Abstract
A 15-y-old captive-born female bottlenose dolphin (Tursiops truncatus) had slowly progressive lethargy, inappetence, and pallor of the oral mucosa. Serial hemograms and serum biochemistry analysis revealed hemolyzed plasma, gradually severe anemia with reticulocytosis, spherocytosis, pronounced neutrophilia with left shift and toxic change, and indirect hyperbilirubinemia. Urinalysis identified both hemoglobinuria and bilirubinuria. Hyperglobulinemia with polyclonal gammopathy was evident by protein electrophoresis, and the saline agglutination test was positive. Splenomegaly and hyperechogenic liver were also observed on ultrasound. These findings meet the diagnostic criteria established for immune-mediated hemolytic anemia (IMHA) in small animals, which is extremely rare in cetacean species, and, to our knowledge, has not been fully documented. Several potential triggers of a secondary IMHA were ruled out, including the most common cetacean virus, exposure to new drugs, and recent vaccination events. Following recommendations from companion animal medicine, immunosuppressive therapy was implemented and proved effective in this bottlenose dolphin; the CBC returned to normal after 3 mo of treatment.
A 15-y-old, born under human care, female bottlenose dolphin (Tursiops truncatus) was presented with mild chronic weight loss and hyporexia of several weeks’ duration. The dolphin was regularly fed with frozen-thawed fish (herring, capelin, sprat, blue whiting, squid) and lived in an outdoor 22,000-m3 pool, shared with 18 other bottlenose dolphins of different sex and ages. The dolphin was eventually moved to a separate pool while experiencing clinical health issues. The health of the pod was unremarkable, except for an adenoviral outbreak 8 y previously and a gastrointestinal disease outbreak of unknown etiology a few weeks earlier.
As defined by the Institution’s preventive medicine program, bi-monthly blood samples for analyses were obtained under operant conditioning from the ventral fluke vasculature (Fig. 1A). Blood drawn into EDTA tube was analyzed in an automated analyzer (XN-1000V, Sysmex; Table 1), in which RIs for bottlenose dolphins were created using the data collected over time from restrained bottlenose dolphins during routine assessments of normal animals in zoologic institutions from around the world (Species 360, https://species360.org/). Reticulocyte counts were performed by the flow cytometry method with polymethine dye (Sysmex XN-1000V). A complete biochemistry profile was obtained from lithium heparin plasma (AU-480; Beckman Coulter) using RIs established for bottlenose dolphins (Species 360; Table 2). Fibrinogen levels were determined quantitatively by the Clauss clotting method (STA Compact-max, Stago; Table 2). Erythrocyte sedimentation rate (ESR) is a traditional marker of inflammation, widely used in cetacean medicine, and was determined by placing a sodium-citrated tube in a strictly vertical position for 1 h at room temperature.
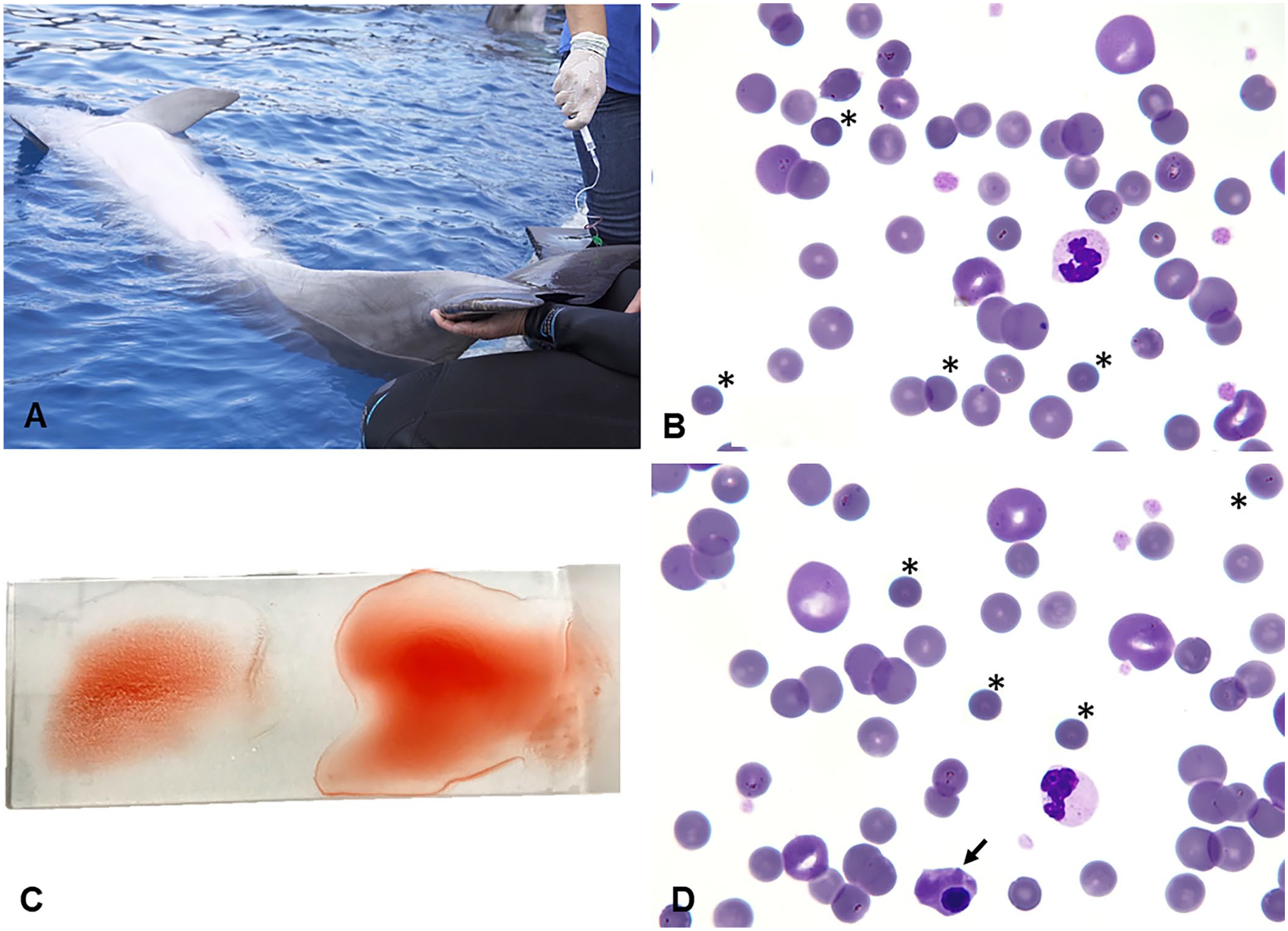
Immune-mediated hemolytic anemia in a female bottlenose dolphin.
Summary of the hematologic results of this bottlenose dolphin from the onset of abnormalities in routine blood analysis (day 1) until recovery; only the most relevant days are presented.
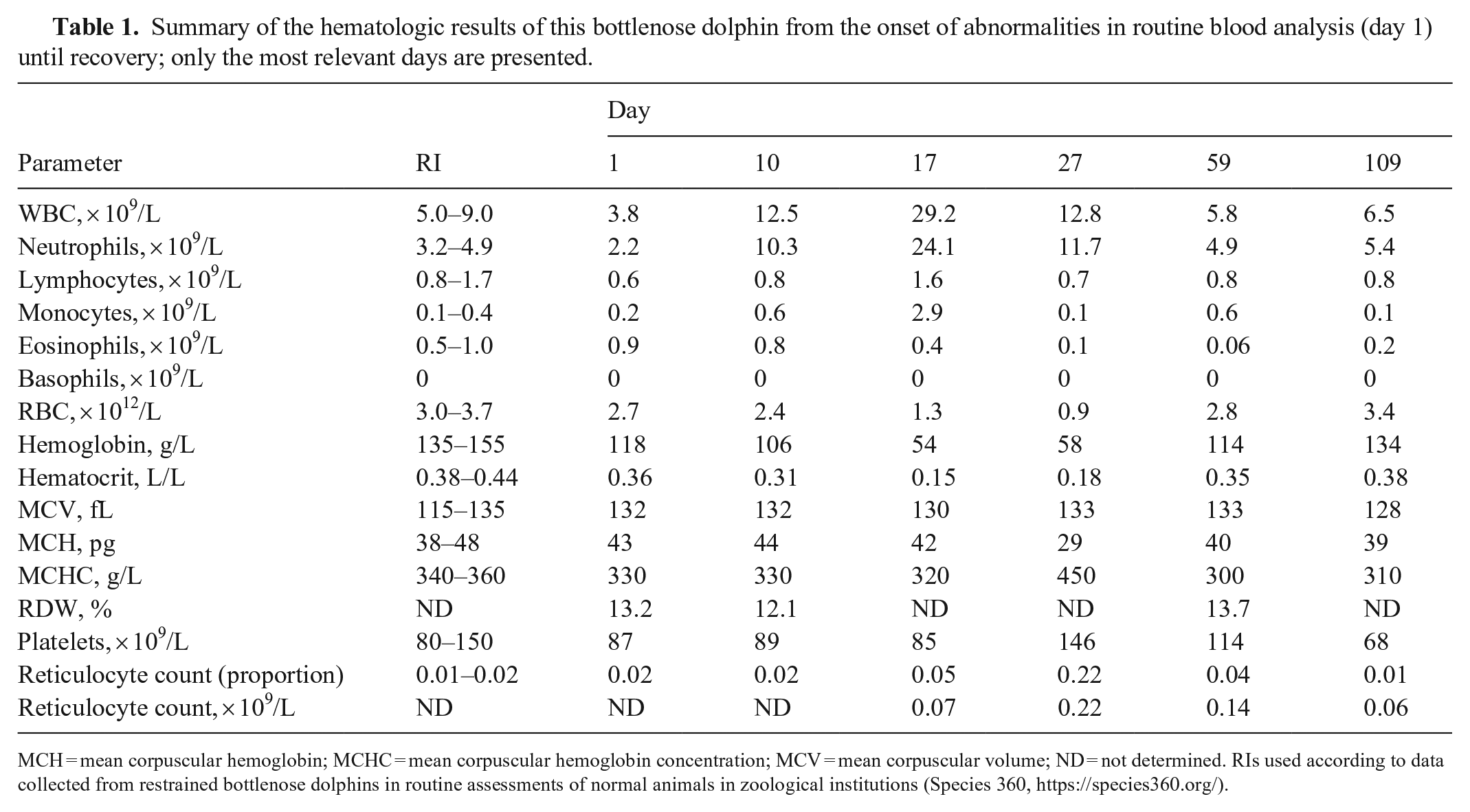
MCH = mean corpuscular hemoglobin; MCHC = mean corpuscular hemoglobin concentration; MCV = mean corpuscular volume; ND = not determined. RIs used according to data collected from restrained bottlenose dolphins in routine assessments of normal animals in zoological institutions (Species 360, https://species360.org/).
Summary of the serum biochemistry results of this bottlenose dolphin from the onset of abnormalities in routine analysis (day 1) until recovery; only the most relevant days are presented.
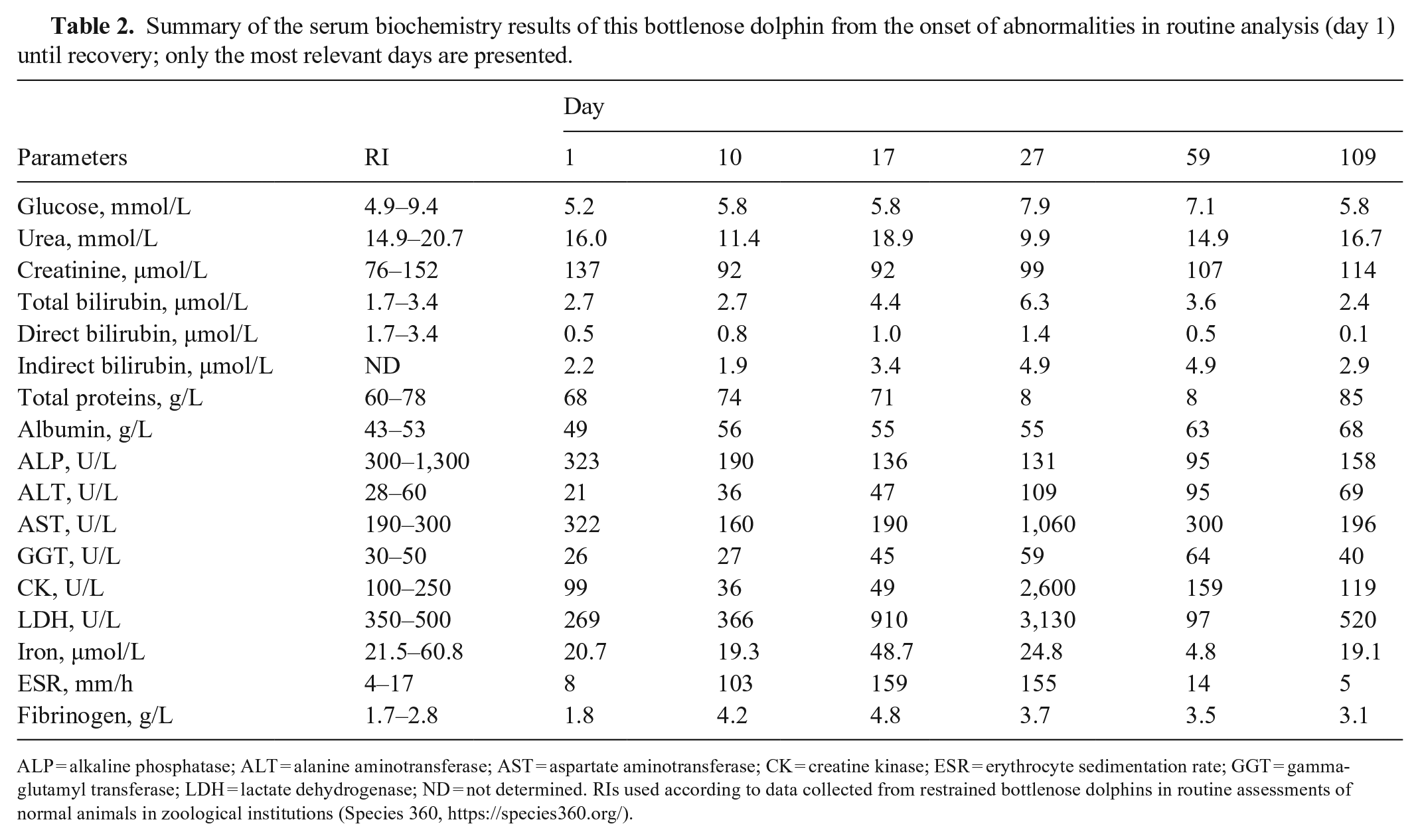
ALP = alkaline phosphatase; ALT = alanine aminotransferase; AST = aspartate aminotransferase; CK = creatine kinase; ESR = erythrocyte sedimentation rate; GGT = gamma-glutamyl transferase; LDH = lactate dehydrogenase; ND = not determined. RIs used according to data collected from restrained bottlenose dolphins in routine assessments of normal animals in zoological institutions (Species 360, https://species360.org/).
Blood smears from EDTA blood were also obtained and stained (Hemacolor; Merck). The monolayer area of the smear was used for manual WBC differential counts and cell morphology evaluation (Fig. 1B). The WBC differential results (Table 1) were obtained from the automated analyzer, and verified manually through microscopic observation by a veterinary team experienced in aquatic mammalian hematology.
When anemia was first detected in routine blood analysis (hereafter, day 1), the dolphin had a subtle decrease in RBC, hemoglobin, and PCV values, along with mild leukopenia. At this time, the dolphin was slightly hypoactive, had a slower response to trainers’ commands, and decreased swimming activity. However, 10 d later, the animal was significantly lethargic, and further tests were performed. On day 10, RBCs (2.4 × 1012/L; RI: 3.0–3.7 × 1012/L), Hb (106 g/L; RI: 135–155 g/L), and hematocrit (0.31 L/L; RI: 0.38–0.48 L/L) had further decreased, and relative reticulocytosis (2%) was observed. Despite moderate leukocytosis (12.5 × 109/L) and neutrophilia (10.3 × 109/L), only occasional band neutrophils (<1.5%) were observed on the blood smear (Table 1). It should be noted that in marine mammals, and particularly in cetaceans, a broader definition of band neutrophil has been recommended to allow early detection of inflammation; a neutrophil is defined as a band if any chromatin broader than a thin filament spans lobules. 14 A healthy cetacean may have up to 5% band neutrophils. Toxic change was evaluated as for other mammals, from 1+ to 3+.14,16
Some of the most important inflammatory markers were displayed on day 10, including a marked increase in fibrinogen concentration (4.2 g/L; RI: 1.7–2.8 g/L) and in ESR (103 mm/h; RI: 4–17 mm/h), which was suggestive of an ongoing inflammatory process (Table 2). 10 Blood-loss anemia from ulcerative gastritis with concurrent infection was suspected considering these analytical results, the high prevalence of gastrointestinal lesions in cetaceans, 1 and the animal’s previous medical history. Several mild episodes of dysbiosis and gastric inflammation had been recorded since weaning, and a more severe episode of hyporexia and hemorrhagic gastritis in a gastrointestinal disease outbreak of unknown etiology occurred a few weeks previously, which was treated with omeprazole and amoxicillin. Eight years previously, an outbreak of adenoviral infection in the dolphinarium led to a severe episode of hemorrhagic gastritis with associated blood-loss anemia. Empirical treatment was implemented, including broad-spectrum antibiotics, analgesics, anxiolytics, antihemorrhagics, vitamin supplements, and gastric protectants to promote erythropoiesis and blood clotting, while resolving upper gastrointestinal bleeding (Fig. 2).
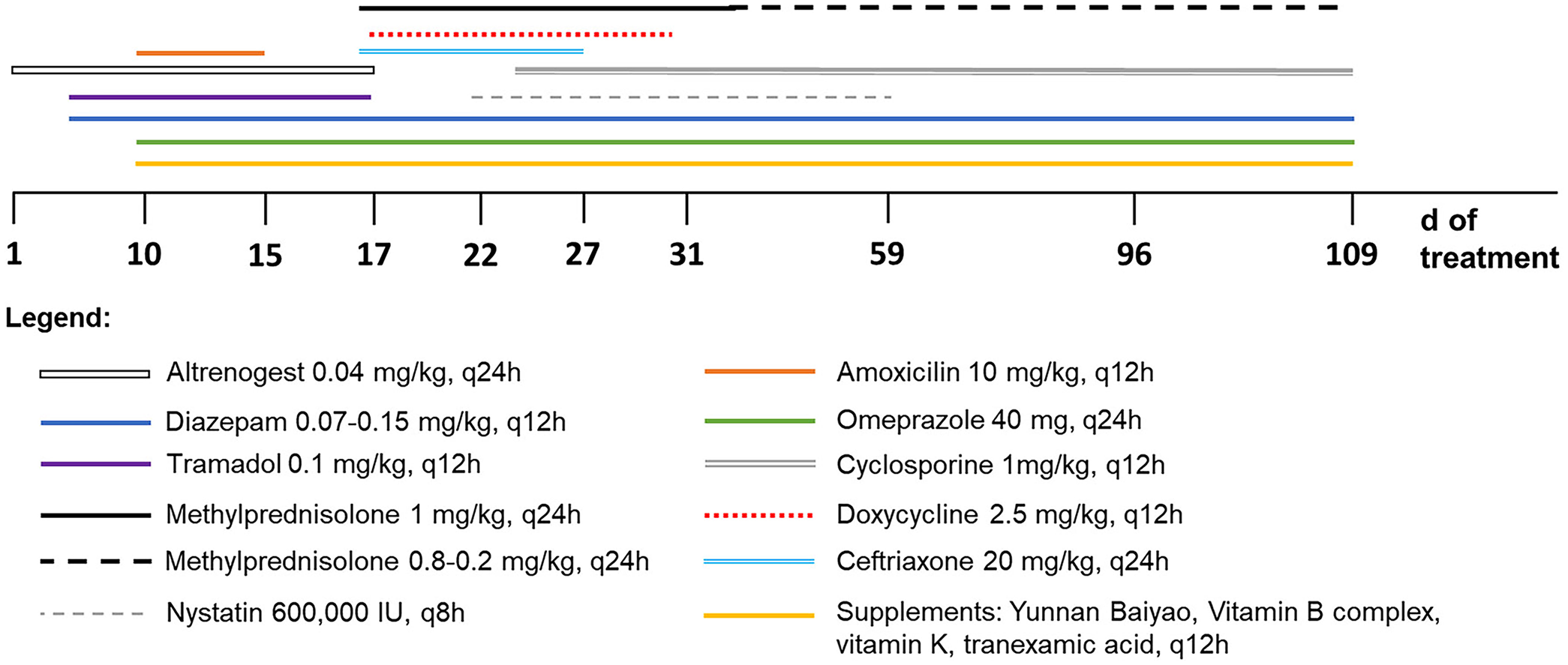
Multistep therapy used in the treatment of a bottlenose dolphin with immune-mediated hemolytic anemia.
On day 15, the dolphin had pale oral mucous membranes and prominent hyporexia. On CBC, the regenerative anemia and leukocytosis with left shift had increased (Table 1), as well as lactate dehydrogenase (LDH) activity. The total bilirubin concentration (6.5 µmol/L) was increased (Table 2), due to increased indirect bilirubin (5 µmol/L); although GGT activities were slightly above the RI, direct bilirubin was normal, making cholestasis less likely. Urinalysis from spontaneous collection also revealed bilirubinuria (2+), hemoglobinuria (1+), and proteinuria (1+) on dipstick examination (Uranotest 11C; Uranovet). Urine sediment analysis revealed no RBCs, thus confirming hemoglobinuria. On an ultrasound examination, no signs of intracavitary blood loss were seen (no pleural or abdominal effusions); however, splenomegaly and hyperechogenic liver parenchyma were noticed (no signs of cholestasis were observed). Slightly hemorrhagic gastric fluid was obtained under operant conditioning and some leukocytes were detected on cytologic examination, thus revealing gastric inflammation. A fecal occult blood test was positive, but this was disregarded as significant because the dolphin was fed with frozen-thawed fish, which may lead to false-positive results. Most common viral infectious diseases in this species, including cetacean morbillivirus, parainfluenza, influenza, adenovirus, and delphinid herpesvirus, as well as SARS-CoV-2, were all excluded by PCR assay of serum (Penta Laboratories). 23
On day 17, due to complete anorexia, the dolphin was moved to the medical pool for assisted feeding and medical support. At this time, anemia had worsened (Table 1), along with leukocytosis and hyperfibrinogenemia (Table 2). Mildly hemolyzed plasma was observed, and true hemolysis was suspected given that preanalytical sampling errors were ruled out, including difficult venipuncture and rough handling of blood tubes. A saline agglutination test (SAT) was performed by adding 4 drops of saline to 1 drop of blood (4:1 dilution), and macroscopic agglutination was observed (Fig. 1C). Blood smears (Fig. 1D) from that day revealed anisocytosis, polychromasia, reticulocytosis, and spherocytosis with ~30% spherocytes observed in the monolayer area of the smear; 4% of nucleated cells were nucleated RBCs (Fig. 1D). Neutrophilia with left shift and Döhle bodies were also present. The serum iron concentration was increased (48.7 µmol/L), probably due to systemic inflammation; polyclonal hypergammaglobulinemia was observed on protein electrophoresis. Bilirubinuria (3+), hemoglobinuria (3+), proteinuria (2+), and glycosuria (1+) had increased on urinalysis. These findings were consistent with immune-mediated hemolysis, as described in human and small animal medicine.3,18,21 RBCs from a normal dolphin were incubated with the plasma of the patient and macroscopic agglutination was observed on a slide (in-house version of an indirect Coombs test; Fig. 1C).
Considering that no treatment for immune-mediated hemolytic anemia (IMHA) has been described for cetaceans, recommendations from companion animal medicine were followed. Drugs considered to be potential triggers of IMHA were discontinued (altrenogest, amoxicillin, tramadol),5,8 and antibiotic therapy was modified on day 17. Anxiolytics, antihemorrhagics, vitamin supplements, and gastric protectants were continued (Fig. 2). Daily intramuscular ceftriaxone (20 mg/kg) was added, along with glucocorticoid therapy (methylprednisolone, 1 mg/kg). Upper digestive tract endoscopy revealed mild gastritis in the first gastric chamber, and gastric fluid cytology showed some yeast pseudohyphae; thus, doxycycline (2.5 mg/kg q12h) and nystatin (600,000 IU q8h) were later included, given their immunomodulatory and antifungal properties, respectively. Cyclosporine, starting at 1 mg/kg q12h dose, was added to reinforce the immunosuppressive therapy on day 24 (Fig. 2).
From day 17 onward, leukocyte numbers and inflammation markers progressively decreased, whereas RBC parameters remained stable for the following 10 d (Tables 1, 2), with reticulocytes peaking on day 27 (0.22 × 109/L). Plasma alanine aminotransferase (ALT) activity increased slightly over time, either due to hepatopathy from high doses of glucocorticoids or due to hypoperfusion resulting from the anemia; thus, a liver protectant supplement (Prolivet; Bioiberica) was added accordingly. Plasma aspartate aminotransferase (AST) activity and creatine kinase (CK) concentration were also increased (Table 2), possibly due to muscular trauma secondary to intramuscular injections.
One month after disease onset, the general attitude of the dolphin along with the anemia started to improve (Table 1), and doxycycline was discontinued after 2 wk of treatment. On day 46, hemoglobinuria and bilirubinuria were no longer detected on urine dipstick, so to avoid further secondary effects from glucocorticoids, methylprednisolone was slowly tapered while gradually increasing the cyclosporine dose. On day 59, RBC parameters were close to normal (Table 1), and an ultrasound examination performed on day 72 revealed a normal-sized spleen.
Three months later (day 109), all RBC parameters were within RIs (Table 1). However, the dolphin developed a moderate hemorrhagic gastritis (with gram-positive bacilli and yeasts in the gastric sample cytology) accompanied by an increase of serum fibrinogen concentration, and decreased iron concentration and ALP activity (Table 2), which resolved after establishing empirical treatment. In the following months, despite the lack of IMHA relapse, the dolphin was not always clinically stable and had recurrent periods of inappetence, presumably due to intermittent episodes of dysbiosis, gastritis, and diarrhea secondary to the immunosuppressive therapy. The dolphin remained underweight and under surveillance. In routine blood sampling, CBC parameters tended to be closer to the minimum RI but no spherocytes were observed up to the time of this writing (~10 mo after the initial diagnosis).
Although anemia of chronic disease and blood-loss anemia are well documented in cetacean literature,10,17 IMHA has not been reported previously, to our knowledge. We retrieved no cases of IMHA in cetaceans in a search of PubMed, Web of Science, Scopus, and Google, except for a recent publication of cold agglutinin-induced anemia. 21 Very few reports of IMHA have been published in marine animals, mainly in birds due to marine oil pollution, 25 and a single postmortem case in a northern fur seal (Callorhinus ursinus). 2 To our knowledge, a case of IMHA in a bottlenose dolphin has not been fully documented previously.
According to a diagnostic algorithm of IMHA in dogs and cats, 5 a diagnosis of IMHA can be considered reliable and consistent when 2 signs of immune-mediated destruction are present (SAT without washing, and spherocytes in our case), along with 1 or more signs of hemolysis (hyperbilirubinemia, hemoglobinemia, and hemoglobinuria in our case). A 1:49 dilution in SAT may increase specificity over a 1:4 dilution, 20 but no other dilutions or washing were performed in our case, even if they could preclude a contribution of hyperfibrinogenemia to the SAT. We used the diagnostic criteria established for dogs, 6 given that no reported cases exist for dolphins. Still, it should be noted that our case shares common features with IMHA in other species, such as cattle, in which spherocytosis and auto-agglutination were also present, along with hyperfibrinogenemia. 9
Fibrinogen is an important acute-phase protein that increases in inflammatory conditions. To account for the influence of hydration status on this parameter, a plasma protein:fibrinogen ratio is routinely used in ruminants; a ratio of <15 is suggestive of a real increase in fibrinogen concentration. 17 Although not calculated routinely in cetaceans, the protein:fibrinogen ratio in our case was <15 on day 17, when the hematocrit had the lowest value, and remained <20 during the acute stage of illness. Polyclonal hypergammaglobulinemia detected on protein electrophoresis suggests systemic inflammation associated with immunologic and hematologic conditions, and has been reported in humans with HIV, hematologic disorders, and autoimmune conditions. 26
Apart from SAT, other procedures that can be used in the diagnosis of IMHA are the Coombs and osmotic fragility tests. The latter has low specificity, is influenced by many conditions (e.g., hyperlipidemia), 11 and is considered an indirect measure of spherocytosis, mainly used in species that lack RBC central pallor. 13 Central pallor is easily recognized in cetaceans, 15 and has been reported in dolphin species, 19 meaning that spherocytes could be recognizable in the monolayer of the smear; spherocyte-like artifacts may arise toward the feathered edge of the smear. 6 In the direct Coombs test, a suspension of washed patient’s RBCs with attached antibodies is incubated with a species-specific antiglobulin reagent and RBC agglutination occurs in a positive test. As no specific reagent has been developed for cetaceans, we could not perform the direct Coombs test. In the indirect variant, washed RBCs from a normal animal are incubated with plasma of the patient. 24 An in-house version of this indirect Coombs test was performed, and macroscopic agglutination was observed. This test is not validated in dolphins, and the indirect Coombs test is less used in the veterinary field because non–cell-bound antibodies are generally low among small animals. 24 Flow cytometry testing for detecting RBC-bound antibodies was also not attempted in our case.
Immune-mediated anemia occurs when RBCs or their precursors are prematurely destroyed.3,18 The normal lifespan of RBCs is ~120 d in dogs and humans, and ~165 d in bottlenose dolphins. 12 In IMHA, through a type II hypersensitivity reaction, immunoglobulin IgG or IgM bind to RBC membranes.3,22 These coated RBCs are removed from circulation by macrophages in extravascular sites, such as the spleen, liver, and bone marrow, leading to extravascular hemolysis. 12 In some cases, the hemolysis may also occur concurrently within blood vessels (intravascular hemolysis) if the complement system is activated; hyperbilirubinemia, hemoglobinemia, and hemoglobinuria may also be present.3,22
In our case, the etiology of the hemolytic anemia remains unclear. IMHA may be primary or secondary.3,12,18 In veterinary species, most IMHA cases occur secondary to infectious, neoplastic, or other immune-mediated diseases or due to drug administration. 3 In cats, cattle, and horses, a long list of triggers has been identified.15,18 In contrast, primary or idiopathic IMHA refers to a condition with a positive Coombs test, in which other causes of hemolytic anemia have been excluded. 3 It is possible that IMHA in our case may have been triggered by the gastrointestinal disease outbreak that took place in that dolphin group a few weeks before the diagnosis of IMHA. There is an increasing number of inflammatory disease–associated IMHA reports in several species, including humans, dogs, and horses.7,15 Our case had a history of chronic intermittent hemorrhagic gastritis since weaning that was usually managed with supportive and empirical treatment (antibiotics, omeprazole, vitamins). Considering the long clinical history of dysbiosis and gastrointestinal disorders (chronic intermittent hemorrhagic gastritis) that preceded and followed the IMHA episode, a subclinical chronic inflammatory disease may have existed and could have been a trigger for an IMHA episode.
Alternatively, the IMHA may have been primary or idiopathic based on the exclusion of other common triggers. Drug exposure was unlikely a trigger in our case, as the animal had already been exposed to all the drugs administered near the onset of the disease and the last inactivated vaccine against Erysipelothrix rhusiopathiae (ER-Bac Plus; Zoetis) was given 2 y before. Neoplasia-associated IMHA was also unlikely, as abdominal and thoracic ultrasound revealed no significant abnormalities. 15 IMHA is a recognized paraneoplastic syndrome in people, 6 and in veterinary medicine several types of neoplasia may play a major role in the development of IMHA, such as lymphoma, hemangiosarcoma, myeloproliferative disease, anaplastic sarcoma, and melanoma.3,18 The main cetacean viral infectious diseases were also ruled out by PCR assay, not supporting the hypothesis of an infection-related IMHA. Blood parasites are another common cause of IMHA among animals,3,15 but they are not described in cetaceans nor were any inclusions observed in our case in the multiple blood smears examined. Lastly, heavy metal serum concentrations were also measured (data not shown), and no significant changes were found. 4
Relapses of IMHA and manifestations of other forms of autoimmune disease are well described in dogs and pose a significant risk in the following months to years after the onset of IMHA.3,6 None of these conditions have been recorded up to the time of writing this report, but our case is still under active surveillance. Negative prognostic indicators identified in dogs include autoagglutination, high serum bilirubin, left shift neutrophilia, thrombocytopenia, hypoalbuminemia, hypokalemia, and elevated serum CK. 6 Although 4 of 7 indicators were present in our case, recovery occurred after intensive supportive treatment. Serial adjustments on the treatment plan have been made daily, using protocols described for small animal IMHA. The possibility of transfusion was also discussed, but it was not attempted due to the lack of studies in cetaceans.
Footnotes
Acknowledgements
We are indebted to the veterinary personnel and caretakers of Oceanogràfic (Valencia, Spain) for their help and advice on the treatment plan.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
