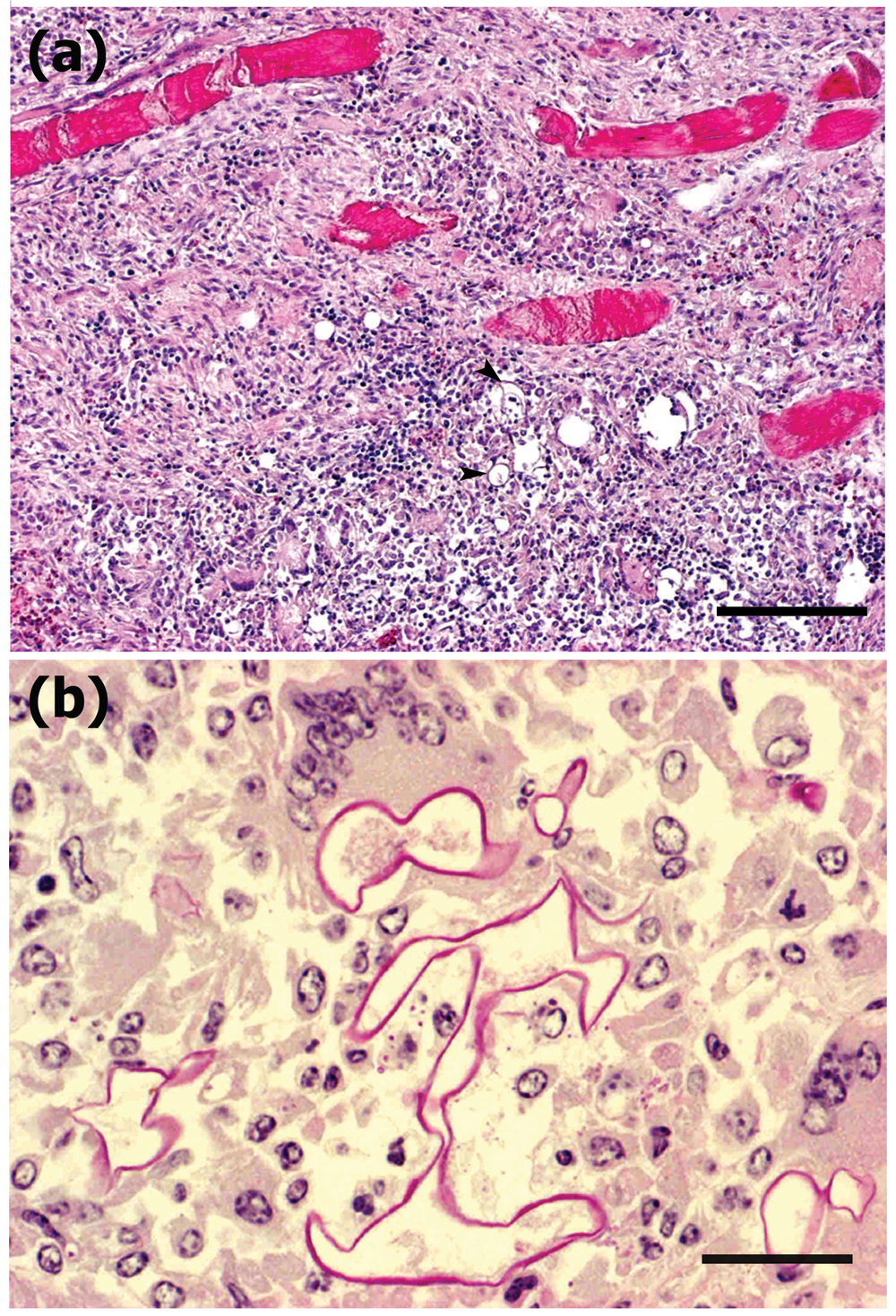
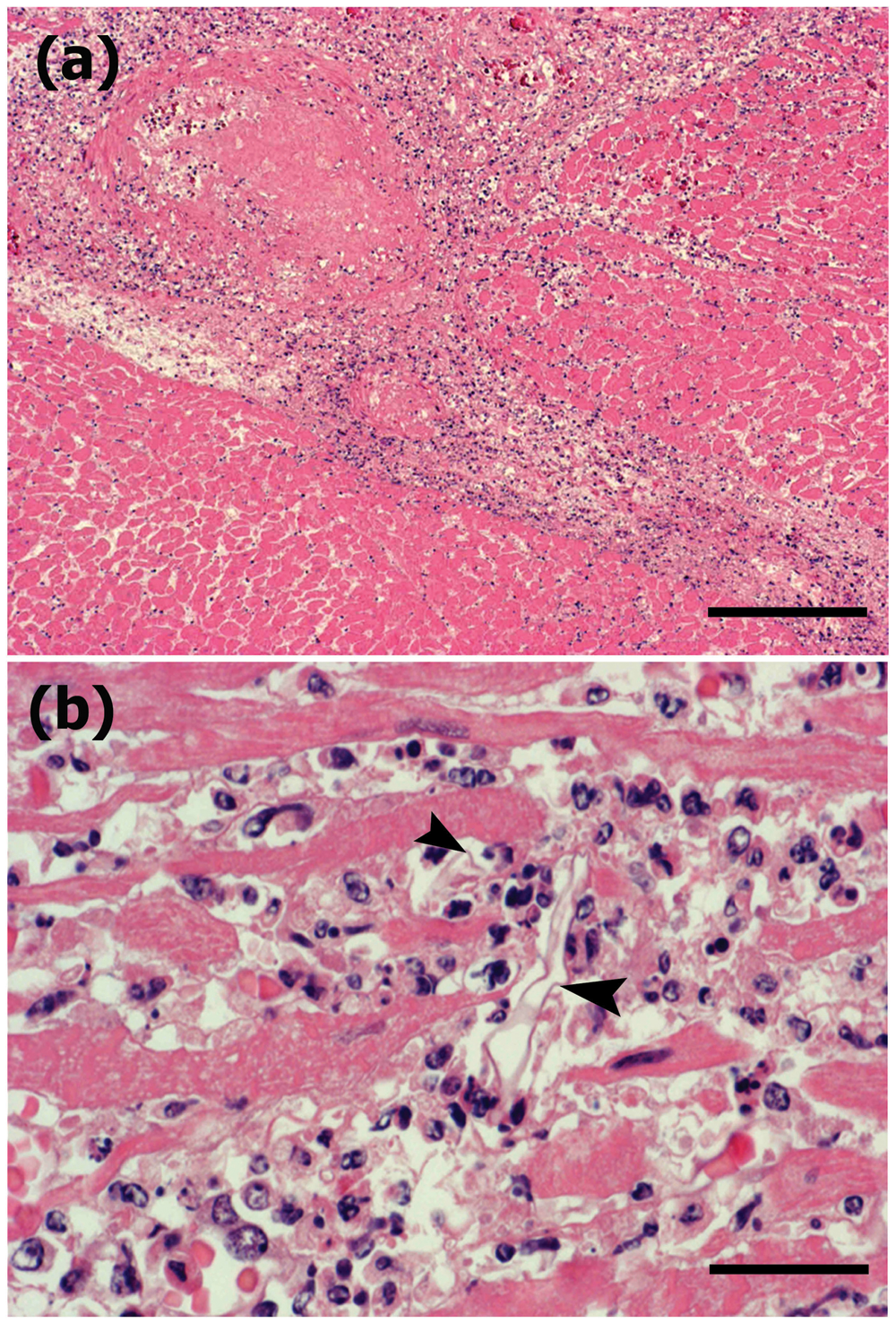
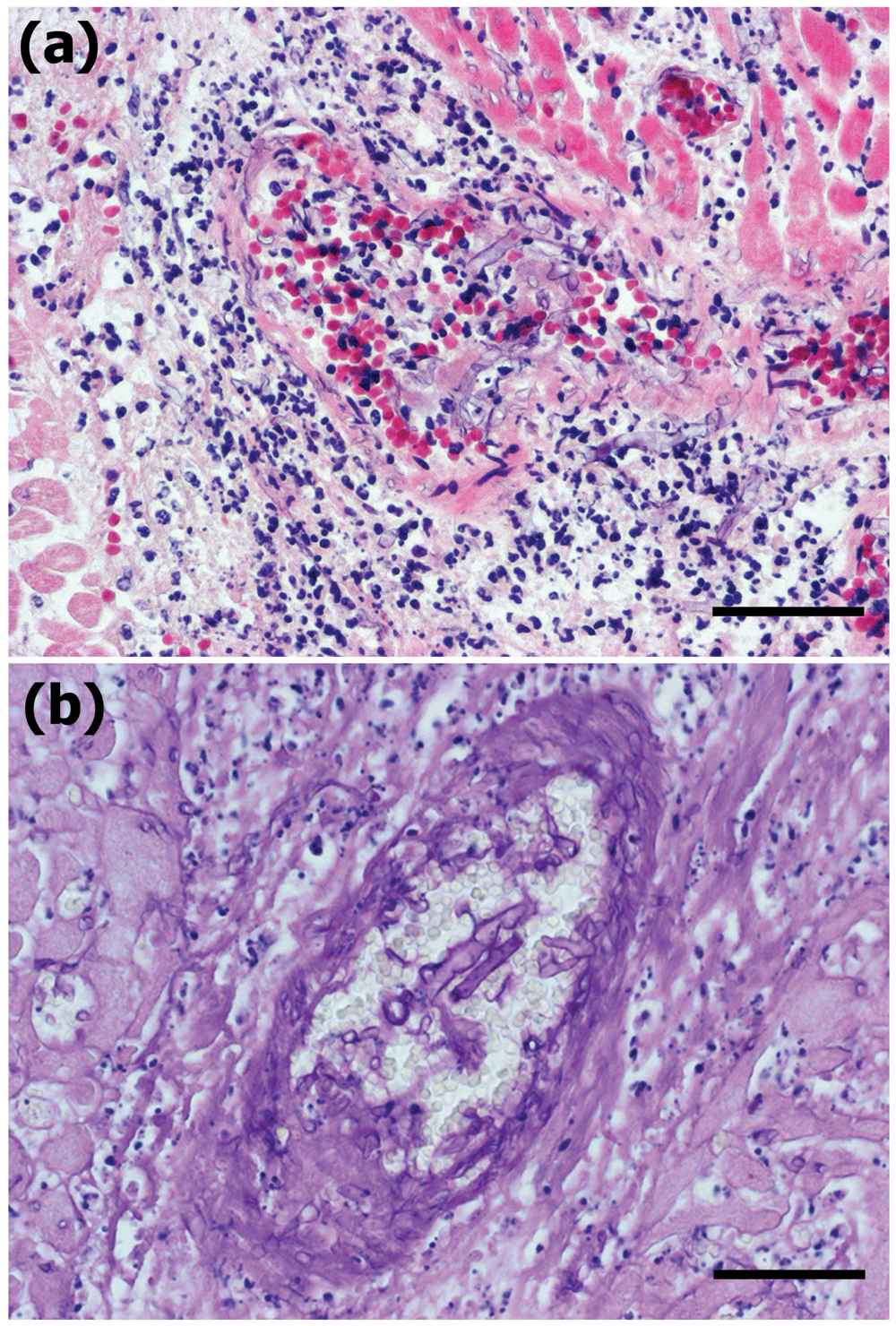
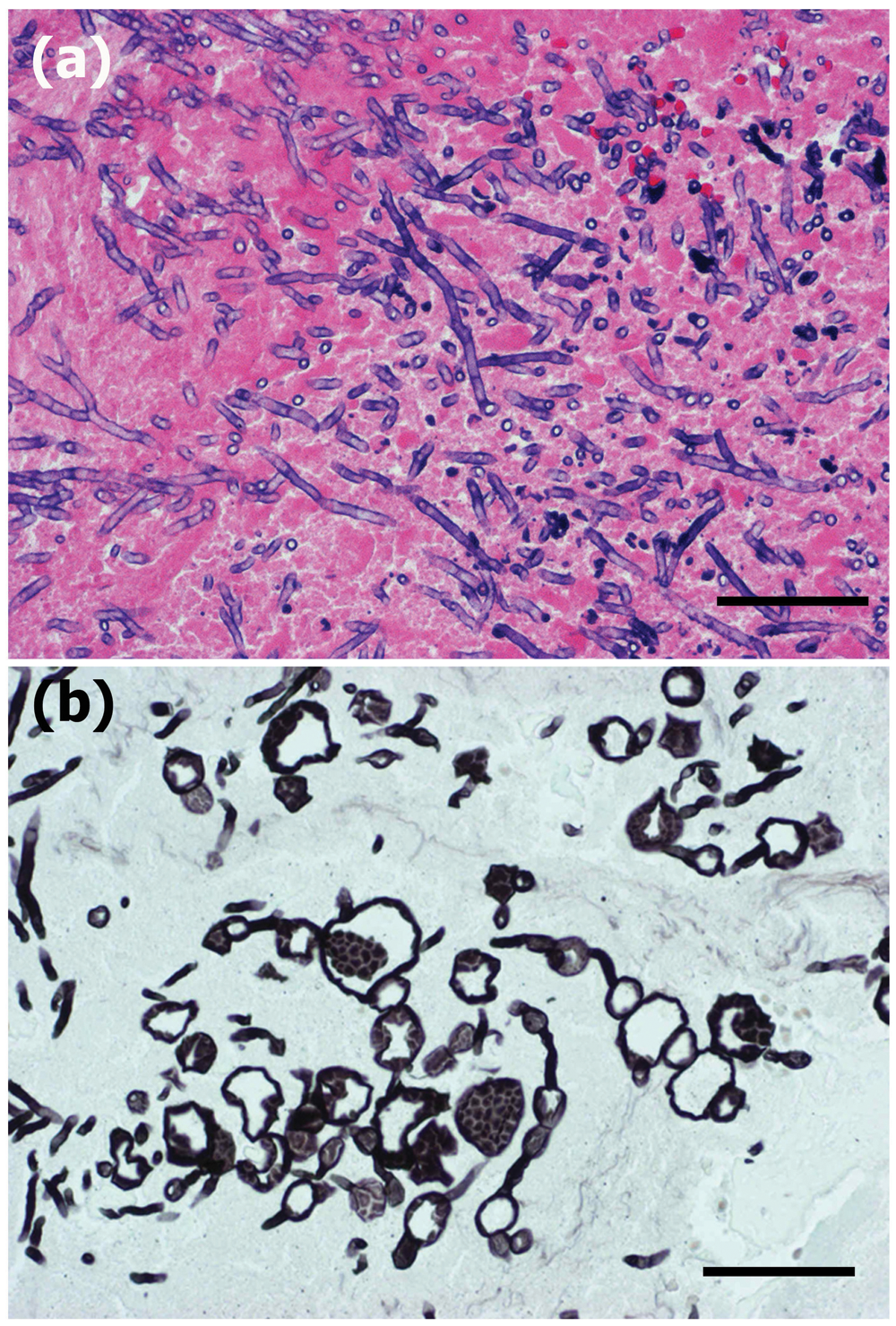
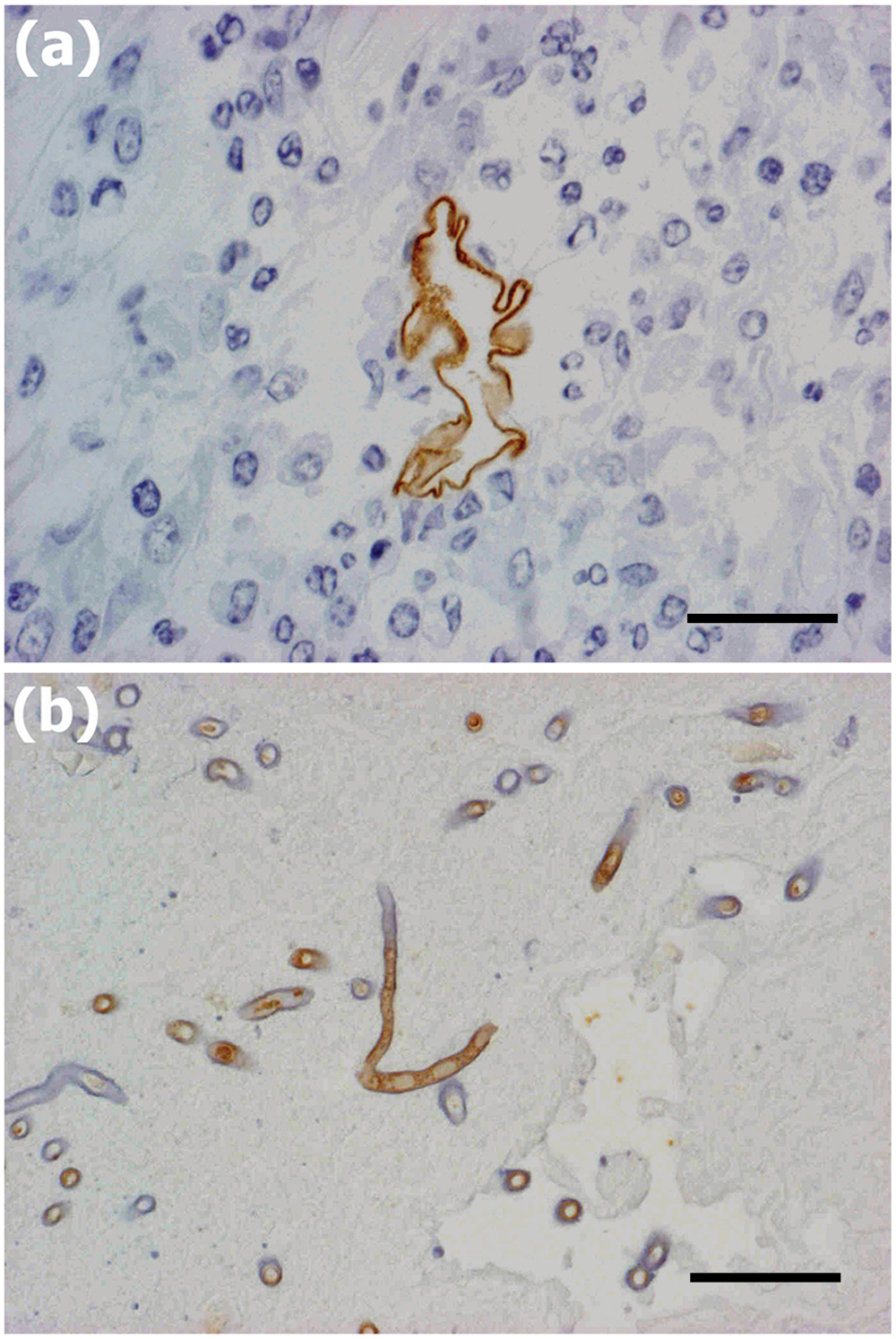
Disseminated fungal pyogranulomatous lesions with varying degrees of necrosis in the back and abdominal muscles corresponded to gross multifocal light tan lesions. The granulomas contained a large number of epithelioid macrophages and foreign body cells engulfing round to spherical structures of degenerative fungal hyphae. There were frequent extracellular fungal organisms, and hyphal elements were scattered and thin walled, frequently with septate and often wrinkled hyphae (Fig. 2). Superficial lymph nodes such as the mandibular nodes showed severe lymphadenitis with marked congestion and edema. In the subcutis of the peduncle, there was severe edema between the muscles and fatty tissues. The thoracic and visceral lymph nodes also showed severe lymphadenitis with marked congestion and edema (thoracic, pulmonary, hepatic, mesenteric, lumbar, and renal nodes). In the heart, multifocal myocarditis was observed with large numbers of neutrophils and macrophages in the myocardium in both the right and left ventricles. In addition to myocarditis, multifocal to diffuse necrosis and prominent infarcts were often observed in the myocardium (Fig. 3), which were more prominent in the left ventricle. In the areas of infarction or myocarditis there were frequent fungal emboli, in which there were thin-walled basophilic hyphae measuring up to 40 µm in diameter. The hyphae were infrequently septate, twisted, and collapsed, had variable branching, and invaded the vessel walls of the small vessels (Fig. 4). The pericardium exhibited moderate degrees of multifocal pericarditis. In the lungs, there was multifocal bronchopneumonia with frequent larger necrotic areas surrounded by infiltrates of neutrophils and mononuclear cells in areas corresponding to macroscopic multifocal consolidation, which contained occasional cavities with numerous typical hyphae of Aspergillus sp. The hyphae were uniform in width, approximately 3–7 µm in diameter, septate, and dichotomously branched, and contained clusters of spore-like structures visible with GMS stain (Fig. 5). Immunohistochemically, Mucor-like structures in the heart and skeletal muscle showed positive reaction to anti–Mucor sp., and Aspergillus-like fungi in the lungs were positive for Aspergillus sp. (Fig. 6). There were also varying degrees of interstitial pneumonia and alveolar edema with marked alveolar and peribronchial infiltration of lymphocytes and macrophages. The liver had slight lymphoplasmacytic infiltrations in dilated sinusoids with occasional extramedullary hematopoiesis. The spleen showed marked congestion with slight lymphoid hyperplasia and extramedullary hematopoiesis.
Killer whale (Orcinus orca).
Killer whale (Orcinus orca).
Killer whale (Orcinus orca).
Killer whale (Orcinus orca).
Killer whale (Orcinus orca).
Within a few days after initial presentation, the predominant findings consisted of the sudden onset of illness and elevation of serum LDH, AST, and ALP activities. Increased LDH activity is a nonspecific indicator of cellular injury 6 and could result from muscle disease, trauma, liver disease, or hemolysis. 33 Aspartate aminotransferase activity generally increases as a result of both hepatic and muscular damage, with an abrupt increase in the case of muscle damage.5,33 Alanine aminotransferase activity elevation has been described as an indicator of liver damage, even though it may also increase in muscle, bone, and kidney injury. 5 Blood samples again revealed increases in serum LDH and AST activities as well as fibrinogen concentration, while ALP activity and the WBC were within reference intervals. Fibrinogen levels often increase with zygomycosis, 25 but this is a nonspecific indicator of inflammation. 32 Despite the treatment, serum LDH, AST, and ALT activities as well as fibrinogen concentration continued to rise progressively throughout the course of the illness. Acute onset of illness, lack of response to antimicrobials, a rapidly deteriorating condition, and increased serum levels of AST and LDH activities are the most common findings in mycoses.19,25 An elevated serum ALT activity is sometimes seen in liver disease, muscle trauma, neoplasia, parasitic infections, and after administration of antibiotics and corticosteroids. 33 Alanine aminotransferase activity was also elevated in a finless porpoise (Neophocaena phocaenoides) with mycotic pericarditis. 19 In addition to a continued decrease in serum ALP activity, a subnormal CK value was also detected in the whale in the present case. In a stranded pygmy killer whale (Feresa attenuata), a low serum ALP value suggested that the animal may have been suffering from a critical illness. 26 Although a higher CK value usually indicates severe necrotizing muscles lesions,5,36 subnormal CK value is sometimes associated with myopathy, muscle wasting, cachexia, or corticosteroid therapy.14,27 Based on the hematological findings, a presumptive diagnosis of muscle damage was made in the present study. Follow-up laboratory testing also revealed leukocytosis in addition to the increased serum enzyme activities. It has been reported that killer whales exhibit a strong leukocytic response to infection. 32 The leukocytosis and elevated serum antibody titer to Simplex virus along with the signs of lymphadenopathy and skin lesions are consistent with herpesvirus infection. Progressive elevation of WBC, increased creatinine and potassium concentrations, and increased LDH, AST, ALT, and CK activities were observed before death, and these are consistent with reports describing mycoses exhibiting an aggressive nature.19,25 Hyperkalemia may be associated with rhabdomyolysis in various animal species, including members of the Tursiops genus. 5 In addition, laboratory test results demonstrated that the hemoglobin, albumin, and globulin concentrations were within reference intervals, while serum iron had decreased.
In the present case, the killer whale was infected with 2 types of fungus: Mucor and Aspergillus species. Aspergillus spp. accounts for the majority of pulmonary mycoses causing pneumonia in marine mammals. Aspergillus fumigatus has been also recovered from the brains of bottlenose dolphins (Tursiops truncatus) and harbor porpoises (Phocoena phocoena), 9 as well as from a northern bottlenose whale (Hyperoodon ampullatus), in which it caused fatal encephalitis. 8 It has also been recovered from the ear of a harbor porpoise in which it caused otitis media. 28 Mucormycosis has also been reported in the brain and other organs of a harbor porpoise found dead on the coast of the German Baltic Sea, 35 and also in a case of granulomatous pericarditis associated with systemic mucormycosis in a finless porpoise. 19
Mycoses may be indicative of underlying immunosuppression. 12 Mycotic infections have been reported as a critical cause of mortality in captive killer whales.13,23,24 Although preexisting immunosuppressive manifestations were uncertain in the present case, the stress associated with captivity may contribute to immune suppression in such animals.
The killer whale may have become infected with Aspergillus sp. from a contaminated aquatic environment; the fungus enters the body through the respiratory tract and can then disseminate to distant sites. Systemic mucormycosis can originate from any of the primary sites of infection, particularly the skin, lung, nasal sinus, and alimentary tract.18,31 It is possible that the fungus initially entered through the skin, causing multiple fungal pyogranulomas, which could have been the primary site of infection, and then spread indirectly as the blood circulated. 25
It has been reported that steroid administration can enhance fungal infection. Dexamethasone induces changes in bottlenose dolphins that include lymphopenia, neutrophilia, eosinopenia, and depressed adrenocorticotropic hormone and cortisol levels within 24 hr of administration. 22 Also, pharmacological doses of the corticosteroid hydrocortisone modestly enhanced the growth of A. fumigatus in cultures. 20 Moreover, it has been found that higher fibrinogen concentrations facilitate specific binding of A. fumigatus conidia to fibrinogen, as well as to the basement membrane component laminin. Thus, higher fibrinogen concentrations could facilitate fungal infection by promoting the adherence of fungal conidia. 7
Based on the skin lesions, lymphadenopathy and serological evidence of Simplex virus, a viral infection was suspected; however, there was no histological evidence of such an infection. The absence of intranuclear inclusion bodies in the infected tissues and tissue culture was also reported in a harbor seal (Phoca vitulina) infected with an outbreak of a herpesvirus. 4
Although it has been widely demonstrated that viruses are species-specific, alphaherpesviruses are considered the least specific among the Herpesviridae family. 17 Herpesviruses have been reported in only a small number of species of cetaceans. To date, most of the recorded herpesviruses were limited to cutaneous and mucosal forms.2,16,29,34 Few reports are available concerning systemic herpesviruses infections; however, a case of encephalitis in a harbor porpoise has been reported, 15 in addition to fatal disseminated herpesviral infections in the cases of 2 immature female bottlenose dolphins. 3 Unfortunately, no further investigations were done to confirm the serological evidence for Simplex virus antibodies; however a novel alphaherpesvirus similar to Simplex virus was isolated from cutaneous and mucosal lesions in an Atlantic bottlenose dolphin, 29 and in addition, a Simplex virus–like infection has been detected in the brain and lungs of a stranded bottlenose dolphin. 10
It should be noted that the source of the herpesvirus infection in the killer whale was uncertain. The current case may have been an immunologically naive animal overwhelmed by initial infection with herpesvirus, or alternatively, recrudescence of latent herpesviral infections secondary to physiologic stress or immunosuppression. 3
In the current case, the most likely direct cause of death was disseminated mycosis and heart failure due to myocardial necrosis and infarction, which was related to fungal embolism, similar to a case previously described in a finless porpoise. 19 Unfortunately, the pathogenesis of angiotropism of mucormycosis remains unknown, but it is known that mucorales are angiotrophic, with a common feature of vascular invasion leading to subsequent infarction of the surrounding tissues. 11
The features of positive immunodetection of Mucor sp. in the pyogranulomas of the skeletal muscles, together with vascular tropism associated with myocardial embolic lesion, allowed the diagnosis of a disseminated mucormycosis combined with pulmonary aspergillosis in a killer whale. Also, the present case emphasizes the pathogenic potential of herpesvirus for killer whale infections.
Footnotes
Notes
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The authors received no financial support for the research, authorship, and/or publication of this article.
