Abstract
Mycoplasma bovis is a major cause of bovine mastitis. Intermittent shedding of the organism for many months is a feature of cows with intramammary infection. A dairy farm in Japan experienced a mastitis outbreak caused by M. bovis in 2016, as well as 2 additional outbreaks and 1 case in 2020–2021. The causative strains in the 3 outbreaks shared a common and identical genetic feature, the insertion of a transposase gene at the same site within the phosphate acetyltransferase-2 gene. Additionally, all isolates were genotyped to closely related sequence types (ST21 and ST141) by multilocus sequence typing, and had similar pulsopatterns by pulsed-field gel electrophoresis. Our results indicate that infection with the same causative strain remained in this herd and environment for 4 y. Treatment with fluoroquinolones, guided by antimicrobial susceptibility test results, eliminated M. bovis from 16 of 20 M. bovis–infected cows, as confirmed by culture and somatic cell counts. However, mastitis caused by other bacteria occurred in 9 M. bovis–free cows within 2 mo of the last treatment.
Keywords
Mycoplasma bovis is involved in a variety of bovine diseases, including mastitis, pneumonia, otitis, and arthritis. 17 Mycoplasmal mastitis is generally characterized by strong infectivity and can appear as large outbreaks in dairy herds. 18 Additionally, M. bovis strains exhibiting reduced susceptibility to first-line antimicrobial agents, such as tetracyclines and 16-membered macrolides, have spread worldwide. 5 The sequence type (ST) 21 clonal complex, established by multilocus sequence typing (MLST), contains many of these low-susceptible strains, most of which have been isolated since the 2000s and have been widely reported. 9 International trade of bovine bioresources (e.g., breeding cows, semen, fertilized eggs) is responsible for the spread of these strains. 9 In fact, the response to antimicrobial therapy against M. bovis infection is poorer than responses to infections by other mycoplasmal species such as M. californicum.11,17 Some reports do suggest that treatment with specific antibiotics, such as valnemulin and enrofloxacin, to which relatively many field strains are susceptible, is effective against M. bovis infection.3,20 Given the current absence of an effective vaccine against M. bovis infection, and in light of the increasing resistance of M. bovis to antimicrobial therapy, early detection and the culling of carrier cows have been the main ways to prevent spread, but these measures cause huge economic losses.4,17
The introduction of subclinical carriers or contaminated semen is thought to be the primary means by which M. bovis–free herds become infected.7,9,17 Once present in a herd, M. bovis can be easily transmitted from carriers to uninfected cows and calves via contaminated secretions, droplets, and the environment. 17 In a dairy herd, udder-to-udder spread is a major means of transmission, and subclinical carriers are frequently confirmed not only in milking cows but also in calves, which can be new sources of contagion that are difficult to detect. 13 Moreover, mycoplasmas can survive for long periods in cool and humid environments, on farm equipment, and on workers in contact with that equipment. 17 The intermittent detection of the causative species over several months is considered a characteristic observed in cows with intramammary infection, and recurrence is sometimes confirmed at the herd level. 17 There are 2 possible causes of recurrence: persistent latent infection within a herd, or invasion of new field strains into a herd.
We examined the genotypes of the isolates obtained from the initial occurrence to the conclusion of the outbreaks to investigate recurrences of M. bovis mastitis at a dairy farm located in Nagano Prefecture, Japan, from 2016 to 2021. After confirming the antimicrobial susceptibility of the causal strains, we attempted antimicrobial therapy. At present, there is no basis for setting the period required to confirm the end of this disease. Furthermore, there are only a few reports on antimicrobial treatment strategies that consider the antimicrobial susceptibility of the causative M. bovis. 3 Our objective was to provide useful knowledge that could inform management and treatment approaches based on surveillance of M. bovis intramammary infection.
The farm housed 240–310 milking cows. Three outbreaks and one case of M. bovis infection (udder and respiratory infections) occurred at this farm: October 2016–July 2017 (outbreak A), March–May 2020 (outbreak B), October 2020–March 2021 (outbreak C), in addition to one case in August 2021 (case D). Cows with infectious diseases, such as diarrhea, pneumonia, and mastitis, were housed separately from healthy cows and are referred to here as the unhealthy herd. Furthermore, mycoplasma-infected cows were isolated as a separate group (mycoplasma herd). Although cows in the unhealthy herd were returned to the healthy herd after treatment, cows in the mycoplasma herd were housed separately from healthy cows to prevent the risk of infection, even after treatment. The milking cows were housed in free stalls and were milked by a double-parallel milking parlor with 8 milking units. The unhealthy herd was milked after the healthy herd, and the mycoplasma herd was milked last. All milking units were disinfected with liquid hypochlorous acid after the end of milking. Cows, heifers, and purchased semen introduced into the herd were not checked by Mycoplasma testing either before or during the study period. Calves (20–50) were kept individually in hutches in another barn to avoid direct contact with milking cows. Additionally, ~80 heifers were housed at another farm and were pastured in the summer. The annual incidence of mastitis on this farm was 34–62% from 2016 to 2022.
Individual milk (i.e., quarter milk or composite milk) was aseptically sampled at the end of milking, lung samples were aseptically collected at autopsy, and nasal samples were collected with a sterile cotton swab and soaked in PBS. After sampling, these samples were stored in an icebox until arriving at the laboratory. Mycoplasma spp. were isolated and identified as follows: the relevant sample (100 µL of milk, a piece of the affected region of a pneumonia-infected lung, or a nasal swab) was cultured in 3 mL of NK broth (Kanto Chemical) at 37°C for 3 d, and 10 µL of culture was further streaked on NK agar (Kanto Chemical), which was incubated in 5% CO2, at 37°C, for up to 5 d. After the isolation and culturing of mycoplasmal colonies, each isolate was stored at −80°C. DNA samples of the isolates were prepared (InstaGene Matrix; Bio-Rad), and the species were identified by PCR assays using species-specific primers.2,16
Effective antibiotics available and approved for use in dairy cows in Japan are limited to tetracyclines, 16-membered macrolides, and fluoroquinolones. 14 Therefore, enrofloxacin (Baytril; Bayer) was selected as the therapeutic antimicrobial. It was administrated for 5 consecutive d in 1 treatment period. Antimicrobial therapy in outbreak B was performed once or twice from May to August 2020. A second treatment was given to 9 cows whose infections had persisted after the first treatment. Moreover, infection status was checked against all cows twice a year until January 2023 at the latest.
To understand the epidemiologic and genetic relationships among isolates, pulsed-field gel electrophoresis (PFGE) 1 and MLST15,19 for M. bovis were performed as reported previously. For MLST, neighbor-joining (NJ) analysis was performed by MEGA5 21 using the profile sequences of the STs of the field strains plus all Japanese field strains registered at the MLST website (https://pubmlst.org/organisms/mycoplasma-bovis). The similarities between PFGE pulsopatterns were calculated using the Dice algorithm, and dendrograms based on the pulsopatterns were constructed by the unweighted pair group method with arithmetic means (UPGMA). To obtain a complete match between duplicate strains, the band position tolerance was set at 1%, and optimization was set at 0.5%. 1
Mutations associated with decreased antimicrobial susceptibility (i.e., tetracyclines, macrolides, lincosamides, spectinomycin, fluoroquinolones) of the causative M. bovis strain were analyzed as described previously. 12 Mutations affecting susceptibility to tetracyclines and spectinomycin, located in the 16S ribosomal RNA gene (rrs), as well as those affecting susceptibility to macrolides and lincosamides, located in the 23S ribosomal RNA gene (rrl), were detected by PCR amplification of rrs and rrl and melting curve analysis using hybridization probes. 12 The coexistence of nonsynonymous mutations in the DNA gyrase gene (gyrA) and topoisomerase IV gene (parC) leads to decreased susceptibility against fluoroquinolones, and these mutations were confirmed by PCR amplification and DNA sequencing of the target regions of gyrA and parC. 12 As well, the MICs of oxytetracycline, tylosin, lincomycin, and enrofloxacin were determined as described previously. 8
Antimicrobial therapy was attempted in 20 infected cows with somatic cell counts (SCCs) of 3.6–453 × 104 cells/mL in outbreak B. Mycoplasma testing by culturing was performed once within one month after antimicrobial therapy, and then once or twice until the end of 2020, and twice per year from 2021 to January 2023. Causative bacteria of bovine mastitis, other than bovine mycoplasmas, were monitored using various culture media, including trypticase soy agar with 5% sheep blood (Becton Dickinson), Edward agar with 5% sheep blood (Kanto), mannitol salt agar (Becton Dickinson), deoxycholate hydrogen sulfide lactose agar (Nissui Pharmaceutical), Sabouraud agar (Eiken Chemical), and X-SA agar (Nissui). Isolates were checked by Gram staining and underwent coagulase, catalase, and oxidase tests.
In outbreak B, intramammary infection caused by M. bovigenitalium was confirmed simultaneously in 2 cows. M. bovis was isolated from mastitic milk with a high SCC (>28.3 × 104 cells/mL) and an abnormal appearance, such as a watery consistency or the presence of clots. The organism was also isolated from normal milk (i.e., milk with a normal SCC [<28.3 × 104 cells/mL] and without the abnormal appearance seen in mastitic milk). In addition, bovine mycoplasmas were isolated from bulk milk samples, nasal swabs, and lung samples of cows with pneumonia. Of the infected cows, 4 were introduced from 2 farms in outbreak B, and 4 were introduced from one farm in outbreak C. These cows were introduced from other farms within one month before confirmation of M. bovis infection. No cases of M. bovis infection had been confirmed at these source farms or at epidemiologically related farms; hence, we speculated that infection occurred after the cows had been introduced to the study farm. The main infection control measures were the segregation and culling of infected cows, mycoplasma testing of milk samples, and feeding pasteurized milk to calves. Moreover, workers from mycoplasma herds were prohibited from entering other herds. M. bovis infections have not been confirmed at this farm since case D (Table 1).
Summary of Mycoplasma bovis infection in 3 outbreaks and 1 case in Japan.

Number of introduced cows are in parentheses.
Details of the main measures implemented for each M. bovis outbreak and case are indicated by the following abbreviations (terms of implementation are in parentheses): a = segregation and culling of infected cows; b = Mycoplasma testing of bulk milk every month; c = Mycoplasma testing of bulk milk every week; d = Mycoplasma testing of all individual milk to confirm uninfected status at end of outbreak; e = Mycoplasma testing of all postpartum milk and mastitic milk; f = feeding pasteurized milk to calves; g = changes to sanitation procedures (changes in traffic lines and milking order, addition of a disinfection step); h = administration of antimicrobial therapy.
All isolates from outbreak A were ST21 and pulsotype A4, with a confirmed transposase gene (tpn) insertion at the same position in the phosphate acetyltransferase-2 gene (pta-2), one of the MLST loci (Table 2). The pta-2 sequence has been deposited at DDBJ/EMBL/GenBank under accession LC760077. The insertion of tpn in pta-2 was also confirmed in 7 isolates each from outbreaks B and C, and these isolates represented ST21 or ST141. ST21 is one of the dominant STs in Japanese isolates; 9 ST141 isolates have not been reported previously from Japan. In the NJ analysis, ST21 and ST141 were closely related, with only 3 bp differences out of the total of 3,015 bp covered by MLST (Fig. 1). Moreover, these isolates had identical or similar pulsopatterns (A1, A2, A3) to isolates from outbreak A (Fig. 2). Because there are <6 fragment differences in these 3 patterns compared with pulsotype A4, these isolates can be considered part of outbreak A. 22 The insertion position of tpn was also identical among these isolates. Our results suggest that all isolates with tpn inserted in pta-2 are likely derived from the same strains found in outbreak A.
Genotyping profiles of Mycoplasma bovis isolates in our study.
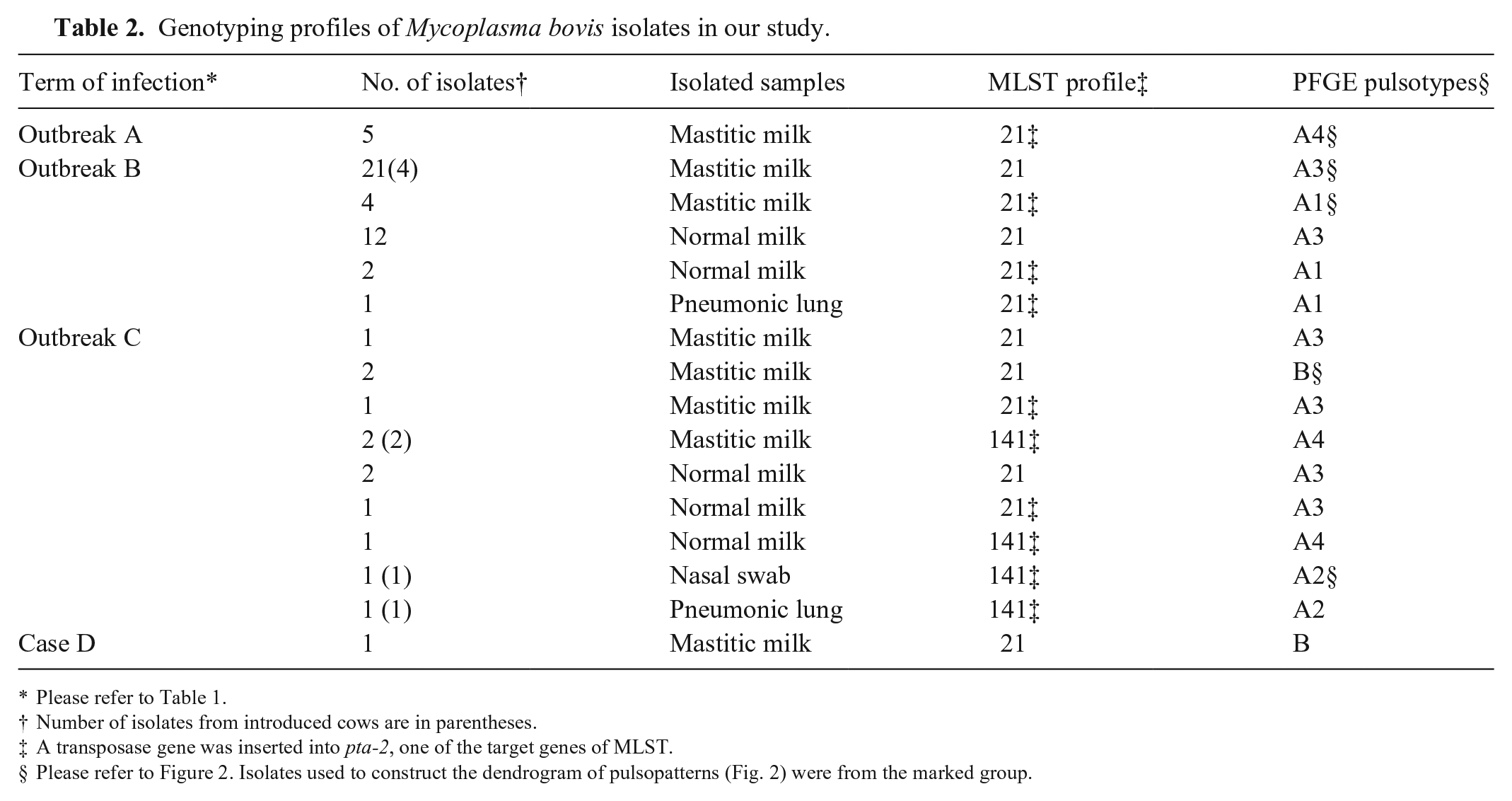
Please refer to Table 1.
Number of isolates from introduced cows are in parentheses.
A transposase gene was inserted into pta-2, one of the target genes of MLST.
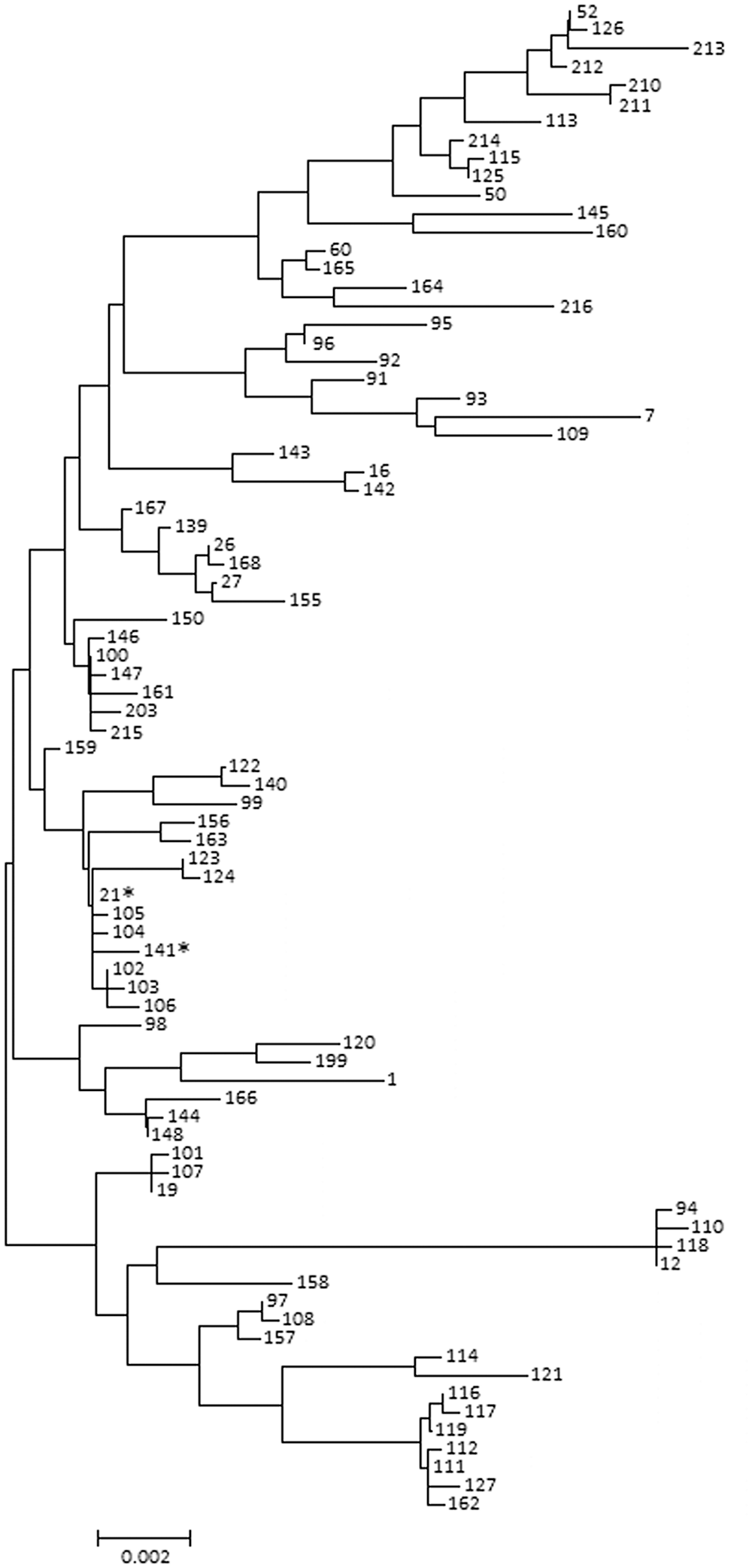
Phylogenetic neighbor-joining tree displaying the sequence types (STs) of Japanese Mycoplasma bovis strains. Numbers indicate specific ST numbers. Asterisks (*) indicate ST21 and ST141 M. bovis isolated in our study.
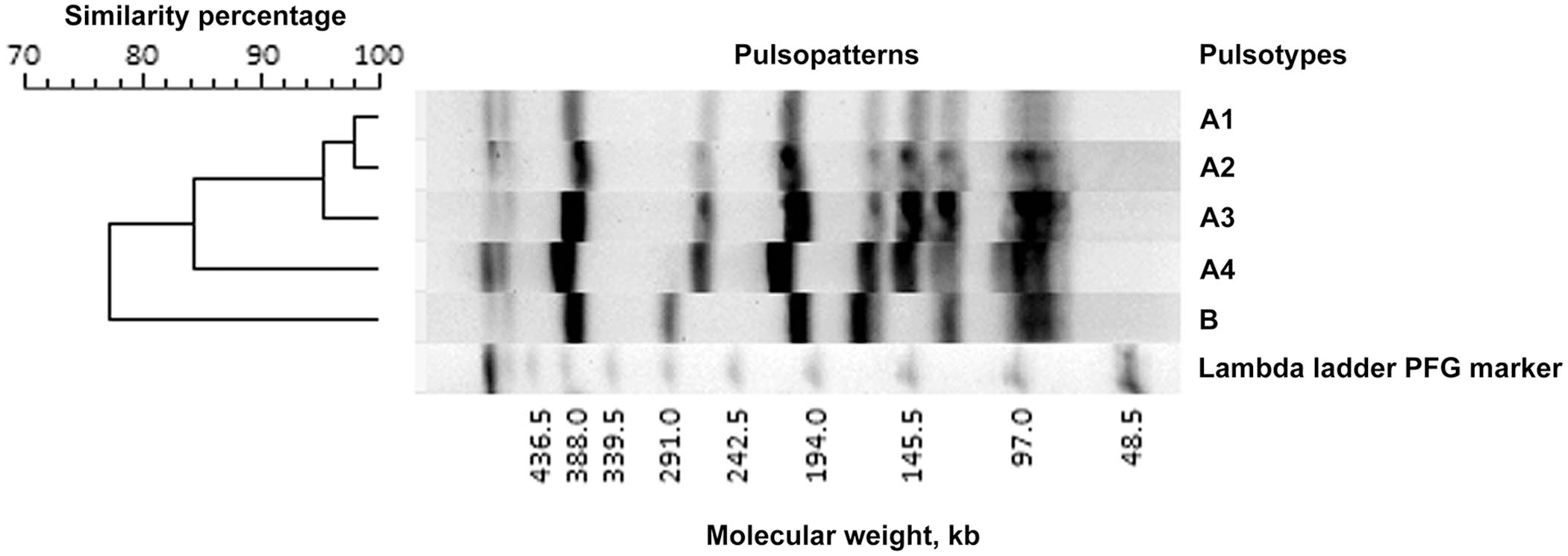
Dendrogram of Mycoplasma bovis pulsopatterns from diverse samples from the monitored dairy farm, based on representative pulsopatterns obtained by MluI-digested total DNA using pulsed-field gel electrophoresis. Pulsopatterns with a similarity of ≥80% are assigned the same alphabetical pulsotype name.
In outbreaks B and C, some of the isolates with the tpn insertion were obtained from normal milk samples without any macroscopic changes and high SCC. These subclinical carriers are suspected to be important sources of herd-to-herd transmission in epidemiologic investigations of M. californicum. 10 Moreover, M. bovis in the dairy environment may also be involved in the persistence of infection between herds. Periodic checks of bulk milk are considered effective for the early detection of these subclinical carriers and useful for maintaining mycoplasma-free herds. 17 Considering our outbreaks, however, it seems that monitoring the herd for infection for at least 4 y following an outbreak is necessary. Isolates with the tpn insertion were also obtained from nasal swabs and lung lesions, indicating an association between mastitis and pneumonia, as reported previously. 13 On the other hand, all isolates without the tpn insertion in pta-2 are also ST21 and have similar pulsopatterns (A3, B), suggesting the possibility that tpn insertion dropped away in some outbreak A isolates. At the same time, given that ST21 is the dominant ST in Japan, 9 the possibility that these isolates had newly invaded the dairy herd cannot be ruled out.
Mutations at rrl A2058 or rrl A2059, and rrs at C1192, are involved respectively in co-decreased susceptibility to macrolides and lincosamides, and decreased susceptibility to spectinomycin. 12 Moreover, the coexistence of missense mutations in both gyrA and parC confers resistance to fluoroquinolones. 12 No mutations related to susceptibility to these antimicrobials were found in any of the isolates tested. The MICs for lincomycin (0.4–0.8 µg/mL) and enrofloxacin (0.2 µg/mL) were similar to those detected using M. bovis PG45T (0.8 µg/mL and 0.2 µg/mL, respectively).
Mutations at rrs A965 and A967 are involved in decreased susceptibility to tetracyclines; those at rrl G768 are involved in decreased susceptibility to 16-membered macrolides. 12 We confirmed these mutations in all of the isolates tested. The MICs for oxytetracycline (3.1 µg/mL) and tylosin (3.1–6.3 µg/mL) for the outbreak isolates were higher than those detected using M. bovis PG45T (0.2 µg/mL and 0.1 µg/mL, respectively).
Mycoplasma testing, conducted by culture, showed that 10 cows were negative for M. bovis infection after the first treatment period, and 6 additional cows were negative after the second. Of the 16 M. bovis–negative cows, 15 had a lowered SCC (<28.2 × 104 cells/mL). The other 4 cows continued to test positive for M. bovis after the second treatment period. Of these 4 animals, 2 had a lowered SCC (<7.0 × 104 cells/mL), but the remaining 2 died within 2 mo after the last treatment because of worsening signs, with increasing SCC (>226 × 104 cells/mL).
M. bovis strains with low susceptibility to first-line antibiotics have been spread around the world, and strains with low fluoroquinolone susceptibility have been isolated from Japanese beef cattle at a high rate. 9 On the other hand, many of the strains derived from dairy cows are fluoroquinolone-sensitive, 9 as in our case. Antimicrobial treatment with fluoroquinolone has been effective in cases of intramammary infection, but the results have been mixed for other diseases (respiratory infections); the therapeutic effect may vary greatly depending on the disease site and antimicrobial susceptibility of the causal strain.3,20
Although 16 cows became negative for M. bovis infection after antimicrobial treatment, 9 cows were confirmed to have mastitis caused by other microorganisms (Arcanobacterium spp., Escherichia coli, Prototheca spp., coagulase-negative staphylococci, and yeast-like fungus) within 2 mo after the last antimicrobial treatment. These microorganisms have long been recognized as the primary causes of mastitis on this farm, and the incidence of mastitis in the treated herd during these 2 mo was comparable to the annual incidence on this farm. Of these 9 cows, 3 were culled because of the deteriorating quantity and quality of their milk. The concentrations of microorganisms in milk samples were 480 to >20,000 cfu/mL. Most of these microorganisms are known as environmental mastitis–causing bacteria. The improvements in sanitary measures taken during our study were mainly against contagious mastitis–causing bacteria, and the changes of measures against environmental mastitis–causing bacteria were insufficient. Even in the case of mycoplasmal mastitis, sanitation measures against environmental mastitis–causing bacteria are important, indicating that a clean environment is an important factor for preventing reinfection by these microorganisms. Additionally, M. bovis–induced mastitis leads to increased expression of immune exhaustion–related genes and to decreased expression of innate immune response–related genes of mononuclear cells. 6 Thus, we speculate that the reduced immune response in mamm-ary tissue caused by M. bovis persists after the elimination or dramatic reduction of M. bovis from mammary tissue, thereby facilitating infection by other microbes and causing mastitis even after antimicrobial treatment.
The multi-year persistence of M. bovis infection at the herd level that we demonstrated reaffirms the importance of periodic monitoring (bulk milk testing) as a control strategy for mycoplasmal mastitis. Moreover, the importance of sanitary measures against environmental mastitis–causing bacteria after antimicrobial therapy was also reaffirmed in mycoplasmal mastitis outbreaks. Future work should look at the recurrence of disease and persistence of infection at the individual level. We have confirmed the importance of selecting an effective antimicrobial agent prior to antimicrobial treatment. Antibiotic treatment is expected to contribute to the reduction of economic losses by reducing preventive culling. Although mastitis treatment with effective antimicrobial agents showed some success, a new problem was revealed: the high frequency of reinfection by other bacteria. To address this problem, the effect of restoring immune function in mammary tissue through treatment during the dry period while concurrently using immunostimulants should be investigated.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
Our study was conducted under the research program entitled “Regulatory Research Projects for Food Safety, Animal Health, and Plant Protection” (JPJ008617.17935699) funded by the Ministry of Agriculture, Forestry, and Fisheries of Japan.
