Abstract
Lactococcus garvieae is the causative agent of lactococcosis in fish and an emerging zoonotic pathogen with high levels of antimicrobial resistance. We report a case of L. garvieae–associated septicemia in a central bearded dragon (Pogona vitticeps) confirmed via whole-blood PCR and direct sequencing. Following a 30-d course of ceftazidime (20 mg/kg IM q72h), the animal’s clinical condition had not resolved; leukopenia persisted, with heterophil toxic change. Coelomic ultrasound findings were consistent with preovulatory follicular stasis, folliculitis, and coelomitis. Following surgical ovariectomy and an additional 30-d course of ceftazidime, the animal’s behavior and appetite returned to normal, the animal tested negative via whole-blood PCR assay, and the CBC was unremarkable. To our knowledge, L. garvieae with L. garvieae–associated clinical disease has not been reported previously in a bearded dragon. We conclude that L. garvieae should be considered as a possible etiologic agent in cases of septicemia in bearded dragons, with the potential for zoonotic transmission warranting further investigation.
Lactococcus garvieae is a gram-positive coccus in the phylum Bacillota, order Lactobacillales, and family Streptococcaceae. It is the causative agent of lactococcosis, a disease of fish.19,25 Although various fish species develop hemorrhagic septicemia, lactococcosis is most economically significant in farmed rainbow trout (Oncorhynchus mykiss). 19 Infected fish commonly have vascular endothelial damage leading to extensive hemorrhage. Molecular techniques have been reported for the identification of L. garvieae targeting genes for 16S rRNA, rRNA internal transcribed spacer, and dihydropteroate synthase.4,21,28 Due to anorexia of infected fish, enteric treatment is challenging. Common antimicrobials prescribed include lincomycin, oxytetracycline, and macrolide antibiotics; however, antimicrobial resistance is high. 14 Additionally, L. garvieae has been classified as an emerging zoonotic pathogen with reports including infective endocarditis, liver abscesses, urinary tract infections, peritonitis, and other conditions in humans.15,16 This bacterium has been isolated from raw seafood, as well as raw milk from cows and camels.5,9 It has also been isolated from green sea turtles (Chelonia mydas), timber rattlesnakes (Crotalus horridus), and mugger crocodiles (syn. marsh crocodile; Crocodylus palustris), among other species, although clinical disease has not been reported in reptiles, to our knowledge.10,17,26
Several species, such as Streptococcus gallolyticus, are known significant pathogens of Dinosauria (Aves); however, reports of clinical disease due to members of Streptococcaceae in non-avian reptiles are limited. S. agalactiae has been associated with sepsis and mortality events in green tree monitors (Varanus prasinus) and saltwater crocodiles (Crocodylus porosus).6,11 Another family in Lactobacillales, Enterococcaceae, contains the significant squamate pathogen Enterococcus lacertideformus.1,7 It is not clear whether older reports of “streptococcal” infection in non-avian reptiles represent Streptococcaceae or related taxa.13,18
Central bearded dragons (Pogona vitticeps) are common pets in private households and are routinely kept as collection animals at zoologic and research institutions. 27 There has been considerable concern regarding the zoonotic potential of contact with reptilian species. Common zoonoses associated with reptiles include salmonellosis, mycobacteriosis, and pentastomiasis, among others. 20 We retrieved no cases of Lactococcus garvieae or L. garvieae–associated clinical disease in bearded dragons in a search of Google, PubMed, CAB Direct, Web of Science, and Scopus, using the search terms “Lactococcus garvieae bearded dragon,” suggesting that this condition has not been reported in bearded dragons to date.
A 4-y-old, intact female, pet bearded dragon was presented to the University of Florida–Veterinary Teaching Hospital (Gainesville, FL, USA) in July 2023 (day 0) for evaluation of acute onset of lethargy, coelomic distension, and inappetence. On initial examination, the animal was quiet, alert, and responsive with normal vital signs. The patient was mildly over-conditioned with a weight of 450 g and apparently euhydrated with no distinct masses or organomegaly appreciated on coelomic palpation. A heparinized blood sample was collected from the ventral coccygeal vein for CBC and biochemical analysis. Additionally, full-body lateral and dorsoventral radiographs were obtained, and a fecal sample was submitted for flotation and direct examination. The animal was hospitalized for monitoring and subcutaneous fluid support (2% bodyweight lactated Ringer solution SC q24h).
Radiographs revealed an intracoelomic mass causing moderate pulmonary compression, compatible with folliculogenesis. Fecal flotation and direct smear did not reveal any parasite ova or motile protozoa. Blood biochemistry was unremarkable. The CBC revealed severe leukopenia of 1.3 × 109/L (RI: 3.9–19.0 × 109/L), severe heteropenia of 0.17 × 109/L (RI: 1.0–11.6 × 109/L), and minimal left shift of 0.09 × 109/L. 12 The heterophils had severe toxic change, including increased cytoplasmic basophilia, markedly decreased granulation, and vacuolation. Numerous chain-forming cocci and coccobacilli were observed extracellularly and were frequently phagocytized by heterophils and monocytes, consistent with septicemia (Fig. 1A). Additionally, a moderate amount of amorphous, basophilic, proteinaceous precipitate was observed along the feathered edge, with lesser amounts noted within the body of the blood films (Fig. 1B, 1C). The material was evaluated further using a phosphotungstic acid–hematoxylin (PTAH) stain to assess for connective tissue, such as fibrin, and periodic acid–Schiff (PAS) stain to assess for glycogen, glycoproteins, carbohydrates, and mucins. The material was negative for PTAH and positive for PAS (Fig. 1D). Based on these results, considerations for the proteinaceous precipitate include yolk (i.e., yolk embolism) or other proteins such as immunoglobulins or acute-phase proteins. Coelomic ultrasound was declined at this time and the animal was sent home on a 30-d course of ceftazidime (20 mg/kg IM q72h) to treat bacteremia.
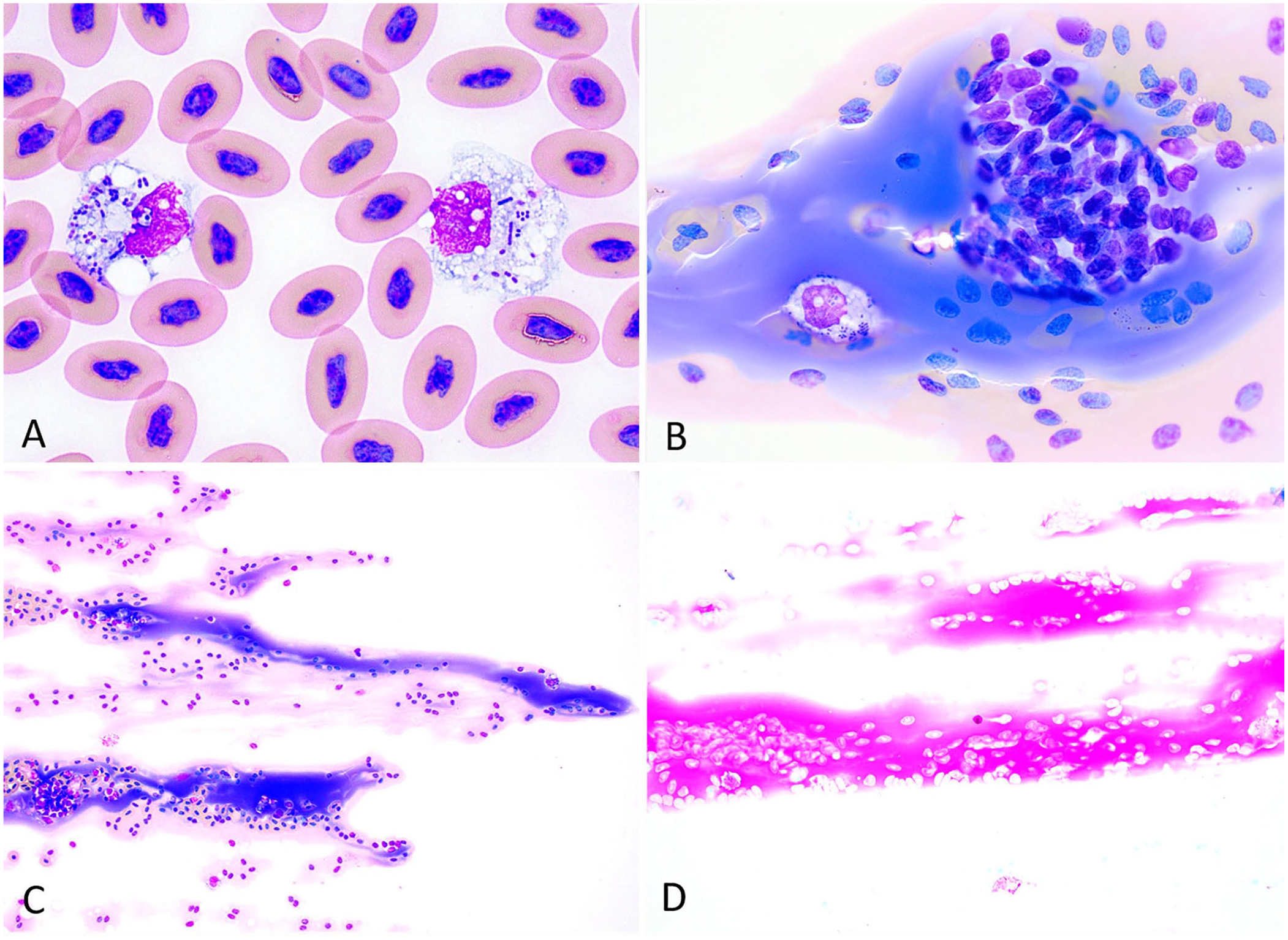
Blood film findings in a central bearded dragon (Pogona vitticeps) with lactococcosis.
DNA was extracted (DNeasy; Qiagen) from the blood. Pan-bacterial PCR targeting the 16S rRNA gene was done using described methods. 23 Sequencing resulted in a 1,420-bp segment of the 16S rRNA gene with 100% nucleotide homology to all type strains of L. garvieae (e.g., JCM 12256, GenBank LC145570) on a BLASTN (https://blast.ncbi.nlm.nih.gov/Blast.cgi) search of GenBank. 2 The sequence was submitted to GenBank (OR820608).
We discussed our findings with the owner and recommended that they continue practicing proper hand hygiene and limit exposure to immunocompromised individuals. On day 33, the animal was eating and acting normally. The patient had much brighter mentation but was still over-conditioned at 598 g (a 33% increase from the evaluation 1 mo prior). The coelom was fluid-distended with several round soft-tissue masses palpated in the caudal coelom, suspected to be follicles. Ultrasound evaluation found numerous follicles of various sizes and mixed echogenicity with an anechoic core and peripheral anechoic rim, consistent with preovulatory follicular stasis and folliculitis. A small amount of coelomic effusion was present and was sampled via ultrasound-guided, fine-needle aspiration. The effusion was slightly hazy and medium-yellow with a total protein of 22 g/L, triglycerides of 0.19 mmol/L, and cholesterol of 1.42 mmol/L. Cytologic evaluation of the effusion revealed a white blood cell estimate of 11.5 × 109/L, which was composed of small lymphocytes (92%) and mononuclear phagocytes (8%), including melanomacrophages. These characteristics were most consistent with a nonchylous lymphatic effusion. Repeat CBC revealed an improved but still severe leukopenia of 1.8 × 109/L (RI: 3.9–19.0 × 109/L) characterized by severe heteropenia of 0.34 × 109/L (RI: 1.0–11.6 × 109/L) with improved toxic change (mild degranulation) and resolved left shift of heterophils. 12 There were no intra- or extracellular bacteria, and the previously observed proteinaceous material was not appreciated.
On day 35, an ovariectomy was performed for concerns of oophoritis. The follicular and ovarian tissue appeared grossly normal. There was a moderate amount of coelomic effusion, but no other obvious evidence of disease. Postoperatively, the animal’s course of ceftazidime was extended for an additional 30 d at a dose of 20 mg/kg IM q72h as well as starting a 10-d course of meloxicam at a dose of 0.2 mg/kg PO q48h.
Cytology of the ovarian tissue revealed yolk, with absence of inflammation, infectious agents, tissue cells, or neoplastic cells. There was no anaerobic or aerobic growth from the coelomic fluid at 5 d post-collection. On day 49, the incision was healing well, and the patient had a normal appetite, no evidence of pain, and a return to normal behavior. On day 77, the skin sutures were removed. CBC revealed a normal white blood cell count (5.6 × 109/L) and was otherwise unremarkable; a 16S rRNA gene PCR assay of blood as performed earlier was negative, hence antibiotic treatment was discontinued. The patient has continued to recover well and was reported to be stable with no evidence of clinical disease at 104 d post-presentation.
Our case demonstrates the potential for infection and resultant disease caused by L. garvieae in bearded dragons. Our knowledge of antibiotic treatment in reptilian medicine is still developing; however, a prolonged course of ceftazidime cleared L. garvieae infection in this individual. Treatment of L. garvieae infection in the human literature has included the use of ceftazidime or other third-generation cephalosporins including ceftriaxone; however, recent antimicrobial susceptibility tests have not included ceftazidime among the other cephalosporins tested.3,8,22,24 Given the zoonotic concerns surrounding L. garvieae and the popularity of bearded dragons as pets, the isolation of this pathogen from the species is important. Future studies to determine the prevalence, ecology, and epidemiology of L. garvieae in the reptile pet trade are indicated.
Footnotes
Acknowledgements
We thank the zoologic medicine veterinary technicians, veterinary students, house officers, faculty, and clinical pathology laboratory technicians who were integral to the management of this case.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
