Abstract
Mycoplasmosis is a well-known cause of morbidity and mortality in small ruminants. Previously recognized outbreaks have involved arthritis, and pneumonia or pleuropneumonia. Modern bacteriology procedures rely less on isolation techniques that require special media for mollicutes given that these species are notoriously difficult to isolate, and rely more on PCR tests. We report an outbreak of arthritis, pleuropneumonia, and mild meningitis affecting dairy goat kids, spanning a period of 3 y, which had unusual epidemiologic characteristics related to husbandry practices. Lesions were characterized by polyarthritis of the appendicular joints, with copious joint fluid and extension of arthritic exudate beyond the joint itself. The cause remained unknown until serendipitous isolation of a mycoplasma on blood agar. Mycoplasmosis was not detected from synovial samples by a general mycoplasma PCR, despite multiple attempts. Isolated colonies were also negative by this general PCR assay. The isolate was identified as Mycoplasma mycoides subspecies capri, using universal 16S primers and amplicon sequencing. Testing of additional isolates from other diseased goats in the herd confirmed that this was the cause of illness. A failure to recognize the distinct nature of organisms of the M. mycoides group of mycoplasmas meant that a PCR test that cannot detect this group of organisms was utilized at first, and the etiology of the illness was overlooked for a period of time. Veterinary pathologists and microbiologists must be aware of the limitations of some PCR assays when confronted with joint disease and pleuropneumonia in small ruminants.
Mycoplasmas are commonly associated with pneumonic and arthritic lesions in goats.5,15,16 Retrospective examination of University of Missouri Veterinary Medical Diagnostic Laboratory (VMDL) case records involving PCR 11 testing of mycoplasmosis in goats produced 77 records, exclusive of the cases described herein. Samples submitted from lung, joints, conjunctiva, genital secretions, milk, and blood were tested over a 13-y period (2005–2018). Of these, 45 (58%) yielded a positive result, and 8 of these were sequenced to identify the species. Detected Mycoplasma organisms were identified as M. ovipneumoniae (5 cases), M. conjunctivae (2), and M. bovoculi (1). None of the M. mycoides group (clade 4) of mycoplasmas was detected by the assay during this period.
Over a 3-y period, a caprine arthritis encephalitis virus (CAEV)-free, 45-doe, multiple breed, dairy goat herd experienced an increased incidence of joint swelling and lameness in young goats. Results of an initial postmortem examination on a 3-wk-old kid in 2015 revealed fibrinosuppurative synovitis involving multiple joints and pleuropneumonia. Bibersteinia trehalosi and α-hemolytic Streptococcus spp. were isolated. Treatment with florfenicol (Nuflor, 40 mg/kg, every 4 d; Merck, Kenilworth, NJ) was partially successful for other members of the herd. The problem abated during the summer and autumn, but reoccurred in kids born into the flock the following spring. During 2016, 14 of 55 kids were observed with lameness and one or more swollen joints, as well as fever (40.3–40.8°C). Six kids were submitted for postmortem examination during this second year. During 2017, a single 3-wk-old goat kid was examined with numerous swollen joints and suspected sepsis. In total, 8 goats, 1–3 wk of age, were submitted for postmortem examination (Table 1).
Signalment and summary of postmortem lesions resulting from Mycoplasma mycoides subspecies capri infections in goat kids.

The farm had a divided system of husbandry for the ~100 goat kids born annually. Male kids were allowed to remain with their dams until weaning, whereas doe kids were separated after birth and given colostrum from either their dam or a pooled colostrum (neither were pasteurized). Thereafter, they were bottle-fed pooled raw milk from 1 of 2 bottles affixed with 1 of 2 nipples. The bottles and nipples were cleaned every other day with dishwashing detergent and dried. Doe kids were housed indoors in separate crates. Although the owner felt that female kids were predominantly affected, 4 male and 4 female kids were submitted for postmortem examination.
Kids presented for autopsy had a median age of 2 wk. At least 2 appendicular joints were swollen in each animal. In these and other joints, synovial fluid was mildly to greatly increased in volume, was variably cloudy, and had decreased viscosity. Carpal joints of 2 goats had circumferences of 10–12 cm. Rayon-tipped swabs (BactiSwab; Remel, Lenexa, KS) were used to collect synovial fluid samples, hence fluid volumes could not be measured. In addition, two 3-wk-old kids had grossly visible fibrinous pleuropneumonia, and one had pericarditis (Table 1).
Multiple tissues were fixed in neutral-buffered formalin for histopathology, and joint swabs were submitted for bacterial isolation (2 of 13 submitted grew aerobic non-pathogens) and PCR detection of general mycoplasmosis (7 goats), using previously published primers. 11 PCR testing of other potential pathogens, including bovine viral diarrhea viruses 1 and 2 (3 goats tested), and bluetongue virus, bovine parainfluenza virus 3, and bovine pneumovirus (1 goat each), were also negative. Four serum samples submitted for CAEV testing by competitive ELISA (VMRD, Pullman, WA) were uniformly negative.
Histologic specimens from the synovium were remarkable for the intensity and extent of inflammation, as has been described by other authors in spontaneous mycoplasmal disease in goats, but not illustrated by them.5,10 In addition to inflammation near the synovial surface, the width of the synovium was heavily infiltrated (Fig. 1A), extending beyond the joint capsule into adjacent muscle in some cases (not shown). Fibrin remained adherent to the synovium in some instances, but often detached during processing. The inflammatory infiltrate contained dispersed mixed leukocytes, with striking large aggregates of degenerate neutrophils (Fig. 1A, 1B). Lymphocytes and plasma cells became more prevalent in some of the older affected kids (not shown). Two kids with pleuropneumonia had a dense layer of fibrin covering the lungs, infiltrated by necrotic leukocytes (Fig. 1C). The associated pneumonia was relatively mild; histiocytic pneumonia without pleuritis was found in 3 other goats. Three of 8 kids also had mild, microscopically identified lymphoplasmacytic meningitis that was not associated with clinical signs (Fig. 1D). Clinical meningitis has been identified previously in an adult goat. 20
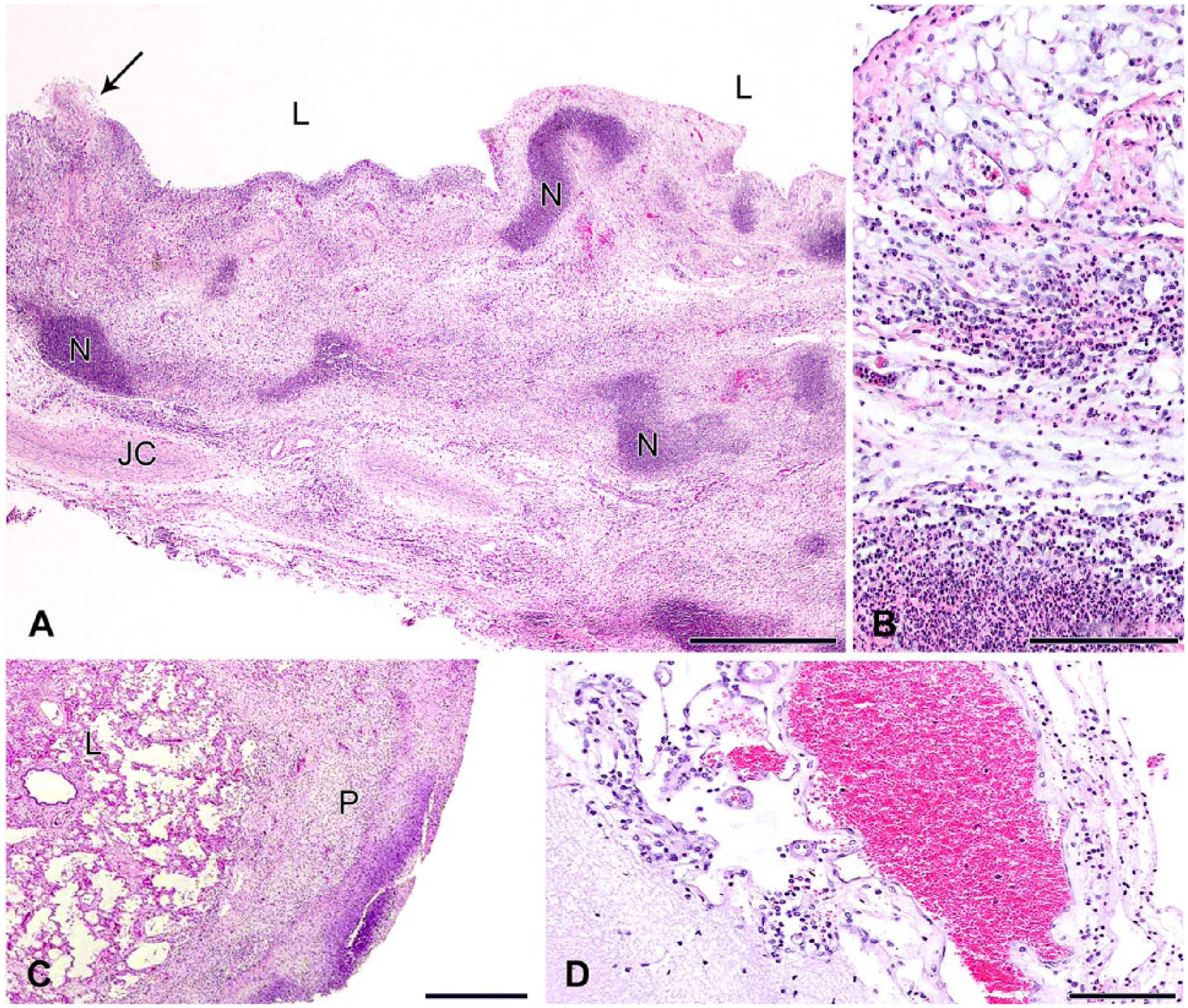
Histologic lesions in goat kids with Mycoplasma mycoides subsp. capri infection.
In 2016, a few small, flat mucoid colonies grew on 5% sheep blood trypticase soy agar (Thermo Fisher-Remel, Lenexa, KS) after 48–72 h of incubation at 37°C from joint swabs of 5 of 6 kids sampled. Tiny, pleomorphic bacteria were found in Gram-stained smears from the colonies. No distinct bacterial cell wall could be distinguished, suggestive of either a bacterial L form or mycoplasma.
To determine whether a mycoplasma etiology might be present, initial end-point PCR testing of joint swabs was performed using primers 11 that span the 16S-23S rRNA spacer region. This region is size-variable between different mycoplasma species, but is typically within the 300–900-bp range. 11 These primers are predicted to be suitable based on the presence of only 1 or 2 mismatches (not at the 3’-termini) for amplification of many of the >100 different mycoplasma species, including 13 species that have been recovered from small ruminants (Supplementary Table 1 and references within). Using the forward primer 5’-ACACCATGGGAGCTGGTAAT and reverse primer 5’-CTTCATCGACTTTCAGACCCAAGGCAT (Integrated DNA Technology, Coralville, IA), this PCR test was uniformly negative in samples from 6 kids.
To identify the recovered organisms, boiled lysates of blood agar–grown colonies were used as templates in a second PCR test (2720 thermal cycler, Thermo Fisher Scientific, Waltham, MA), utilizing universal 16S rRNA forward and reverse primers. 17 Sequence analysis (BigDye terminator cycle sequencing kit, Life Technologies, Grand Island, NY; 3730xl DNA analyzer, Life Technologies) of the 1.4-kb resulting amplicons identified the isolates as M. mycoides subsp. capri. Based on the closely related 16S rrn sequences from M. mycoides cluster taxa, the identity as M. mycoides subsp. capri was further confirmed by sequencing portions of the rpoB and metG reference genes that had been PCR-amplified using primers designed for our study based on M. mycoides subsp. capri genome sequences in GenBank. The primers used for amplification of these targets were rpoBF (5’-CGTAATGTAGAACGTCGTGATTA), rpoBr (5’-TATCATTTTCATCAACATAAGTTA), metGf (5’-ATGTCRAAGAAATTTTATATAACC), and metGr (5’-GAACRTTTTCTAATCCAGATTGTTC), with “R” representing both purines. The resulting sequences were then used to query, by using BLASTn, the complete portfolio of M. mycoides cluster type strain genomes in the National Center for Biotechnology Information databases, which contain multiple complete genomes for each of the M. mycoides cluster representatives. For rpoB, the sequence was 99.2% identical to that from M. mycoides subsp. capri PG3 (GenBank NZ_JFAE00000000.1; 29 single nucleotide polymorphisms [SNPs] in 3,711 bp) with the second closest match being M. mycoides subsp. mycoides SC PG1 (GenBank NC_005364.2; 76 SNPs; 98% identity). For metG, the sequence was 98.2% identical to that from strain M. mycoides subsp. capri PG3 (NZ_JFAE00000000.1; 24 SNPs in 1,352 bp), while exhibiting 97% identity to strain M. mycoides subsp. mycoides SC PG1 (GenBank NC_005364.2; 41 SNPs).
The occurrence of disease was reduced during 2017 (Table 1), after implementing more strict sanitation of milk bottles. Other husbandry modifications included allowing all kids to nurse their dams, not separating kids from their dams until 4–6 wk of age, continued use of florfenicol by the owner, and replacing the herd buck. The occurrence of another case in the third year suggests that the organism was not completely eliminated. Testing of ear swabs from adult goats 1 is ongoing to remove subclinically infected animals. No additional cases occurred in 2018.
Mycoplasmas from the M. mycoides group (clade 4) affect cattle and small ruminants, 6 sometimes with high morbidity and mortality. 12 Species within this group of closely related taxa are thought to have emerged ~10,000 y ago, at the time when ruminants were being domesticated. 8 Some group members show minimal variability between isolates, notably M. mycoides subsp. mycoides small colony biotype, the cause of the OIE-listed disease contagious bovine pleuropneumonia, and M. capricolum subsp. capripneumoniae. 8 Although representatives within the M. mycoides cluster share genetic, taxonomic, and antigenic similarity, as a group they are distantly related to other animal mycoplasmas. 13 PCR assays that are mycoplasma species–specific are primarily used with sequencing to detect differences in geographically diverse isolates of a single species. 22 Other assays or combination s of assays are presently available, aimed at detecting multiple clade 4 species,1-3,10,14 but are often targeted at separating 1 or 2 group pathogens from each other.1,2,10 Some assays use an innovative bead-based multiplex method. 18 The gradient gel motility PCR offers the greatest ability to detect a range of clade 4 and other organisms at the species level, but is technically somewhat challenging. 14
There is controversy over the frequency of M. mycoides subsp. capri infections of goats in the United States, given that some authors have described the infection as uncommonly encountered, 9 yet a high incidence of disease has been reported in individual large herds. 5 Two outbreaks in Mexico of M. mycoides subsp. capri involving thousands of goats resulted in morbidity of ~20% and mortality of ~10%. 21 Both adults and kids were affected over a period of ~2 wk. Respiratory signs predominated, but intra-articular inoculation of the isolate resulted in arthritis and mild meningitis as well. In an outbreak in California with high mortality in dairy goat kids, the authors noted that mycoplasmosis was contagious in kids housed in confinement and suggested that the infection of young kids occurred through colostrum or milk. 10 The husbandry conditions used in the flock in our report produced a control group for kids nursing their dams in which infection was not observed. Nannies were not tested by ear swabs, but pooling of raw milk fed to female kids, in addition to the use of common utensils, most likely resulted in the high incidence of disease reported by the owner.
Experimental infections involving M. mycoides subsp. capri, M. mycoides large colony (subsequently reassigned to the subsp. capri), and M. capricolum produce lesions in multiple organs. 19 In some outbreaks of natural M. mycoides subsp. capri, disease only involved the thoracic cavity.21,22 Experimentally infected kids typically had fever, inappetence, and collapsed at 3–7 d post-inoculation. 15 Interestingly, arthritis was the predominant manifestation of illness in the flock in our study, and only in kids, whereas in other outbreaks, pleuropneumonia has been emphasized.5,21,22
Other clade 4 mycoplasmas also cause disease in sheep and goats in the United States, and cause similar clinical signs. In an outbreak linked to M. capricolum in a goat herd affected during the kidding season, some mortality occurred in yearlings and adults, but all kids died. 4 Both adults and kids developed joint swelling, with a clinical course of days to weeks. Leukocytes also extended into structures adjacent to the joints, as determined by microscopy, 4 very similar to our results (Fig. 1).
Negative findings with a widely used mycoplasma PCR assay 11 led us to re-assess the suitability of this assay for detection of the ruminant pathogens in the M. mycoides group. Phylogenetically, this group species encompasses a very tight cluster of bacteria similar to the Spiroplasma group of mollicutes. 8 Given that the phylogenetic ranking of the M. mycoides group is based on 16S rRNA sequence analysis,13,14 it is perhaps not surprising that this group should be less amenable for detection using primers developed for more typical mycoplasma species. The basis of the general mycoplasma PCR used at our laboratory is amplification of the rrn intergenic spacer region, also referred to as the internal transcribed spacer, using primers that correspond to the 3’-terminal region of the 16S rRNA gene and the 5’-terminal region of the 23S rRNA gene. 11 The resulting amplicon will vary in size depending on the Mycoplasma species from which it is derived. The forward primer has 18 of 20 nucleotide matches to the target sequence in the M. mycoides subsp. capri PG3T 16S rRNA gene. However, the reverse primer has interspersed and limited similarity to its target within the 23S rRNA gene of this type strain (19 of 27 nucleotide matches, longest region of 9 nucleotides). This highlights the need to either further modify the existing primer set to be inclusive of M. mycoides cluster organisms or to use an additional primer pair to selectively identify organisms from this group.
The test we have developed has unfortunately not been adapted directly to fluid or tissue samples, and thus considerable effort must go into making primary mycoplasma isolations prior to testing. Additional sequencing is needed to determine similarities or differences of isolates in a single herd or geographic region.7,22 Given that the etiologic agents of 2 devastating foreign animal diseases, contagious bovine pleuropneumonia and contagious caprine pleuropneumonia, are both key taxa in the M. mycoides group, there is a critical need to have at hand molecular detection assays that can identify these fastidious bacteria.
Supplemental Material
DS1_JVDI_10.1177_1040638719835243 – Supplemental material for An outbreak of Mycoplasma mycoides subspecies capri arthritis in young goats: a case study
Supplemental material, DS1_JVDI_10.1177_1040638719835243 for An outbreak of Mycoplasma mycoides subspecies capri arthritis in young goats: a case study by Gayle C. Johnson, William H. Fales, Brian M. Shoemake, Pamela R. Adkins, John R. Middleton, Fred Williams, Mike Zinn, W. Jeff Mitchell and Michael J. Calcutt in Journal of Veterinary Diagnostic Investigation
Footnotes
Acknowledgements
We thank Don Connor and Barbara Calvin for assistance with illustrations, and the technicians of the University of Missouri VMDL histology laboratory for preparing histopathology specimens. Some of the material in this article was presented orally by G Johnson at the 59th Ann Conf Am Assoc Vet Lab Diagnosticians, 13–19 Oct 2015, Greensboro NC.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplementary material
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
