Abstract
Lipofuscin is a complex mixture of highly oxidized, cross-linked macromolecules that accumulates in neurons with age and some neurodegenerative diseases. Equine dysautonomia (ED) is a polyneuropathy that mainly affects autonomic and enteric nervous systems, resulting in alimentary tract dysfunction. Our main aim was to determine whether neuronal lipofuscin increased with increasing duration of ED. We investigated the prevalence of lipofuscin in cranial cervical ganglia of horses with acute (AED), subacute (SED), and chronic ED (CED), young controls (of similar age to ED cases), and aged controls (n = 8 per group). We used Schmorl stain for histologic detection of lipofuscin and assessed its accumulation in neurons using image analysis software. The percentage of neurons positive for lipofuscin increased with age in individual groups and all groups combined (p < 0.001). There were fewer positive neurons in AED and SED compared to aged controls (p < 0.001) and more in CED than AED cases (p = 0.042) and young controls (p = 0.012). We found a strong positive correlation between percentage positive neurons and percentage positive area of the neuron containing lipofuscin for combined groups (p < 0.001). Although neuronal lipofuscin increased in cranial cervical ganglion in CED cases, it remains to be determined whether this is a cause or consequence of neuronal degeneration.
Equine dysautonomia (ED), or grass sickness, is a neurodegenerative disease affecting the central and peripheral nervous systems, particularly the autonomic nervous system. 13 It is most prevalent in northern Europe, southern Argentina, and Chile, and almost exclusively affects horses at pasture, with young horses (2–7-y-old) being predisposed.13,21 It is fatal in the acute (AED, duration <2 d) and subacute (SED, duration 2–7 d) forms but ~50% of chronic (CED, duration >7 d) cases may survive. 13 The etiology is uncertain but hypotheses include Clostridium botulinum type C toxicoinfection, and mycotoxins ingested at pasture.9,12 Clinical signs include colic, dysphagia, nasogastric reflux, muscle tremors, and sweating, with marked weight loss in CED. Diagnosis is confirmed in autonomic ganglia postmortem by identification of characteristic neuronal degenerative changes of chromatolysis, loss of Nissl substance, swelling, vacuolation, and pyknosis or loss of nuclei. 16 Such changes occur in prevertebral and paravertebral ganglia, the ventral horn of the spinal cord, intermediolateral nucleus, dorsal root ganglia, and several brainstem nuclei. 5 In the enteric nervous system, the ileal submucosal and myenteric plexuses are most severely affected, and ileal biopsies can provide an antemortem diagnosis. 13 Several studies demonstrated accumulation of metabolites in degenerating autonomic neurons, including protein 8 and synaptophysin, 22 and an increase in expression markers for apoptosis and neurotoxin exposure, 18 consistent with widespread derangement of cellular function.
Lipofuscin consists of complex, cross-linked aggregates of protein, lipid, iron, and other minerals. 19 It is normally found in lysosomes, but accumulation of highly insoluble, reactive aggregates impairs autophagy and lysosomal degradation. This results in accumulation of lipofuscin in the cytoplasm of degenerating and senescent neurons in humans and laboratory animals, adversely affecting their function.10,15 This is a feature of aging and some human neurodegenerative diseases (e.g., Parkinson and Alzheimer diseases). 15 In horses, there are very few studies on lipofuscin in autonomic ganglia. In normal horses, lipofuscin is described in the spinal and sympathetic ganglia. 2 In ED, a subjective increase in lipofuscin has been reported in autonomic ganglia, although this appeared associated with age rather than disease. 8 Neuronal lipofuscin also increases in the CNS in older horses, 4 in the pars nervosa in pituitary pars intermedia dysfunction, 7 and in the CNS in equine degenerative myeloencephalopathy and equine motor neuron disease.6,14 We retrieved no previous reports of quantification of lipofuscin in autonomic neurons with aging or ED in horses in a search of Google, PubMed, Web of Science, and Scopus using the search terms “lipofuscin”, “horses”, “autonomic neurons”, and “ganglia”, suggesting that such studies have not been performed previously in horses.
Our hypotheses were 1) that neuronal ganglionic lipofuscin accumulation is quantitatively increased in longer duration cases (CED) compared to AED and SED, and 2) that neuronal lipofuscin increases with age in ED and control horses. Such changes could potentially contribute to neuronal dysfunction in the chronic form of ED.
Our study was approved by the University of Edinburgh Veterinary Ethical Review Committee (approval 62.21). Cranial cervical ganglia (CCG), which are paravertebral sympathetic ganglia, were collected postmortem from AED, SED, and CED cases (8 per group), 8 young control horses, and 8 aged controls without ED or other multisystem neurologic disease (Suppl. Table 1). All horses were euthanized for welfare reasons. The CCG were fixed in 10% neutral-buffered formalin, embedded in paraffin wax, and 4-µm thick sections routinely processed to H&E sections to confirm the presence or absence of ED and qualitatively assess granularity. All ganglia were sectioned longitudinally except 1 young and 1 aged control that were sectioned transversely. CCG were used as they are consistently affected in ED and used routinely for ED diagnosis in our laboratory.
Many stains have been reported for lipofuscin including Sudan black, Fontana–Masson, Schmorl, Ziehl–Neelsen (ZN), and the gold standard of autofluorescence (unavailable for our study), none of which is completely specific. A preliminary trial of a small subsection of the CCG sections was stained with Masson, Oil Red O, ZN, PAS, and Schmorl stains. Only PAS and Schmorl stain were clearly positive for typical granules in the appropriate area of the cell. As Schmorl stain allowed more distinct differentiation between the positively stained areas and the background cytoplasm, we used it for subsequent analyses, using routine methods. Images were taken at 3 sites (approximately the middle and near each end of the ganglion using the naked eye) using a BX41 microscope with a ×20 objective and DP72 camera (3 mm2/picture; Olympus). The image processing software Fiji 17 was used to count the number of positively and negatively staining neurons in each of the 3 images; the average percentage of neurons positive for lipofuscin was calculated, regardless of the amount of lipofuscin present in positive neurons. In addition, to semi-quantify the amount of lipofuscin, the area of cytoplasm positive for lipofuscin in each neuron containing the pigment was calculated using image analysis software QuPath (v.0.3.2, https://qupath.github.io/), 1 expressed as a percentage of total neuronal area.
Statistical analyses were performed with R (https://www.r-project.org/). Generalized linear models with binomial errors (GLMb) were carried out on differences in the percentage of positive neurons and percentage lipofuscin-positive area between groups. Post-hoc Tukey pairwise comparisons were undertaken to assess pairwise differences in these percentages between individual groups. Ages of the groups were also compared by post-hoc Tukey pairwise comparisons using an ANOVA. The relationship between age and percentage of positive neurons and between lipofuscin-positive area (%) and percentage of positive neurons were also assessed with GLMb, with odds ratios (OR) and associated 95% CIs calculated. A p-value ≤0 .05 was considered statistically significant.
Each group comprised approximately equal numbers of males and females, as well as a variety of large and small breeds (Suppl. Table 1). The ages of each group were as follows: AED (median: 6.5 y; range: 2–16 y), SED (6.5 y; 2–17 y), CED (4.5 y; 1–11 y), and controls (young: 9 y, 2–10 y; aged: 17 y, 11–28 y). The aged controls were older than all other groups (p < 0.002), but the ages of the other groups did not differ from each other (p > 0.844).
In the H&E-stained sections, typical degenerative changes were present in the CCG of all ED cases and absent from the controls (Fig. 1). Yellow-brown granular pigment consistent with lipofuscin, mainly at the periphery of one pole of the neuron, was present in H&E-stained sections. This corresponded to areas of dark-blue granular staining against a pale-pink background in Schmorl-stained sections (Fig. 2). In the initial staining trial on a small subset of CCG, we found PAS-positive staining for the material in the same position in the neuron. This combination of features was considered strongly indicative of lipofuscin. Qualitative examination by a pathologist (E.M. Milne) did not reveal any apparent difference in intracellular pattern or size of granules between groups.
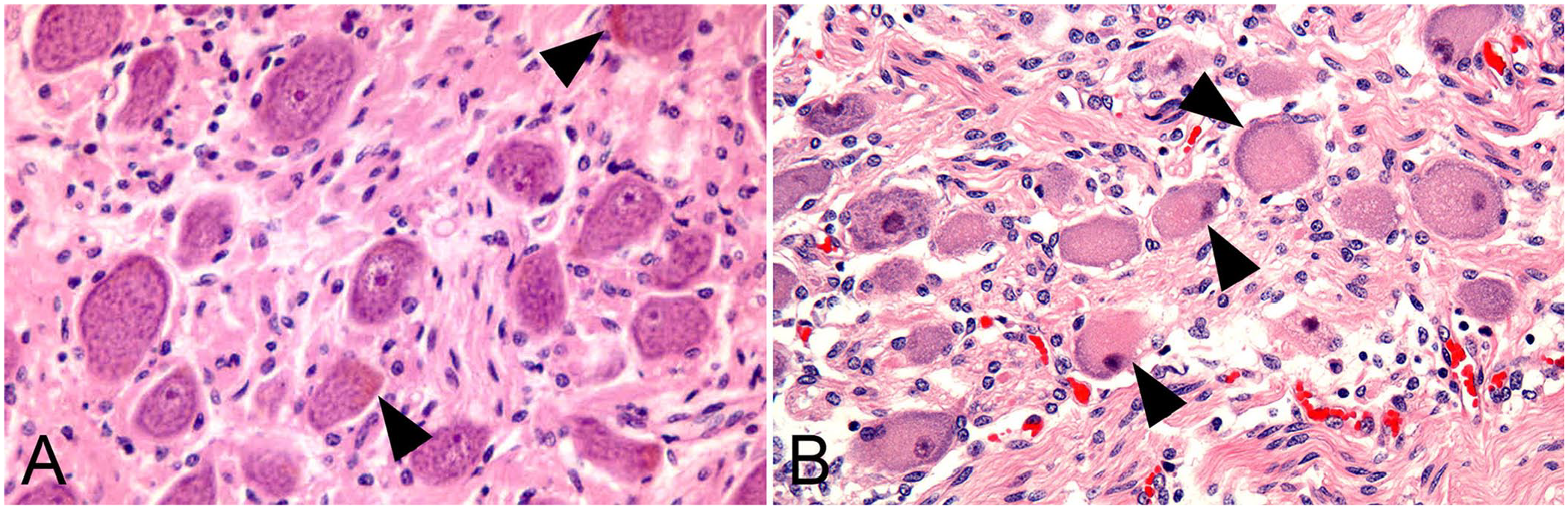
Cranial cervical ganglionic neurons.
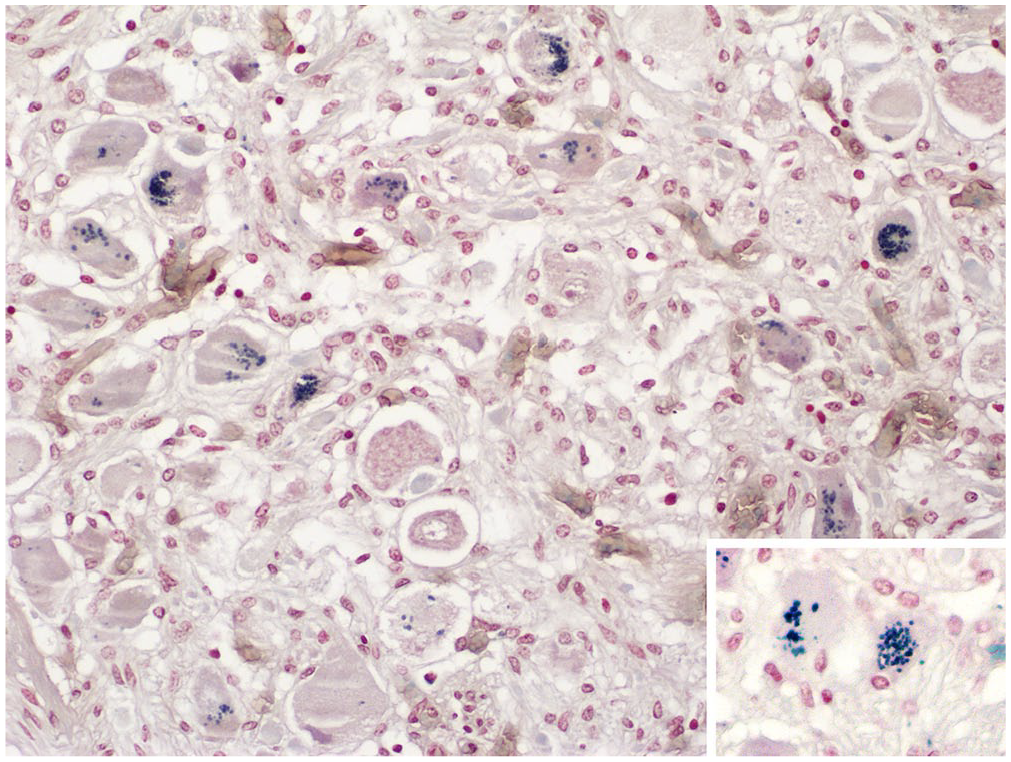
Cranial cervical ganglionic neurons from a case of acute equine dysautonomia. Lipofuscin granules are stained dark-blue in the cytoplasm of several neurons. Inset: higher magnification image from the same section. Schmorl stain.
Age had a positive curvi-relationship with percentage of neurons positive for lipofuscin for all groups combined and each individual group (p < 0.001, OR > 1.12 in each case; Fig. 3A). There was an apparent increase and then plateauing of the relationship between percentage area positive for lipofuscin with increasing age for all data (p = 0.020); however, the relationship was confounded by the 2 oldest control horses, which had a relatively low lipofuscin area for their age. Excluding these 2 horses resulted in the relationship changing to a positive linear one (p < 0.001, OR = 1.12 [1.07–1.18]; data not shown).
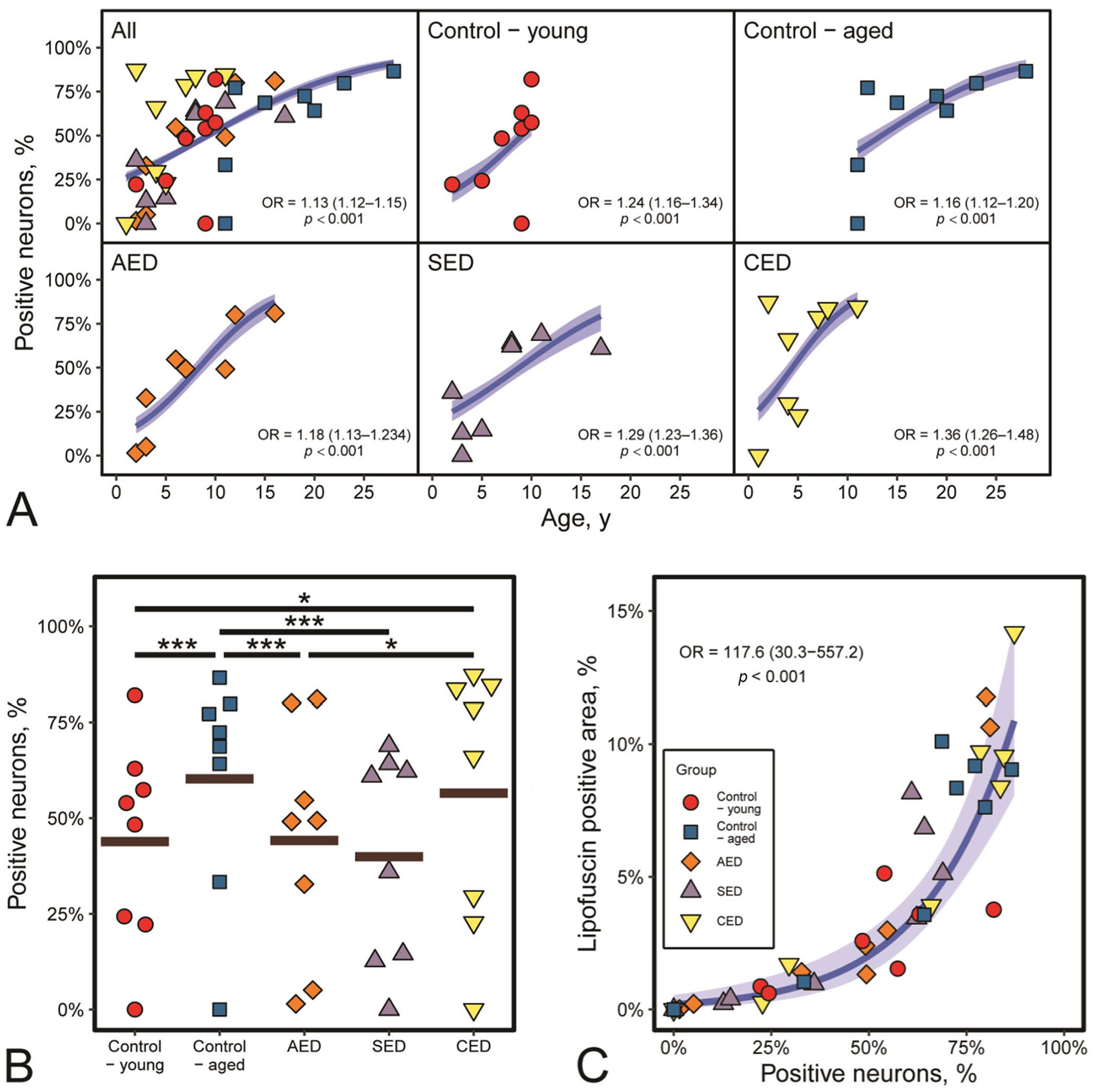
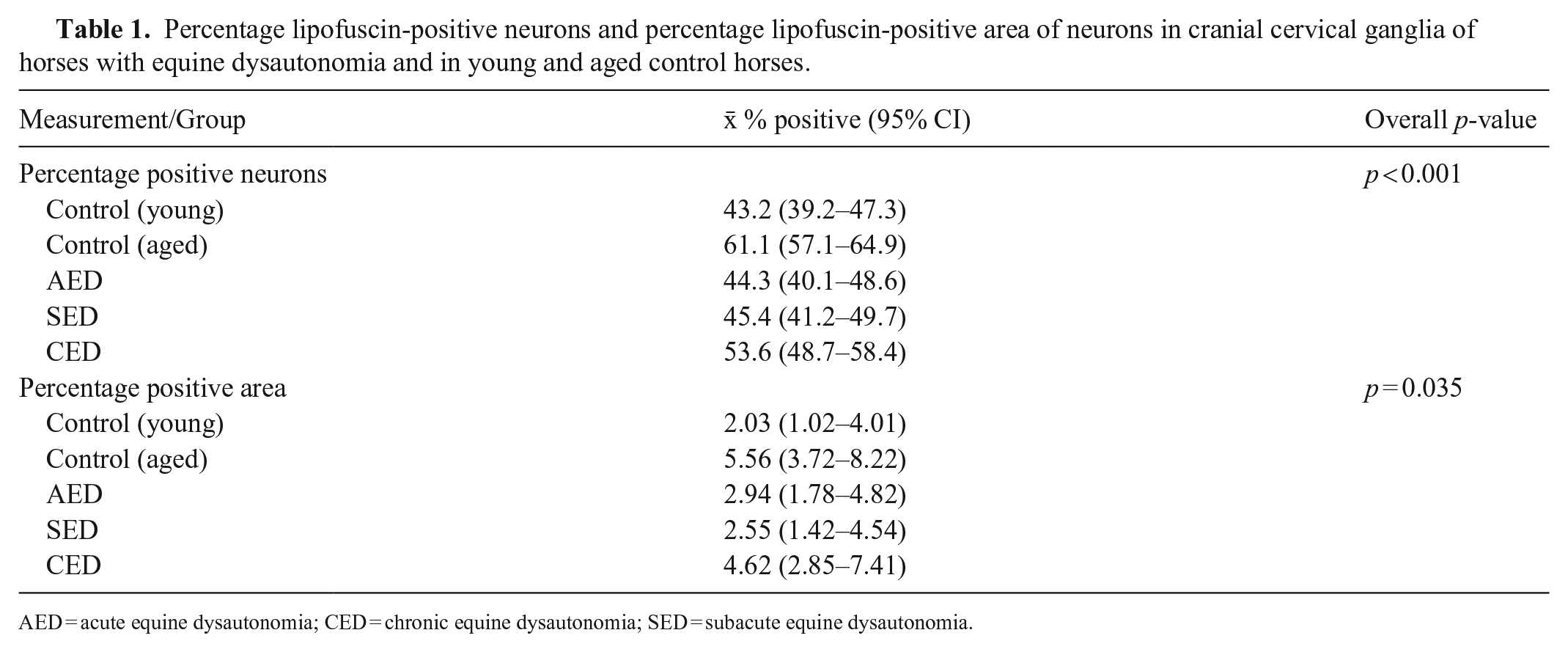
The percentage of neurons positive for lipofuscin differed between groups (p < 0.001; Fig. 3B), with lower percentages of positive neurons in AED and SED compared to aged controls (p < 0.001), a higher percentage in CED cases compared to both AED and young controls (p < 0.014), and higher percentages in the aged controls compared to young controls (p < 0.001; Table 1). However, there was considerable individual variation in percentages of positive neurons, with marked overlap between groups (Fig. 3B).
Percentage lipofuscin-positive neurons and percentage lipofuscin-positive area of neurons in cranial cervical ganglia of horses with equine dysautonomia and in young and aged control horses.
AED = acute equine dysautonomia; CED = chronic equine dysautonomia; SED = subacute equine dysautonomia.
Apparent overall differences in percentage positive area of lipofuscin in each group were observed (p = 0.035; Table 1); however, post-hoc Tukey pairwise analyses of differences did not indicate any differences between individual groups (p > 0.087, with again marked overlap between groups [Table 1]).
There was a positive relationship between the number of positive neurons and percentage area positive for lipofuscin in all groups combined (p < 0.001, OR = 117.6; Fig. 3C). This relationship was curvilinear, with an increasing steepness of the curve, and therefore relatively larger amounts of lipofuscin, as percentage of positive neurons increased.
Schmorl stain is not specific for lipofuscin but also stains other reducing substances, such as melanin, bile pigments, and hematoidin. PAS staining was also positive in a subset of cases, and although not specific, is expected for lipofuscin. The reason for positive PAS staining is uncertain but it is thought to be due to the formation of 1,2-glycols by hydroxylation of epoxides during early stages of lipofuscin formation. These can be converted to Schiff-positive dialdehydes during staining. It is therefore possible that additional inclusion of PAS staining could aid the assessment of temporal progression of lipofuscin accumulation with age. Overall, intraneuronal location, appearance on H&E sections, and positive staining with Schmorl reagent (and PAS in a subset of cases) made it highly likely that the material evaluated was lipofuscin. These methods are generally accepted as suitable for detecting lipofuscin, and Schmorl staining co-localizes with the more specific method of autofluorescence, which was unavailable.4,11 The possibility that some of the granules comprised neuromelanin cannot be completely discounted, although in any case, neuromelanin is considered to be melanized lipofuscin in equine CCG. 2
The mechanisms of lipofuscin accumulation and its effects are complex and profoundly affect cell metabolism. Accumulation of highly insoluble, reactive aggregates impairs autophagy and lysosomal degradation resulting in accumulation of lipofuscin in the cytoplasm. Autophagy is a protective mechanism, and its impairment causes excessive production of reactive oxygen species (ROS) from mitochondria and increased oxidative stress. 20 Oxidative stress of lysosomes adversely affects their function, and release of lysosomal contents into the cytoplasm results in apoptosis. Lipofuscin can also damage proteins, leading to their aggregation, and form a reservoir for metal ions, further leading to ROS generation and cell death. Lipofuscin is known to accumulate with age, particularly in long-lived, largely post-mitotic, neurons, especially the larger ones controlling movement; by contrast, proliferative cells dilute lipofuscin during cell division.3,19 In neurodegenerative diseases, lipofuscin aggregation increases with neuronal loss, 15 and lipofuscin-positive neurons have fewer dendritic processes and reduced neurotransmitter production. 10 However, the extent to which lipofuscin accumulation is a cause or consequence of such neurodegenerative changes remains uncertain. The increase in lipofuscin in CCG with age in horses with and without ED in our study is consistent with other species and other parts of the nervous system in horses 4 and confirms our second hypothesis.
The lower number of positive neurons in AED and SED compared to aged controls is likely to be age-related given that the AED and SED cases were younger than the aged controls and that ED preferentially affects younger horses. 13 However, it was of interest that the CED cases had a higher percentage of lipofuscin-positive neurons than AED and young control horses, despite the lack of age differences, confirming our first hypothesis. There were large individual differences; however, this trend suggests that intraneuronal lipofuscin accumulation in autonomic ganglia may be a feature of CED. The fact that this was not the case for AED and SED suggests that lipofuscin accumulation requires time, and duration of signs of ED of <7 d may be insufficient for the process to be evident by light microscopy, even though neuronal degeneration is already present at an early stage. 16 This could have also resulted from lipofuscin accumulation secondary to sublethal neuronal injury in CED cases, compared with the rapid neuronal degeneration and loss in AED and SED. It remains unclear whether lipofuscin is the cause or consequence of neurodegeneration in CED, but the presence of severe degeneration without a significant increase in lipofuscin in AED and SED is more consistent with it resulting from degeneration. Accumulation of lipofuscin, in addition to other metabolites,8,18,22 may play a role in impaired neuronal function in ED similar to that observed in neurodegenerative diseases in humans in which it is associated with neuronal loss or cellular alterations related to oxidative stress and lysosomal and mitochondrial dysfunction. 15 In addition, the possibility that there were biochemical changes in the lipofuscin that affected its visualization with Schmorl stain with age or ED cannot be completely discounted.
Our results for the percentage area of the neuron cytoplasm positive for lipofuscin were inconclusive despite the apparent positive relationship between this variable and percentage positive neurons. The latter suggests that as the number of neurons containing lipofuscin increases, either due to increasing age or ED, the amount of lipofuscin accumulating per neuron also increases.
The main limitations of our study are the small number of horses per group and that it was not possible to use the gold standard staining method of autofluorescence, even though the granules stained were very likely to be lipofuscin. A larger study on lipofuscin in ED using detection by autofluorescence is warranted, including the prevertebral and paravertebral ganglia, and enteric plexuses, all of which are associated with degenerative changes in ED. Such studies should consider the effects of aging on lipofuscin accumulation in autonomic neurons.
Supplemental Material
sj-pdf-1-vdi-10.1177_10406387241265715 – Supplemental material for Lipofuscin accumulates in ganglionic neurons in chronic equine dysautonomia
Supplemental material, sj-pdf-1-vdi-10.1177_10406387241265715 for Lipofuscin accumulates in ganglionic neurons in chronic equine dysautonomia by Lydia Tan Yi Shean, Elspeth M. Milne, Darren J. Shaw, Scott Maxwell and Jorge Del-Pozo in Journal of Veterinary Diagnostic Investigation
Footnotes
Acknowledgements
We thank the clinicians and owners for their care of the cases and for agreeing to postmortem examinations.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
Lydia Tan Yi Shean received an Equine Grass Sickness Fund John Gilmour Summer Research Scholarship.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
