Abstract
Many neoplasms have been reported in goats; however, neoplasia of the rumen is rarely reported. A 9-y-old castrated male pygmy goat was presented with a history of respiratory stertor, fever, and anorexia. A respiratory diagnostic work-up including skull and thorax radiographs and endoscopy revealed minor enlargement of the arytenoids but no other abnormal findings. After a month of little improvement on symptomatic treatment and worsening general health, the goat was euthanized. On autopsy, the forestomachs, liver, spleen, diaphragm, and the ventral and lateral aspects of the cranial third of the walls of the peritoneal cavity were adhered to one another by fibrinous and fibrous adhesions. Numerous firm, white, up to 2 cm diameter nodules were found throughout the liver. A large sessile mass extended from the rumen wall into the lumen. The rumen mass was a gastrointestinal stromal tumor with metastasis to the liver.
Neoplasia of the rumen, reticulum, or omasum are rarely reported in small ruminants, 11 but are reported in cattle. 16 Reported tumors of the stomach compartments of cattle include papilloma, fibroma, fibropapilloma, squamous cell carcinoma, and lymphoma. 16 These tumors occur less commonly in sheep and have not been reported in goats. 16 Leiomyoma has been reported in the omasum of a goat as an incidental finding. 16 Fibrosarcoma of the rumen has been reported in one adult sheep, with metastasis to the liver. 16 Clinical signs of forestomach neoplasia vary by species and tumor type. In cattle, papilloma, fibropapilloma, and fibroma often cause chronic bloat given their location near the ruminoreticular groove. 16 Squamous cell carcinoma may cause abdominal pain, bloat, and drooling in cattle, and lymphoma generally causes ill-thrift. 3 Most forestomach neoplasms in sheep and goats are discovered at autopsy without premonitory specific clinical signs. 16
A 9-y-old castrated male pygmy goat was presented to the Field Service section at the University of Pennsylvania’s New Bolton Center (UPNBC) with a history of stertor. On physical examination, the goat was bright, alert, and responsive with a body condition score of 4 of 5. Vital parameters included a rectal temperature of 38.5°C (normal range: 38–40°C 5 ) and a heart rate of 120 beats per minute (normal range: 70–90 beats/min 5 ). The goat had a normal respiratory rate of 24 breaths per minute (normal range: 15–30 breaths/min 5 ) with referred upper airway noise upon auscultation of the chest. Airflow was present from both nostrils and a stertorous noise was heard on auscultation of the trachea. Peripheral lymph nodes were normal on palpation. A complete blood count revealed a non-regenerative anemia (hematocrit of 0.21 L/L, reference interval [RI]: 0.32–0.50 L/L 9 ), and a chemistry panel showed hypocalcemia (1.8 mmol/L, RI: 2.3–2.9 mmol/L; 7.3 mg/dL, RI: 9.2–11.6 mg/dL 10 ) and hypoalbuminemia (21 g/L, RI: 29–43 g/L; 2.7 g/dL, RI: 2.9–4.3 g/dL 9 ). Plasma fibrinogen concentration was increased at 138 g/L, RI: 2–3 g/L (1,390 mg/dL, RI: 200–300 mg/dL 9 ). Differential diagnoses for the anemia included gastrointestinal parasitism and anemia of chronic disease. Differential diagnoses for the upper respiratory signs included nasal mass, sinusitis, pharyngeal/retropharyngeal abscess, or thymoma. Sinusitis and pharyngeal/retropharyngeal abscess were considered as differential diagnoses for hyperfibrinogenemia. Additional diagnostic procedures including upper airway endoscopy and radiographic evaluation of the neck were declined at that time. Treatment was initiated with florfenicol (20 mg/kg, IM every other day for 2 treatments, then SC every other day until reexamination; Nuflor, Merck Animal Health, Whitehouse Station, NJ). The goat was also administered ivermectin (0.3 mg/kg SC once; Ivomec, Merial, Duluth, GA).
Reexamination of the goat 10 d following presentation revealed unchanged stertor and worsening of the hyperfibrinogenemia (155 g/L, RI: 2–3 g/L; 1,550 mg/dL, RI: 200–300 mg/dL 9 ). Based on these findings, the goat was referred to the UPNBC for further diagnostic procedures. In-house physical examination revealed little significant change from the presenting general condition seen in the field.
Radiographic evaluation of the skull and neck revealed minor soft tissue swelling in the laryngeal region but no evidence of a retropharyngeal or cervical mass. Upper airway endoscopy showed normal nasal passages and trachea, and bilaterally slightly enlarged arytenoid cartilages. Laryngeal function was found to be normal. These findings led to a diagnosis of possible laryngitis. The goat was sent home and treatment recommendations included continued florfenicol as previously described and meloxicam (45 mg, PO, q24h for 10 d; Generic Meloxicam, Zygenerics, Zydus Pharmaceuticals, Pennington, NJ). Given the lack of response to therapy, and the worsening of the goat’s general health, it was euthanized 2 wk following discharge from the hospital, and the goat was brought to the UPNBC for autopsy.
At autopsy, the forestomaches, liver, spleen, diaphragm, and cranial abdominal wall were adhered to one another by fibrinous and fibrous attachments. The rumen contained a 15 × 15 × 9 cm white-to-tan exophytic verrucous mural mass on the mucosal surface covered with green-black friable material (i.e., normal rumen content covering the ulcerated surface; Fig. 1). Further examination revealed the abomasum to be thickened by submucosal edema. The liver had numerous firm, white, pinpoint to 2 cm diameter nodules scattered throughout the parenchyma (Fig. 2). One of the mediastinal lymph nodes was firm, white, and enlarged with loss of architecture. The arytenoid cartilages were grossly normal.
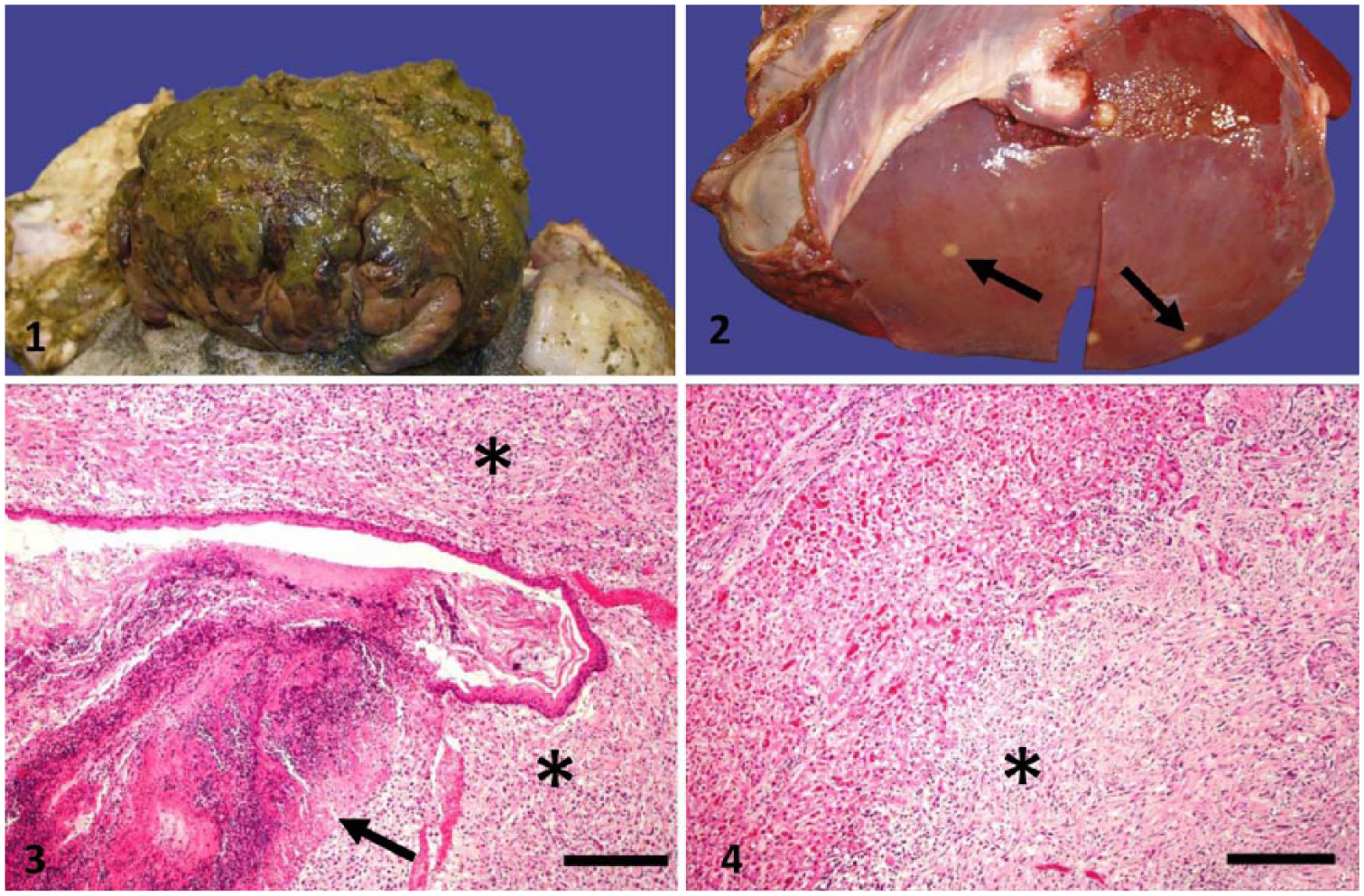
Ruminal gastrointestinal stromal tumor with metastasis to the liver.
On histologic examination, a non-encapsulated, moderately cellular nodule of interlacing bundles of neoplastic cells embedded in a loose fibrous stroma effaced the associated rumen mucosa, submucosa, and muscularis externa (Fig. 3). The cells were spindle-shaped with indistinct margins, scant eosinophilic cytoplasm, and a medium-to-large, oval-to-round nuclei that contained dense-to-stippled chromatin. There was mild anisocytosis and anisokaryosis. There were 5 mitotic figures per 10 high power fields of 400× magnification. There was coagulative necrosis of the ruminal mucosa at the junction of non-neoplastic and neoplastic tissue, with cell debris and mixed bacterial organisms.
Metastatic nodules of similar composition were scattered throughout the liver. The masses infiltrated and occasionally compressed the hepatic parenchyma and often tracked along the portal areas surrounding the bile ducts (Fig. 4). The enlarged mediastinal lymph node had mild diffuse edema with accumulation of amorphous eosinophilic material in the subcapsular sinus. The right and left arytenoid cartilages were unremarkable on histologic examination.
These findings led to the diagnosis of spindle cell tumor, the initial differential being ruminal fibrosarcoma with metastasis to the liver. This differential was high on the list given the morphology and location of the tumor and the species affected. Fibrosarcomas are found most commonly in dogs and cats, and have been reported in llamas, alpacas, cattle, ferrets, sheep, goats, and horses.2,7,8,12,13,16,17 Fibrosarcomas are not commonly found internally and are more common in the skin or subcutis with defined predilection sites in dogs (skin, subcutis, oral, and nasal mucosa) and cats (subcutis) with no defined predilection sites for other domestic species. 15 These tumors more commonly affect adult and geriatric animals, but there have been reports in cats and dogs <6 mo of age; a fibrosarcoma of the nasal and maxilla bones was reported as a congenital finding in a horse.8,15
To confirm the diagnosis of fibrosarcoma, further tests were pursued. Immunohistochemistry was chosen as the test of choice to identify c-kit expression (CD117) and the presence of smooth muscle actin (SMA) to differentiate between spindle cell tumors such as fibrosarcoma, gastrointestinal stromal tumor (GIST), and leiomyoma. The specific expression of CD117 (Polyclonal rabbit anti-human CD117, Dako, Santa Clara, CA) confirmed a GIST (Fig. 5). GISTs of equine and feline origin were stained both with and without the primary antibody as positive and negative control tissues. The control tissues stained as expected. Approximately 95% of the neoplastic cells exhibited moderate-to-strong cytoplasmic staining for CD117 (c-kit). Approximately 30% of the neoplastic cells exhibit strong cytoplasmic staining for SMA (Monoclonal IgG2a mouse anti-human antibody, Dako). The same staining pattern was found in the metastases in the liver.
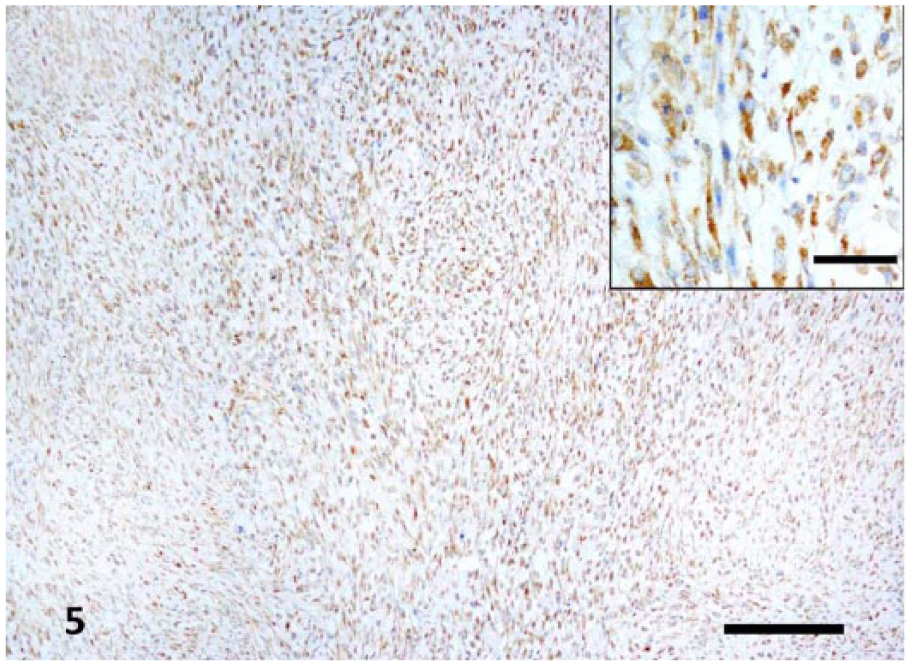
Strong immunostaining for CD117 differentiated the gastrointestinal stromal tumor (GIST) in the rumen from fibrosarcoma. Bar = 200 μm. Insets show higher magnification of the ruminal GIST. Bar = 50 μm.
Although GISTs have been reported in other domestic species, such as the dog, horse, and donkey,4,6,14 to our knowledge, GIST has not been reported previously in a goat. GISTs most commonly occur in the intestines and have been reported in the stomach of both horses and dogs.4,6 In horses and dogs, GISTs are often incidental findings; however, they have been associated with vomiting, weight loss, and anorexia in some dogs.4,6 In both horses and dogs, GISTs are known to be positive for c-kit (especially CD117) 1 with <50% also exhibiting positive reactivity to SMA staining. Other spindle cell tumors, such as leiomyomas, do not stain positively for c-kit. 6
The respiratory noise heard on presentation was attributed to the fibrous adhesions between the rumen and diaphragm interfering with normal function of the diaphragm. The unusual clinical presentation with primary respiratory signs and the lack of gastrointestinal signs is a notable feature of our case. The clinical findings of non-regenerative anemia can be attributed to anemia of chronic disease; the hyperfibrinogenemia was the result of inflammation caused by fibrinous peritonitis.
Footnotes
Acknowledgements
We thank Robert Ewing from the Information Technologies department at New Bolton Center (University of Pennsylvania School of Veterinary Medicine) for his help with editing the images in this manuscript.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
