Abstract
Giardia duodenalis is a common parasite of the gastrointestinal tract of dogs, with an especially high prevalence in dogs <1-y-old. Methods for detecting G. duodenalis are point-of-care (POC) tests such as lateral-flow tests or fecal flotation. The Vetscan Imagyst (Zoetis) is a new POC device for the detection of G. duodenalis in fecal samples using zinc sulfate flotation, automated slide scanning, and image recognition with artificial intelligence. Vetscan results are the number of Giardia cysts per coverslip. We compared the performance of the Vetscan and another POC test (SNAP Giardia test; Idexx) with a direct immunofluorescence assay (IFA) performed in a specialized parasitology laboratory as the reference test. We included 164 dogs <19-mo-old. We used pooled fecal samples from 3 defecations gained within 2–3 d and tested the repeatability of the Vetscan by triplicate measurement. Compared to IFA, Vetscan had a diagnostic sensitivity of 88.4% and specificity of 98.1%; SNAP had a diagnostic sensitivity of 74.4% and specificity of 98.1%. A variation coefficient of 67.0% was determined for the Vetscan results. The performance of the Vetscan is acceptable for the qualitative evaluation of fecal samples (Giardia positive or negative), and the device can be used by untrained personnel. Given its high variation coefficient, we do not recommend the Vetscan for monitoring the number of cysts.
Giardia duodenalis is one of the most common parasites of the gastrointestinal tract in dogs and cats.2,19,23,25,30,35 Infection with G. duodenalis may either cause clinical signs or remain subclinical. 5 Typical clinical signs of giardiasis in dogs and cats are acute or chronic intermittent, thin-to-watery diarrhea, with or without mucus. Nausea, vomiting, malabsorption, weight loss, and lethargy are also observed frequently.1,10,34 Worldwide, the mean prevalence of G. duodenalis has been reported as 15% in adult dogs and 12% in adult cats. 5 In animals <6-mo-old, the prevalence can be up to 50%.4,38 Prevalence may vary depending on the test method used5,12 as well as the amount of fecal samples tested, due to the intermittent shedding of Giardia cysts.9,16,17,21,36
Given its high sensitivity and specificity, several studies consider the direct immunofluorescence assay (IFA) as the reference method for the detection of G. duodenalis in dogs.3,14,15,31,32 The IFA is a direct detection method that uses specific monoclonal antibodies against epitopes of the Giardia cyst wall. Combined with fluorescence microscopy, this technique is considered to have an exceptionally low detection limit. 13 For point-of-care (POC) testing of fecal samples, lateral-flow rapid tests or centrifugal flotation with zinc sulfate solution are recommended due to their ease of use as well as their good sensitivity and specificity.32,33 The SNAP Giardia test (SNAP; Idexx) is a lateral-flow test that is used frequently in veterinary practices.3,8,11 The principle of the SNAP test is based on the detection of soluble Giardia antigen, which occurs freely in the feces and is not attached to the cyst wall.18,29
The Vetscan Imagyst (Vetscan; Zoetis) is a POC device for the detection of Giardia cysts in fecal samples. 28 The system is based on centrifugal flotation of the fecal sample with zinc sulfate solution. The centrifuged sample is applied to a glass slide and coverslipped. The area under the coverslip is automatically scanned by the Vetscan, and the composed digital image of the area is screened for cysts using artificial intelligence. The Vetscan provides a quantitative result of the cysts found per slide.26,28 A 2021 study examined 100 samples for G. duodenalis and found a sensitivity of 76% and a specificity of 97% for the Vetscan compared to conventional centrifugal flotation and microscopic analysis of sample slides by an experienced parasitologist. Sensitivity increased to 95% when samples that contained <10 cysts per gram of feces were excluded. 28
SNAP and Vetscan are used as POC tests that, unlike the IFA, can be performed quickly and without any special prior knowledge or training. The Vetscan also offers the possibility of having questionable results evaluated by an experienced parasitologist from Zoetis. Our objectives were to compare the Vetscan and SNAP tests to a reference test (IFA) for the detection of G. duodenalis and to test the repeatability of Giardia cyst quantification by the Vetscan.
Material and methods
Study overview
The collection and use of fecal samples was approved by the ethics committee of the veterinary faculty of Ludwig-Maximilians-University Munich (LMU Munich, Germany; 270-12-05-2021). We examined fecal samples for G. duodenalis in the laboratory of the Small Animal Clinic of LMU Munich using Vetscan and SNAP tests. In addition, an IFA was performed at the Institute of Experimental Parasitology (IEP) of LMU Munich. For each measurement, 3 fecal samples were collected as a pooled sample. The sample collection and pooling method used in our study reflects the method used in practice for Giardia tests: scoops from the 3 samples were taken separately and pooled; sample weight was not standardized. We included pooled samples from 164 dogs of which 150 were measured by all 3 detection methods. To determine the repeatability of the Vetscan results, we performed the measurement 3 consecutive times per sample. For 14 of the 164 samples, we only performed the triplicate measurement using the Vetscan given the small amount of sample material.
Fecal specimen collection
We collected fecal samples between January 2022 and August 2023 in the wider Munich area. Dogs of all breeds and both sexes, up to 19-mo-old were enrolled regardless of fecal scoring. The dog owners collected 3 fecal samples from each dog over 2–3 d and brought them immediately to LMU Munich. The fecal samples were stored in sample tubes at 4°C and tested within 72 h of the start of individual sample collection using the Vetscan and SNAP tests. Technicians at the IEP at LMU Munich examined the samples within 14 d of submission using IFA. Fecal consistency was recorded using the Purina Fecal Scoring Chart (Suppl. Material).
POC testing with the Vetscan Imagyst
As a POC test, the Vetscan is intended to be a simple time-saving tool for testing fecal samples for intestinal parasites. The Vetscan sample preparation kit for Giardia cysts includes stir sticks, glass slides, sample tubes with zinc sulphate solution (specific gravity 1.18) and an attached scoop, a transfer loop, coverslips, and collection tubes and caps. From each of the 3 fecal samples per dog, we placed a scoop (which is attached to the collection tube of the sample material of the Vetscan) of feces in the sample tube and stirred with the stir stick. We then sealed the tube, and centrifugation was performed at 500 × g for 2 min. After centrifugation, we applied the top layer of the sample to the glass slide with the transfer loop and placed the coverslip on top. The glass slide was inserted into the Vetscan Ocus 40 scanner (Grundium), and the scanning process was started via the Vetscan online platform. We tested each pooled fecal sample 3 consecutive times. At the end of the scanning process, the images from the scan were uploaded to a cloud-based server, analyzed, and the results uploaded to the online platform. In the case of a positive test result, photos of the objects found were provided by the Vetscan, and the number of objects classified as Giardia cysts was indicated. The Vetscan workflow requires confirmation of each result before it is logged into the patient file. For each sample that the Vetscan marked positive, and to avoid false-positive results, a trained veterinarian checked the photos to see whether the structures shown were Giardia cysts. If only structures other than Giardia cysts were visible, the measurement was assigned negative. We considered pooled samples with 2 or 3 positive measurements positive, and samples with 1 or no positive result negative.
POC testing with the SNAP Giardia test
We tested 150 samples with the SNAP test, and then tested the same samples with the IFA reference method. The SNAP lateral-flow rapid test is an enzyme immunoassay for the qualitative detection of Giardia antigen in feces. We coated the swab included in the test kit with the material from 3 fecal samples per dog and placed the tube over the swab. We then mixed the conjugate solution with the sample material by pressing the plastic bulb according to the manufacturer’s instructions. We used the swab as a pipette and added 5 drops of conjugate solution to the sample well of the SNAP device. After pressing down the activator, we waited 8 min and then read the result according to the manufacturer’s instructions. The positive control must be positive with every test.
Reference test using an immunofluorescence assay
The testing of the fecal samples for G. duodenalis using the direct IFA was carried out by parasitologists of the IEP LMU Munich. We sent sample material of 3 fecal samples per dog to the IEP as a pooled sample. A spatula tip sample of the pooled fecal sample was used for testing. Soft-to-liquid feces were mixed before sampling; if the feces were solid, the sample material was collected from several locations. The laboratory uses a commercial Cryptosporidium/Giardia test kit (Merifluor; Meridian Bioscience) and fluorescence microscopy at a wavelength of 495 nm. Fluorescence-labeled antibodies bind to antigens on the cell wall of Giardia cysts. Thereby the cysts can be visualized on a slide under a fluorescence microscope. The fecal samples were scored by the parasitology laboratory according to the number of cysts as follows: negative (–); low (+): 1–50; moderate (++): 51–500; high (+++): >500 (each per slide).
Statistical analysis
Data collection and statistical analysis were performed using Excel v.16.76 (Microsoft) and Prism v.5.04 (GraphPad). We calculated the sensitivity and specificity of the Vetscan and SNAP in comparison to the IFA using contingency tables with a 95% CI. We determined the repeatability of the Vetscan measurements of the 3 test results per sample using the CV.
Results
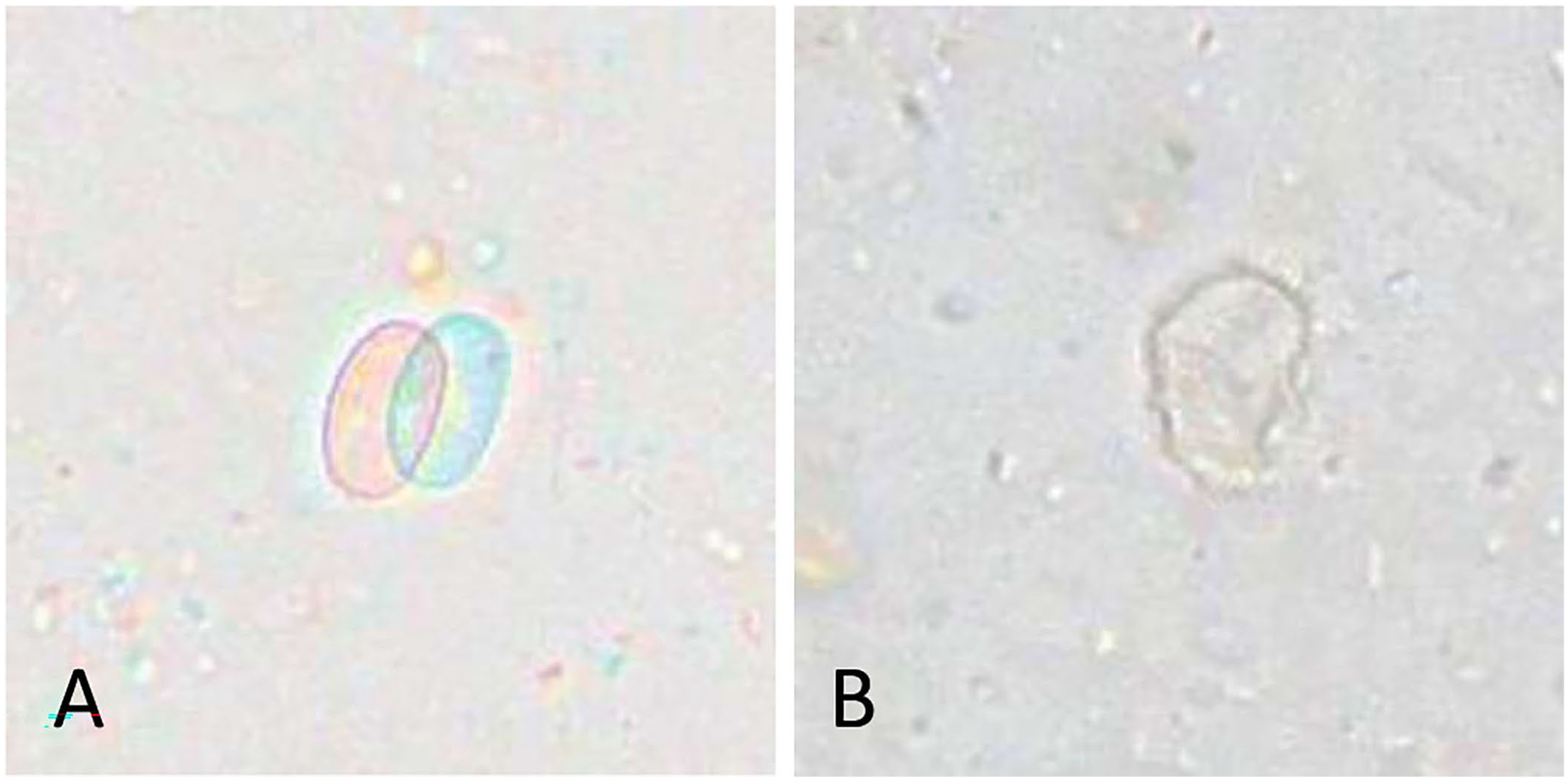
We collected fecal samples from 164 dogs. In the study population, 79 (48.2%) of the dogs were male (4 castrated); 80 (48.8%) of the dogs were female (3 spayed); the sex of 5 (3%) dogs was not specified. We included dogs 1–18-mo-old (median 9.0) in our study. The fecal consistencies of 156 of the 164 samples were recorded according to the Purina Fecal Scoring Chart; a score of 1–3.5 (no diarrhea) was found in 95 (60.9%) dogs. The 61 (39.1%) remaining dogs had a fecal score of 4–7 (diarrhea). Samples from 150 dogs were tested with all 3 tests: IFA, Vetscan, and SNAP. The remaining 14 samples were analyzed exclusively with the Vetscan. Of these 14 samples, 7 samples were positive (3 of 3 measurements positive) and the other 7 samples were negative (3 of 3 measurements negative) for Giardia cysts. Of the 150 samples examined with all 3 detection methods, 107 were negative with the reference method (IFA). The remaining 43 samples were positive with the reference method, representing ~28.7% of the study population. Semiquantitative measurement by IFA showed a low number of cysts in 37.2% of the positive samples, a moderate amount in 53.5%, and a high number of cysts in 9.3% of the measurements. The Vetscan test was positive in 40 samples (Fig. 1) and the SNAP in 34 of the 150 pooled samples. For the 150 samples that were tested with all 3 methods, results were concordant with all 3 methods in 134 samples (89.3%); results were discordant in 16 samples (Table 1). Most of the 16 samples with discordant results either tested negative or had a low cyst count with the IFA (Table 2). Two of the 150 samples analyzed with each test method were positive solely with the Vetscan. The results of the triplicate measurement showed 23, 11, and 31 cysts and 4, 1, and 4 cysts in these samples. Because size and form of the cysts in these samples did not differ from real Giardia cysts (Fig. 2A–D), we decided to have them tested by PCR in a specialized laboratory (Anicura Tieraerztliches Labor, Freiburg, Germany). The result of the PCR assay (target: 18S rRNA gene) was negative for Giardia DNA for both samples.
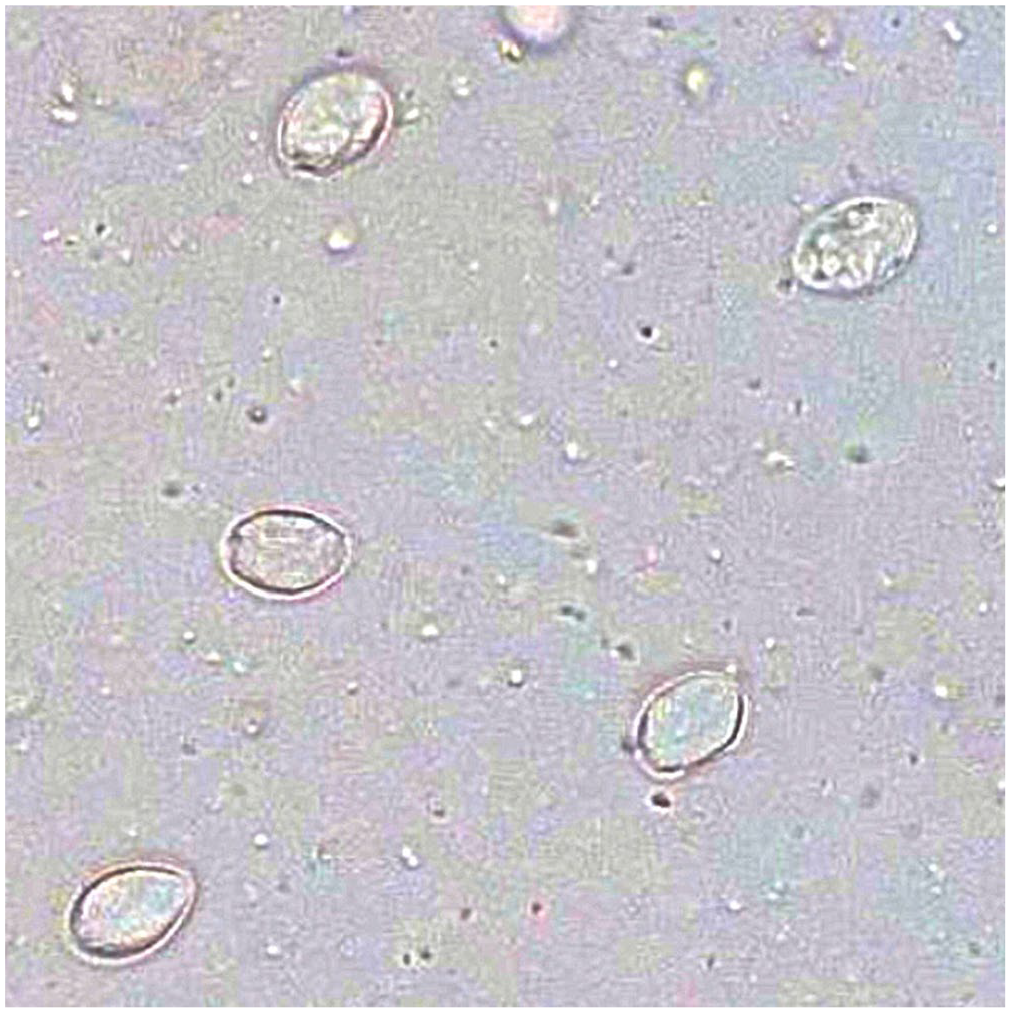
Objects counted as Giardia cysts by the Vetscan. True positive measurement with multiple cysts in a sample that was also Giardia positive by direct immunofluorescence assay and SNAP test.
Combination pattern of qualitative test results for Giardia duodenalis in canine fecal samples.

+ = positive; – = negative; IFA = direct immunofluorescence assay for Giardia; SNAP = SNAP Giardia test (Idexx); Vetscan = Vetscan Imagyst, Giardia test kit (Zoetis).
Fecal samples with discordant results, with qualitative results of the Vetscan, SNAP, and IFA, interpretation and quantitative results of the triplicate Vetscan measurements, and interpretation and semiquantitative results of the IFA measurements.
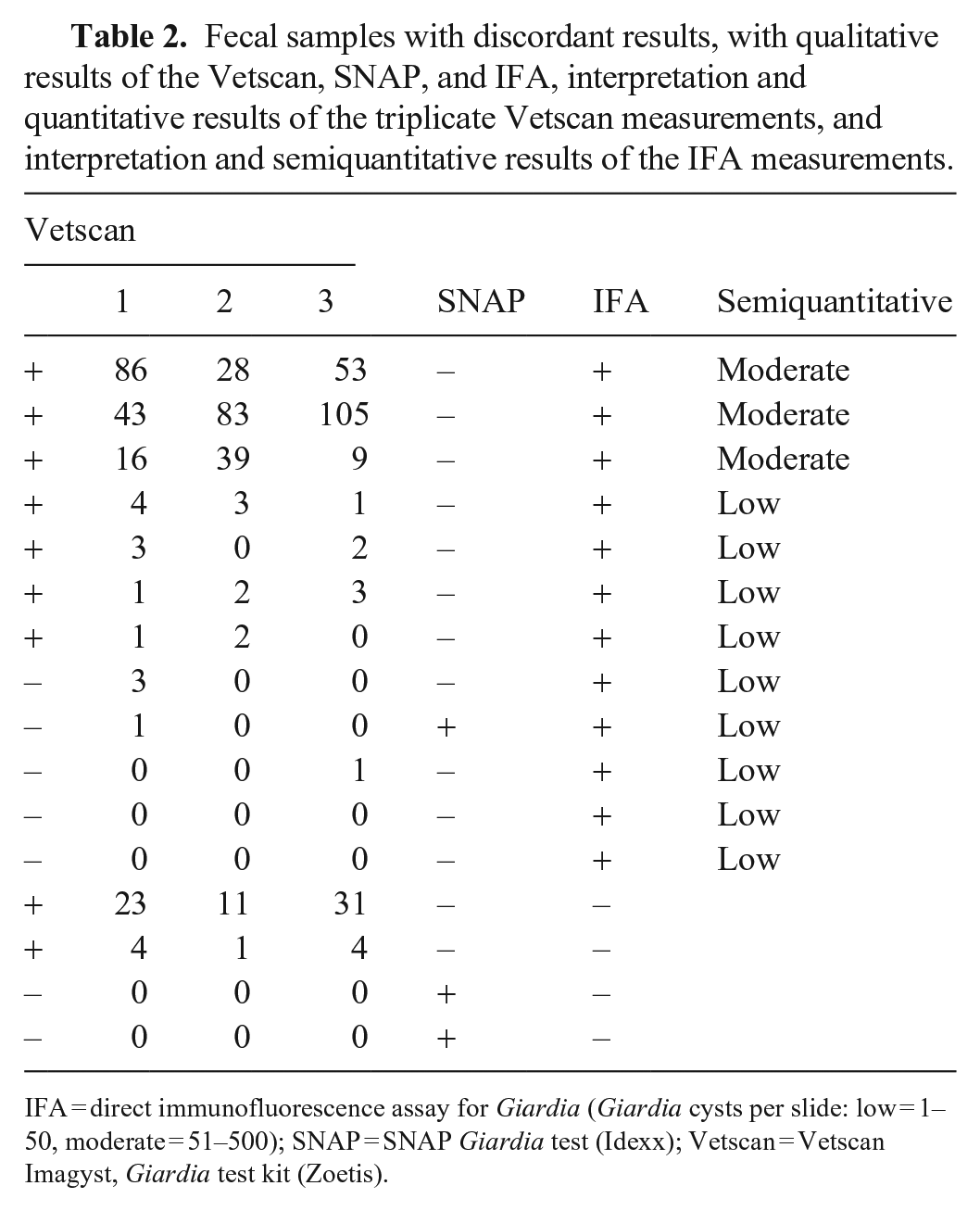
IFA = direct immunofluorescence assay for Giardia (Giardia cysts per slide: low = 1–50, moderate = 51–500); SNAP = SNAP Giardia test (Idexx); Vetscan = Vetscan Imagyst, Giardia test kit (Zoetis).
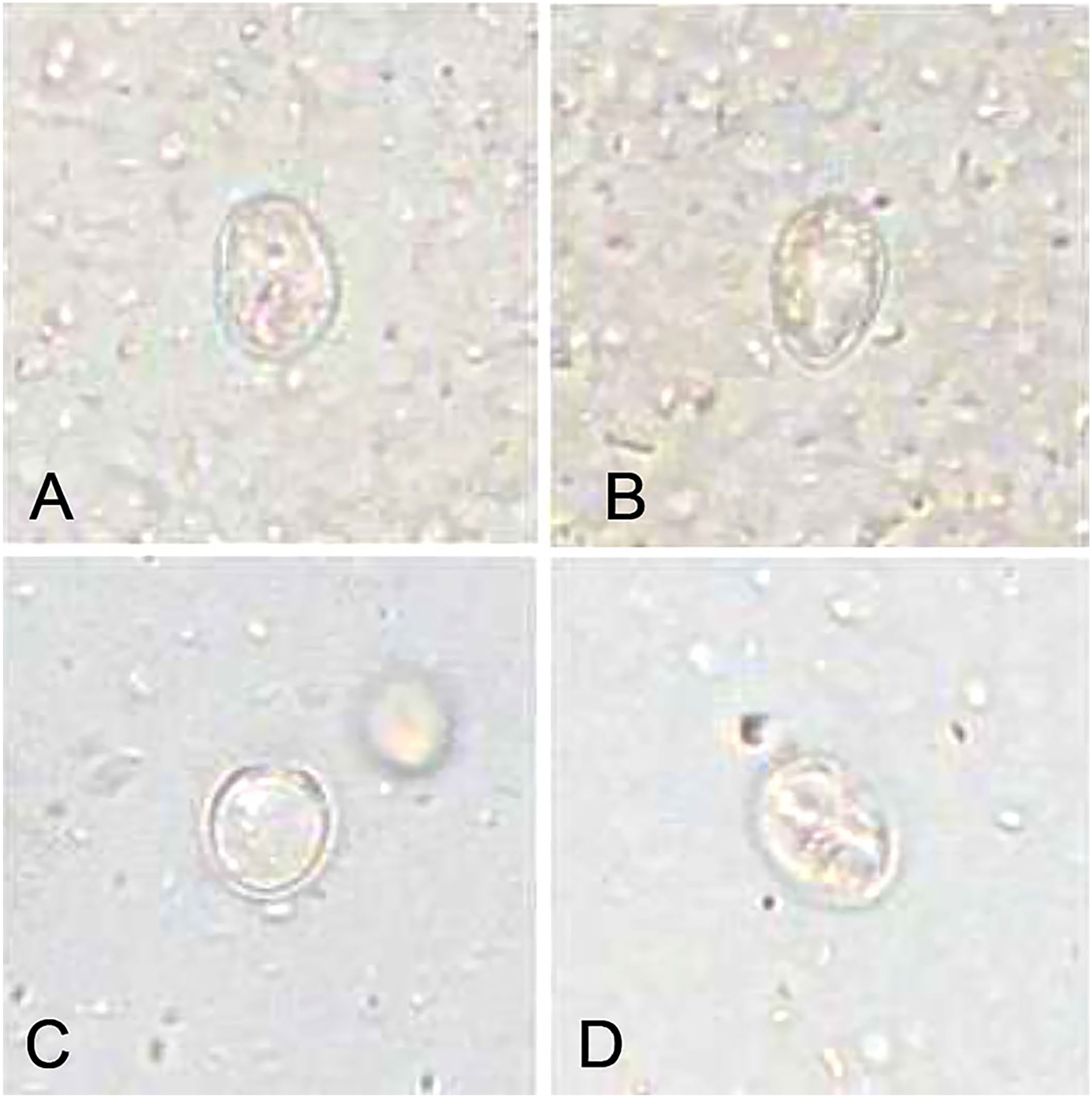
Objects consistent with cysts and counted as Giardia cysts by the Vetscan in 2 samples (sample 1:
Diagnostic sensitivity and specificity of the Vetscan and SNAP were compared to the IFA. The Vetscan had higher sensitivity than the SNAP test compared to the reference method in our study. The specificity was equal for both the Vetscan and SNAP tests (Table 3).
Sensitivity and specificity of the Vetscan and SNAP tests compared to IFA as the reference method for the detection of Giardia cysts in canine fecal samples.
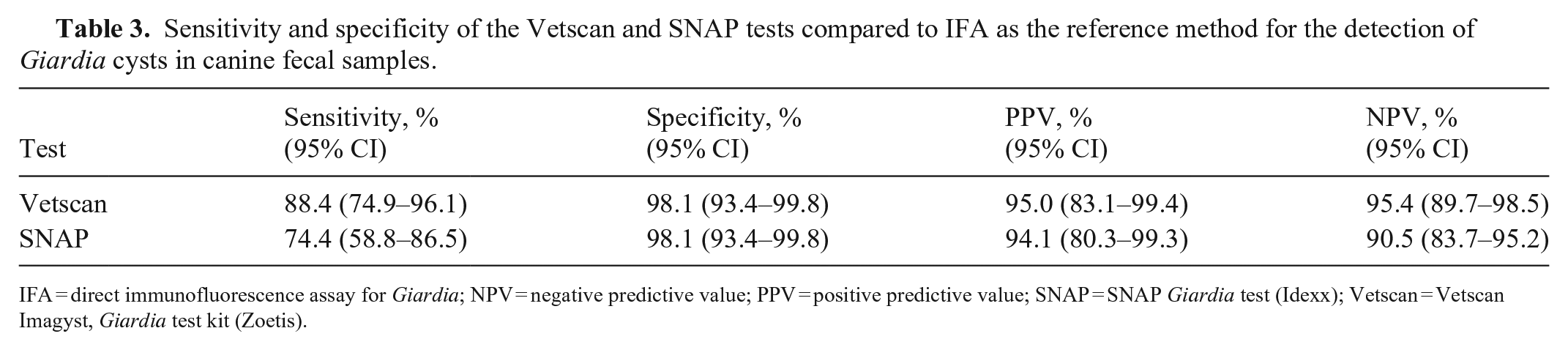
IFA = direct immunofluorescence assay for Giardia; NPV = negative predictive value; PPV = positive predictive value; SNAP = SNAP Giardia test (Idexx); Vetscan = Vetscan Imagyst, Giardia test kit (Zoetis).
To test repeatability, we measured the pooled samples of all 164 dogs 3 times with the Vetscan for a total of 492 measurements. In 7 of the 492 measurements, the Vetscan showed irregularly shaped structures that did not resemble Giardia cysts (Fig. 3). In these false-positive measurements, 4 structures other than Giardia cysts were found in 2 measurements and 1 structure in each of the 5 other measurements. A false-positive result was displayed in 1.4% of the measurements. Consequently, we were able to confirm 47 pooled samples as correctly positive. In 42 samples, 3 of 3 measurements were positive; in 5 samples, 2 of 3 measurements were positive. Seven pooled samples had 1 positive and 2 negative test results, and 110 samples had 3 negative results. For the quantitative results (cysts per slide), the CV was 20.0–141%, with an average of 67.0%.
Single objects that do not resemble Giardia cysts but counted as Giardia cysts in 2 samples (sample 1:
Discussion
In most measurements, the qualitative Vetscan results were concordant with the IFA results. In specimens in which the IFA was positive and the Vetscan result was negative, only low cyst counts could be detected with the IFA. For samples in which the IFA cyst counts were moderate or high, results were always positive with the Vetscan. The Vetscan image recognition detected structures that, in our opinion, were not Giardia cysts in 1.4% of our measurements; these false-positive results were rejected manually. This problem only occurred in samples with a low count of structures (<5), which are also difficult to assign as positive or negative with manual microscopic methods.
Our calculated sensitivity and specificity of the Vetscan and SNAP tests are comparable with the results from the literature. Comparisons of zinc sulfate flotation with IFA have reported a sensitivity of 49–88.1% and a specificity of 65–94%.31,32 In one study, the Vetscan sensitivity was 75.8%; after excluding samples with <10 cysts per gram, the sensitivity increased to 95.2% compared to the results of microscopic evaluation by a parasitologist. 28 Although we excluded no samples with a small number of cysts in our study, the sensitivity of 88.4% is higher than in the aforementioned study.
In another study, the performance of the Vetscan with 2 different scanners, the Ocus 40 and the EasyScan One, was compared with the results of an expert parasitologist; 174 canine and feline fecal samples were examined with the Ocus 40 and 87 samples with the EasyScan One scanner. Due to a recording error, the results of the canine and feline samples were combined. The Ocus 40 achieved a sensitivity of 92.1% and a specificity of 98.8%, whereas the sensitivity of the EasyScan One was 73.6% and the specificity 100% compared to the expert parasitologist. The authors concluded that the Ocus 40 was able to achieve better sensitivity due to the higher quality of the scanned images. 27 Despite the fact that we used the Ocus 40 in our study, the sensitivity was lower at 88.4%. This may be because we used the IFA as the reference method, whereas the previous study 27 compared the Vetscan to manual examination of the same fecal slide by experienced personnel without immunofluorescent labeling.
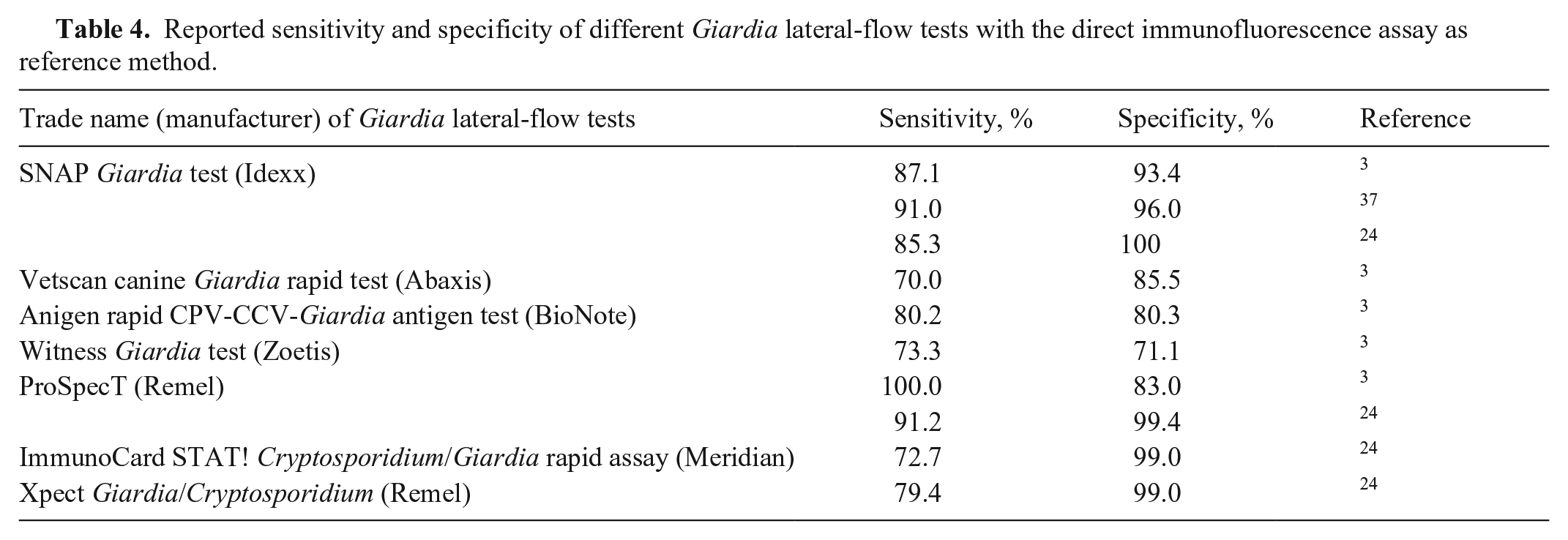
We collected the published performance parameters of 7 frequently used lateral-flow tests for Giardia detection with the IFA as the reference method. The sensitivity was 70–100%; specificity was 71–100% (Table 4). Compared to the IFA in previous studies, the sensitivity of the SNAP test was 85–91% and specificity was 96–100%.3,24,37 The sensitivity of the SNAP is lower in our study at 74.4%, whereas the specificity of the SNAP test that we found was similar to the aforementioned studies. Test performance varies with the disease prevalence in the study population and with the patient spectrum, which might explain the differences. 22
Reported sensitivity and specificity of different Giardia lateral-flow tests with the direct immunofluorescence assay as reference method.
Two samples in our study had cysts that were consistent with Giardia cyst morphology in all 3 measurements with the Vetscan, but were negative with all other test methods applied, including PCR for the target 18S rRNA gene. These 2 samples are the false-positive Vetscan measurements. Cysts may be distributed irregularly in feces, which may occasionally lead to discrepant results in the same sample. Information about the sensitivity of the PCR assay used for these samples is not available, but the sensitivity of some PCR assays has been shown to be low compared to IFA in previous studies.36,37 Given that the SNAP and IFA were also negative, the objects may also be cysts of parasites other than Giardia that do not infect dogs, but are found in dog feces by ingestion of mice or feces of other animals. The Vetscan may not be able to differentiate these cysts and may then yield false-positive results.
Repeated measurements using the Vetscan had a CV of 67%, although the measurements were performed with sample material from the same 3 fecal samples per dog. One possible reason for these discrepancies could be that the sample material was taken from different parts of the fecal samples and the cysts may not be distributed evenly. It is also possible that the consistency of the feces and possible admixtures such as mucus or blood could affect the results. As Giardia cysts are small, transparent, and fragile, cysts may not have been detected, leading to differences in quantitative measurements. The automated cyst count of the Vetscan may also not be consistent or may not include cysts that are slightly tilted or damaged. In 12 samples, the triplicate measurements of the Vetscan yielded 1 or 2 negative results. In these dogs, low-level infection might have been missed with a single measurement despite the use of a pooled fecal sample.
Due to the scatter of results, we believe that it is not possible to determine an exact quantitative Giardia cyst count with the Vetscan. Current recommendations advise retesting dogs with clinical signs of giardiasis and a positive Giardia test after treatment, especially in dogs that continue to have clinical signs.7,9 Studies have shown that some dogs were still positive after standard therapy with fenbendazole or metronidazole, yet the clinical signs of the dogs had improved significantly.6,20 The World Association for the Advancement of Veterinary Parasitology defines therapeutic success in canine giardiasis as a reduction in Giardia cysts of ≥90% using a quantitative immunofluorescence approach. 15 Based on our results, we believe that quantitative determination of the number of cysts with the Vetscan should not be used to monitor cyst reduction.
The nomenclature of Giardia species is currently being critically reconsidered. 39 As no PCR tests were performed in our study to determine the assemblages, we cannot say with certainty which genotypes were involved in our fecal samples.
The design of our study was based on the procedures for Giardia tests in clinical practice. The Vetscan and SNAP tests were performed according to the manufacturer’s instructions, without standardized sample weight, as was the IFA method used in the external parasitology laboratory. This lack of standardization with all 3 methods contributes to the variation in results. However, >95% of the qualitative Vetscan results are concordant with the reference test, making it a useful qualitative POC test for Giardia detection in canine fecal samples.
Supplemental Material
sj-pdf-1-vdi-10.1177_10406387241279177 – Supplemental material for Performance of the Vetscan Imagyst in point-of-care detection of Giardia duodenalis in canine fecal samples
Supplemental material, sj-pdf-1-vdi-10.1177_10406387241279177 for Performance of the Vetscan Imagyst in point-of-care detection of Giardia duodenalis in canine fecal samples by Sabrina Kanski, Kathrin Busch, Regina Hailmann and Karin Weber in Journal of Veterinary Diagnostic Investigation
Footnotes
Acknowledgements
We thank the staff of the Small Animal Clinic of LMU Munich, who helped with the preparation of fecal samples. Our special thanks go to the colleagues from the laboratory who helped with the testing procedures and sample storage. Many thanks to the colleagues of the IEP of the Faculty of Veterinary Medicine of LMU Munich for their cooperation and support.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The positions of Sabrina Kanski and Regina Hailmann were supported by the Federal Ministry for Economic Affairs and Climate Action (BMWK) based on a decision by the German Bundestag.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
