Abstract
Canine leishmaniosis is a widespread, potentially fatal disease caused by infection with Leishmania infantum. We evaluated 5 commercial point-of-care tests (POCTs) for the detection of L. infantum antibodies in dog sera and compared their performance with the immunofluorescence assay (IFA) as the reference method. We tested serum samples from 160 dogs that either had lived in or traveled to leishmaniosis-endemic areas using the following immunochromatography POCTs: the trüRapid Leish (Biotech), FASTest Leish (Megacor), Leishmania Ab (Bionote), and Uranotest Leishmania 2.0 and 3.0 (Uranovet). Practicality, sensitivity, specificity, positive predictive value, and negative predictive value were determined. The prevalence of L. infantum antibodies in all dogs was 28.1% as determined by IFA. The highest sensitivity among POCTs was achieved by the Leishmania Ab and the Uranotest Leishmania 2.0 (88.9%), followed by the trüRapid Leish (86.4%), the Uranotest Leishmania 3.0 (84.4%), and the FASTest Leish (48.9%). The highest specificity was achieved by the FASTest Leish (100%) followed by the Leishmania Ab (98.3%), the trüRapid Leish (96.5%), the Uranotest Leishmania 3.0 (94.8%), and the Uranotest Leishmania 2.0 (90.4%). Given its comparatively high sensitivity and specificity, the Leishmania Ab may be considered for screening dogs from endemic areas and for confirming Leishmania infection in clinical dogs.
Keywords
Canine leishmaniosis (
For the detection of L. infantum antibodies, various methods are available, such as immunofluorescence assay (IFA), ELISA, western blot (WB), direct agglutination test (DAT), or rapid chromatographic immunoassays for in-house use (point-of-care tests [
POCTs offer an economical and fast alternative for qualitative antibody detection suitable for veterinary practice. Although sensitivity can vary widely (52.9–100%), specificity is generally high (84.9–100%), which ensures their practical value for confirming CanL in dogs with compatible clinical signs.15,30,31,48 Although several POCTs for the diagnosis of CanL are available commercially, comparative validation studies under standardized conditions are lacking. This gap limits evidence-based recommendations for clinical practice.
Our aim was to address this gap by evaluating 5 POCTs for the detection of L. infantum antibodies in dog sera and comparing their performance with the IFA reference method. We determined practicality, sensitivity, specificity, positive predictive value (PPV), and negative predictive value (NPV).
Material and methods
Serum samples
The sample size for the McNemar test using G*Power software was calculated to detect a moderate difference in paired test outcomes (expected proportion of discordant pairs ≈ 0.4; odds ratio = 0.5). These assumptions were guided by prior comparative studies of POCTs for the detection of L. infantum antibodies and reference serologic assays, which reported comparable discordance rates between rapid tests and IFA or ELISA.18,36,45 With a power of 80.0% and alpha of 0.05, a minimum of 145 samples was required. We added 10% as a buffer to account for potential unusable samples, resulting in a total sample size of 160. We included serum samples from dogs that either had lived in or traveled to a CanL endemic area. We collected samples from October 2022 to July 2023 from privately owned dogs presented for preventive health care or in the infectious disease service at the LMU Small Animal Clinic, Centre for Clinical Veterinary Medicine (Munich, Germany). All dogs with confirmed CanL infection had tested positive for Leishmania by IFA, ELISA, or PCR ( Table 1 ). The dogs had clinical signs compatible with CanL at various times before being included in our study, but were mostly subclinical at the time our samples were taken. All clinically healthy dogs had no signs of disease and/or Leishmania infection in the clinical health check. Blood tests, if available, also showed no anomalies. All dogs with other diseases had various clinical or laboratory abnormalities but did not have a positive Leishmania test prior to study sampling; some tested positive for other infectious diseases. We performed data collection and statistical analysis (Excel v.2401, Microsoft; Prism v.5.04, GraphPad). We stored 2 aliquots of each sample at −80°C until testing. The use of the serum samples was approved by the ethics committee of the veterinary faculty of the LMU Munich (reference 311-09-06-2022).
Sample groups with result of reference method (IFA) and additional testing.

CanL = canine leishmaniosis.
Point-of-care tests
We used 5 commercial POCTs for the detection of L. infantum antibodies ( Table 2 ). The cost of a single POCT is US $12–23 (EUR €10–20) per test. For testing, all samples were numbered consecutively from 1 to 160 and then tested with all 5 POCTs, according to the manufacturers’ instructions. All tests were performed by a single trained veterinarian, who was blinded to the results of the reference method. The POCTs used in our study were provided as complete batches, and all tests originated from the same production lot. All POCTs were based on immunochromatography (lateral flow immunoassay) in which the antibody in the sample reacts with an immobilized antigen in a line across a nitrocellulose membrane strip. In the case of a positive result, antibodies specific to the target analyte conjugate with the colored particles and produce a visible test line. For a valid result, the control line must appear, which indicated the proper liquid flow through the strip. The control line contained species-specific anti-immunoglobulin antibodies. 25 A test with visible test and control lines was counted positive, regardless of the intensity of the test line. In the event of an ambiguous result, a second person was consulted for interpretation.
Leishmania immunochromatography point-of-care tests used in our study.
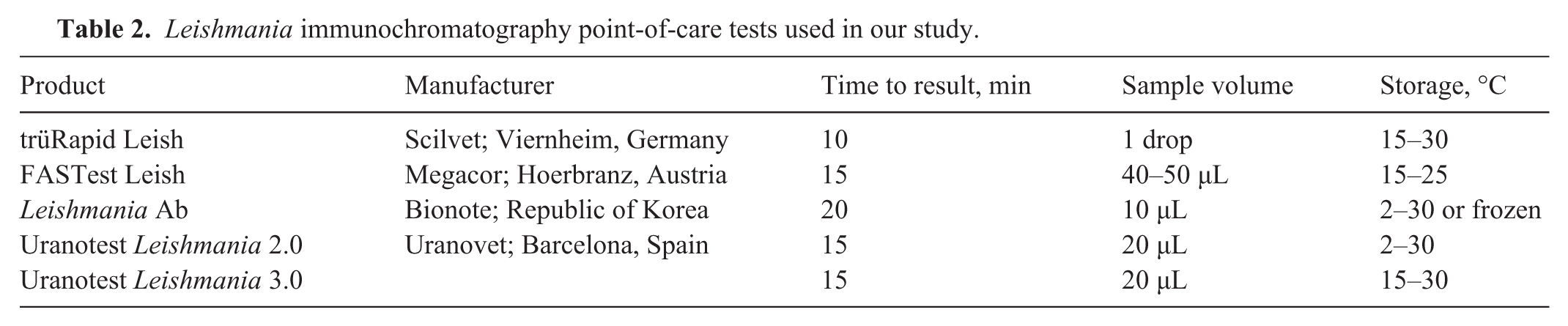
Reference test
After performing the 5 POCTs, all samples were sent as a batch to a reference laboratory (Anicura, Freiburg, Germany) for detection of antibodies by IFA (MegaScreen FLUOLEISH; Megacor). As target antigen, slides were coated with a mixture (1:1 ratio) of promastigotes of L. infantum (zymodem MON-1) and L. tropica. The positive cutoff for the IFA was a dilution of ≥1:200. The samples were analyzed by at least 2 independent laboratory technicians, who were blinded to the results of the POCTs.
In a limited number of cases, we had access to the results of an additional ELISA and PCR testing performed in external laboratories (Table 1). These samples originated from dogs that had been presented for infection consultation, for follow-up examinations of CanL, for screening after travel abroad, or for vaccination preparation.
Results
Detection of L. infantum antibodies by IFA
With the reference method, 45 of 160 (28.1%) serum samples were positive for L. infantum antibodies. High antibody titers (>1:200) were detected in 36 of 45 samples, with moderate antibody titers (1:200) in the remaining 9 positive samples. Of the 115 negative samples, 2 samples had a titer of 1:100, 9 samples a titer of 1:50, and no titer was detectable in the remaining 104 samples.
IFA results and additional confirmatory tests
At the time sampling, for 89 of 160 samples, additional test results were available (Table 1). All 54 dogs of the CanL group were tested with at least one additional test (ELISA and/or PCR), 7 of the 9 CanL IFA-negative dogs were also negative with additional tests. For the remaining 106 samples (from clinically healthy dogs and dogs with other diseases), 44 additional test results from 35 samples were available and all were negative.
Practicality of the POCTs
All 5 POCTs can be stored at room temperature and therefore do not require a warm-up phase. The tests were equally easy to perform; the time to result varied from 10–20 min. Because pipettes are supplied with each POCT by the manufacturer, no external materials are required. There was one invalid result with no control line in a trüRapid Leish test. We repeated the test with different sample dilutions, and a valid positive result was achieved with a 1:3 dilution. This result was not included in the statistics. In 36 of the 45 positive samples (80.0%), test and control lines were clearly detectable. The FASTest Leish had a light test line (classified as positive) in 4 of the positive samples; the Uranotest Leishmania 2.0 and 3.0 had a light test line in 5 samples each. One sample with the trüRapid Leish and 2 samples with the FASTest Leish turned positive only after the end of the reading time specified by the manufacturer. These 3 samples were classified as negative according to the manufacturer’s instructions.
Comparison of the POCTs with the IFA
The sensitivity of the 5 POCTs was 48.9–88.9% and specificity was 90.4–100% ( Table 3 ). PPV was 78.4–100%; NPV was 83.3–95.6%. The level of agreement between the reference method (IFA) and the POCTs was 85.6–95.6%. The Leishmania Ab had the highest agreement with the IFA. Of the 160 samples analyzed, 100 (62.5%) were negative by IFA and yielded concordant negative results across all POCTs. The FASTest Leish had the highest number of false-negative results but also had the highest number of true-negative results compared with the other POCTs. The Uranotest Leishmania 2.0 had the highest number of false-positive results ( Table 4 ). In 4 samples that tested positive by IFA, none of the POCTs were positive, even though all samples had high antibody titers (>1:200). In 15 of 115 (13.0%) IFA-negative samples, one or more POCTs were positive ( Table 5 ). Of the 11 samples with low titers (1:100, 1:50), 4 tested positive with ≥1 POCT; in the remaining 104 samples without detectable antibodies, 11 tested positive with ≥1 POCT. The 2 negative samples with titers of 1:100 were from the group of confirmed infected dogs. For one of these samples, only the Uranotest Leishmania 2.0 had a positive result; for the other sample, all POCTs were negative. Of the 9 negative samples with a titer of 1:50, 6 tested negative with all POCTs, 2 tested positive with 1 POCT, and 1 sample tested positive with 4 of 5 POCTs.
Leishmania point-of-care tests (POCTs) performance compared to immunofluorescence assay.
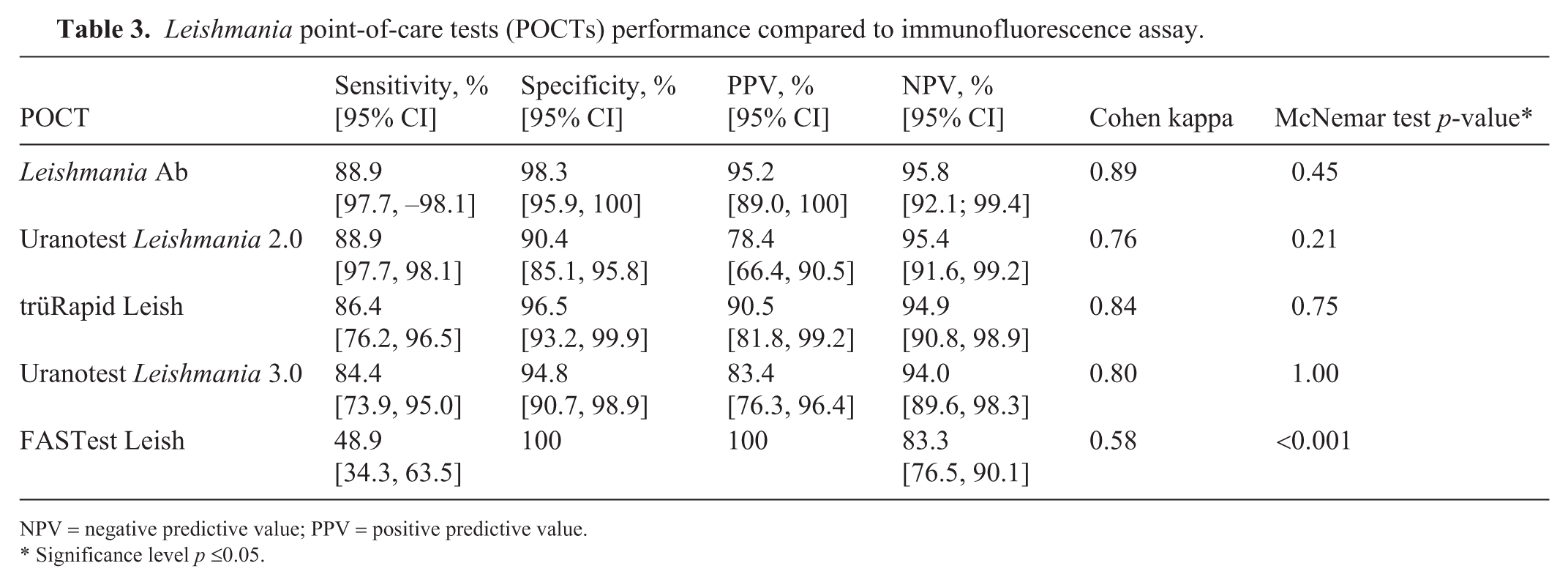
NPV = negative predictive value; PPV = positive predictive value.
Significance level p ≤0.05.
Results of the anti–Leishmania infantum antibody testing, using immunofluorescence assay (IFA) as the reference method.
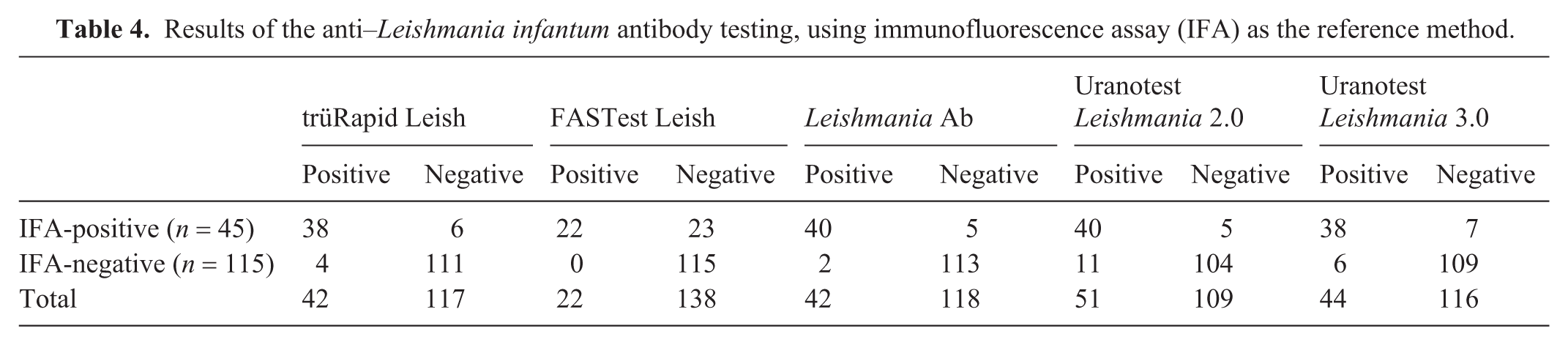
Combination patterns of Leishmania point-of-care test and immunofluorescence assay (IFA) results.

+ = positive; – = negative.
Comparison of the 5 POCTs
The agreement between the individual tests was highly variable ( Table 6 ). The highest agreement was observed between the trüRapid Leish and the Uranotest Leishmania 3.0 (κ = 0.92) and Leishmania Ab (κ = 0.89). The comparison between the FASTest Leish and the Uranotest Leishmania 2.0 had the lowest agreement (κ = 0.51). The McNemar test results had significant differences (p ≤0.05) for several test comparisons, especially when the FASTest Leish was compared with other POCTs (Table 6). In contrast, there was no significant difference (p >0.05) between the trüRapid Leish and the Uranotest Leishmania 3.0, or between the Leishmania Ab and both Uranotest Leishmania 2.0 and 3.0.
Comparison of all point-of-care tests (POCTs) using the McNemar test.
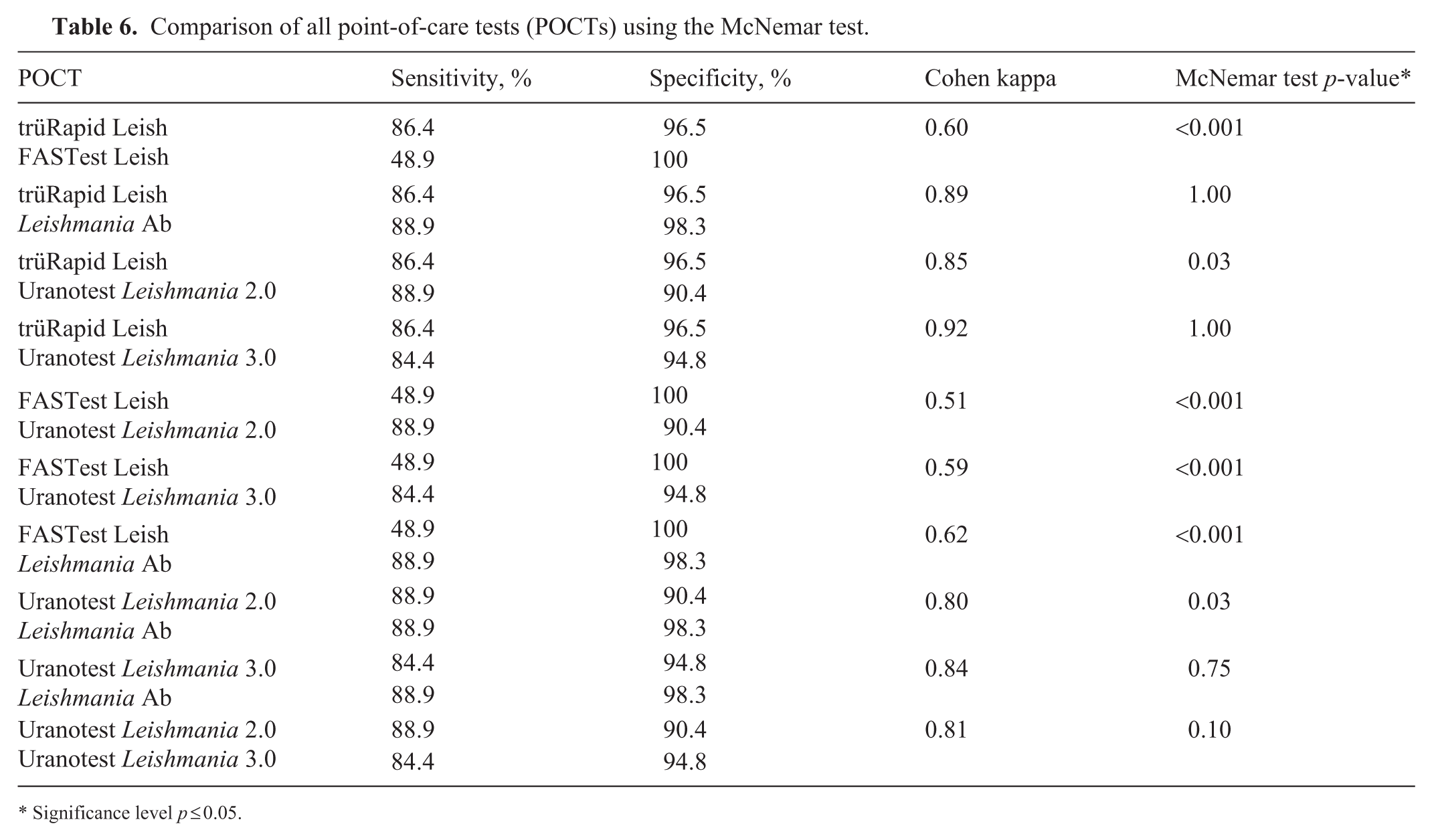
Significance level p ≤ 0.05.
Discussion
The Leishmania Ab and the Uranotest Leishmania 2.0 were the POCTs with the highest sensitivity (88.9%). A 2016 study that evaluated the Leishmania Ab compared with the IFA revealed similar results with a sensitivity of 89.7%. 35 For the Uranotest Leishmania 2.0, a 2022 study using an in-house ELISA and an in-house IFA as reference methods reported a higher sensitivity of 97%. 48 For the FASTest Leish, 2 studies with the IFA as reference method had a much higher sensitivity (92–100%) than demonstrated in our study (48.9%).47,48 Reasons for the differences in the sensitivity could be test modifications by the manufacturer, differences in the quality of the batches, and different prevalences of study sample sets, which applies to both studies with the FASTest Leish. The composition of the study population also influences the performance parameters of tests for Leishmania antibody detection: In 2 studies, the sensitivity of in-house ELISAs was lower for subclinical dogs (52.9–66.0%) than for dogs (96.0–100%) with clinical CanL.30,34 This difference could be caused by low antibody titers at the beginning of an infection with L. infantum or in dogs with a predominantly cellular immune response. 19 A large study of dogs living in a CanL-endemic area of Brazil, reported decreasing test sensitivity of an antibody-based test: 93.8% in clinical dogs, 89.2% in oligoclinical dogs, and 75% in subclinical dogs. As reference standard, direct parasitologic examinations (culture techniques, histopathology, immunohistochemistry) were used to analyze the prevalence of infection in these groups, which was high in clinical dogs (32.0%), but much lower in oligoclinical (5.1%) and subclinical (2.7%) dogs. 17
All 45 dogs (28.1%) that tested positive by the reference method in our study were confirmed infected with Leishmania. At the time of blood sampling, all 45 dogs were undergoing treatment and showing few-to-no signs. Only one dog with acute CanL, which had not yet received any treatment, was included during our study. This low number may be explained by the fact that samples were collected at a referral clinic for infectious diseases, potentially introducing referral bias.
Overall, the sensitivity of the POCTs should be improved. High sensitivity and therefore low numbers of false-negative tests are particularly important if the test is used for screenings in dogs who lived in or traveled to a region where CanL is considered endemic. Screening is often performed by animal welfare organizations importing dogs from endemic to non-endemic areas before dogs are rehomed. In a study analyzing samples from 2004 to 2009, 4,681 dogs living in Germany—with either a travel history to an endemic area for CanL or those that were rehomed from an endemic area—12.2% of the dogs tested positive for L. infantum antibodies by IFA. 29 In another study, which analyzed dogs imported to Germany from Mediterranean and Southeast European countries between 2007 and 2015, 314 dogs were tested for Leishmania infection by direct or indirect test methods, with a positive rate of 21%. 39 In our study, all dogs that tested positive by the reference method were born in an endemic area for CanL and were rehomed later. About 40% of dogs that tested negative by IFA had only spent a short period of time in endemic areas, for instance during travel, and 60% were from endemic areas.
The specificity of all POCTs in our study was high (>90%). The FASTest Leish had the highest specificity (100%). This is consistent with other studies, in which specificity for the FASTest Leish was >98%.7,47,48 Similar results were also obtained for the specificity of Leishmania Ab (>98%) in another study. 35 In our study, the Uranotest Leishmania 2.0 had a lower specificity of 90.4% compared with a previously reported value of 98% and had the highest rate of false-positive results among all POCTs. 48 A high specificity in POCTs—and the resulting low numbers of false-positive results—is particularly important for confirming or rejecting the diagnosis of CanL in suspected cases, because false-positive results could lead to unnecessary treatment. Highly specific POCTs are furthermore required for screening for L. infantum antibodies before Leishmania vaccination. One reason for low specificity and thus a high number of false-positive results, such as the 15 samples in our study, could be cross-reactions with antibodies against other Leishmania species or protozoa such as T. cruzi, which predominantly occur in South America.14,34,46 Cross-reactivity between Leishmania target-antigen and antibodies of E. canis, Babesia spp., or Hepatozoon canis has also been reported and can lead to false-positive results.14,34,41 In fact, in our study, we detected antibodies against E. canis and/or B. canis in 3 of 7 of the samples that tested false-positive with the Uranotest Leishmania 2.0. Cross-reactivity, resulting from antibodies against T. cruzi, might have occurred in one dog from our study that originated from Colombia and had a positive result with the Uranotest Leishmania 2.0.
In CanL dogs that were subclinical at the time of sampling, false-positive results may be explained by low antibody concentrations, which produce faint test lines that must be interpreted as positive according to the manufacturers’ instructions. In addition, the immunologic profile of subclinical infections often has a cellular rather than a humoral immune response, which may further explain discrepant or misleading serologic results. 33 In our study, the 3 samples that yielded positive results after the manufacturers’ reading period with 2 POCTs (and were therefore counted as negative) originated from dogs that were positive by the reference method and were in the group of CanL dogs with a positive result by an additional ELISA. This introduces some uncertainty, given that a positive POCT result outside the specified reading time could indicate a potential infection.
Each POCT test in our study was performed only once, except for one sample with a high IFA antibody titer (>1:200) that was tested with the trüRapid Leish and had an invalid result without a control line. With a 1:3 serum dilution, the test had a valid positive result. An additional ELISA also was positive. We suspected that the high antibody concentration in the undiluted sample might have stopped the flow of the conjugate, or a high-dose hook effect occurred in the control zone. This phenomenon is observed especially in sandwich lateral-flow immunoassays. 38
In 4 samples, all of which had a high antibody titer (>1:200) by IFA and came from the CanL group, none of the POCT results were positive. One reason for the discrepancies of the test results could be differences in the antigenic target. The recombinant protein rK39, which is an epitope expressed by tissue amastigotes of L. infantum, is often used as target antigen in POCTs for the detection of L. infantum antibodies.9,30,31 In our study, the IFA target antigen was a mixture of 2 antigen fractions of promastigotes. Samples in which antibodies are not directed against the specific epitope (e.g., rK39) may test negative with a POCT but positive by IFA because more epitopes are offered. Another reason could be cross-reactions and, therefore, false-positive results by IFA because of infection with different pathogens (e.g., E. canis, B. canis). We had no information regarding infections with other pathogens in any of the 4 dogs. An additional Leishmania antibody-ELISA in 2 of these 4 cases had a negative result and therefore was unable to confirm our IFA findings. For the other 2 samples, additional Leishmania DNA-PCR tests were performed; 1 sample was positive, and the other was negative.
In general, a Leishmania infection cannot be ruled out based on one negative L. infantum antibody test. L. infantum antibodies can be detected as early as one month after experimental infection.10,13,23 In the case of a subclinical L. infantum infection, in which the immune response is dominated by Th1 (T helper) cells, dogs sometimes do not produce antibodies at all.24,32 We included one dog that had tested negative by IFA but was confirmed for CanL by a PCR test of lymph node aspirate; the Leishmania Ab was the only POCT to give a positive result in this case. In a 2016 study, 20 of 92 (21.7%) Leishmania-infected dogs that tested negative by a Leishmania antibody test (DPP CVL rapid test) had at least one positive PCR result in various sample types (oral swab, conjunctival swab, blood, lymph node aspirate). 3 Especially in cases of suspect dogs with clinical signs, a negative antibody test should be interpreted critically and, if possible, should be repeated using an additional detection method (ELISA, PCR).
Additional confirmatory testing in our study had high concordance with negative test results by IFA. Only 3 of the IFA-negative samples additionally tested with ELISA had a positive result. Two of these 3 samples were from CanL subclinical dogs, which supports the result of another study in which ELISA was more sensitive than IFA in subclinical dogs. 30
Limitations of our study include the choice of IFA as the (only) reference method; we used IFA based on its use as a reference method in other studies.2,16,28,49 Considering lack of standardization (e.g., for cutoffs), cross-reactions, and limited sensitivity in subclinical infections, IFA is not a gold standard. As additional ELISA testing was only performed in a subset of samples for clinical reasons (infection consultation, follow-up, travel screening, or vaccination preparation), our results cannot be generalized. Nevertheless, our discrepant findings highlight possible differences in sensitivity between serologic methods. Standardized testing of all samples with a second reference method, plus testing for coinfections, could have led to greater comparability and accuracy. Given our inconsistent additional testing, information that might be important for assessing POCT performance is missing. Another potential limitation is the composition of our study population. Because we mainly included confirmed infected dogs that were receiving treatment, the sensitivity of the tests may be different than it would be in the general dog population (including clinical animals or dogs in early stages of infection). In addition, ours was a single-center study, which means that the results may not be easily transferable to other populations or regions. To achieve greater significance, further studies should include a more diverse study population.
Footnotes
Acknowledgements
We thank the staff of the LMU Small Animal Clinic, Munich, who helped with the collection of serum samples. We also thank the manufacturers of all POCTs in our study for providing the tests.
Declaration of conflicting interests
The authors declared no potential conflict of interest with respect to the research, authorship, and/or publication of this article. Although the POCTs were provided for free by Megacor, Bionote, Uranovet, and Scilvet, the funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript; or in the decision to publish the results. No commercial conflict of interest exists, as the information generated here is solely for scientific dissemination.
Funding
The positions of Regina Hailmann and Sabrina Carstens were supported by the Federal Ministry for Economic Affairs and Climate Action (BMWK) based on a decision by the German Bundestag.
