Abstract
Two adult male Puerto Rican crested anoles (Anolis cristatellus cristatellus) housed in a research facility were presented with debilitation and were euthanized. On autopsy, anole 1 had a large cystic white structure in the left pelvic limb, which protruded through the ruptured epidermis, and a large, poorly demarcated swelling in the right caudal abdomen. Anole 2 had masses in the mid-dorsum, caudal dorsum, left pelvic limb, and tail. These masses contained variably sized cestode larvae, which ruptured into the coelomic cavity. Evaluation of the larvae revealed a thickened and wrinkled anterior end, with a cleft-like invagination, consistent with either a plerocercoid sparganum or a tetrathyridium. Histologically, several cestode larvae were contained in the body wall of both anoles. These were up to 650 μm in diameter, with a thin tegument and a spongy parenchyma. The spongy parenchyma contained numerous, up to 30 μm diameter, sharply demarcated, basophilic-to-black structures (calcareous corpuscles). There was pneumonia and hepatitis in anole 2, suggestive of potential secondary infection subsequent to immunosuppression. Molecular amplification of the cytochrome C oxidase subunit 1 revealed 100% homology for the COX1 gene of the diphyllobothriid tapeworm Spirometra erinaceieuropaei, also known as Spirometra mansoni.
Two wild-caught adult male Puerto Rican crested anoles (Anolis cristatellus cristatellus), used in experimental behavioral studies at a local university for several years, were presented to the institution’s laboratory medicine service for evaluation of debilitation, and were subsequently euthanized by the veterinary team. Grossly, both individuals contained multiple (2 in anole 1; 4 in anole 2) well-demarcated, raised, soft, nodular, up to 1-cm diameter masses distributed over the left pelvic limb and right lateral body wall (anole 1; Fig. 1A), and the dorsum, left pelvic limbs, and base of the tail (anole 2). Nodules occasionally expanded through the ruptured epidermis in anole 1, and, on sectioning, all of the nodules contained abundant clear gelatinous material admixed with small, ~2-mm diameter, flat, white, larval cestodes (metacestodes; Fig. 1B).
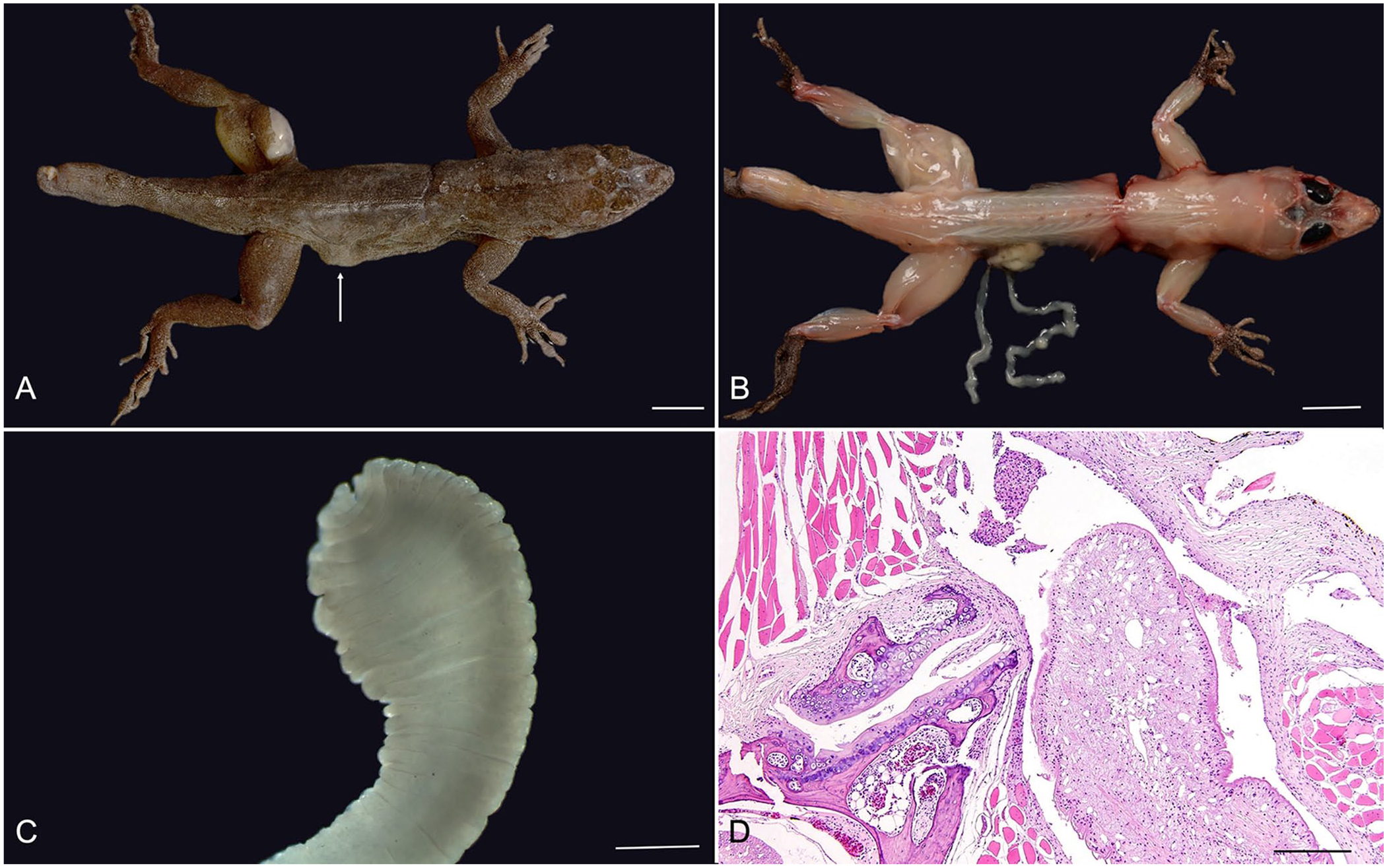
Cestodiasis in 2 Puerto Rican crested anoles.
Nodules associated with the body wall of both anoles had perforated through the body wall musculature, and free metacestodes were found in the caudal coelomic cavity. Stereoscopic examination of the extracted metacestodes revealed a solid body, with no appreciable scolex (Fig. 1C). The parasites were considered at this time to be either a sparganum (plerocercoid) of Spirometra spp. or a tetrathyridium of Mesocestoides spp.
Histologic review of tissues for both individuals revealed metacestodes in the muscle. The metacestodes were ~0.5-mm wide, with a thin, ~10-μm wide, poorly demarcated, eosinophilic tegument, with multiple invaginations and no external spines. The spongy parenchyma lacked a coelomic cavity, and contained numerous, up to 30-μm diameter, sharply demarcated, irregularly round, basophilic foci (calcareous corpuscles; Fig. 1D). There were no digestive or reproductive organs noted. The examined metacestodes lacked a scolex in all examined sections. The coelomic cavity adjacent to the metacestodes was expanded by fibrin and mononuclear cells, and the vertebrae in these areas had bony remodeling with scalloped, irregular edges. Adjacent myocytes were multifocally degenerate, with shrunken, hypereosinophilic muscle fibers containing pyknotic nuclei and karyorrhectic debris. Additionally, myocytes were multifocally surrounded by a moderate population of lymphocytes and macrophages. The surrounding fascia was similarly infiltrated by moderate numbers of lymphocytes and macrophages. The remainder of the examined organs in anole 1, including brain, liver, kidney, stomach, intestine, and eye, were within normal limits. Evaluation of the remainder of the organs for anole 2 revealed suppurative pneumonia and granulomatous hepatitis.
Precise characterization of the parasites was achieved through amplification and sequencing of the COX1 (cytochrome c oxidase subunit 1) gene. 2 The recovered sequences were a 100% match in GenBank for the COX1 gene of Spirometra erinaceieuropaei (also known as S. mansoni).
S. erinaceieuropaei is a diphyllobothriid tapeworm first described in the European hedgehog (Erinaceus europaeus), for which it is named. The lifecycle of S. erinaceieuropaei begins with the passage of eggs from a final host, which may be a cat or dog.3,5,12,24 Eggs passed in feces survive in water, and ciliated coracidia (larvae) hatch from these eggs at varying rates, from a few days to 5 y under experimental conditions. Larvae are ingested by copepods (e.g., genera Mesocyclops and Eucyclops), and develop to procercoids within the copepod’s body cavity. The second intermediate hosts are vertebrates, which are infected by consumption of copepods containing procercoids; these subsequently penetrate the gut of the vertebrate and develop into plerocercoids within the tissues of this new host. Second intermediate hosts are most commonly frogs and grass snakes, although mammals (including humans) may also be infected either via ingestion of copepods or tissues containing plerocercoids, in the latter case acting as paratenic hosts.12,15 Dogs and cats are final hosts, and ingest plerocercoids in the flesh of vertebrate intermediate and paratenic prey hosts. The cycle continues when eggs are passed in feces 15–18 d later. 3
S. erinaceieuropaei causes subclinical infections as an adult tapeworm in both the dog and cat, with no reports of clinical signs as a result of infection in either species. 3 It is uncertain to what degree S. erinaceieuropaei causes disease in anoles. Both of our cases had body wall perforations caused by the cestode nodules. This may represent a characteristic finding of cestode infection in anoles or may represent a specific gross pathologic manifestation in this cestode species; however, additional specimens must be examined to further characterize this presentation.
Metacestode infections with plerocercoids (also known as spargana) have been shown to cause a variety of symptoms in humans, in a condition known as sparganosis. Given that eggs passed in the feces of cats and dogs must first hatch in water to infect copepods, and then, further development in these invertebrate hosts occurs before becoming a form that can infect vertebrates, zoonotic transmission directly from cats and dogs is not possible. Humans may become infected via ingestion of infected copepods in drinking water, or from vertebrate intermediate or paratenic hosts containing plerocercoids. Additionally, traditional poultices of raw meat from frogs and snakes used in wound dressing and eye applications can lead to sparganosis. 21 Following infection, plerocercoids can migrate to a variety of anatomic locations, including the skin, muscles, visceral organs, and CNS. Spargana may cause skin and soft tissue swelling and urticaria, followed by CNS lesions, leading to seizures and hemiparesis.3,4,21
Sparganosis has been reported worldwide; however, the disease is most frequently diagnosed in east Asia, particularly in China, Japan, Korea, Thailand, and Vietnam, with most cases arising in Japan.7,10,22 Infection with adult stages of the parasite in people has also been reported in 5 cases. 20 The distribution of reported cases of sparganosis corresponds with the parasite distribution, which is predominantly in Asia, although reports from Europe and the Americas, including Hawaii and Puerto Rico, also exist.1,7–9,14,16,23 A related species, S. mansonoides, is more commonly distributed in the eastern United States, and can cause mild disease in cats, with clinical signs including weight loss, stunted growth, and diarrhea.6,13 S. mansonoides is also known to cause sparganosis in people.
Interestingly, in experimentally infected mice, S. erinaceieuropaei has been associated with significant weight gain that cannot be fully attributed to the mass of the parasites alone, with an average weight difference of 17.6% in infected mice compared to control mice, and a weight gain of up to 30% in some infected mice. 11 Additionally, one study found that green anoles infected with plerocercoids had a weight gain of up to 32% compared to uninfected controls, and injection of lizards with whole-worm homogenate also resulted in weight gain, although to a lesser extent; infected lizards had less reserve fat than control animals, suggesting increases in body weight were the result of overall growth and not fat accumulation. 18 In our 2 cases, captured anoles were indeed subjectively larger than expected, especially given the history of debilitation; however, body weight was not recorded at the time of autopsy.
A cause for these changes in growth was elucidated with the discovery of plerocercoid growth factor (PGF), a neutral cysteine proteinase with agonistic effects for human growth factor.17,19 Although PGF is known to mimic a mammalian growth factor, it may be that sufficient homology exists in reptilian growth factor receptors. Indeed, Puerto Rican an-oles found in urbanized areas are anecdotally bigger than their forest counterparts. It has been hypothesized that urbanized areas contain additional resources, allowing for more growth in these anoles. However, the potential for increased plerocercoid infection given contamination of urban environments with canine and feline feces, and subsequent in-creased growth as a result of PGF expression, is an interesting notion.
We have described here 2 cases of Spirometra infection in Puerto Rican crested anoles. Characterizing sparganosis in reptiles is important not only in cataloging the overall parasite life cycle, but also in developing and expanding the phylogenetic tree, and tracking the overall geographic distribution of the parasite. Sparganosis in reptiles has not been well characterized, and further understanding of this infection in reptiles could have direct implications in determining the extent of parasitism and resulting pathology, potentially allowing for better prognostication and treatment. 8
Footnotes
Acknowledgements
We thank Drs. Laura Coffee, Brian Powell, and Manuel Leal for their invaluable contribution to making this publication a reality.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
