Abstract
We examined a case of congenital idiopathic megaesophagus (CIM) in a 5-wk-old female Gordon Setter puppy by means of contrast radiography, autopsy, histopathology, and immunohistochemistry. Clinical and radiologic findings included weight stagnation and marked generalized esophageal dilation with ventral displacement of the heart and lungs. These findings were confirmed at autopsy, and segments of the thoracic esophagus were sampled for histopathology. On histopathology, diffuse esophageal muscular atrophy, mucosal erosions, mononuclear inflammation, and a marked reduction in the number of myenteric plexus structures and number of ganglion cells were present (aganglionosis). The latter was determined immunohistochemically using an anti-peripherin antibody as the primary reagent, which provides a strong tool for the histologic confirmation of CIM. The histologic findings share some similarities to lesions associated with megaesophagus in Friesian foals, as well as esophageal achalasia and Hirschsprung disease in humans.
Megaesophagus (esophageal ectasia) is defined as dilation of the esophageal lumen or parts thereof, resulting from atony and flaccidity of the esophageal musculature. 16 The condition occurs in several animal species, including dogs, cats, horses, ruminants, and rodents, and may be either congenital or acquired.4,9,11,15,16 In most species, the pathogenesis of the congenital condition remains rather obscure. In dogs, however, a link to an organ-specific sensory dysfunction has been confirmed resulting from a defect in the distention-sensitive vagal afferent system innervating the esophagus. 7 Although the prevalence of congenital idiopathic megaesophagus (CIM) has increased in some breeds of dogs (e.g., Great Danes, Labradors, Irish Setters), an inheritance pattern has only been confirmed in Miniature Schnauzers, Wire-haired Fox Terriers, and German Shepherd dogs.3,16 CIM is well documented with regard to clinical presentation, diagnostic imaging, and treatment options; however, descriptions of the histologic manifestations of this disease in dogs are scant. We present here the clinical and pathologic manifestations of a severe case of CIM in a 5-wk-old female Gordon Setter puppy.
The puppy originated from a litter of 9 (3 females, 6 males), of which one female was euthanized immediately after birth because of gastroschisis. After the introduction to solid food (day 21), one of the female puppies slowly started to deteriorate. The owners noticed gradual weight stagnation compared to the littermates, despite a voracious appetite (Suppl. Fig. 1), and clinical signs of nasal discharge (rhinitis), coughing, recurrent vomiting, and regurgitation. The rhinitis cleared within a week, but the weight stagnation continued despite assisted feeding; at 5-wk-old, the puppy was subjected to contrast radiography, using orally administrated barium sulfate. Radiography revealed marked dilation of the esophagus occupying most of the neck and thoracic cavity, with a size exceeding the gastric volume (Fig. 1A). Given a poor prognosis, the puppy was euthanized and submitted for autopsy.
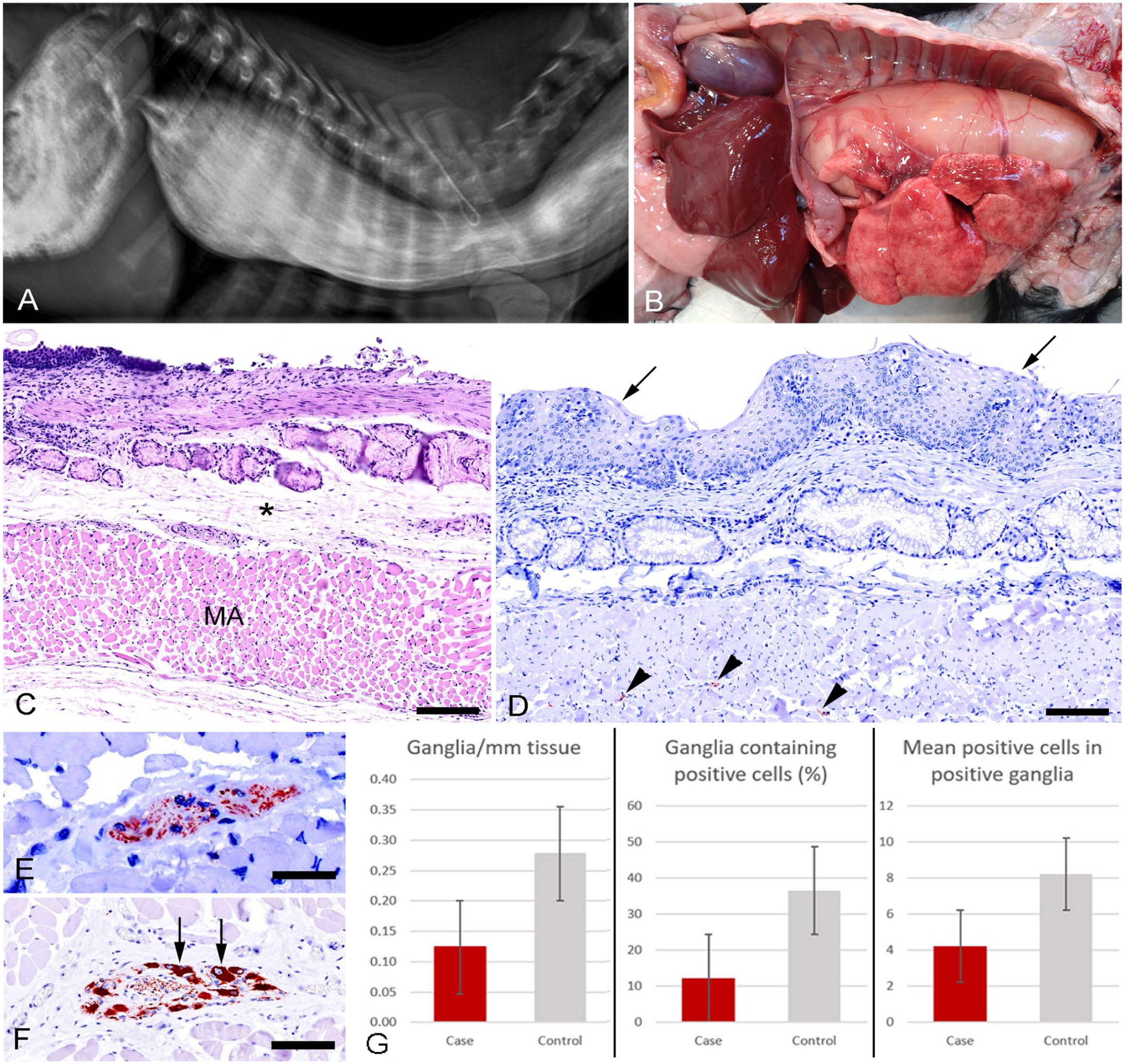
Morphologic characteristics of congenital idiopathic megaesophagus (CIM) in a 5-wk-old Gordon Setter puppy.
During autopsy, marked dilation of the entire esophagus was confirmed (Fig. 1B; Suppl. Figs. 2, 3), with ventral displacement of the heart and lungs. The animal had a body condition score slightly below average (WSAVA BCS 3 of 9). The rest of the autopsy was unremarkable. Several segments of the thoracic esophagus were fixed in 10% neutral-buffered formalin and processed for histopathology using standard protocols. By applying primary polyclonal anti-peripherin antibodies (PA316723 Invitrogen; Thermo Fisher), a protocol for the visualization of ganglion cells within the myenteric plexus of the esophagus was developed (Suppl. Text 1). Immunostaining was performed (UltraVision LP detection system HRP, Fisher; TL-125-HL, Epredia). In brief, sections were deparaffinized and pretreated in a microwave oven with buffered citrate (pH 6), before being blocked for endogenous peroxidase activity with 3% H2O2. A protein block (UltraVision) was then applied prior to incubation with the primary antibody (diluted 1:9,600 in 1% bovine serum albumin and Tris-buffered saline) at 4°C overnight. Post incubation, sections were treated with a primary antibody enhancer, large-volume HRP polymer, and 3-amino-9-ethylcarbazole (AEC; Vector). Sections were counterstained with Mayer hematoxylin and mounted on glass slides with coverslips using glycerol gelatin. A section of esophagus from a healthy young dog was used as a positive control, and the negative control included substitution of the primary antibody with a nonsense antibody (IgG X0903, Dako; diluted 1:192,000, to reach the same content of protein as the primary reagent).
Histopathology revealed generalized muscular atrophy or hypoplasia of the external muscle layers of the esophagus (Fig. 1C; Suppl. Fig. 5), together with moderate interstitial edema. In the epithelial layer, erosions and areas of squamous hyperplasia were present (Fig. 1D; Suppl. Fig. 4). Mononuclear inflammatory infiltrates were found in the submucosa below epithelial erosions (Suppl. Fig. 4). The ganglia of the myenteric plexus, located between the transverse and longitudinal external muscle layers, were reduced in number and size (Fig. 1E) compared to esophagus samples from a healthy dog (Fig. 1F; Suppl. Fig. 5), with segmental complete absence of peripherin-positive ganglion cells (segmental aganglionosis), identified by the intense staining of the cell cytoplasm. Ganglia were found to be farther apart (number of ganglia/mm was reduced by 55.5%), fewer were peripherin-positive (66.4% reduction in the percentage of positive ganglia), and the average number of ganglion cells within these positive ganglia was also greatly reduced (48.5% reduction; Fig. 1G). All measurements (i.e., number of ganglia and positive cells counted manually, and measurement of total tissue length with the polyline tool) were carried out with digital image software (v.0.4.3; QuPath 1 ). Measurements and counts were repeated 3 times and averaged before being used for further processing in Microsoft Excel.
Loss of enteric neurons in the myenteric plexus of the esophagus has been reported in the veterinary literature, albeit sporadically. In a study of a German Shepherd puppy, complete aganglionosis was associated with congenital esophageal achalasia, 5 and similar observations were found in a Friesian foal with megaesophagus. 2 Interestingly, in another study of megaesophagus in Friesian foals, the esophageal dilation was associated with marked caudal esophageal smooth muscle hypertrophy, but with no signs of myenteric plexus involvement, suggesting the presence of different pathogenic pathways for the development of the morphologic phenotype in Friesians. 9 Ganglionic cell loss has also been reported in association with megaesophagus in Rassf1a-null mice and a colony of transgenic rats, emphasizing their use as models of human achalasia.11,17 Achalasia (meaning “lack of relaxation”) is the human equivalent to megaesophagus, in which dysfunctional regulation of the lower esophageal sphincter and lack of progressive peristalsis causes decreased passage of food from the esophagus into the stomach, with resulting secondary esophageal dilation. 13 As for the veterinary counterpart, the exact pathogenesis for development of achalasia remains unknown, but is hypothesized to be caused by immune-mediated destruction of ganglionic structures following exposure to an environmental trigger (antigenic mimicry), which might explain the frequent findings of myenteric inflammation in combination with ganglion cell loss in this disease.6,13,14 The early onset and lack of myenteric inflammation associated with canine CIM suggests that the pathogenesis differs from that of human achalasia.
Another condition associated with complete or segmental aganglionosis in humans is congenital intestinal aganglionosis or Hirschsprung disease (HD), caused by a malformation of the enteric nervous system, presumably a result of incomplete craniocaudal migration of neural crest cells during fetal development. 8 A similar condition is known in horses, namely congenital colonic aganglionosis or lethal white foal syndrome (LWFS).10,16 Even though HD and LWFS affect another part of the gastrointestinal tract (distal rectum and a variable length of the neighboring intestinal tract), they share several pathologic characteristics with canine CIM, including the age at presentation and the non-inflammatory loss of ganglion cells within the myenteric plexus.
As emphasized by several authors, identification of ganglion cells within the myenteric plexus using H&E-stained slides, independent of the underlying disease, may prove difficult given the morphology of immature ganglion cells in neonatal tissues, which may be confused with endothelial cells, other mesenchymal cells, or even inflammatory cells.8,18 To accommodate for this challenge, and confirm the diagnosis of aganglionosis, several immunohistochemical techniques have been described in the literature. These include staining for calretinin, peripherin, neuron-specific enolase, neurofilament, and negative-image staining with S100 a.o.,8,12,18 but their use in esophageal pathology diagnostics is less well documented, and their application in animals has been limited to mice and rats.11,17
Our case may be the first description of the application of anti-peripherin antibody for visualization of ganglionic neuronal loss in association with esophageal disease in any species (no retrieved reports on a search of Google, Web of Science, and PubMed, using the search terms “peripherin” AND “immunohistochemistry”). The highly specific and intense cytoplasmic staining of ganglion cells, with only faint granular staining of the surrounding nerve fibers, makes peripherin immunohistochemistry a strong tool for histologic confirmation of canine CIM. The application of this particular antibody may in the future be extended to other species, and for the diagnosis of other animal neuropathies, including LWFS and grass-sickness in horses.
Supplemental Material
sj-pdf-1-vdi-10.1177_10406387241226950 – Supplemental material for Clinicopathologic evaluation of congenital idiopathic megaesophagus in a Gordon Setter puppy: a case report and development and application of peripherin immunohistochemistry for detection of ganglion cells
Supplemental material, sj-pdf-1-vdi-10.1177_10406387241226950 for Clinicopathologic evaluation of congenital idiopathic megaesophagus in a Gordon Setter puppy: a case report and development and application of peripherin immunohistochemistry for detection of ganglion cells by Cecilie B. Becker and Henrik E. Jensen in Journal of Veterinary Diagnostic Investigation
Footnotes
Acknowledgements
We thank the veterinary team at Odsherreds Animal Hospital (Asnæs, Denmark) for their clinical assistance with this case. Observations from this case were presented as a poster at the joint European Congress of Veterinary Pathology and Clinical Pathology, 31 Aug–2 Sep 2023, Lisbon, Portugal.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
