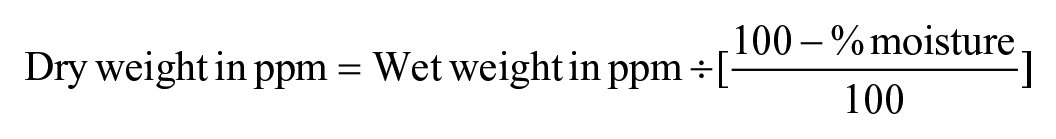Abstract
Copper is a trace element that plays an essential role in neurodevelopment and neurologic function. Acquired copper deficiency has a range of neurologic manifestations, with myelopathy being the most common association. We describe here the clinical, radiologic, histopathologic, and toxicologic findings of a rabbit with malnutrition, neurodegeneration, and suspected copper deficiency. A stray, adult female dwarf rabbit cross (Oryctolagus cuniculus) in poor body condition developed ataxia and pelvic limb weakness progressing to lateral recumbency and urine retention. The clinical findings suggested multifocal brainstem disease with right-sided central vestibular involvement; however, microscopic examination identified thoracic and lumbosacral spinal cord myelopathy. Differentials for the spinal cord changes included neurodegenerative disease, nutritional deficiency, neurotoxin, trauma to the lumbosacral region, and ischemia. Hepatic copper levels were suboptimal at 18 ppm dry weight (RI: 24–150 ppm dry weight). While speculative, copper-deficiency myelopathy is a treatable cause of non-compressive myelopathy that may occur in this species.
Keywords
Copper influences health and growth performance in rabbits. High and low levels of copper have pathologic sequelae. The leading consequence of copper deficiency in rabbits is microcytic, hypochromic anemia; however, inadequate dietary copper is also associated with achromotrichia, alopecia, dermatosis, cardiovascular disease, bone abnormalities, reduced growth rate, and decreased hemoglobin and hematocrit.12,13,23,26,29,31 Many species have neurologic manifestations of copper deficiency, including myelopathy and peripheral neuropathy, 36 but copper-associated myeloneuropathy has not been reported in rabbits.
Copper is a cofactor for biologic processes that are essential for proper CNS development. Deficiencies of copper during the perinatal period in unweaned lambs and goat kids result in enzootic ataxia or swayback disease, a progressive neuropathy of demyelination and microcavitation of the neuropil in the white matter of the spinal cord. Copper is a critical component of several metalloenzymes and proteins necessary for maintenance of the structure and function of the nervous system, including cytochrome c oxidase (mitochondrial respiratory chain), superoxide dismutase (antioxidant defense), ceruloplasmin (iron transport), tyrosinase (melanin synthesis), lysyl oxidase (cross-linking of collagen and elastin), dopamine β-monooxygenase (catecholamine synthesis), and peptidylglycine alpha-amidating monooxygenase (maturation of neurotransmitters and neuropeptides). 10 Acquired copper-deficiency myeloneuropathy has a predilection for the dorsal columns and corticospinal tracts. 17 The clinical and radiologic findings of copper-deficiency myelopathy (CDM) in humans are indistinguishable from subacute combined degeneration (SCD) due to vitamin B12 (cobalamin) deficiency. 18
Extrahepatic copper distribution is tightly regulated by the liver, which uptakes copper absorbed into the portal circulation by the stomach and small intestine and facilitates incorporation into copper-requiring proteins for subsequent secretion into the blood. Several factors influence copper absorption. In humans, copper absorption is impaired by gastrointestinal surgery, malabsorption syndromes, and elevated dietary intake of divalent cations (zinc, iron, tin, and molybdenum) and carbohydrates. 33 Zinc, molybdenum, and sulfur have a major influence on copper absorption and storage in ruminants. 6 There are mixed reports on whether high dietary phytic acid affects copper absorption, which may be secondary to the effects of phytates on zinc absorption or precipitation of phytate–copper complexes.3,21 In rabbits, dietary ascorbic acid can accentuate copper-deficiency signs by further reducing cytochrome c oxidase function due to reduced iron absorption. Iron is essential for the coordinated function of copper ions and heme groups that form cytochrome c oxidase (complex IV or translocase), the final enzyme in the respiratory electron transport chain. 12 Excess molybdenum induces secondary copper deficiency by reducing copper bioavailability and increasing excretion. 1 The normal concentration of hepatic copper for rabbits is 8–50 ppm wet weight (WW) or 24–150 ppm dry weight (DW). 25
Copper is found in a variety of cells and tissues with the highest concentration in the liver and brain. 20 The first indications of CDM in humans are gait abnormalities and paresthesia from dorsal column dysfunction. 14 Spasticity and urinary signs occur less frequently. Less well-established human conditions associated with copper deficiency include isolated peripheral neuropathy,9,35 motor neuron disease, 34 CNS demyelination,24,30 myopathy, 19 and optic neuritis.9,30
A stray, adult female dwarf rabbit cross (Oryctolagus cuniculus) in poor body condition was surrendered to a humane society with no apparent neurologic signs. Body condition did not improve despite being syringe fed 4–6 times per day (Critical Care Herbivore, Oxbow; Appetite Restore, Sherwood), as well as having access to greens, timothy hay, and alfalfa hay. In humans, there is documentation of hypercatabolic responses after spinal cord injuries that result in loss of lean body mass, which can compound malnutrition caused by inadequate intake of nutrients. 5 No supplemental copper or vitamin B12 were provided, to our knowledge. The animal developed ataxia and pelvic limb weakness within ~3–4 wk of surrender. Neurologic signs progressed over the next 2 mo to marked cervical neck extension and thoracic limb extensor rigidity, culminating in lateral recumbency and urine retention.
On presentation, the rabbit was obtunded and in lateral recumbency. It was assessed to have a body condition score of 2 of 9 with diffuse muscle atrophy. The right ocular conjunctiva was erythematous with notable blepharospasm. Both globes were rotated dorsally. Intermittent twitching or spasms were seen at the base of both ears, but otoscopic examination revealed no abnormalities. Incisor malocclusion was noted. The pelvic limbs had grade I of VI pododermatitis on the plantar surface of the hocks. On neurologic assessment, the rabbit had non-ambulatory tetraparesis. When the rabbit was placed in an upright position, the animal became opisthotonic with marked cervical neck and thoracic limb extensor rigidity. The head tilted to the right with appropriate physiologic nystagmus and occasional non-positional rotary nystagmus. Horner syndrome was suspected based on the finding of right miosis and partial ptosis. Cranial nerve, dazzle, and pupillary light reflexes were intact. Conscious proprioception was lacking in all limbs, but withdrawal response was present. The left patellar reflex was decreased. The history, physical examination, and neurologic examination findings suggested progressive neurologic disease involving the brainstem and/or the cerebellar region. Differentials for the neurologic signs included an infectious process, inflammatory disease, vascular event, or neoplasia. Toxicant exposure was considered, but lateralization of the neurologic signs is unusual in toxic and metabolic myelopathies.
The CBC and biochemistry panel performed by the referring veterinarian were unremarkable. Serologic tests were negative for IgM and IgG antibodies to Encephalitozoon cuniculi antibodies, and an RT-PCR assay (Research Associates Laboratory) did not detect Sarcocystis neurona, Baylisascaris procyonis, Toxoplasma gondii, or herpesvirus. The rabbit underwent cardiorespiratory arrest during general anesthesia for MRI scan and CSF tap. An abbreviated postmortem MRI was performed, with only T1, T2, and FLAIR images acquired. Images from the postmortem MRI showed T2 and FLAIR hyperintensity around the mesencephalic aqueduct and focally from the level of the left caudal peduncle of the cerebellum along the dorsal medulla oblongata (Fig. 1). Cytology of a CSF sample collected from the cisterna magna postmortem revealed lymphoplasmacytic inflammation only.
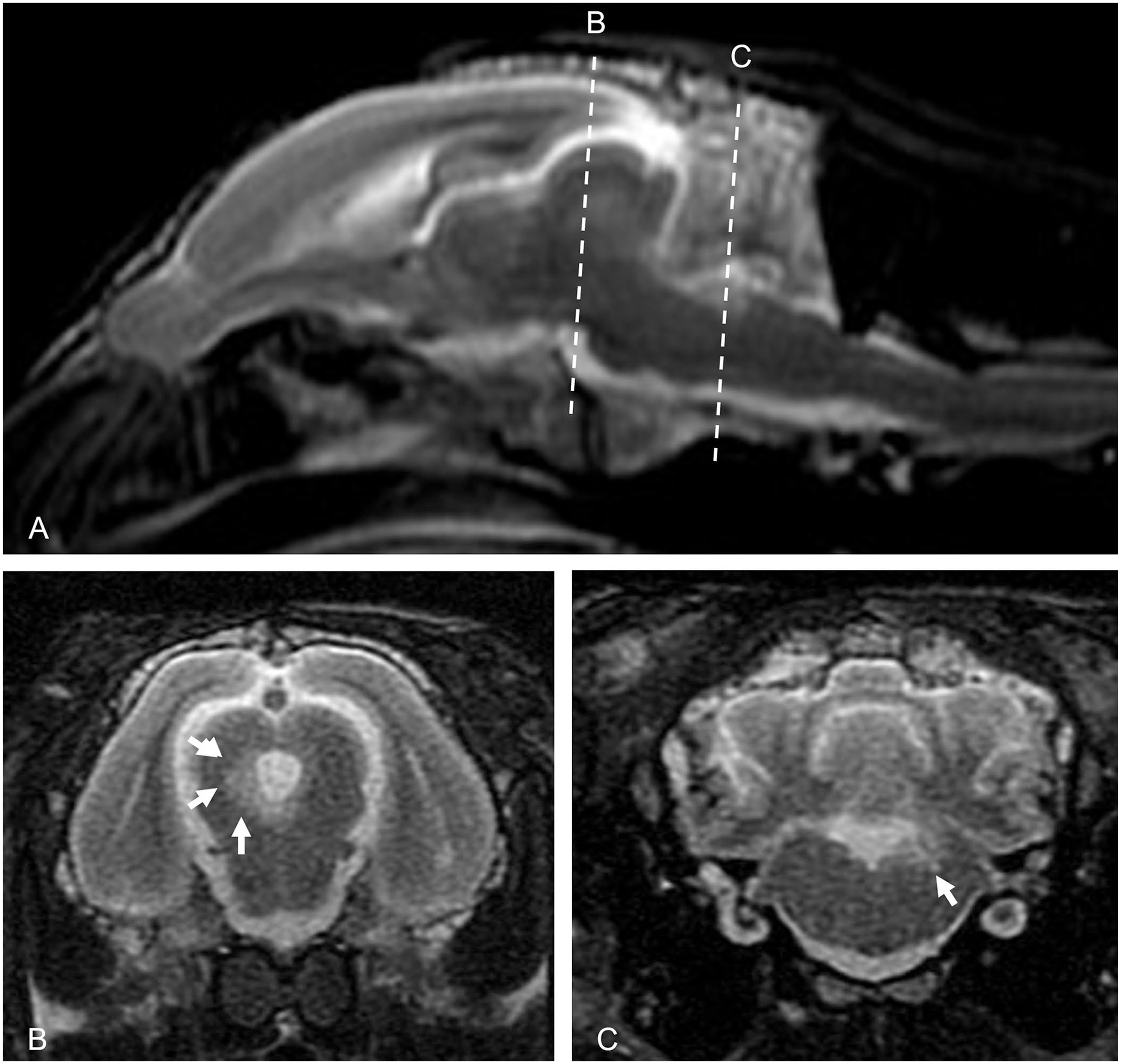
Sagittal and transverse plane T2 weighted MRI images of a rabbit.
A postmortem examination was performed. The brain and spinal cord appeared grossly normal. Histologically, severe, segmental myelin sheath swelling, axonal degeneration, gliosis, and demyelination with digestion chambers were present, predominantly within the dorsal funiculi of the caudal cervical and proximal thoracic spinal cord (Fig. 2). The asymmetric lumbosacral spinal cord changes were centered on motor pathways. White matter changes were most severe in the lateral and medial corticospinal region. Neuronal degeneration with chromatolysis and gliosis of motor columns predominated in lamina VII, VIII, and IX of the gray matter. A glial nodule was observed in the brainstem. Incidentally, the animal had atrophy or hypoplasia of the right kidney. The localization of spinal cord lesions within the motor pathways triggered the submission of liver for heavy metal analysis. For heavy metal analysis, 1 g of liver tissue was analyzed by inductively coupled plasma–optical emission spectrometry following the laboratory’s standard operating procedure. The reporting limits in DW, ppm, were: arsenic 25; cadmium 7.5; copper 7.5; iron 25; lead 25; manganese 2.5; mercury 25; molybdenum 10; zinc 7.5. The copper was found to be low at 18 ppm DW (RI: 24–150 ppm DW). Arsenic, cadmium, lead, mercury, and molybdenum were not detected. The liver contained 1,500 ppm DW of iron, 11 ppm DW of manganese, and 110 ppm DW of zinc. No other postmortem testing was conducted, but in retrospect, vitamin B12 testing should have been considered.
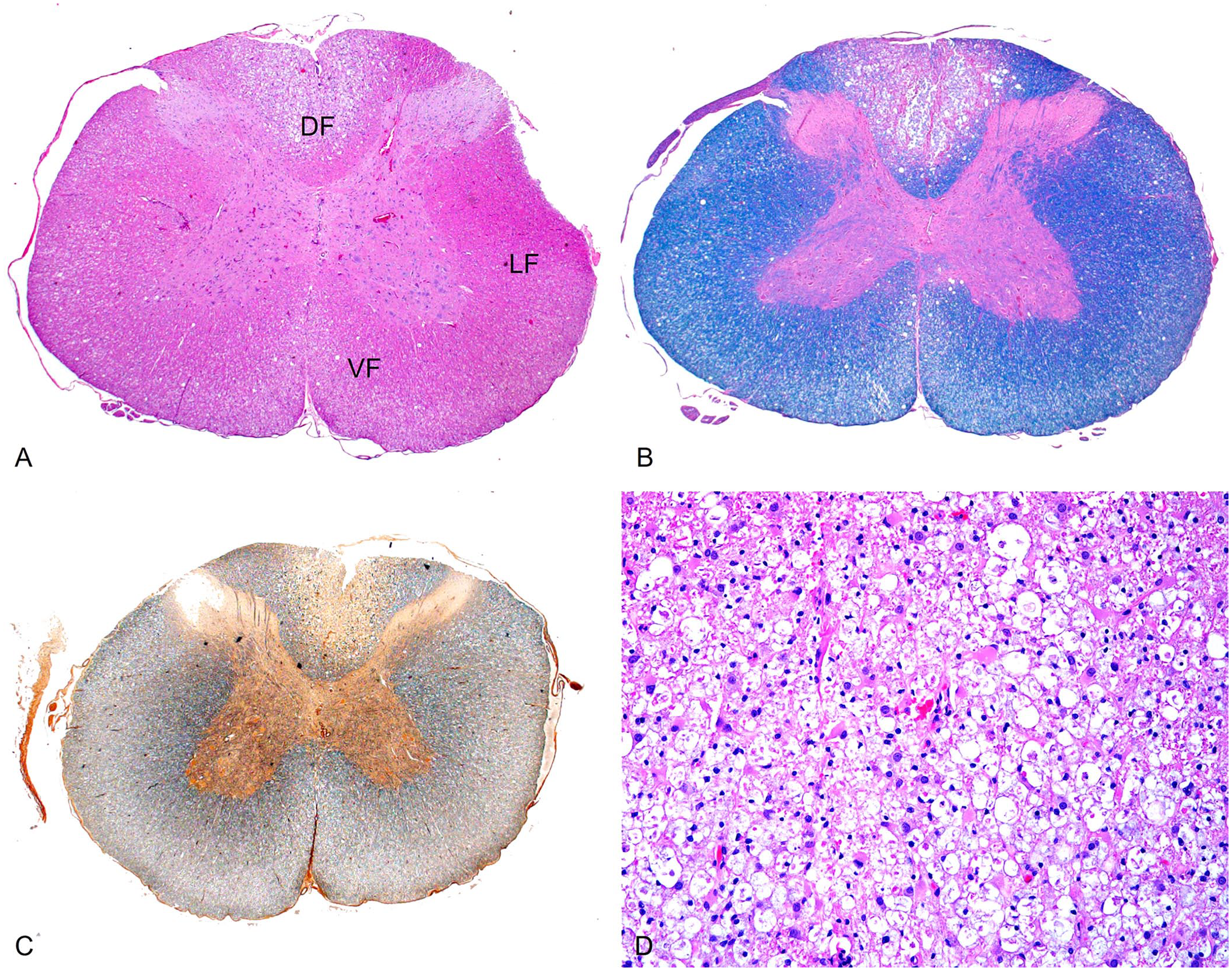
Demyelination of the dorsal thoracic spinal cord in a rabbit.
We retrieved no cases of myelopathy in rabbits with low copper levels in a search of Google, PubMed, CAB Direct, Web of Science, and Scopus, using search terms “rabbit, myelopathy, neurodegeneration, and copper,” suggesting that this condition has not been reported in rabbits. The clinical presentation of this rabbit was that of a progressive syndrome with histologic evidence of myelopathy predominantly of the dorsal and dorsolateral funiculi of the thoracic and lumbosacral spinal cord. Differentials for the spinal cord changes include neurodegenerative disease, nutritional deficiency, toxicant exposure, trauma to the lumbosacral region, and ischemia. The character of the spinal cord lesions was akin to those seen in acquired copper-deficiency disorders, such as enzootic ataxia in sheep (nutritional neuroaxonal dystrophy), or CDM (human swayback) and SCD due to vitamin B12 deficiency in humans. Lesions with enzootic ataxia predominantly affect the dorsolateral and ventromedial white matter,2,28 whereas human swayback and SCD are known to affect dorsal columns. 8
Neuroimaging studies of copper deficiency in humans had hyperintense T2 MRI signal within lacunar regions, including the neuropil of the thalamus, pons, and medulla oblongata. 16 T2 and FLAIR hyperintensity for our case were in the same regions (around the mesocephalic aqueduct and the left caudal peduncle of the cerebellum along the dorsal medulla oblongata). Our findings indicate that there was increased fluid in these regions, which could be edema from peri- and/or postmortem changes, trans-ventricular fluid in the case of the mesencephalic aqueduct, associated with infarction, or a sequala to copper deficiency.
The copper concentration in the liver of this rabbit was suboptimal (18 ppm DW; RI: 24–150 ppm DW). The liver minerals were calculated based on a dry matter basis due to the small fresh sample available for toxicologic analysis. Conventionally, wet weight RI can be multiplied by 3 when you assume a moisture content of 70% for a given tissue to get a crude estimate of the dry weight (based on the calculation below). A conversion factor of 3.5–4.0 has been proposed, 25 but a conversion factor of 3 seems more accurate based on the average water weight of 71% reported for 19 rabbit livers (68.7–76.0% in non-feverish animals). 11 The measured hepatic copper of 18 ppm DW (6 ppm WW based on the conversion factor of 3) is within the marginal range (4.0–10.0 ppm WW) for hepatic copper in rabbits. This is below the adequate range of 8.0–50.0 ppm WW. 25
The clinical and radiologic findings of CDM in humans are indistinguishable from SCD due to vitamin B12 deficiency; however, confirmed cases of this disease in other animals are scarce.4,27 B-complex vitamins are synthesized by bacteria in the hindgut of rabbits and consumed during coprophagy, which provides 42% more vitamin B12 than would otherwise be available. 15 Primary vitamin B12 deficiencies in rabbits are rare due to the large amounts synthesized by the fecal flora, 32 but levels were not assessed in our case. Rather, an infectious or inflammatory disease was considered more likely given the neurologic examination findings. Serologic tests for E. cuniculi, S. neurona, B. procyonis, T. gondii, and herpesvirus were negative. This is supported by the postmortem CSF analysis that reported lymphoplasmacytic inflammation with no signs of infectious agents or neoplastic cells. Lymphoplasmacytic inflammation in the CSF is a nonspecific finding that can be caused by a broad number of underlying etiologies including, but not limited to, infection, trauma, inflammation, ischemia, or neoplasia, especially in lagomorphs in which we know very little about CSF characteristics in specific disease states.
CDM is a treatable cause of non-compressive myelopathy. 14 Interestingly, the CBC and biochemistry profile for this animal were within normal limits. The literature on copper deficiency in rabbits describes anemia as a common finding; however, in humans, the neurologic syndrome associated with hypocupremia can be present prior to or without the hematologic manifestations. 16 Supplementation allows prompt normalization of hematologic disturbances and may prevent further neurologic deterioration. It is possible that the provided diet, which contained the minimal nutritional requirements of copper and vitamin B for rabbits, helped normalize the hematologic findings given that the copper concentrations are considered marginal and not deficient. Unfortunately for this animal, neurologic recovery typically takes time, is rarely complete, and preferentially involves sensory systems. 7 Full recovery, even with copper supplementation in this case, was unlikely given the severity of neurologic clinical signs and histologic lesions.
Known causes of acquired copper deficiency include excessive zinc absorption and malabsorption; however, often the cause is unclear, as was evident with our case. Other detected liver minerals were within acceptable ranges for this species, including cadmium, iron, molybdenum, and zinc, which are known to affect copper absorption and/or availability. Speculatively, decreased copper levels may be due to consumption of copper-deficient forage given that this animal was a stray in Northern California where forage concentrations of copper vary from low (<5 ppm) to normal (5–15 ppm). 22 Progression despite supplementation after receiving nutritional support could be due to insufficient nutrient intake and the need for additional supplementation. Since vitamin B12 levels were not measured, it is unknown whether the rabbit was deficient in both vitamin B12 and copper given that these deficiencies can coexist and have similar clinical features and neuroimaging findings.
Our case report describes the clinical, radiologic, histopathologic, and toxicologic findings of a rabbit with malnutrition, neurodegeneration, suspected copper deficiency, and unknown vitamin B12 status. In many species, copper deficiency manifests clinically with hematologic and neurologic signs. In copper-deficient regions, copper and vitamin B12 level assessments may allow for early supplementation, potentially limiting neurologic deterioration.
Footnotes
Acknowledgements
We thank Dr. Noa Safra, the clinical pathology resident who assisted in the interpretation of the postmortem CSF, and the William R. Pritchard Veterinary Medical Teaching Hospital (University of California, Davis) neurology service for conducting the premortem neurologic examination and collecting the postmortem cisternal CSF.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors declare that they received no financial support for their research and/or authorship of this article.