Abstract
Natural oak toxicity, a phenomenon sporadically reported in the United States, is due to consumption of any part of most oak trees (Quercus spp.). Ruminants, mainly cattle, are disproportionately susceptible to oak toxicity. Toxicity is attributed to degradation of the oak plant hydrolysable tannins by rumen microbes and enzymes into absorbable low-molecular-weight metabolites, which are postulated to bind and damage endothelial cells by unknown mechanisms. The clinical manifestations of acute toxicosis are nonspecific or broadly suggestive of renal disease due to acute tubular injury. Here we document the clinical, gross, histopathologic, and novel ultrastructural features of natural acute oak nephrotoxicity in 3 beef calves on 2 farms in Colorado, USA. Gross postmortem findings included perirenal edema with renomegaly and hemorrhagic gastroenteritis. Histologically, renal tubular epithelial necrosis was severe, with hemorrhage and intratubular hyaline casts. Transmission electron microscopy revealed extensive involvement of proximal and distal convoluted tubules, with predominantly intact basement membranes, and glomerular and interstitial endothelial injury and necrosis. The ultrastructural details of toxic nephropathy and vasculopathy induced by oak metabolites in natural cases of bovine oak toxicosis have not been described previously, to our knowledge.
Natural oak toxicity in cattle caused by consumption of Quercus spp. is a phenomenon that is reported sporadically worldwide.8,9,11,12 Oak toxicity is more commonly reported in countries in which oak is utilized as part of the feed ration or potentially available to forage on rangeland.8,12 In the United States, naturally occurring oak toxicity in cattle has been reported in the Southwest, Northeast, and Midwest.1,13,16 Any part of the plant is potentially toxic, including the leaves, buds, and acorns.2,11 Although the exact mechanisms by which oak tannin metabolites lead to major clinicopathologic findings has not been completely elucidated, there are many known circumstantial factors that favor an increased risk of naturally occurring oak toxicity. 11 The species, season, and maturity of the plant can affect potency. 11 Additionally, younger animals may favor grazing acorns in autumn and oak leaves in spring. 2 Droughts and storms leading to unfavorable grazing conditions or feed restriction followed by consumption of large amounts of the oak plants can also predispose to toxicosis. 2 In an experimental study in cattle, severe feed restriction was found to be a significant factor that favored toxicosis related to the quantity of oak ingested; prior feed restriction may allow absorption and systemic distribution of larger amounts of tannin metabolites.8,11
Oak toxicity is attributed to hydrolyzed metabolites derived from the degradation of tannins.4,14 Tannins are a heterogeneous group of polyphenolic plant metabolites that are able to interact with, bind with variable affinity, and precipitate macromolecules such as proteins, polysaccharides, microbial cell membranes, and enzymes involved in protein and carbohydrate digestion.4,8,14 Tannins can be classified into hydrolysable and non-hydrolysable (condensed) groups based on their chemical structure.4,14 In the gastrointestinal tract, microbial and enzymatic degradation of hydrolysable tannins forms low-molecular-weight metabolites that include tannic acid, gallic acid, ellagic acid, and pyrogallol.4,11 Tannin metabolites are postulated to induce direct gastrointestinal mucosal irritation leading to ulceration and hemorrhage.6,11 Once absorbed, the metabolites exert systemic effects including hepatocellular degeneration and endothelial damage–associated perirenal edema, hydrothorax, and ascites.6,11 Tannin metabolites are also thought to impair protein reabsorption in renal tubular epithelium and cause cortical tubular necrosis with formation of intratubular protein casts and hemorrhage.11,14 Life-threatening systemic complications include uremia and disseminated intravascular coagulation.6,11
Natural oak toxicity has been reported in cattle, sheep, goats, llamas, horses, moose, avian species, and a dog.1,3,5,7,10,15,17 Although many species are susceptible, cattle are the species reported most commonly to be clinically impacted by the negative effects following consumption of oak elements. 11 The increased susceptibility of cattle may be partly due to decreased amounts and decreased affinity of tannin-binding salivary proteins (TBSPs) compared to species such as goats and pigs. 14 Salivary TBSPs form stable complexes with oak tannins in the oral cavity and hamper their ability to interact with other molecules. 11 TBSPs may prevent interaction between tannin and digestive enzymes and thus prevent degradation into toxic metabolites.4,8,11,14
Common clinical signs associated with oak toxicity include anorexia, depression, dehydration, brisket edema, polyuria, polydipsia, hematuria, ruminal stasis, constipation, and mucoid or hemorrhagic diarrhea. Common gross findings include perirenal edema and ulcerative and hemorrhagic gastroenteritis.4,8,11 Histologically, groups of necrotic cortical tubules are expected, and tubular hemorrhage is a distinctive feature that elevates oak on the differential list of acute renal nephrotoxins. 6 Chronic cases may develop chronic interstitial nephritis and fibrosis. 6
Here we describe the clinical, gross, histopathologic, and novel ultrastructural features of the renal lesions associated with acute oak toxicity in 3 beef calves from 2 farms in Colorado.
Farm 1. Calf 1 was a 3-mo-old male Angus mixed-breed steer calf from a farm in Fremont County, Colorado (elevation 1,520–3,660 m), evaluated in early June 2022. The attending veterinarian reported 6 of 30 calves with signs including bruxism, labored breathing, dehydration, difficulty rising, difficulty passing manure, and unspecified neurologic signs. The herd was pastured on a native grass meadow with oak trees. A severe late snowstorm had covered the pasture with 1 m (3 ft) of snow the week before the onset of clinical signs. The calves were reported to have sheltered under the oak trees during the storm.
Farm 2. Calves 2 and 3 were 3.5-mo-old heifer beef calves of unspecified breed from a farm in Huerfano County, Colorado (elevation 2,040–4,150 m). The owner reported multiple calves with anorexia, depression, and dullness, and 4 dead calves during late May 2022. The number of unaffected animals in the herd was not reported. Calves were reported to have been exposed to oak buds (Quercus spp.) for several days because of a late snow 10 d prior. Adult cows on the property were not affected.
Field autopsies were performed on the 3 calves by the referring veterinarians (Fig. 1). The time between death and autopsy was not reported. Gross postmortem lesions are not available for calf 1. Calves 2 and 3 had marked perirenal edema, swollen dark-purple kidneys that bulged on cut section, and prominent white streaks through the cortex. These calves also had ventral perianal and vulvar edema, hydrothorax, hydropericardium, ascites, mesenteric edema, and renal lymphadenomegaly.
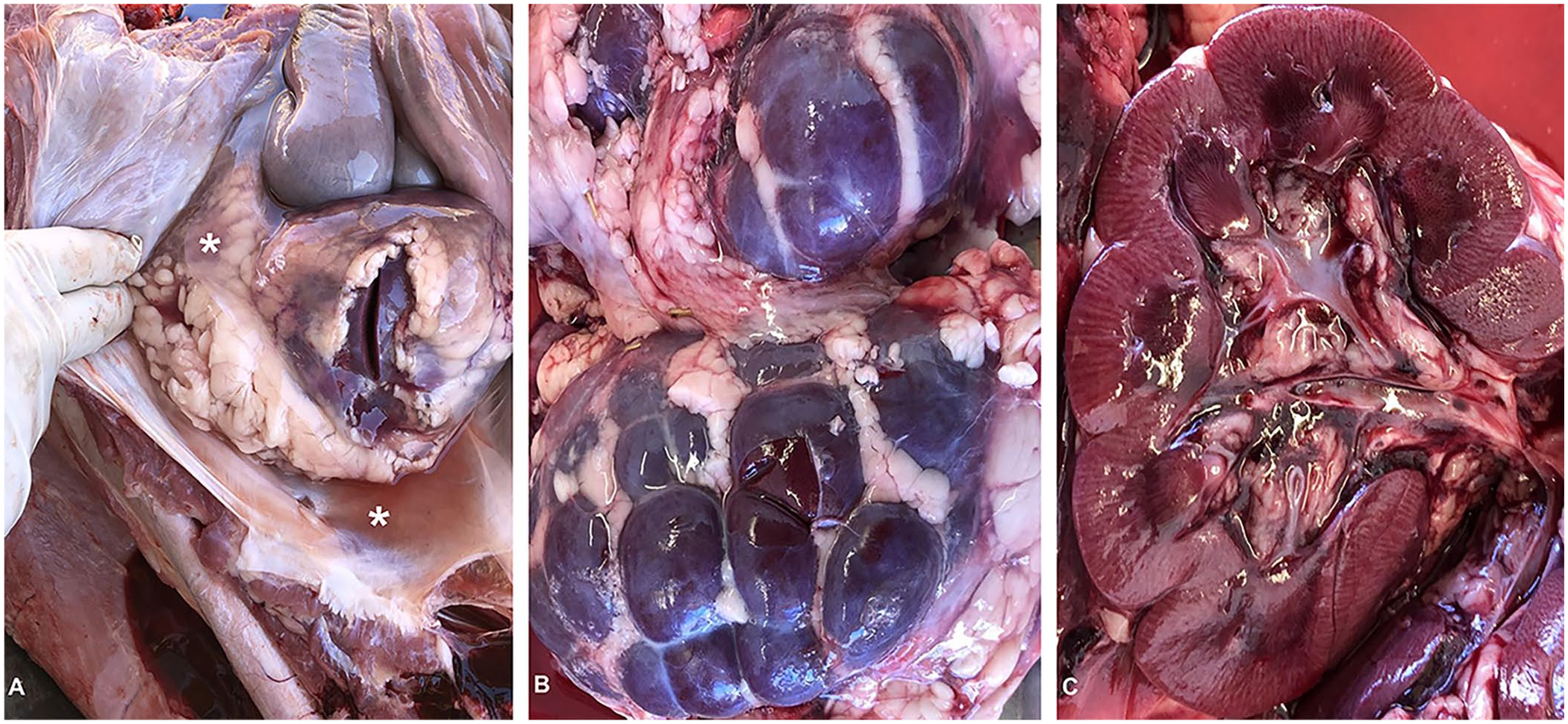
Acute oak nephrotoxicity in beef calves in Colorado (calf 2).
Formalin-fixed tissue samples of liver and kidney from calf 1; lung, liver, and kidney from calf 2; and lung, spleen, kidney, and heart from calf 3 were submitted in 10% neutral-buffered formalin, processed routinely to 4-μm sections, and stained with H&E. Microscopically, tissues of all 3 calves had mild-to-moderate autolysis, and there was moderate-to-severe acute multifocal tubular necrosis with intratubular sloughed swollen epithelium, hyaline casts, and hemorrhage (Fig. 2A, 2B). The tubular damage was more pronounced in cortical than medullary regions. Additional serial sections were stained routinely with the periodic acid–Schiff (PAS) reaction. These preparations highlighted both intact and effaced basement membranes of affected cortical tubules (Fig. 2C). Endothelial cell detachment and tunica media leiomyodegeneration were evident in interstitial capillaries and small arterioles. Affected leiomyocytes were vacuolated or had hypereosinophilic cytoplasm, and occasionally pyknotic nuclei. Extravasated erythrocytes dissected the perivascular interstitium (Fig. 2D). Mild-to-moderate interstitial lymphoplasmacytic infiltrates were present multifocally throughout the cortex. There were subjectively increased numbers of leukocytes in hepatic sinusoids. Neither hepatocellular degeneration nor necrosis were identified.
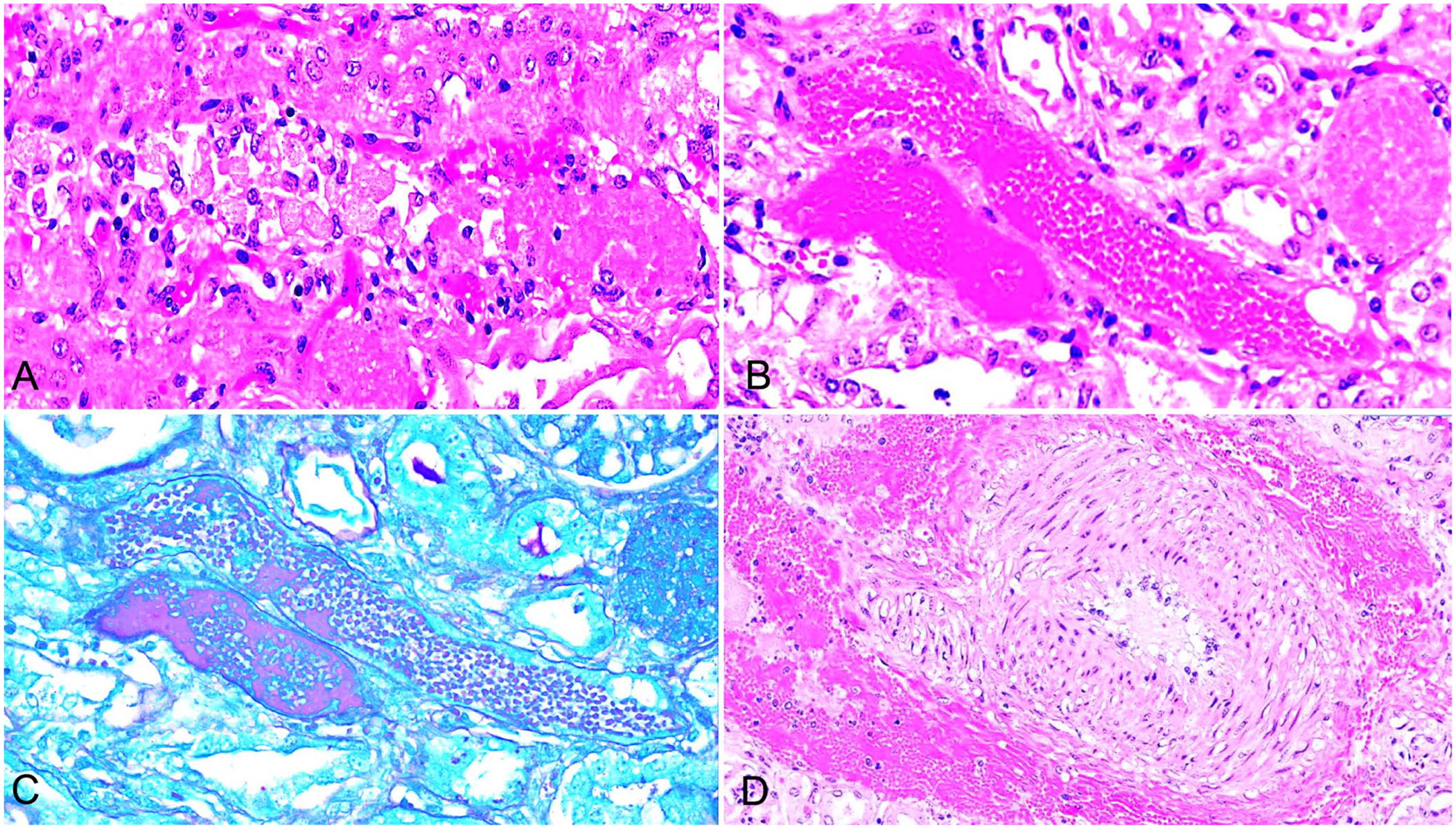
Acute oak nephrotoxicity in beef calves in Colorado (calf 2).
Formalin-fixed samples of kidney cortex from all 3 calves were submitted to the Electron Microscopy Services at the University of Georgia (Athens, GA, USA) for transmission electron microscopy (TEM). Tissues had been formalin-fixed for ~3 mo. Tissues were routinely processed for TEM after post-fixation in a solution of 2% paraformaldehyde and 2% glutaraldehyde in phosphate buffer, followed by osmication and 0.5% uranyl acetate, and embedding in Mollenhauer Epon–Araldite resin (Electron Microscopy Sciences). Toluidine blue–stained thick sections were used to determine areas of interest for ultra-thin sections, which were stained with lead citrate and examined on a transmission electron microscope (Fig. 3). Severe tubular epithelial degeneration and necrosis involved multiple segments of nephrons based on the thickness of basement membranes. Degenerate epithelial cells were swollen with electron-lucent cytoplasm and degenerate organelles, and rafts of necrotic cells were detached from intact basement membranes. Many tubules contained prominent casts of necrotic debris or intratubular hemorrhage. Glomerular changes included capillary endothelial damage with cytoplasmic electron-lucency and degeneration of organelles or overt endothelial necrosis. Podocytes were swollen and lucent with vacuolated cytoplasm and swollen organelles consistent with degeneration. Podocytes occasionally contained intracytoplasmic fragments of erythrocytes consistent with erythrophagocytosis. Interstitial capillaries and small arterioles had endothelial degeneration and necrosis with endothelial cell detachment and local perivascular hemorrhage. There was also medial leiomyodegeneration and necrosis of small arterioles with endothelial detachment.
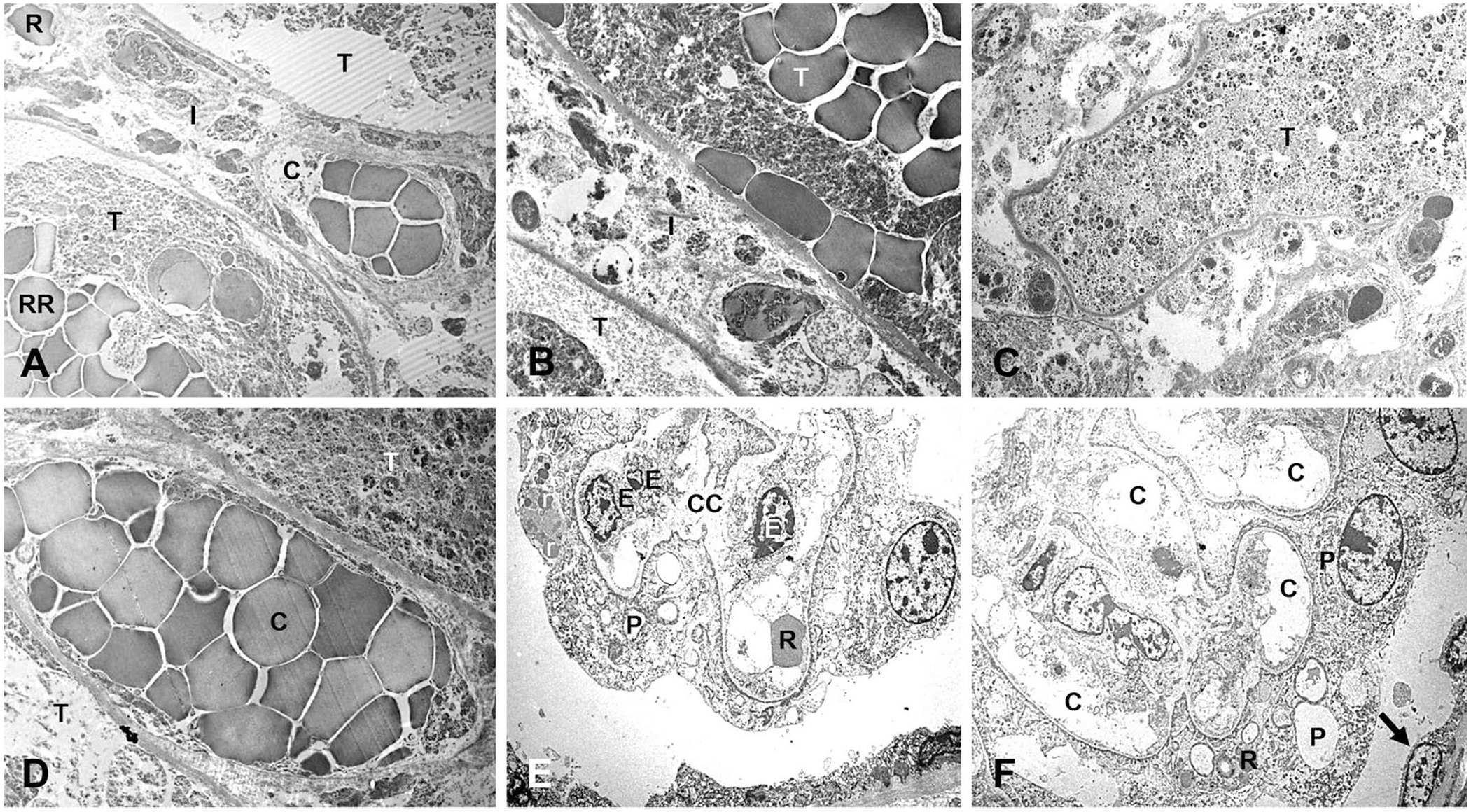
Acute oak nephrotoxicity in beef calves in Colorado (calf 2).
Clinical laboratory data were not available for calves from either farm. Rumen contents from calf 1 were submitted to the California Animal Health and Food Safety Laboratory System (Davis, CA, USA) for gallic acid detection via qualitative gas chromatography–mass spectrophotometry analysis (GC-MS). Results were reported as strongly positive.
Naturally occurring acute oak toxicity in these calves occurred after snowstorms that temporarily limited pasture access, a circumstantial factor that restricted feed and favored prolonged ingestion of available oak tree parts, presumably buds given the time of year. The clinical, gross, and microscopic findings in all calves of both farms were consistent with renal failure from oak toxicity, which was confirmed by detection by GC-MS of gallic acid in calf 1 from farm 1. This test is reported as positive or negative based on conversion of gallotannins to gallic acid for detection. Gallotannins are a broad class of hydrolysable tannins that may be present in different concentrations within oak, and because the specific gallotannins may convert to gallic acid at different rates, the concentration of all tannins cannot be extrapolated in reverse. There is insufficient literature to correlate detection of gallic acid alone with levels of intoxication (Robert Poppenga, pers. comm., 2023). Unfortunately, information regarding the precise species of oak trees present on the affected properties was not available. Gambel oak (Quercus gambelii) is the only species native to Colorado, although nonnative species are also present in the region and include burr oak (Q. macrocarpa), red oak (Q. rubra), pin oak (Q. palustris), English oak (Q. robur), and swamp white oak (Q. bicolor).
Ultrastructural examination of oak toxicity cases has been limited to experimental cohorts in rodents and sheep. 18 We performed an advanced search query via Google, CABI Direct, Web of Science, Scopus, Sage Journals, and PubMed biomedical literature databases using combinations of the following key terms: “oak”, “Quercus”, “toxicosis”, “toxicity”, “nephrotoxicosis”, “nephrotoxicity”, “ultrastructure”, and “electron microscopy”. This systematic search did not yield published literature that documented the ultrastructural examination of oak toxicity in natural cases of bovine oak toxicity, suggesting that these features of oak toxicity have not been reported previously in cattle. Extensive involvement of both proximal and distal renal tubules was present in our cases, which contrasts with experimental findings in sheep in which only proximal tubules were involved. 18
Tubular changes were extensive histologically and ultrastructurally, which greatly interfered with the distinction of which segment of tubule was involved. Intact basement membranes visible in PAS preparations and ultrastructurally suggest classical toxicity. However, the histologic and ultrastructural evidence of endothelial damage and leiomyocyte damage suggests that ischemic injury could contribute to the pathogenesis of oak toxicity. Furthermore, occasional effacement of basement membranes could also support vascular injury contributing to disease. Whether the mechanism of oak nephrotoxic vascular injury chiefly targets the vascular endothelium with sequela to the smooth muscle vascular wall or both simultaneously is unclear and could further be hypothesized to act in a dose-dependent manner. We speculate that direct vascular injury results in vasoconstriction and a decreased vasodilation response of small renal arterioles, tubulo-glomerular feedback in response to pre-glomerular arteriolar vasoconstriction, enhanced leukocyte-endothelial adhesion and activation, and local activation of the inflammatory and coagulation systems known to contribute to renal ischemia and reperfusion injury. The extensive tubular damage could be the result of direct toxic tubular damage compounded with renal ischemia or reperfusion injury further to the toxic vascular damage. Further studies may provide a better understanding of vascular injury in oak toxicity.
Our renal ultrastructural findings suggest that naturally occurring oak toxicity affects multiple cell types in the kidneys of cattle, including glomerular and interstitial endothelial cells, podocytes, and renal tubular epithelial cells across multiple tubular segments. Identification of endothelial damage and necrosis in the glomeruli would explain the intratubular hemorrhage that is a useful histologic feature of this particular toxicity. Our findings contribute to the understanding of natural oak toxicity in cattle.
Footnotes
Acknowledgements
We thank Chelsea Sykes (Toxicology Section, California Animal Health and Food Safety Laboratory, School of Veterinary Medicine, University of California, Davis, CA, USA) for assistance in interpretation of the gallic acid GC-MS results. We thank Mary Ard (Electron Microscopy laboratory at the Department of Pathology, College of Veterinary Medicine, University of Georgia, Athens, GA, USA) for assistance in sample preparation for electron microscopy.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
