Abstract
Osteogenic melanoma is a rare variant of metaplastic malignant melanoma in human medicine and appears to be a similarly rare variant in dogs. Two dogs with oral malignant melanoma with neoplastic bone formation are reported in this study. Both tumors were characterized by malignant melanocytes that transitioned into neoplastic bone at the deep margins of the neoplasm. Immunohistochemical analysis revealed S100- and Melan-A–positive neoplastic cells adjacent to, and occasionally embedded within, an osteoid and chondroblastic matrix. Scattered clusters of neoplastic cells were also positive for osteocalcin. The findings indicate that in dogs, as in humans, neoplastic melanocytes have metaplastic potential and can be osteogenic.
The combination of neoplastic bone and malignant melanoma is a rare entity in both human and veterinary medicine. This combination was first described in human medicine in 1984 21 and remains rare, in that only 20 have been reported since that time. 4,7 In humans, most of these tumors arise within lentiginous melanomas, and more than 50% of the cases are subungual. 12 Cases of osteogenic melanoma arising in the skin, oral cavity, and nasal cavity have also been reported in humans. 1,4,7,8,12,14,15,18,21 Only 15 cases of malignant melanoma with osteoid formation have been reported in the dog. 3,13,16,19 Because osteosarcoma and malignant melanomas have different prognoses and require different treatments, the recognition of the potential for malignant melanocytes to produce osteoid is important, especially if few melanin-containing neoplastic cells are present and the osteosarcoma-like phenotype is predominant. Two cases of oral osteogenic melanoma in dogs are reported with histologic and immunohistochemical characterization of this unusual tumor.
The first case was an 11-year-old spayed female Shih Tzu that presented to the University of Georgia, College of Veterinary Medicine, Veterinary Teaching Hospital (UGA-VTH; Athens, GA) for a recurrent maxillary gingival melanoma. The mass was incompletely excised by the referring veterinarian 2 months before presentation. Physical examination revealed a pedunculated, partially pigmented mass (2.8 cm x 2 cm x 1.3 cm) in the area of the left maxillary fourth premolar and first molar, as well as an enlarged left mandibular lymph node. Aspirate and cytologic examination of the enlarged mandibular lymph node showed lymphoid hyperplasia, with no evidence of metastatic melanoma. Thoracic and abdominal radiographs were normal. Computed tomography of the maxilla revealed a mass closely associated with the tooth roots of the left fourth premolar and first molar with a radiolucent zone noted near one tooth root. Debulking surgery was performed with removal of the mass and adjacent teeth. The tumor site and regional lymph nodes were radiated with 4 weekly 8-Gy (Gray unit) fractions with a cobalt-60 teletherapy unit. Two weeks after completion of radiation therapy, thoracic radiographs were normal, and chemotherapy was initiated with carboplatin a (150 mg/m 2 intravenously every 3 weeks). Eight weeks after radiation therapy, the tumor had recurred locally, and lomustine b was administered (55 mg/m 2 orally). Follow-up information from the referring veterinarian revealed that the dog developed rapidly progressive local disease and died at home 2 months later (4 months after completing radiation therapy).
The second case was a 9-year-old spayed female Scottish Terrier that presented 2 weeks after incomplete excision of a gingival mass. The mass was pedunculated and located at the level of the right mandibular first premolar. Computed tomography of the mandible showed loss of the mandibular incisors. Slight bony lysis and defects of the mandible were noted in the area of the sockets for the mandibular incisors. Periosteal new bone was noted at the level of the canine teeth. Changes were most prominent on the right. On dorsal plantar reconstructions, a small mineralized opacity was noted within the soft tissues adjacent to the right rostral portion of the mandible. Neoplasia was not evident on abdominal ultrasound or in thoracic and abdominal radiographs. Biopsy samples were obtained from multiple areas of proliferative gingival tissue, a pigmented area in the region of the mandibular canine teeth, and the right mandibular lymph node. All oral biopsies were diagnosed as gingival hyperplasia except for an area near the right second premolar, which was diagnosed as a fibromatous epulis. Additionally, sections of the tumor removed by the veterinarian were obtained for review and immunohistochemical staining. On the basis of the diagnosis of incompletely excised junctional melanoma with metastasis to the mandibular lymph node and underlying osteoblastic osteosarcoma, adjunctive coarse fractionated radiation therapy and chemotherapy were recommended. The tumor site and mandibular lymph node bed were radiated with 4 weekly 8-Gy fractions with a cobalt-60 teletherapy unit. No chemotherapeutic treatment was pursued. Four months after radiation therapy, recurrence of the oral mass was not evident, and thoracic radiographs were normal. Follow-up information from the referring veterinarian revealed that the dog did well for 25 months after radiation therapy. At that point, the dog developed difficulty rising and lying down. Thoracic radiographs revealed diffuse pulmonary metastasis. The dog was euthanized 1 month later. A postmortem examination was not performed.
Tissues from both dogs were fixed in 10% buffered formalin and routinely processed for histologic examination. The tissue from dog 2 was decalcified before being processed. Samples of the masses were embedded in paraffin, sectioned at 4 μm, stained with hematoxylin and eosin, and viewed with light microscopy.
In the biopsies from both dogs, the gingival tissue was effaced by nests of pleomorphic neoplastic melanocytes within the mucosa, at the junction of mucosa and lamina propria, and throughout the rest of the submitted tissue. Most tumor cells were oval to elongate, had large, oval nuclei, prominent nucleoli, scant to abundant eosinophilic cytoplasm, and variable amounts of cytoplasmic melanin. Occasional megalocytic and multinucleated neoplastic cells also were noted throughout both tumors. Neoplastic cells at the deeper aspects of the masses were more stellate and spindle-shaped, and fewer contained melanin. These cells were adjacent to, or trapped within, thick bands of irregular cartilaginous-like matrix or osteoid, which was sometimes mineralized (Fig. 1). Cells immediately adjacent to osteoid had more cytoplasm and a slight perinuclear halo, giving them an osteoblast-like appearance (Fig. 2). Fifteen mitotic figures per 10 fields at 400x were seen in both tumors. The osteogenic region was more distinctly separated from the area of more classical melanoma. Junctional activity was very pronounced in the tumor from dog 2. Inflammatory cells, in particular lymphocytes and plasma cells, also were prominent in some areas along the junction of the dermis and epidermis in the tumor from dog 2. In comparison, the tumor from dog 1 was characterized by an indistinct transition between melanoma and osteogenic tumor. The initial biopsy from dog 1 consisted predominantly of neoplastic melanocytes, although some neoplastic osteoid was present, whereas the majority of the tumor was osteogenic in the second biopsy (Fig. 1). Differences between the first and second biopsies might have been related to the invasiveness, in that the first biopsy was more superficial. Thrombi containing neoplastic cells also were observed in lymphatic (dog 2) and blood (dog 1) vessels in the tissues from both dogs.
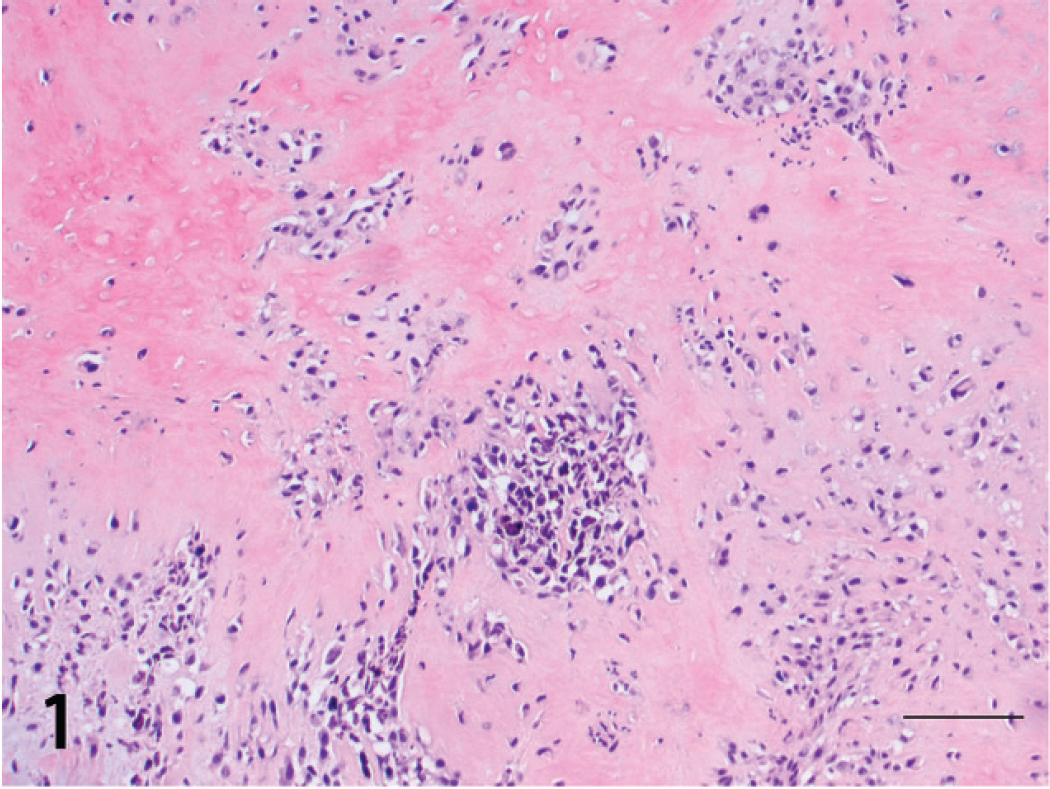
Gingival osteogenic melanoma; dog 1. Aggregates of neoplastic melanocytes surrounded by an osteoid matrix. Hematoxylin and eosin. Bar = 100 μm.

Gingival osteogenic melanoma; dog 2. Melanin-containing neoplastic cells closely adjacent to an osteoid matrix. Hematoxylin and eosin. Bar = 30 μm.
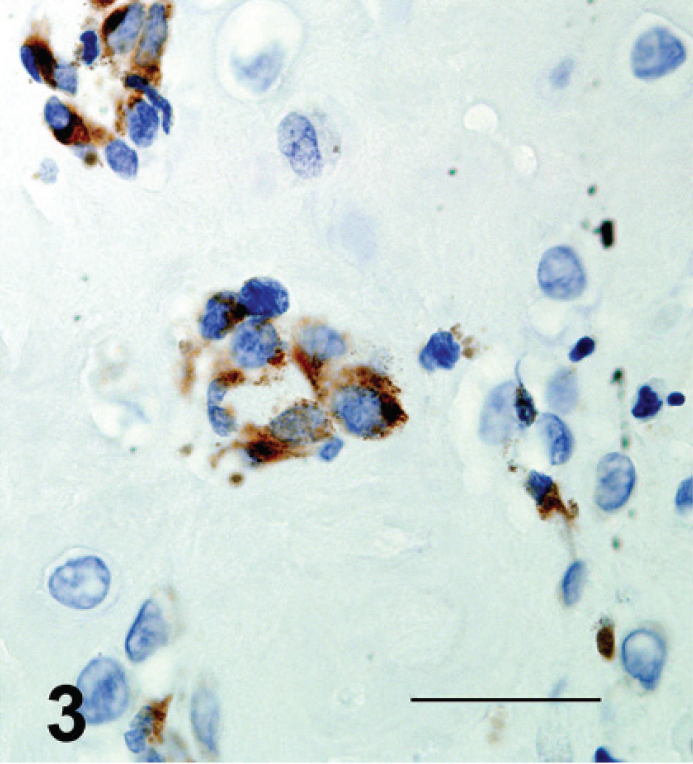
Gingival osteogenic melanoma; dog 1. Clusters of neoplastic cells embedded in osteoid showed positive cytoplasmic immunolabeling for Melan A with a peroxidase detection system. Bar = 30 μm.

Gingival osteogenic melanoma; dog 2. Scattered aggregates of neoplastic melanocytes near the osteoid matrix are positive for S100 by immunohistochemical analysis with peroxidase detection. Bar = 30 μm.

Gingival osteogenic melanoma; dog 1. Neoplastic melanocytes positive for osteocalcin by immunolabeling with alkaline phosphatase detection. Bar = 30 μm.
Immunohistochemical evaluation with antibodies for S100, Melan-A, and osteocalcin was used to characterize the neoplastic cells. S100 is a calcium-binding protein that is present on a variety of cells, including melanocytes, Langerhans cells, and chondrocytes. 2 Approximately 89% of amelanotic melanomas in dogs have been reported to exhibit weak to moderate cytoplasmic staining for S100. 11 Melan-A is considered a highly specific marker for malignant melanoma, although it is generally not expressed in canine amelanotic melanomas. 11 In human tissue, osteocalcin reportedly has at least 70% sensitivity in identifying osteosarcoma and 100% specificity in identifying bone-forming tumors. 5
Immunohistochemical evaluation was performed with S100 (polyclonal rabbit anti-bovine, 1:1,000), c Melan-A (monoclonal mouse anti-human, 1:10), c and osteocalcin antibody (monoclonal anti-bovine, 1:200). d A streptavidin–peroxidase method e was employed with S100 and Melan-A staining. The alkaline phosphatase Super Sensitive method f was used with osteocalcin staining.
Staining for all 3 markers was multifocal in both tumors. In dog 1, the majority of tumor cells were negative for Melan-A. Local, intense, cytoplasmic staining was observed, however, primarily in spindle-shaped cells. Neoplastic cells in dog 1 had multifocal to diffuse cytoplasmic staining with S100. Cells within the neoplastic cartilaginous matrix consistently exhibited intense staining. Low numbers of neoplastic cells in less differentiated areas were stained intensely, whereas the majority were only slightly stained. S100 and Melan-A staining was less pronounced in decalcified tissue from dog 2 and was present primarily in neoplastic cells infiltrating junctional areas. In both cases, Melan-A– and S100-positive neoplastic cells adjacent to osteoid matrix were observed (Figs. 3, 4). Scattered clusters of tumor cells near osteoid often stained positively for osteocalcin (Fig. 5). On the basis of the histologic features, immunohistochemical results, and reports of similar cases, both tumors were diagnosed as osteogenic melano-mas. 1,3,7,8,12,14,15,18,21
Several mechanisms for bone formation in malignant melanoma have been proposed. 1,12,14,15,17,18,21 Embryologic studies have established that certain skeletal, dermal, and muscular elements of the head are derived from the cranial aspect of the neural crest. 17 Thus, metaplastic changes observed with malignant melanoma have been attributed to pluripotentiality of neuroectodermal cells. 17 Metastasis of malignant melanocytes with neoplastic bone formation to regional lymph nodes has been documented in several cases of osteogenic melanoma in human medicine. 8 This supports the hypothesis that bone production is secondary to mesenchymal metaplasia of melanocytes, rather than the presence of a second tumor, 8 such as an osteosarcoma. This interpretation is further supported by the observation that osteosarcomas usually do not metastasize to lymph nodes. 6,9,20
Other potential causes of bone formation in melanomas include stromal response to injury or inflammation (e.g., to previous excisional surgery or biopsy procedures), pseudosarcomatous proliferation and subsequent ossification of connective tissue stroma invaded by malignant cells, 12 and production and release of factors from tumor cells that stimulate stromal mesenchymal cells to differentiate into chondroblasts and osteoblasts. 3 Specific factors have not been identified in osteogenic melanomas. Bone morphogenetic proteins, transforming growth factor-β1 (TGF-β1) and TGF-β2 of the TGF superfamily, however, are considered possible factors responsible for bone formation in other human malignancies. 3 Finally, bone formation in melanomas might be the result of a collision of a melanoma and an osteosarcoma.
In the cases presented in this study, the presence of neoplastic bone is most likely due to metaplasia of malignant melanocytes, because neoplastic cells containing melanin were observed directly adjacent to, and occasionally embedded in, osteoid. Bone production secondary to inflammation or tissue injury is unlikely because inflammation was minimal and generally limited to the ulcerated surface of the tumor (dog 1) or was multifocal at the epidermal–dermal junction (dog 2). The neoplastic appearance of the cells associated with osteoid also makes a reparative process unlikely. In dog 1, the marked increase in neoplastic bone and cartilage with fewer melanin-containing cells in the recurrent mass was probably due to the increased depth of the second biopsy, although increased malignant metaplasia stimulated by surgical trauma cannot be excluded completely. 10 The separation between classical melanoma and osteosarcoma phenotypes was distinct in dog 2, making a collision tumor composed of malignant melanoma and osteosarcoma a possible differential. This differential was ruled out on the basis of the gradual transition from melanin-containing cells to fibroblast-like cells to osteoblast-like cells. Mesenchymal metaplasia of melanoma cells with production of both fibroblastic and osteoblastic cells and their associated matrices has been reported in human melanomas. 14 Further evidence supporting the presence of populations of tumor cells with varying phenotypes is the scattered positive staining observed for all 3 of the markers used.
Too few cases of osteogenic melanoma have been described in both the human medical and veterinary literature to know whether this phenotype has a behavioral pattern distinctly different from other malignant melanomas. Currently, expectations, prognoses, and therapeutic plans for these masses are the same as for other malignant melanomas. 12,19 In human malignant melanoma, expression of bone sialoprotein (BSP) has been correlated with local and regional tumor spread. 18 In vitro studies have found that direct bone formation by melanoma cells is mediated by expression of BSP and the transcription factor Cbfa1/Runx2, which are normally only expressed in cells of mesenchymal origin. 18 Increased Cbfa1 expression has also been shown to induce the expression of specific metallo-proteinases and collagenases, which could potentially cause matrix degradation and tumor invasion. Because the presence of neoplastic bone in malignant melanoma suggests apparent mesenchymal conversion, it is possible that osteogenic malignant melanoma has a greater metastatic potential. In dogs, the majority of oral melanomas have already metastasized by the time the lesion is noticed. 22 Evidence of vascular invasion and ipsilateral mandibular lymph node metastasis was present in both dogs in this study. Both dogs were treated for malignant melanoma. Dog 1 rapidly developed local recurrence despite therapy and died at home approximately 6 months after being diagnosed. Dog 2 was euthanized more than 2 years after initial diagnosis for uncharacterized pulmonary metastasis.
On the basis of the features of the canine and human cases, experimental evidence demonstrating the ability of melanocytes to make mesenchymal elements, and the fact that osteosarcoma–malignant melanoma collision tumors have yet to be documented in either the human medical or veterinary literature, melanomas with neoplastic bone should be considered to be osteogenic melanomas. The cases of osteogenic melanoma reported herein and those reported previously 3,16,19 were in older dogs. Small breeds were more commonly affected. With one exception, tumors were oral and were primarily gingival. Features common to these cases of canine osteogenic melanoma are the absence of primary bony involvement, presence of junctional activity, and neoplastic cells with distinct melanin granules or Melan-A– and S100-positive cells adjacent to or embedded in tumor osteoid, or both. Ultrastructural identification of melanosomes in tumor cells also could potentially be used to confirm the diagnosis of melanoma. 12 On the basis of the small number of reported cases, the biological behavior of canine osteogenic melanoma appears to be similar to that of oral malignant melanoma.
Footnotes
a.
Paraplatin®, Bristol-Myers Squibb Co., New York, NY.
b.
CeeNU®, Bristol-Myers Squibb Co., New York, NY.
c.
Dako North America Inc., Carpinteria, CA.
d.
Zymed Laboratories Inc., San Francisco, CA.
e.
LSABTM 2, Dako North America Inc., Carpinteria, CA.
f.
Super SensitiveTM Alkaline Phosphatase Complete, BioGenex Laboratories Inc., San Ramon, CA.
