Abstract
American pikas (Ochotona princeps) are small lagomorphs that live in mountainous talus areas of western North America. Studies on the histopathology of American pikas are limited. We summarize here the clinical histories, and gross and histologic findings of 12 American pikas, including 9 captive (wild-caught) and 3 wild animals. Death was often attributed to stress (transport, handling, anesthesia) with few-to-no premonitory clinical signs. Infection was the cause of death in 2 cases: 1 had bacterial pyogranulomatous dermatitis, cellulitis, and lymphadenitis with sepsis; the other case had oomycete-induced necrotizing colitis. Incidental parasitic infections included sarcocystosis, nematodosis (oxyurids), and ectoparasitism. Most animals with adequate nutritional status had periportal hepatic lipidosis; this finding was absent in all animals with adipose atrophy, and it is possible that periportal hepatic lipidosis is non-pathologic in American pikas. Three cases had myocardial necrosis that was considered the cause of death; the cause of necrosis was not determined, but it may have been caused by stress or vitamin E–selenium deficiency. Esophageal hyperkeratosis was noted in animals with a history of anorexia and negative energy balance; accumulation of esophageal keratin can result from lack of mucosal abrasion by ingesta. Several histologic findings that are likely normal in American pikas include splenic extramedullary hematopoiesis, thymic tissue in adults, and Clostridium sp. in the enteric lumen.
American pikas (Ochotona princeps), also known as rock rabbits or whistle hares, are 1 of 30 species of pikas within the Ochotonidae family of the Lagomorpha order. They are relatively small lagomorphs with adults weighing 121–176 g. Their egg-shaped silhouette is composed of short non-cursorial digitigrade legs that are all of similar length terminating in relatively short hind feet and furred soles, oval ears with fur on both surfaces, and a short inapparent tail. Males and females lack visible external genitalia, with the reproductive and excretory systems joining terminally in a cloaca-like structure.12,40 The skull has a rounded profile with a broad, flat interorbital region, and adults >1-y-old have notable sexual dimorphism in skull size. They have 26 teeth with a dentition pattern of 2(i 2/1, c 0/0, p 3/2, m 2/3) as opposed to domestic rabbits, which have 28 teeth with 2(i 2/1, c 0/0, p 3/2, m 3/3). 40 Their normal body temperature is high for a mammal (40.1°C [104.2°F]). 12
American pikas are distributed in a discontinuous pattern across western North America from central British Columbia to central New Mexico, often in mountainous talus areas (particularly with >50% rocky debris). They have a predilection for higher elevations at the southern end of their range and lower elevations at the northern end of their range due to intolerance of high environmental temperatures in most populations. They are primarily diurnal and vigorously defend relatively large territories of 410–709 m2. Pikas are facultatively monogamous with their neighbors of the opposite sex, producing up to 2 litters of ~3 kits per year, and they reach a maximum age of 7-y-old in the wild. Juveniles do not travel far to establish their own territories due to low success rates with long-distance dispersal, and they frequently inhabit territories adjacent to their natal home range. American pikas eat a flexible and diverse variety of meadow plants and engage in “haying” during the summer months following spring breeding season; they gather plant matter, dry it on talus outcroppings, and store it for partial sustenance during the winter (at which time they are also actively foraging via snow tunnels). 40
Climate change since the last ice age and more rapid climate change within the last century has contributed to the isolation of American pika populations. Within the fossil record, pikas inhabited a contiguous swath of the North American west, to include sea-level habitats. Drying and warming of their environment has led to upslope migration of these populations and subsequent genetic adaptation to high altitude. 38 In major cordilleras such as the Rocky, Sierra Nevada, and Cascade Mountain ranges, a large amount of territory is viable, and the populations are able to mix via rare long-range dispersals of juveniles. Extirpations (local population extinctions) have been documented in areas with discontinuous mountain ranges (precluding repopulation) that contain smaller isolated populations, such as the Great Basin, and in areas that have experienced prolonged droughts.6,12,15,39
There are several reports on the pathology of Afghan pikas (Ochotona rufescens rufescens), which were bred and used as laboratory animals in the 1970s and 1980s.25,26,28,37 However, little is known about the pathology of American pikas. 12 This is likely due to infrequent access to these animals; no Association of Zoos & Aquariums–accredited zoos currently keep pikas in captivity (reference: Rebecca Young, Taxon Advisory Group chair, written communication, Feb 2023). Studies on the histology and histopathology of American pikas are scarce. There are 2 histologic studies of the reproductive tract of the American pika, and no references on other organ systems (PubMed search 2023 Mar 9).13,22 There are also studies on relative size or weight of body parts in American pikas, but none with histology.31,51 Two pathology studies have been reported, including a single case of adiaspiromycosis in 1 of 240 American pikas in Alberta, Canada, due to infection with Emmonsia crescens causing bilateral pulmonary granulomas, and 6 cases of botfly myiasis in Oregon and Montana due to infection with rodent-associated Cuterebra spp. larvae during the summer.2,3,42 Other studies on the parasites of American pikas are limited to identification of parasites without investigation into pathologic effects on the host.8,10,17 Although little is known about viral diseases of American pikas, they are suspected to be susceptible to rabbit hemorrhagic disease virus 2 (genus Lagovirus), a fatal caliciviral disease of lagomorphs. 34 Plateau pikas (syn. black-lipped pika; Ochotona curzoniae) are known carriers of influenza viruses H7N2, H9N2, and H5N1, and an alphacoronavirus termed “plateau pika coronavirus,” although the susceptibility of American pikas to these viruses is unknown. 52 We retrieved no cases of metabolic conditions or toxins affecting American pikas in a search of PubMed, using the search term “American pika” and “Ochotona princeps.” Hair selenium concentrations were not related to body condition index in American pikas of the northern Rocky Mountains. 32
Due to the paucity of literature on the histology and histopathology of American pikas, we aim to provide insight into the health and disease of these rarely studied animals via our multi-institutional, retrospective study. Our objective is to describe clinical histories, gross findings, and histologic lesions in American pikas.
Materials and methods
We searched the pathology archives of the Smithsonian’s National Zoo and Conservation Biology Institute (NZCBI; Washington, DC, USA) and the U.S. Geological Survey’s National Wildlife Health Center (NWHC; Madison, WI, USA) for cases of American pikas. Formalin-fixed, paraffin-embedded (FFPE) blocks from 3 NWHC cases and 9 NZCBI cases were pulled, and duplicate H&E slides were made from 5-µm thick sections at the Joint Pathology Center Laboratory (within Walter Reed National Military Medical Center, Bethesda, MD, USA). 3 H&E histology from all cases was reviewed. Additional histochemical special stains, including Brown and Brenn Gram stain, Congo red, Gomori methenamine silver (GMS), periodic acid–Schiff (PAS), von Kossa, and Ziehl–Neelsen (ZN), were performed when diagnostically relevant (e.g., to screen for infection, confirm amyloid, or confirm mineral). PCR assays using panfungal primers targeting a portion of the 18S ribosomal gene were performed on FFPE scrolls on one case in which a hyphal organism was identified histologically (PCR was commercially available and performed by the Washington Animal Disease Diagnostic Laboratory, Pullman, WA, USA). Case histories and gross necropsy findings from all cases were collated.
Results
We included 12 pikas in our study, including 7 females and 5 males ranging from 6 mo to “adult” (exact age was unspecified in 7 animals listed as “adults”; Table 1). Body weights at autopsy for 6-mo-old animals had a mean of 109 g and median of 103 g (range: 94–130 g), the two 12- and 13-mo-old animals had a mean and median of 159 g (134 and 184 g), and animals listed as “adult” had a mean of 153 g and median of 154 g (range: 125–176 g). Nine animals were captive and 3 were wildlife-trapped for tagging and monitoring. The most common history was found dead during or within 24 h of stress (n = 5); reported stressors included anesthesia (n = 2), capture (n = 1), transport (n = 1), cage cleaning (n = 1), and lactation (n = 1). The next most common history was found dead without premonitory signs or evidence of an environmental stressor (n = 3), anorexia and/or lethargy for at least 1 d (n = 3), and cagemate fighting (n = 1).
Signalment, history, and cause of death in 12 American pikas.
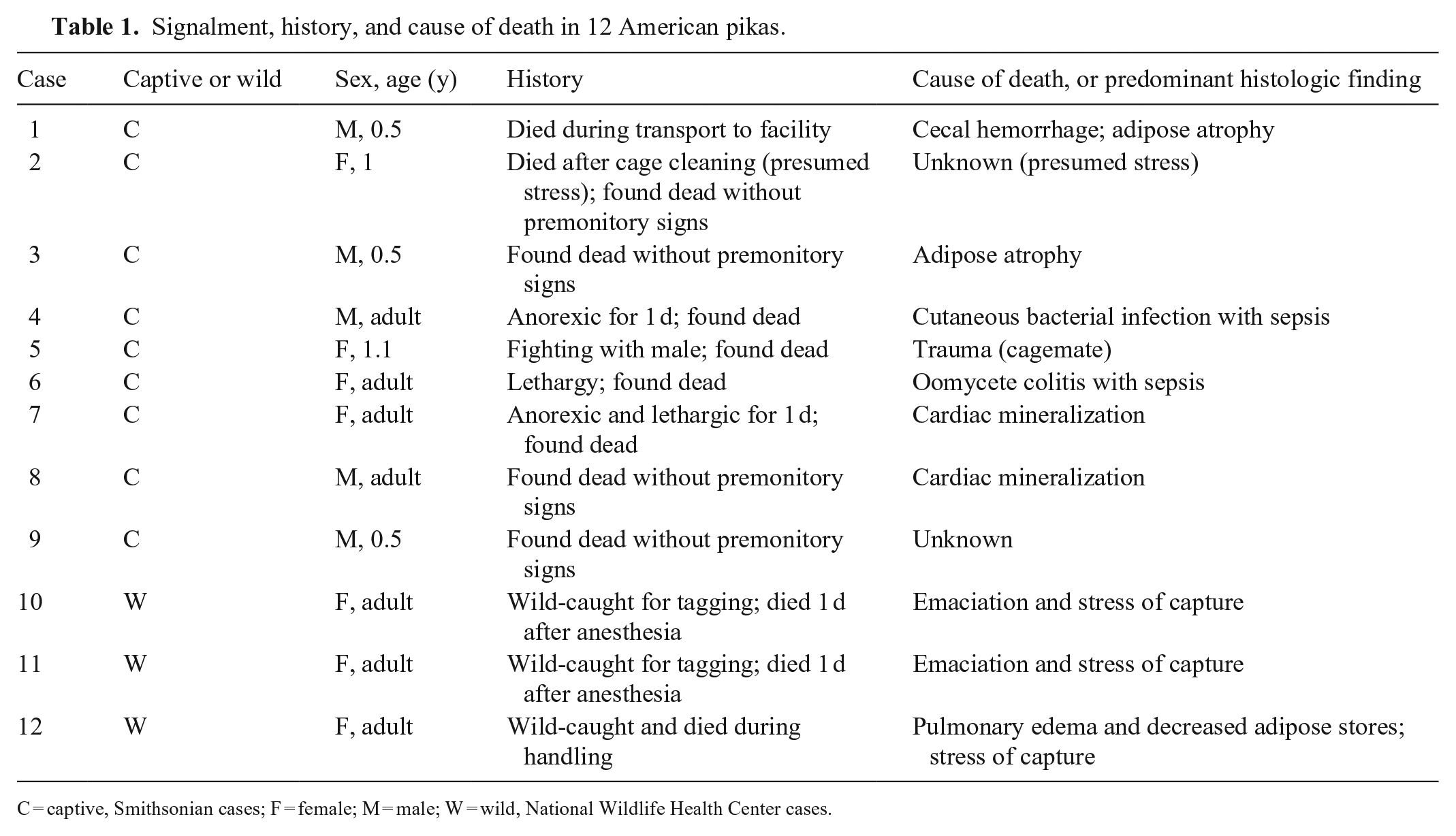
C = captive, Smithsonian cases; F = female; M = male; W = wild, National Wildlife Health Center cases.
At autopsy, 6 animals had decreased adipose tissue (cases 1, 3, 4, 10–12; histologic correlate: adipose atrophy), 4 animals had adequate adipose tissue (cases 2, 6, 7, 9), and 2 animals had excessive adipose tissue (cases 5, 8). Additional gross findings included hepatic enhanced reticular pattern (cases 6–8; histologic correlate: periportal hepatic lipidosis), ectoparasitism with fleas (cases 10, 11) and ticks (case11; possible histologic correlate: epidermitis), hyperemic cecum (case 1; histologic correlate: cecal hemorrhage), multifocal gastric reddening (case 9; histologic correlate: gastric serositis), cutaneous lacerations (case 5; lacerated tissue not sampled for histology), small tan spherical masses in the subcutis and skeletal muscle (case 12; histologic correlate: sarcocystosis), blood filling the tracheal lumen and nares (case 12; no identified histologic correlates), and purulent draining tracts in the stifle, skeletal muscle, and skin with pulmonary nodules (case 4; histologic correlate: multisystemic pyogranulomatous inflammation with gram-positive rods; bacterial culture was not performed). The prosector noted that lymph nodes were difficult to identify in case 10. Organ weights were provided only for the liver and only in 2 cases (10, 11), both of which were emaciated. The liver weighed 5.65 g (4.52% of body weight) and 6.89 g (body weight not recorded) in cases 10 and 11, respectively.
Cardiovascular system
Lesions in the heart in 3 of 11 cases included acute, moderate-to-severe myocardial necrosis with fragmentation of myofibers, mild edema, cellular swelling, and cellular fragmentation; 2 of 3 cases had mineralization of the sarcoplasm confirmed by von Kossa stain (Table 2; Fig. 1). In all cases, the right ventricular free wall was most severely affected, followed by the interventricular septum and left ventricular free wall; the atria were unaffected. Lesions were not evident in blood vessels in any of the 12 cases.
Histologic lesions in 12 American pikas.
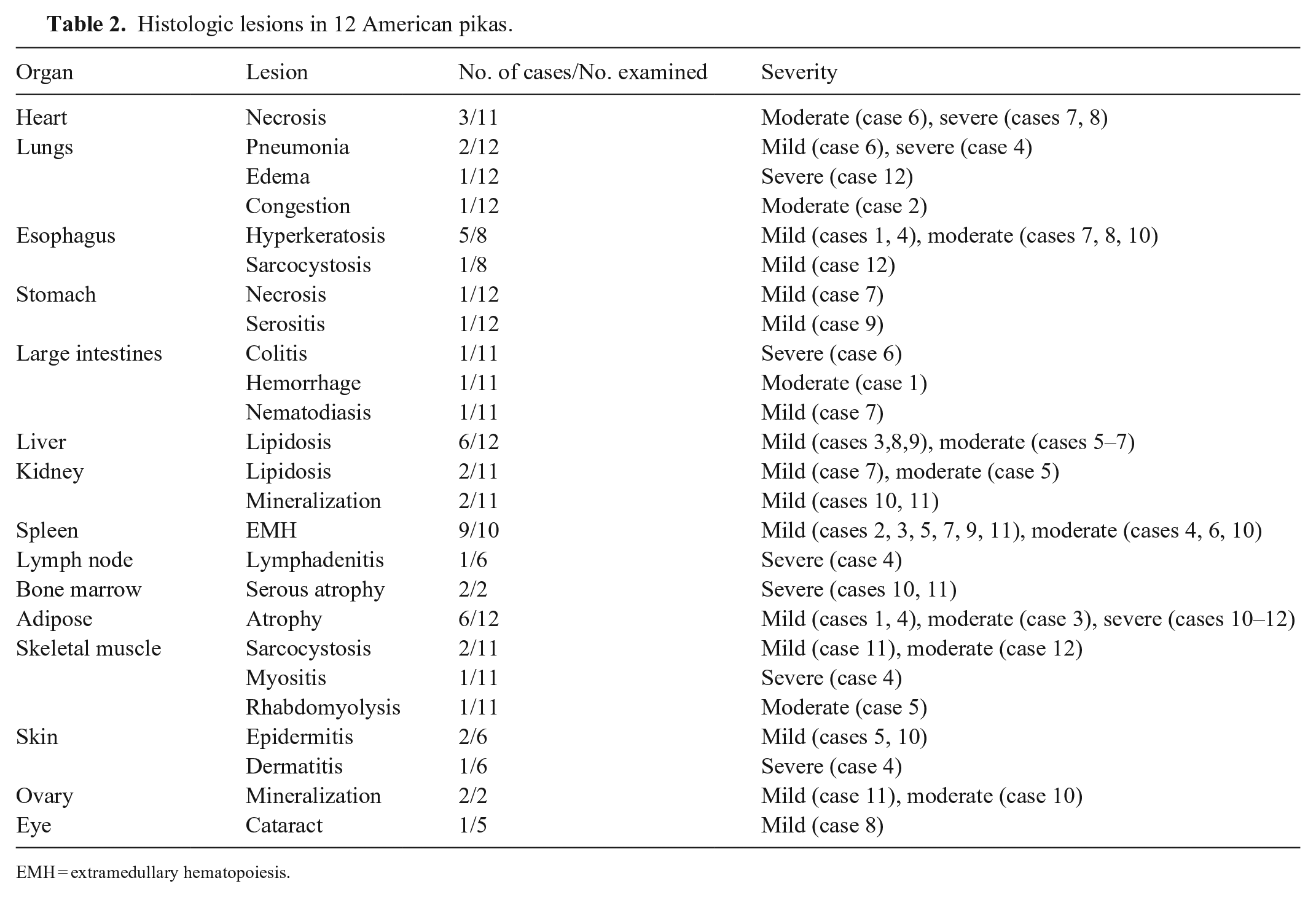
EMH = extramedullary hematopoiesis.
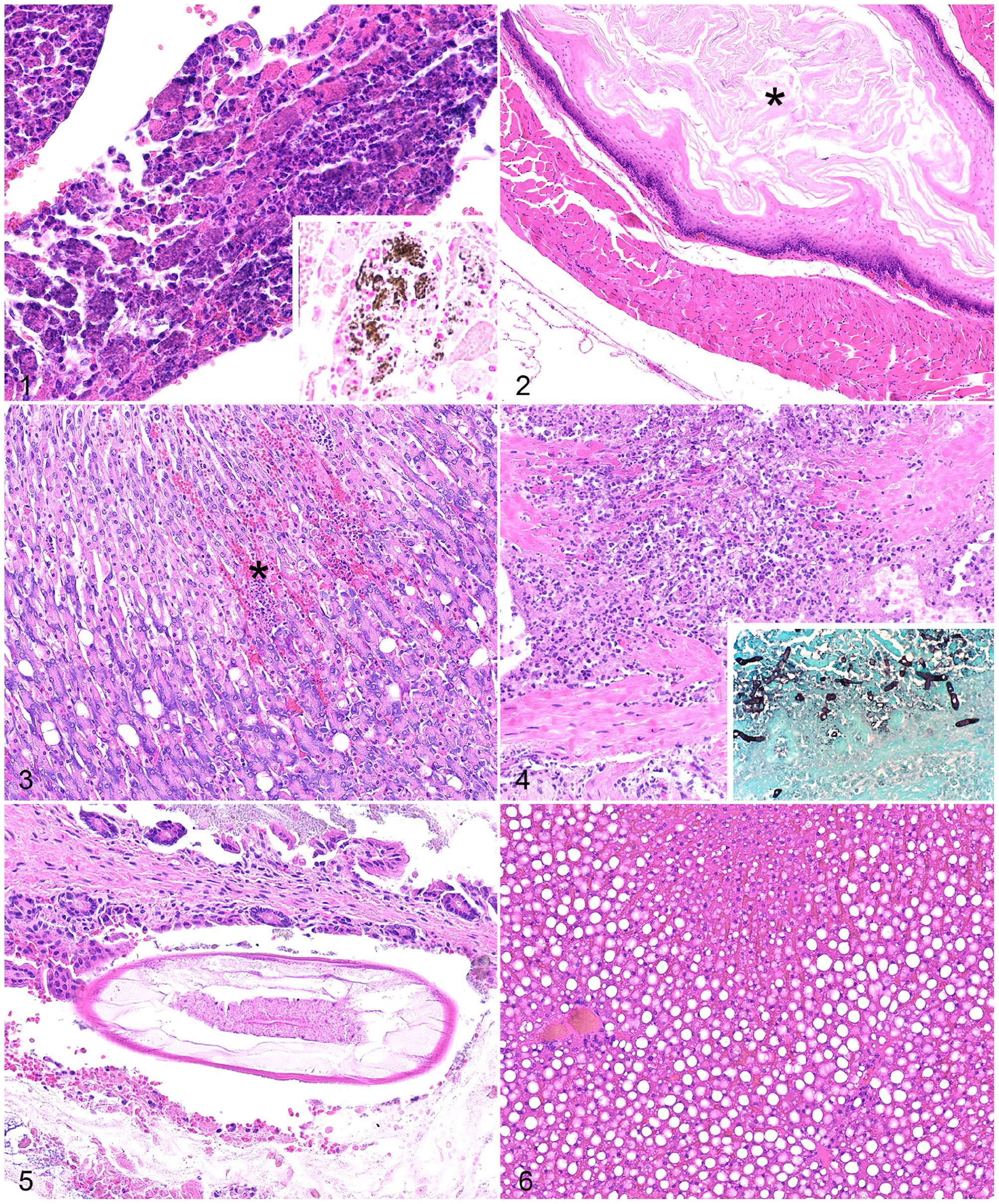
Histologic lesions in American pikas (Ochotona princeps).
Respiratory system
The lungs had lesions in 4 of 12 cases. Case 4 had pyogranulomatous pneumonia characterized by multifocal-to-coalescing, large regions of lytic necrosis surrounded by a few fragmented neutrophils and further surrounded by abundant macrophages, fewer fibroblasts, lymphocytes, and plasma cells. Inflammation was centered on colonies of bacilli. Gram stain revealed numerous filamentous gram-positive rods; molecular testing was not performed. Similar bacteria and inflammation were seen in the lymph nodes, stifle, skeletal muscle, and skin of case 4, and the pulmonary lesion was considered a septic embolus. Case 6 had a small, discrete nodule of pneumonia with moderate numbers of neutrophils and necrotic cellular debris. This animal also had severe ulcerative colitis, and the pulmonary lesion was considered a septic embolus. Case 2 had moderate diffuse congestion of the large and small blood vessels. Case 12 had severe pulmonary edema. Lesions were not evident in the trachea in any of the 12 cases.
Alimentary system
The esophagus had lesions in 6 of 8 cases. In 5 cases, the mucosa was overlain by a small-to-moderate amount of orthokeratotic (n = 2), parakeratotic (n = 2), or combination (n = 1) keratin, consistent with hyperkeratosis (Fig. 2); this was interpreted as recent inanition and was present in both animals with a history of anorexia. Case 12 had rare schizonts within skeletal myocytes of the esophagus that formed cysts up to 170 µm in greatest dimension. Schizonts were composed of dozens of 1–2-µm, elongate zoites that formed tight clusters within parasitophorous vacuoles in the sarcoplasm, consistent with Sarcocystis sp.
The stomach had lesions in 2 of 12 cases. Case 7 had foci of hemorrhage in the mid and superficial gastric mucosa that were associated with small regions of lytic necrosis of mucosal epithelial cells (Fig. 3). In case 9, the serosa was expanded by a small nodule of inflammatory cells composed predominantly of macrophages with fewer plasma cells and lymphocytes. Gram, ZN, and GMS stains did not reveal infectious agents in this lesion. The Gram stain revealed abundant gram-positive, spore-forming bacilli in the lumen of the intestines.
The large intestines had lesions in 3 of 11 cases. In case 6, the colon had segmental mucosal lytic necrosis with infiltration by numerous fragmented neutrophils and hyphae that extended into the muscularis. The hyphae were 6–10 µm diameter and had non-parallel walls, irregular branching, and lacked septa. The mucosa in this region was replaced by hemorrhage, necrotic cellular debris, fibrin, neutrophils, and hyphae. GMS and PAS stains confirmed that the hyphal walls were argyrophilic and PAS-positive (Fig. 4). PCR was performed, and a portion of the 18S ribosomal RNA gene was amplified using universal oomycete primers. Sequencing matched the oomycete Leptolegnia caudata (100% sequence identity to GenBank AJ238659). In case 7, cross-sections of nematodes were present in the colonic lumen without associated colitis. Nematodes were 50–130 µm in diameter with large lateral cords, a pseudocoelom, platymyarian musculature with pronounced cytoplasm, an enteric tract with uninucleate, columnar-to-cuboidal cells, and a triradiate esophagus (Fig. 5). In case 1, the cecal mucosa had moderate perivascular hemorrhage in the lamina propria and submucosa and severe mucosal congestion.
The small intestines, pancreas, tongue, and salivary glands were examined in 11, 8, 6, and 6 cases, respectively, and lesions were not evident. Abundant spore-forming, gram-negative bacilli were present in the small intestinal lumen of case 9, consistent with Clostridium sp.
Hepatobiliary system
The liver had lesions in 6 of 12 cases. Five cases had mild-to-moderate periportal and/or midzonal microvesicular-to-macrovesicular hepatic lipidosis (Fig. 6). All cases with periportal hepatic lipidosis (cases 5–9) had adequate or excessive adipose tissue at autopsy, and cases 5 and 7 also had renal tubular lipidosis. One case had mild centrilobular hepatic lipidosis (case 3), and this animal had decreased adipose tissue at autopsy. Lesions were not evident in the gall bladders in the 3 cases examined.
Urinary system
The kidneys had lesions in 4 of 11 cases. Cases 5 and 7 had tubular lipidosis characterized by expansion of the cytoplasm in proximal convoluted tubular epithelial cells. Rare tubules were mineralized in the renal papilla in cases 10 and 11. Lesions were not evident in the urinary bladder in the 6 cases examined.
Hematopoietic system
The spleen had lesions in 9 of 10 cases. In all cases, the red pulp was expanded by small-to-medium foci of extramedullary hematopoiesis (EMH). Mild EMH was found in animals 6–13-mo-old and in adults. Moderate EMH was found only in adults. The only animal without splenic EMH was 6-mo-old (case 1).
The lymph nodes had lesions in 1 of 6 cases. In case 4, the lymph nodes were obliterated by necrosis and pyogranulomatous inflammation centered on bacilli, similar to that described in the lung.
The bone marrow had lesions in 2 of 2 cases. In cases 10 and 11, bone marrow adipose was completely absent and replaced by scant myxomatous matrix. The myeloid:erythroid ratio could not be determined due to prolonged postmortem interval.
Lesions were not evident in the thymus in the 3 cases examined. The thymus was still present and robust in cases 2, 10, and 11, which were 1-y-old, adult, and adult, respectively.
Body wall and adipose tissue
The adipose tissue had lesions in 6 of 12 cases. Adipose atrophy was characterized by shrinkage of the adipocyte cytoplasm due to reduced lipid volume. Atrophic adipocytes were variably sized. In cases of severe atrophy, adipocytes were difficult to recognize due to complete loss of cytoplasmic lipid and relative prominence of interstitial tissue (Fig. 7). Brown adipose tissue can look similar histologically to severely atrophied white adipose tissue; however, brown adipose tissue was ruled out in these cases as there was no evident lipid vacuoles in any section, including the bone marrow.
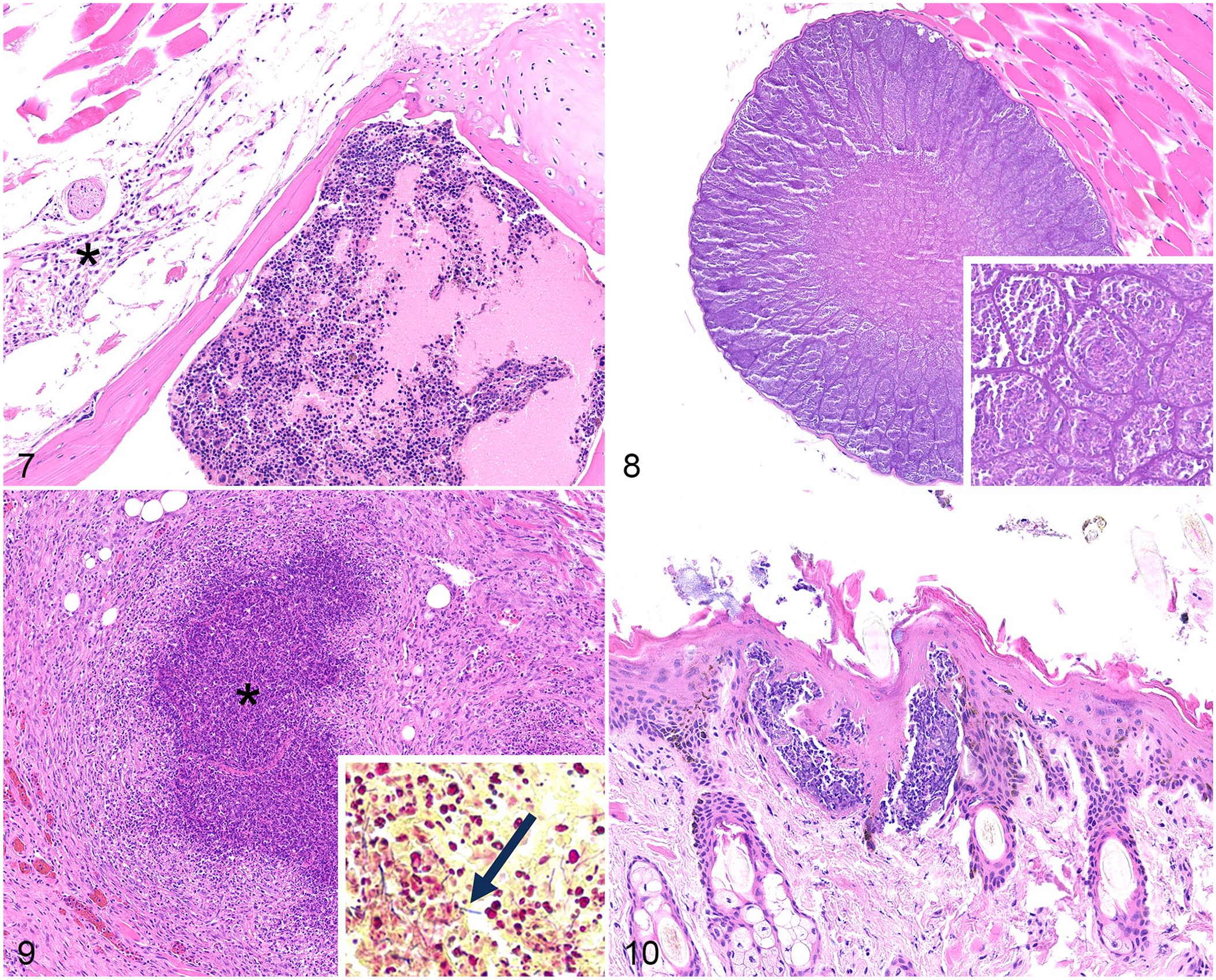
Histologic lesions in American pikas (Ochotona princeps).
The skeletal muscle had lesions in 4 of 11 cases. In case 12, skeletal myocytes and adjacent connective tissues were multifocally expanded by megaloschizonts that were ≤70 µm diameter and contained myriad, 1–2-µm, elongate, basophilic zoites that formed tight clusters (Fig. 8). In case 11, fewer skeletal myocytes were multifocally expanded by smaller tissue cysts of ≤20 µm diameter and contained similar appearing zoites. The morphology was consistent with Sarcocystis sp. in both cases. In case 4, myositis was characterized by large tracts of lytic necrosis surrounded by pyogranulomatous inflammation (Fig. 9), similar to that described in the lung. In case 5, rhabdomyolysis was observed in an unspecified section of skeletal muscle. It was composed of individual myocytes that had fragmentation of the sarcoplasm, loss of cross-striations, and nuclear fragmentation.
The skin had lesions in 3 of 6 cases. In cases 5 and 10, the pustules in the spinous, granular, and corneal layers consisted of small clusters of fragmented neutrophils that expanded the epidermis (Fig. 10). In case 4, the dermis, subcutis, and deeper layers were expanded by abundant necrosis and pyogranulomatous inflammation as was present in the lung.
Mammary tissue was hyperplastic in case 11, but presumably due to postpartum lactation (physiologic hyperplasia).
Reproductive system
The ovary had lesions in 2 of 2 cases. In cases 10 and 11, the ovarian interstitium had oval-shaped foci of mineralization roughly the size of an immature follicle; mineral was confirmed with von Kossa stain. The ovary in both cases had amyloid within the ovarian parenchyma; amyloid was confirmed with apple green birefringence on Congo red stain. Lesions were not evident in testes in 4 cases or in the uterus in 2 cases.
Special senses
The eyes had lesions in 1 of 5 cases. The lens in case 8 had loss of fiber definition in the subcapsular cortex and replacement by globular eosinophilic material (cataractous change). Lesions were not evident in the lacrimal gland in the case examined.
Nervous system
Lesions were not evident in the brain in the 10 cases examined.
Discussion
The most common cause of death in our cases was undetermined and attributed to secondary effects of stress (e.g., catecholamine release and myocardial dysfunction). 9 Most animals had minimal premonitory clinical signs before death, which may reflect the propensity for prey animals to hide signs of disease to avoid predation. Several animals died after a stressful event (anesthesia, transport, or handling); it may be that these non-domesticated lagomorphs do not adapt to captive manipulation, resulting in acute deaths. Anesthesia can cause coronary vasoconstriction, ischemia, and myocardial necrosis. 9 Two cases (10, 11) were anesthetized within 24 h of death, but no cardiac lesions were found in these cases; the time between the stressor and death was likely too short for the development of histologic lesions. In the domestic rabbit, death following handling is caused by catecholamine release and subsequent myocardial necrosis. 9 Three animals (6–8) died within 72 h after transport. All 3 animals had myocardial necrosis, and 2 of 3 had moderate-to-severe cardiomyocyte mineralization; it is possible that death was due to catecholamine release. 9 Another potential cause for transport death in these pikas is thermal stress. Domestic rabbits and hares, especially those that are young or pregnant, can succumb to thermal stress at temperatures >35°C (95°F) and have nonspecific lesions at autopsy. 16 American pikas may have an even greater sensitivity to thermal stress as this species typically lives in alpine talus and has a reported core temperature of 40.1°C (104.2°F). 40 Minimizing handling and ensuring cool transport temperatures may help avoid stress-associated mortality in pikas.
Histologic lesions were seen in most cases. Moderate-to-severe inflammation was found only in a few cases, all of which were associated with infection. In lagomorphs, neutrophils are often termed “heterophils” because they contain eosinophilic granules that make them difficult to differentiate from eosinophils histologically. Lagomorph heterophils function similarly to neutrophils in other mammalian species, unlike avian and reptilian heterophils that lack myeloperoxidase. Therefore, some texts use the terms neutrophil and heterophil interchangeably in the domestic rabbit. 43 One report on disease in an Asian pika species used the term heterophil. 41 In American pikas in our series, eosinophilic granules were not visible in the segmented leukocytes associated with oomycete and bacterial infections. Therefore, we interpreted these as neutrophils.
Death was attributed to infection in 2 cases, specifically a cutaneous bacterial infection (case 4) and a colonic oomycete infection (case 6). Parasitism was considered incidental based on lack of or minor host response and relatively low burden of infection. Neither viral nor fungal infections were identified in any case by histologic or molecular methods. Cutaneous bacterial infections in other lagomorphs, specifically domestic rabbits, are usually caused by trauma or laceration with secondary bacterial invasion. 43 Abscesses secondary to fighting or bite wounds are common in pair-housed or group-housed animals that are sexually mature, which was reported in case 5 with cutaneous bacterial infection 43 ; the cutaneous wound with granulation tissue at the stifle indicated traumatic disruption of the skin barrier with secondary infection and systemic dissemination. Commonly isolated agents in rabbit cutaneous infections include Staphylococcus aureus, Pasteurella multocida, and Pseudomonas aeruginosa; abscesses associated with oral flora are commonly the result of secondary infection by Fusobacterium sp., Streptococcus spp., Actinomyces sp., or Trueperella sp. 43 Although bacterial culture results were not available in case 4, the gram staining and morphology are most consistent with either Actinomyces sp. (such as A. israelii) or Trueperella sp. (such as T. pyogenes). Both Actinomyces and Trueperella are in the family Actinomycetaceae and are gram-positive, GMS-positive, PAS-negative, pleomorphic branching rods. Both genera have been found in oral, pulmonary, and multisystemic abscesses in domestic rabbits (Oryctolagus cuniculus).5,18,44 Fusobacterium necrophorum infection may occur similarly in rabbits, but is an unlikely cause of the lesions in case 4 as it is a gram-negative pleomorphic rod. 43 Cutaneous bacterial infections in rabbits most frequently cause a caseous and necrotic inflammatory response, which is similar to the morphology seen in case 4 with large caseous centers. 43
Oomycete and fungal infections in lagomorphs are less common than bacterial infections. 5 Reported deep and systemic mycoses of rabbits are generally confined to the lungs and include Aspergillus fumigatus, A. niger, and A. flavus. 11 The only case of fungal or oomycete infection in our series was an oomycete-induced colitis. PCR confirmed this to be Leptolegnia caudata, which is an oomycete closely related to Saprolegnia. L. caudata is a rarely reported oomycete that has been identified in water samples and insects. 35 We found no previous reports of L. caudata as a pathogen in mammals.
Evidence of sepsis was present in both cases 4 and 6 with an infectious cause of death (cutaneous bacterial infection and colonic oomycete infection, respectively). These animals both had embolic pneumonia seen as randomly distributed, interstitial aggregates of neutrophils with necrosis. Of note, neither case had inflammation or necrosis in the liver, which is commonly targeted in sepsis in lagomorphs. 43 In case 4 with cutaneous bacterial infection, bacteria may have traveled via lymphatics as well as blood vessels as infection was present in the lymph nodes. Bacteria may have entered the lungs before the liver if traveling via lymphatics.
Several parasitic infections were noted in these American pikas, none of which were considered the cause of death. Protozoa consistent with sarcocystosis were diagnosed histologically in cases 11 and 12. Sarcocystis sp. infection has been reported in pikas and is considered an incidental finding.4,14 Although they are similar morphologically to S. leporum of rabbits, there is not a published speciation of Sarcocystis sp. in pikas, to our knowledge. 4 Cecal and colonic nematodosis was diagnosed histologically in case 7. These parasites were considered incidental due to their presence in the lumen, lack of mucosal attachment, and lack of associated inflammation or other tissue changes in the large intestines. Oxyurids that have been reported in pikas include Dermatoxys sp., Cephaluris coloradensis, Labiostomum rauschi, and L. talkeetnaeuris (L. coloradensis),21,36 and are considered incidental findings. Ectoparasitism with fleas and/or ticks was noted grossly in 2 wild pikas. Fleas reported in the American pika include Oropsylla tuberculata and O. francisi. 8 Reported ticks in the American pika include Dermacentor andersoni, Ixodes angustus, I. ochotonae, and I. spinipalpis.1,7
Lipid accumulation was common in these American pikas. Periportal hepatic lipidosis often appeared grossly as an enhanced reticular pattern. Nearly all animals with adequate and excessive adipose stores had periportal hepatic lipidosis; only one animal with adequate body fat lacked this finding. Conversely, periportal hepatic lipidosis was absent in all animals with adipose atrophy. All cases of hepatic lipidosis occurred in captive pikas (1 wild pika was in fair body condition and 2 wild pikas were emaciated). It is unclear whether periportal hepatic lipidosis is pathologic (e.g., associated with chronic stress of captivity, dyslipidemia) or non-pathologic in American pikas. 20 As almost all animals without periportal hepatic lipidosis had negative energy balance, the absence of this histologic finding may warrant further investigation of the nutritional status of an animal. In the domestic rabbit, hepatic lipidosis is considered pathologic and may occur during periods of acute stress, gastric stasis, gastric ulceration, and generalized ileus. 33 The significance of hepatic lipidosis in American pikas warrants further investigation.
Moderate-to-severe myocardial necrosis was the cause of death in cases 6–8. The right ventricular free wall was consistently most severely affected, and this section of the heart warrants being consistently sampled for histology at autopsy. Myocardial mineralization was likely caused by myocyte necrosis in 2 of 3 cases (7, 8), which can be caused by stress, exertion, or nutritional deficiency. Transport stress and/or nutritional deficiencies of vitamin E–selenium causing low antioxidant capacity may have led to cardiac failure and death. The 3 affected animals died with minimal-to-no premonitory signs within a few days after transport to a new facility. In the domestic rabbit, cardiomyocyte necrosis can be a stress-induced change associated with catecholamine release. 9 The lesion is typically limited to one region of the myocardium and is less commonly multifocal. Often no premonitory signs occur prior to death, 9 similar to our cases. Vitamin E and selenium concentrations could not be determined as the only remaining tissues were formalin-fixed and paraffin-embedded, and vitamin E is destroyed by formalin. Additionally, selenium concentrations in formalin-fixed tissue may vary substantially from those in frozen tissues due to possible leaching or contamination. 30 Metastatic mineralization (e.g., excess dietary calcium, vitamin D toxicity, hyperparathyroidism) was considered an unlikely cause for myocardial necrosis in these cases as mineralization was not observed in the lung or stomach, which are more commonly affected by mineralization than the heart in most species.
Esophageal hyperkeratosis was a common lesion in our cases. Passage of food causes mild abrasion of the esophageal mucosa, preventing accumulation of superficial keratin. During anorexia, there is no stimulus for keratin removal, and keratin may accumulate on the mucosal surface. 24 In pikas with esophageal hyperkeratosis, anorexia and adipose atrophy were commonly reported in the history and observed histologically, respectively, and anorexia was presumed to be the cause of this lesion. In rats, vitamin imbalance, zinc deficiency, and anorexia are all reported causes of esophageal hyperkeratosis. Zinc deficiency specifically causes parakeratosis, which has been noted in some American pikas with esophageal hyperkeratosis. 24 Zinc deficiency may also cause cutaneous parakeratosis and esophageal epithelial hyperplasia, which were not noted, and zinc deficiency is considered unlikely in our cases. Therefore, esophageal hyperkeratosis in these cases was presumed to be secondary to anorexia.
Two cases had ovarian mineralization, which has not been reported previously in the pika but has been reported in mice and rats.13,22,49 This may reflect systemic mineral homeostasis (both cases also had scant renal tubular mineralization) or dystrophic mineralization associated with cell damage or necrosis, such as corpora lutea or follicle atresia as reported in mice and rats. 49 Ovarian amyloidosis has been reported in mice and rats and is generally associated with old age or chronic inflammation. 48 A similar interpretation is made in these 2 adult female American pikas (cases 10, 11), which were presumed to be >1-y-old.
Several histologic findings were identified that may be normal for American pikas, including splenic EMH, robust thymic tissue in adult animals, and Clostridium within the alimentary canal. Mild-to-moderate splenic EMH was present in nearly all of our cases and subjectively increased with age. Splenic EMH is likely normal for this species. This is similar to domestic rabbits in which EMH is reported in the spleen in healthy animals.19,47 Robust thymic tissue was present in our adult American pikas. It is likely that the thymus does not regress or involutes later in life in the American pika. In the domestic rabbit, thymic involution begins with sexual maturity at 16–25-wk-old, and substantial involution occurs by 13–19 mo.9,27,29 We found no data on timing of sexual maturity in American pikas; although this species reaches adult size in 3 mo in a laboratory setting, these seasonal breeders do not mate until the following spring or summer.40,50 Abundant spore-forming, gram-negative bacilli were identified in the intestinal lumen in the single case with Gram staining performed. Bacilli were consistent with Clostridium sp., which are typical flora of the cecum of domestic rabbits, although some Clostridium spp. may be opportunistically pathogenic.9,45,46 A study on the fecal microbiota of American pikas noted a large percentage of firmicutes; this phylum includes clostridia but further speciation was not provided. 23 Given the lack of tissue reaction in the pika, these clostridial organisms are likely normal flora with potential postmortem overgrowth.
Footnotes
Acknowledgements
We thank the individuals who performed initial pathology workup on these cases, including David Earl Green from NWHC, and Dick Montali from NZCBI, Barbara Bodenstein from NWHC for epidemiologic contributions, Charles Alex from Wildlife Conservation Society for assistance with study design, and Chris Gardiner, consultant for the Joint Pathology Center, for assistance with parasite identification.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Disclaimer
The views expressed in this manuscript are those of the authors and the U.S. Geological Survey and do not necessarily represent the views of the Department of Defense, the Smithsonian Institution, or the U.S. Government. The use of trade, firm, or product names is for descriptive purposes only and does not imply endorsement by the U.S. government.
