Abstract
Schwannosis is a rare, non-neoplastic, perivascular proliferation of aberrant Schwann cells within the CNS with simultaneous partial myelination of axons. A single report exists in veterinary medicine of schwannosis in the spinal cord of 3 foals and 1 calf. Here we describe a case of schwannosis in the brain of a 1-d-old Holstein-Friesian calf, submitted for autopsy due to arthrogryposis and premature death, with no other gross abnormalities observed. Histologically, the brain had multifocal, mainly perivascular, spindle-cell proliferations within the white matter of the medulla oblongata and focally within the gray matter of the midbrain. These cells immunolabeled with periaxin, myelin protein zero, SOX10, S100, and equivocally for vimentin, indicating Schwann cell origin. No changes were identified within other organs. Ancillary tests did not support an infectious etiology. Schwannosis should be considered as a differential diagnosis when investigating cases of arthrogryposis in calves with negative ancillary tests for infectious conditions.
A 1-d-old, female, Holstein-Friesian calf, from a herd of 224 animals, was submitted for postmortem examination to the Kilkenny regional laboratory of the Department of Agriculture, Food and the Marine (Republic of Ireland), with a history of arthrogryposis and premature death. The carcass was mildly autolytic and was of expected body condition for its age. No other gross abnormalities were seen at autopsy.
Fresh liver, lung, and brain tissue were collected and tested for infectious agents. The tissue was negative for bovine herpesviruses 1 and 4, bovine viral diarrhea virus (BVDV), Schmallenberg virus, Anaplasma phagocytophilum, Campylobacter fetus, Chlamydia abortus, Coxiella burnetii, Leptospira spp., and Listeria monocytogenes. The abomasal content was sterile on both anaerobic and aerobic culture. Samples of brain (coronal sections at the level of the frontal cortex, thalamus, hippocampus, midbrain, cerebellum, and medulla oblongata), cervical spinal cord, and portions of liver, lungs, kidneys, and heart were fixed in 10% neutral-buffered formalin and processed routinely; 4-µm sections were stained with H&E and Luxol fast blue and examined by light microscopy.
Formalin-fixed, paraffin-embedded (FFPE) tissue sections from the affected areas of the brain were used for immunohistochemistry (IHC) against periaxin, a marker of Schwann cells 17 (rabbit polyclonal anti-periaxin antibody, catalog HPA001868; MilliporeSigma); myelin protein zero (P0), a marker of peripheral nerve myelin and Schwann cells 13 (rabbit polyclonal anti-P0 antibody, catalog AB31851; Abcam); SOX10, a marker of neural crest 12 (rabbit monoclonal anti-SOX10 antibody, clone EPR4007-104, catalog ab180862; Abcam); S100, a protein that exhibits positivity throughout the nervous system, particularly in glial and Schwann cells8,13 (rabbit polyclonal anti-S100 antibody, catalog Z0311; Dako); and vimentin, an intermediate protein found in many mesenchymal cells, including neuroepithelial cells4,13 (mouse monoclonal anti-vimentin antibody, catalog M7020; Dako) using an automated IHC staining system (Bond Max; Leica), according to the manufacturer’s instructions and as described by others.10,13,15,18 A negative control omitting the primary reagent, as well as a positive control showing the normal pattern of distribution of the antigen in question, was run with each batch of slides.
Periaxin, expressed by myelinating Schwann cells, is crucial for peripheral myelination. 17 It initially localizes to the adaxonal membrane during myelin development and shifts to the abaxonal membrane as myelination matures. 17 P0, encoded by the MPZ gene, is the main structural component of peripheral myelin, primarily expressed by Schwann cells and essential for myelin formation. 7 SOX10 is a transcription factor used as a marker for neural crest origin cells, though it is also found in myoepithelial cells of various glands. 12 In veterinary species, SOX10 labels Schwann cells, autonomic ganglia, melanocytic neoplasms, and some non-neural crest cells. 15 SOX10 is almost always positive in neurofibromas, schwannomas, and most melanomas, indicating its utility in identifying tumors of Schwannian and melanocytic origin. 15 Proper SOX10 signaling is crucial for normal peripheral nervous system (PNS) myelination. 15 S100 proteins are calcium-binding proteins that are found in various cell types 8 and are present in nearly all nerve sheath tumors and some tumors with controversial origins. 8 S100 proteins have been linked to tumorigenesis and cancer progression across different cancer types. 8 Vimentin, a major type III intermediate filament protein, is crucial for cell migration, proliferation, and division. 4 It is overexpressed in many tumors, including brain tumors, in which high vimentin levels indicate poor prognosis. 4 Additionally, vimentin promotes Schwann cell migration and inhibits myelination. 4
Histologically, multifocally, in the midbrain (predominantly gray matter) and medulla oblongata (predominantly white matter), perivascular proliferations of 7–15-μm spindle cells minimally infiltrated the adjacent neuroparenchyma. The spindle cells had variably distinct cellular borders, a moderate amount of eosinophilic, fibrillar cytoplasm (Fig. 1A, 1B), and oval, central nuclei with finely stippled chromatin, and up to 4 nucleoli. No mitotic figures were evident. Anisokaryosis and anisocytosis were mild. The cells stained faintly blue with Luxol fast blue (Fig. 1C). No other histologic changes were noted within the CNS or other examined organs.
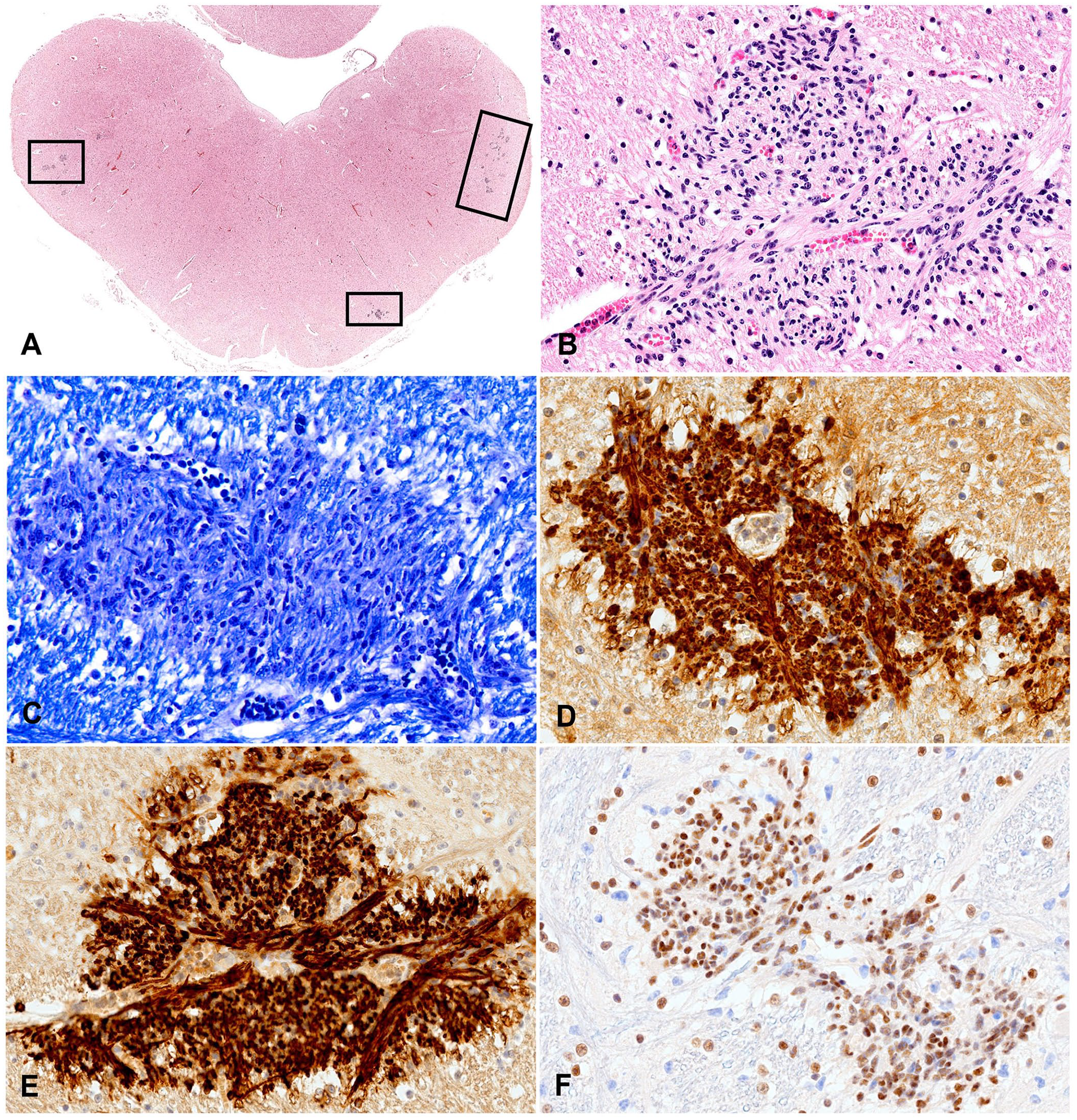
Schwannosis in the medulla oblongata of a 1-d-old calf.
All of the proliferating spindle cells had diffuse, granular cytoplasmic immunolabeling for periaxin (Fig. 1D), diffuse granular membranous and cytoplasmic immunolabeling for myelin P0 (Fig. 1E), equivocal immunolabeling for vimentin (data not shown), and granular intranuclear immunolabeling for SOX10 (Fig. 1F). The proliferating cells had intracytoplasmic immunolabeling with S100 (data not shown); as S100 immunolabels all CNS cells, the proliferating cells could not be distinguished from the adjacent neuroparenchyma with this marker. Based on the histologic features and immunolabeling pattern, a morphologic diagnosis of schwannosis was established.
Schwannosis is defined as an invasion and proliferation of ectopic or aberrant Schwann cells from the PNS to the CNS, with attempted myelination. 13 Schwannosis is a rarely described entity in the veterinary literature, hence clinical implications are not fully known. We retrieved one publication regarding schwannosis in animals in a search of Google, PubMed, CABI Direct, Web of Science, and Scopus, using search terms “schwannosis”, “Schwann cell proliferation”, and “perivascular Schwann cell proliferation’’, suggesting that this condition has only been reported once in a calf and 3 foals. 13 The authors reported 4 cases of spinal schwannosis with concurrent hydrocephalus in a calf, and with scoliosis, syringomyelia, syringohydromyelia, and small and asymmetric gyri and folia in 3 foals. 13 In our case, the calf was born with arthrogryposis and died soon after. No other pathologic changes were seen in any internal organs, and all ancillary testing targeting perinatal infections were negative.
Arthrogryposis is a term used to describe contractures of the joints in more than one area of the body, usually with bilateral symmetry, present at the time of birth. 5 A complex pathogenesis results in decreased fetal movement during a critical developmental window. 5 Causes of arthrogryposis in bovids include in utero infections with Aino virus, Akabane virus, bluetongue virus, BVDV, or Schmallenberg virus, as well as genetic causes and toxicity by alkaloid-containing plants ingested by the dam, such as seen with lupin (Lupinus sp.) toxicity.3,5 Our case was negative for BVDV and Schmallenberg virus on PCR; additionally, no gross changes, such as lissencephaly, hydranencephaly, porencephaly, or others suggestive of a viral cause of arthrogryposis, were noted. 3 As Ireland is under surveillance for bluetongue virus by the National Disease Control Centre (NDCC-considered free), this condition was unlikely. Histologically, neither perivascular cuffing nor demyelination were noted. Maternal toxicity as a cause of arthrogryposis was considered unlikely and was excluded, as the remaining calves from the herd were unaffected. It is possible that the schwannosis in our case could have led to some degree of fetal paralysis and arthrogryposis. There have been documented instances in humans in which brain malformations, or even the absence of malformations, are accompanied by arthrogryposis and future borderline intellectual function, 6 which has not been described in veterinary medicine.
A similarly named neoplastic condition, known as schwannomatosis, was initially linked with neurofibromatosis type 2 (NF2) in humans. 11 Schwannomatosis sets itself apart from NF1 and NF2 due to its minimal skin manifestations, potential for malignant transformation, and absence of bilateral vestibular nerve involvement. 9 NF2 is an autosomal dominant disorder with hyperplastic or hamartomatous lesions, benign proliferation of Schwann cells (resulting in schwannomas and schwannosis), meningothelial proliferation (leading to meningiomas and meningioangiomatosis), and glial proliferation (resulting in ependymomas and glial hamartomas). 16 In NF2 patients, schwannosis typically manifests in the spinal cord at dorsal root entry zones or within the parenchyma. 16 Conversely, in non-NF2 individuals, schwannosis may arise from infarction or trauma to the spinal cord, with trauma occurring in most cases of chronic spinal cord injury. 16 The condition has been described in humans with spinal cord injuries, particularly in individuals who survived >4 mo after the injury. 2 In normal conditions, Schwann cells should not be present within the CNS. It has been suggested that Schwann cells are introduced into the injured cord via a breach in the pial surface, potentially displaced from spinal roots, with the ability to proliferate within the affected area, particularly if gliosis was reduced. 2
Several hypotheses have been proposed to elucidate the presence of schwannosis lesions in the brain and spinal cord. One of the hypotheses is that these lesions could originate from ectopic peripheral Schwann cells, which may migrate to the CNS during development. 13 Initially, it was believed that Schwann cells, engaged in remyelinating CNS axons, were ectopic in nature, responding to demyelination signals by migrating from PNS sources into the CNS, possibly due to disruption of the glia limitans. 13
Alternatively, another theory states that Schwann cells might arise from the differentiation of resident brain oligodendrocyte progenitor cells. 20 In individuals with multiple sclerosis experiencing demyelination, there are instances of spontaneous myelin regeneration facilitated by oligodendrocytes and Schwann cells. 20 These cells can originate from adult oligodendrocyte progenitor cells. 20 In the cases of schwannosis reported in humans following spinal cord injury, it is speculated to be an unsuccessful attempt at neuronal remyelination. 2 In rats with spinal injury, Schwann cells access the dorsal funiculi via the dorsal root entry zone and the lateral funiculi via rootlets adhered to the lateral spinal cord after inflammation. 10 Some CNS remyelinating Schwann cells originate from the PNS; however, potentially, most stem from the differentiation of resident oligodendrocyte progenitor cells within the spinal cord. 13 This evidence suggests the existence of a native Schwann cell population within the CNS. 13 Most of the evidence suggests that schwannosis, characterized by the proliferation of Schwann cells, is of reactive etiology, often occurring as a response to nerve injury or chronic inflammation. 13
A variety of different Schwann cell disorders have been described in human and veterinary medicine ranging from hyperplasia to neoplasia. Schwannomas are reported in many animal species and usually exhibit positive immunolabeling for S100 and vimentin; some cases also have a point mutation in the neu oncogene. 18 Among unusual cases in veterinary medicine is a cluster of malignant schwannomas in cattle in Argentinian Patagonia, between 1998 and 2001, suggesting an unidentified virus, 14 cutaneous triton tumor (with rhabdomyosarcomatous differentiation) in a cat, 19 and a subcapsular schwannoma with bone differentiation in a dog. 1
The cause of schwannosis in our case remains unknown. A growing availability of histochemical stains and immunohistochemical markers, such as those that we used (myelin P0, periaxin, SOX10, S100, and vimentin) may increase our understanding of this condition.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
