Abstract
HistoGel™ is an aqueous specimen-processing gel that encapsulates and suspends histologic and cytologic specimens in a solidified medium. HistoGel-embedded specimens can then be processed and evaluated by routine histologic and immunohistochemical methods. This methodology has been used in human diagnostic pathology and is especially useful for small, friable, or viscous tissue samples that are difficult to process. In addition, special histochemical stains or immunohistochemistry can be performed on HistoGel-embedded cytologic specimens using standardized methods developed for histopathology. The current report describes several applications for HistoGel, including use with cytologic specimens, bone marrow aspirates, retention of tissue orientation for endoscopic biopsy specimens, and evaluation of friable tissues. Samples were encapsulated in HistoGel, fixed in 10% neutral buffered formalin, routinely processed, paraffin embedded, and sectioned for histochemical and immunohistochemical evaluation. The results of this study support the use of HistoGel in veterinary diagnostic pathology.
Processing small or delicate specimens and exudates can be one of the many challenges in diagnostic veterinary pathology. HistoGel™ a is an inert aqueous processing gel that can be used to encapsulate a variety of cytologic specimens and unfixed or formalin-fixed tissues in a solidified agar-like medium prior to processing.
Evaluation of cytologic specimens is routinely performed on aspirates, direct smears, and touch prep applications using prototypical staining techniques, such as Romanowsky stains. However, there are relatively few special staining techniques available for routine cytology, thus limiting the phenotypic characterization of cytologic preparations. For example, lymphoma is routinely diagnosed via cytology; however, immunocytochemistry or surgical pathology with subsequent immunohistochemistry is necessary for immunophenotypic characterization of the neoplastic cell population. HistoGel is a readily available media that can be used for double embedding techniques to incorporate cytologic specimens into a solid medium. In smaller or hypocellular samples, cell blocks can be readily prepared from specimens with singly scattered loose cells using conventional centrifugation. 3 Double embedding provides improved tissue support such that specimens can be processed as a routine histologic sample for further histochemical and immunohistochemical evaluation. HistoGel double embedding methodology and immunohistochemistry have been previously used on peripheral blood samples in people with chronic lymphocytic leukemia (CCL).1,3 In a previous study, 1 HistoGel cell blocks were used to evaluate MUM1/IRF4 expression in peripheral blood CCL cells. While MUM1/IRF4 expression was variable, the study demonstrated that surface antigens expressed on circulating leukemic cell lines could be identified in HistoGel-prepared specimens.
The present study describes the use of HistoGel for encapsulating a variety of fresh and formalin-fixed tissues, as well as highly cellular cytologic, fluid samples. This strategy helps to maintain the integrity of friable sample and orientation of small tissues during processing. Cytologic specimens can retain elements of tissue architecture that are often lost in standard cytologic preparations and are amenable to standard methods for immunostaining. The intent of the current study was to support the use and explore potential applications of HistoGel in veterinary diagnostic pathology.
Multiple tissues were collected from a 2-year-old, male Domestic Shorthair cat that presented for postmortem examination following postoperative anesthetic-related death. Collected tissues included fragments of nasal turbinates, sections of gastrointestinal tract, and cerebellum. These representative tissues are frequently encountered during routine surgical biopsy service and may be small or extremely friable and therefore more labor intensive to process and section. Five sections of each tissue were collected using endoscopic biopsy forceps to simulate samples that are often encountered in surgical pathology submissions. Two sections of each tissue were fixed by immersion in 10% neutral buffered formalin for 24 hr prior to encapsulating the tissues into HistoGel. Two sections of each tissue were directly embedded into the gel before fixation, and the fifth section of each tissue was routinely processed without the addition of HistoGel. HistoGel was prepared following the manufacturer’s recommendations. Briefly, HistoGel is a solid at room temperature and must be liquefied for use by heating to 50ºC in a water bath. The liquefied HistoGel was placed in a sterile, flat, 12 mm in diameter polyethylene centrifuge tube cap and allowed to cool to a semi-solid state at room temperature. All tissues were embedded in the gel with appropriate orientation, and additional liquefied HistoGel was added to completely encapsulate the tissue. The samples were then refrigerated at 4°C for 5 min to completely solidify the embedded sample. HistoGel-encapsulated samples were transferred to a processing cassette. Unfixed tissues were fixed by immersion in 10% neutral buffered formalin for 24 hr prior to embedding in HistoGel. Following fixation, HistoGel-encapsulated tissues were routinely processed, paraffin embedded, sectioned, and stained with hematoxylin and eosin for evaluation.
The cytologic specimen evaluated in the present study was fluid from traumatic catheterization of a dog with transitional cell carcinoma. Half of the fluid was prepared as a cytologic direct smear on a glass slide, allowed to air dry, and stained with modified Wright stain. The remaining portion of the fluid from the traumatic catheterization was embedded in HistoGel as follows: 1.5 ml of sediment prepared from the fluid was combined with an equal volume of liquefied HistoGel. Samples were gently mixed to distribute cells evenly within the gel matrix prior to solidification. HistoGel-encapsulated samples were transferred to a tissue processing cassette and fixed by immersion in 10% neutral buffered formalin for 24 hr. Following fixation, the sample was routinely processed and evaluated as described previously.
To demonstrate the retention of immunoreactivity in HistoGel-embedded specimens, sections of nasal turbinate, gastrointestinal tract, and cerebellum were examined im-munohistochemically with a polymer detection kit b using an autostainer c and commercially available antisera for cytokeratin, d glial fibrillary acid protein, e muscle specific actin, f and vimentin g with appropriate control tissues for each antibody (Table 1). HistoGelembedded sections of the fluid sediment were immunolabeled with commercially available antisera for cytokeratin and vimentin. Table 1 summarizes the immunohistochemical antibodies and antigen retrieval technique.
Antigens and immunohistochemical protocols used in the current study.*
HIER = heat-induced epitope retrieval. Source of antigens and HIER: Dako North America Inc., Carpentaria, CA.
Both fresh and formalin fixed sections of nasal turbinates, gastrointestinal tract, and cerebellum were found to be easily embedded into HistoGel with preservation of tissue architecture and proper orientation. All tissues were fixed in formalin for 24 hr prior to processing; however, fixation time may vary with tissue type and should be evaluated on an individual basis. In all instances, tissues were easily sectioned, and there was excellent retention of cell morphology and immunoreactivity with several commonly used immunohistochemical antibodies. These results are consistent with previous studies that evaluated bone marrow aspirates in dogs with hematopoietic tumors (presented at the 29th annual meeting of the Veterinary Cancer Society, Austin, Texas, 2009). During this previous study, bone marrow aspirates from 21 dogs with hematopoietic tumors were evaluated for clinical staging. Results demonstrated that bone marrow aspirates could be readily embedded into HistoGel and routinely processed as a histologic specimen. Phenotypes of neoplastic cell lines were further characterized by immunohistochemistry using the same methodology applied to staining of tissue sections.
There were no histocytologic differences in tissues placed in HistoGel prior to fixation or following fixation when compared to tissues that were not placed in HistoGel. However, embedding fresh tissues in HistoGel prior to fixation increased preservation of friable specimens, and proper anatomic orientation was easier. In addition, while there were no differences in tissue processing techniques including processing times, histochemical staining, or immunohistochemical reactivity, double embedding friable and small tissues in HistoGel as compared to non-embedded samples provided improved tissue support and sectioning qualities. In some instances (i.e., sections of cerebrum and gastrointestinal tract), specimens that were embedded in HistoGel were easier to section compared to standard, non-HistoGel–embedded tissue.
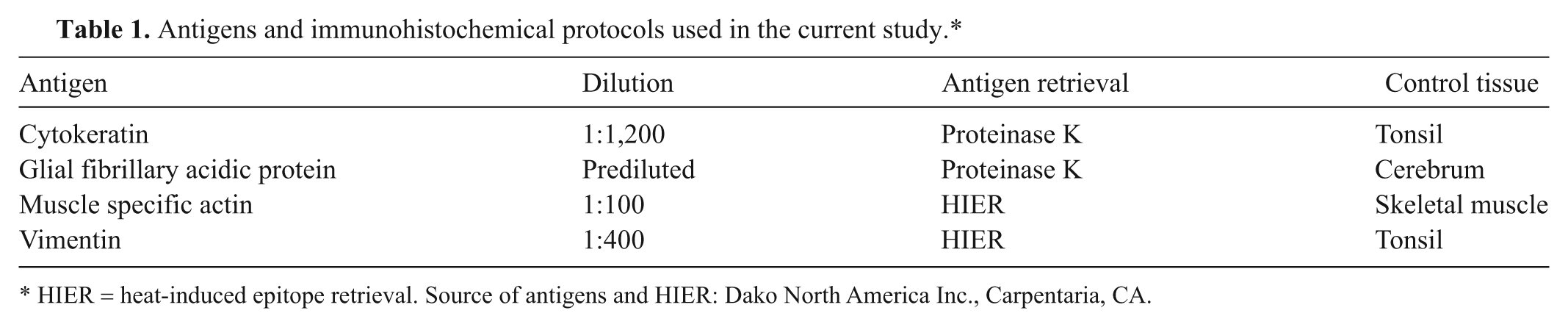
Tissues with high lipid content or those that are heavily myelinated (i.e., brain and spinal cord) are extremely friable and often fragment with tissue handling, processing, and sectioning. In the present study, sections of cerebellum were collected and placed in HistoGel prior to fixation in order to evaluate preservation of tissue integrity in friable specimens. The encapsulated sections of cerebellum were completely fixed in formalin within 24 hr and remained intact during processing, embedding, and sectioning. Histologic sections of embedded cerebellum were well preserved with excellent tinctorial contrast when embedded in HistoGel before or after fixation in 10% neutral buffered formalin as compared to non-HistoGel–embedded sections (Fig. 1). The non-HistoGel–embedded sample required 10 additional serial sections to achieve nonfragmented sections; however, nonfragmented sections were readily obtained with a single section of those samples embedded in HistoGel.
Cerebellum; cat. There is excellent retention of cerebellar architecture with preservation of cell morphology and tinctorial properties in all treatments. Note that fragmented samples of cerebellum were retained in HistoGel™ encapsulation matrix and not lost upon sectioning. Additional sectioning of the samples that were not embedded in HistoGel was required for comparable results. a, sections of cerebellum were encapsulated in HistoGel prior to fixation. Hematoxylin and eosin (HE). Bar = 100 µm. b, sections of cerebellum were encapsulated in HistoGel following fixation. HE. Bar = 100 µm. c, sections of cerebellum were not embedded in HistoGel. HE. Bar = 100 µm.
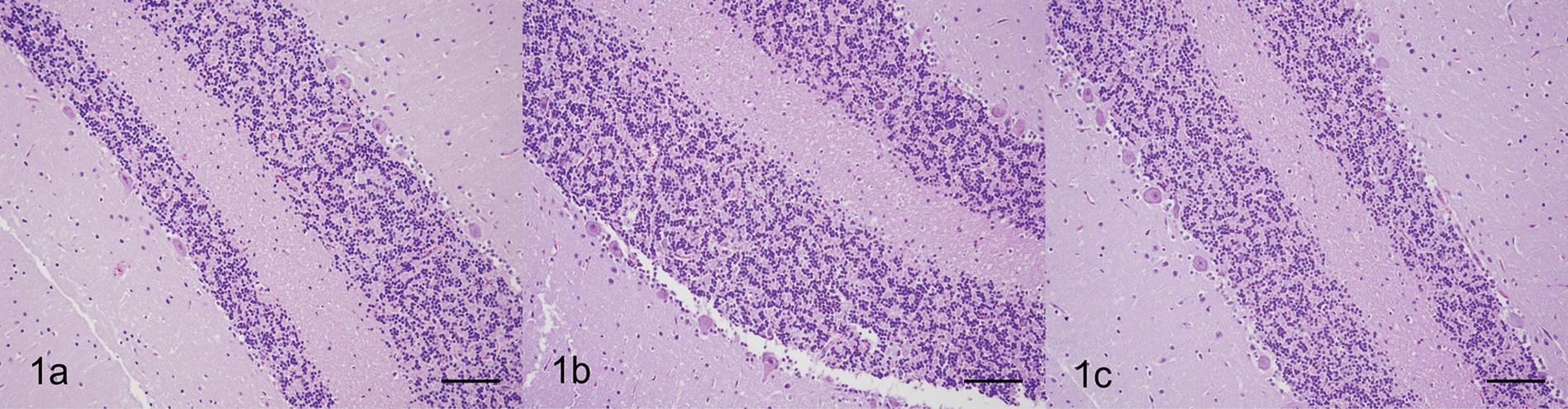
Thin, delicate tissues, such as nasal turbinates, may be difficult to orient when embedding directly into paraffin and tend to readily fragment during sectioning. Incorporating the tissue into HistoGel prior to processing allows proper orientation and provides improved tissue support for sectioning. Sections of fresh and formalin-fixed nasal turbinates were easily embedded into HistoGel, preserving tissue orientation and maintaining mucosal integrity, thus validating the use of HistoGel in delicate specimens (Fig. 2). Ciliated mucosal epithelial cells were distinct, and strong immunoreactivity with cytokeratin was maintained in the mucosal epithelium.
Nasal turbinates; cat. Thin, delicate nasal turbinates were easily oriented into HistoGel™, preserving tissue orientation and maintaining mucosal integrity that can be lost during processing of small, friable tissues. Formalin-fixed sections of nasal turbinates that were not embedded in HistoGel appeared similar histologically, but were more difficult to orient and embed during processing. a, sections of respiratory tract were encapsulated in HistoGel following fixation. Hematoxylin and eosin (HE). Bar = 100 µm. b, sections of respiratory tract were encapsulated in HistoGel prior to fixation. HE. Bar = 100 µm. c, sections of respiratory tract were not embedded in HistoGel. HE. Bar = 100 µm.
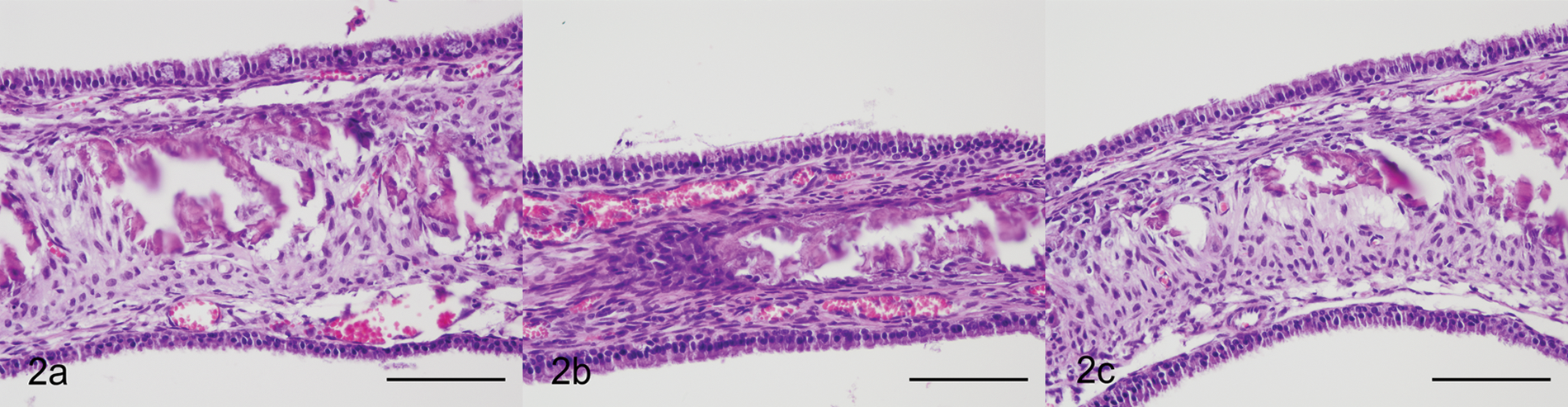
Endoscopic biopsies of the gastrointestinal tract are commonly encountered in diagnostic surgical pathology. Anatomic orientation can be difficult when dealing with small endoscopic-guided biopsy samples (i.e., gastrointestinal or respiratory tract). Proper orientation is necessary to evaluate the villus-to-crypt ratio, and the mucosal epithelium lining the villus tips is extremely delicate. Full-thickness sections of unfixed intestine were directly embedded into HistoGel following collection to demonstrate a potential intraoperative use for maintaining tissue orientation and preservation of tissue architecture. Proper orientation of the unfixed sections of gastrointestinal tract was readily achieved and maintained in HistoGel (Fig. 3). The villus-to-crypt ratio could be assessed, and the mucosal integrity was intact.
Gastrointestinal tract; cat. Proper orientation of small unfixed sections of gastrointestinal tract was readily achieved and maintained in HistoGel™. a, sections of gastrointestinal tract were oriented and encapsulated in HistoGel prior to fixation. Hematoxylin and eosin (HE). Bar = 100 µm. b, sections of gastrointestinal tract were oriented and encapsulated in HistoGel following fixation. HE. Bar = 100 µm. c, sections of gastrointestinal tract were not embedded in HistoGel. HE. Bar = 100 µm.
Perhaps the most striking potential for HistoGel in veterinary diagnostic pathology involves a multitude of cytologic applications. While routine cytology provides tremendous insight into etiologies and disease mechanisms, limitations exist with regard to the amount of diagnostic information that can be deduced from a cytologic specimen. Preparation of the cytologic samples embedded in HistoGel was rapid, and no special equipment was required. Once embedded into HistoGel, samples were fixed in formalin and processed exactly as for tissue biopsies, and no special handling of the sections was required for immunostaining. The use of HistoGel for cytologic samples may require additional sample preparation compared to strategies described for immunocytochemistry. 2 However, a principal advantage of HistoGel-encapsulated cytologic specimens over routine cytology is the ability to further evaluate and phenotypically classify cell types using an extended array of histochemical and immunohistochemical techniques. Moreover, additional sections can readily be obtained from cytologic samples in HistoGel. With the continuing evolution of clinical treatments directed toward specific cellular phenotypes (i.e., neoplastic cell lineages), immunohistochemical categorization of cell types is critical. HistoGel-encapsulated cytologic specimens address this dilemma by functionally transforming a cytologic sample into a solid media that can be processed as a routine histologic sample. Special histochemical stains and immunohistochemical antibodies can then be applied to the sample using routine procedures that have been developed and standardized for histopathology.
An additional consideration is retained stromal architecture in HistoGel-embedded cytologic specimens. It was found that in contrast to the conventional cytologic sample, sections of the fluid sample that were encapsulated in HistoGel maintained some stromal integrity, as large sheets of neoplastic transitional epithelium were organized around a fibrovascular stroma (Fig. 4). This allows the pathologist to assess the interaction between the neoplastic cell population and the neighboring tissue, which is very important when considering localized and vascular invasiveness that is frequently associated with several neoplasms. Because collection of cytologic samples is relatively noninvasive, HistoGel-encapsulated cytologic specimens may provide the diagnostician with additional information with minimal patient discomfort as compared to surgical biopsy procedures.
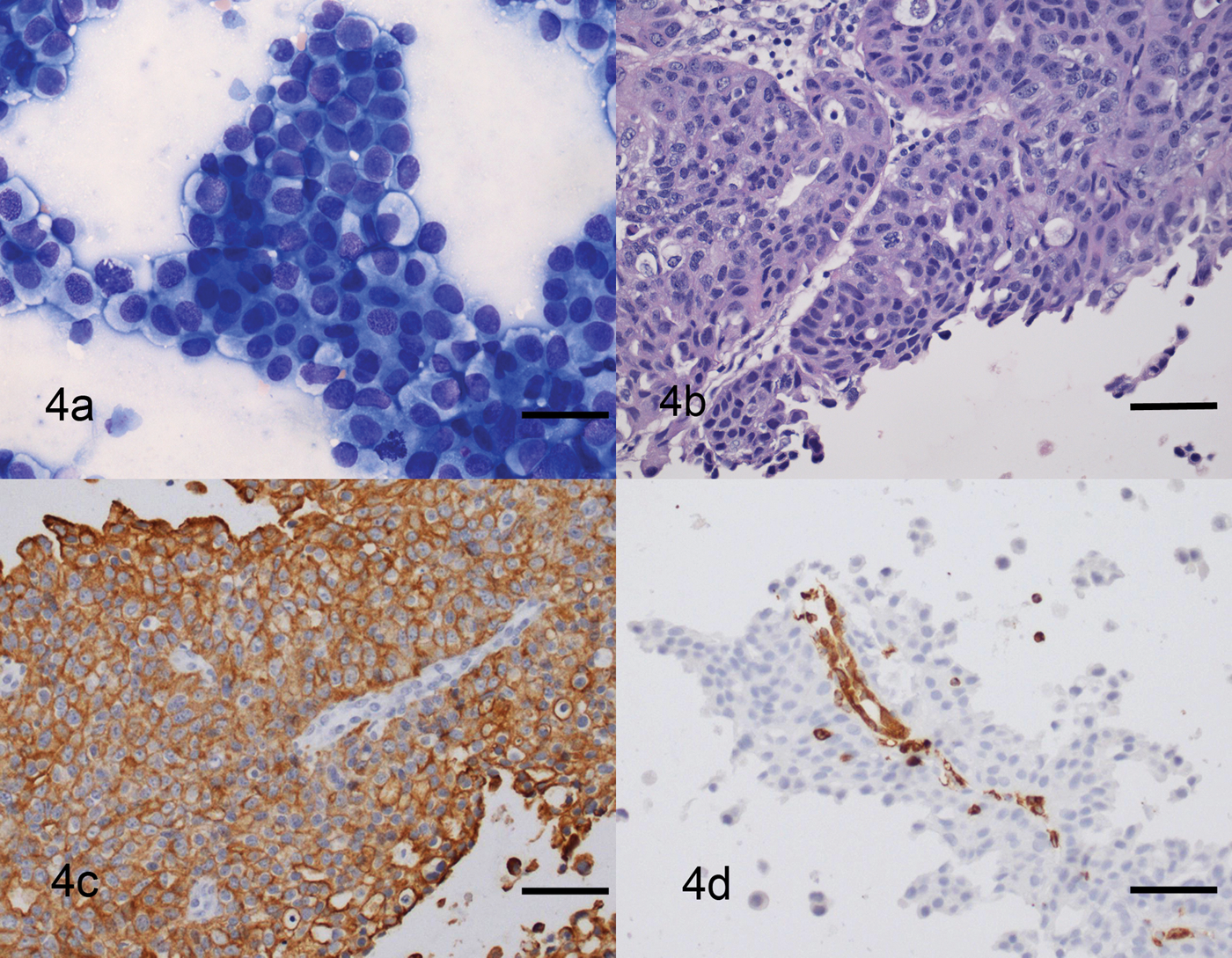
Sediment made of fluid from traumatic catheterization of the urinary bladder; dog. a, smear made from the sediment is abundantly cellular and contains many large sheets of epithelial cells that show moderate anisocytosis and anisokaryosis. Binucleated cells and mitotic figures are occasionally seen. Modified Wright stain. Bar = 100 µm. b, sections made of the fluid sediment encapsulated in HistoGel™ show many sheets of neoplastic epithelial cells that are organized around a fibrovascular stroma. Hematoxylin and eosin. Bar = 100 µm. c, neoplastic cells are strongly immunoreactive with cytokeratin. Bar = 100 µm. d, vascular endothelial cells are strongly immunoreactive with vimentin. Dual link system. Hematoxylin counterstain. Bar = 100 µm.
The findings also illustrate the use of immunohistochemical antibodies in embedded cytologic specimens. Neoplastic transitional epithelium was strongly reactive with cytokeratin, and the vascular endothelial cells were strongly immunoreactive with vimentin (Fig. 4). In addition, encapsulated sections of cerebellum showed strong immunoreactivity with glial fibrillary acid protein whether fixed in formalin prior to or after embedding in HistoGel (Fig. 5). Similar immunoreactivity to cytokeratin and muscle-specific actin was present in processed sections of nasal turbinates and gastrointestinal tract, respectively (data not shown). Optimized techniques were identical to those used for standard histologic specimens. Because some laboratories are limited to immunohistochemistry and do not perform immunocytochemistry, this technique provides an additional diagnostic tool.
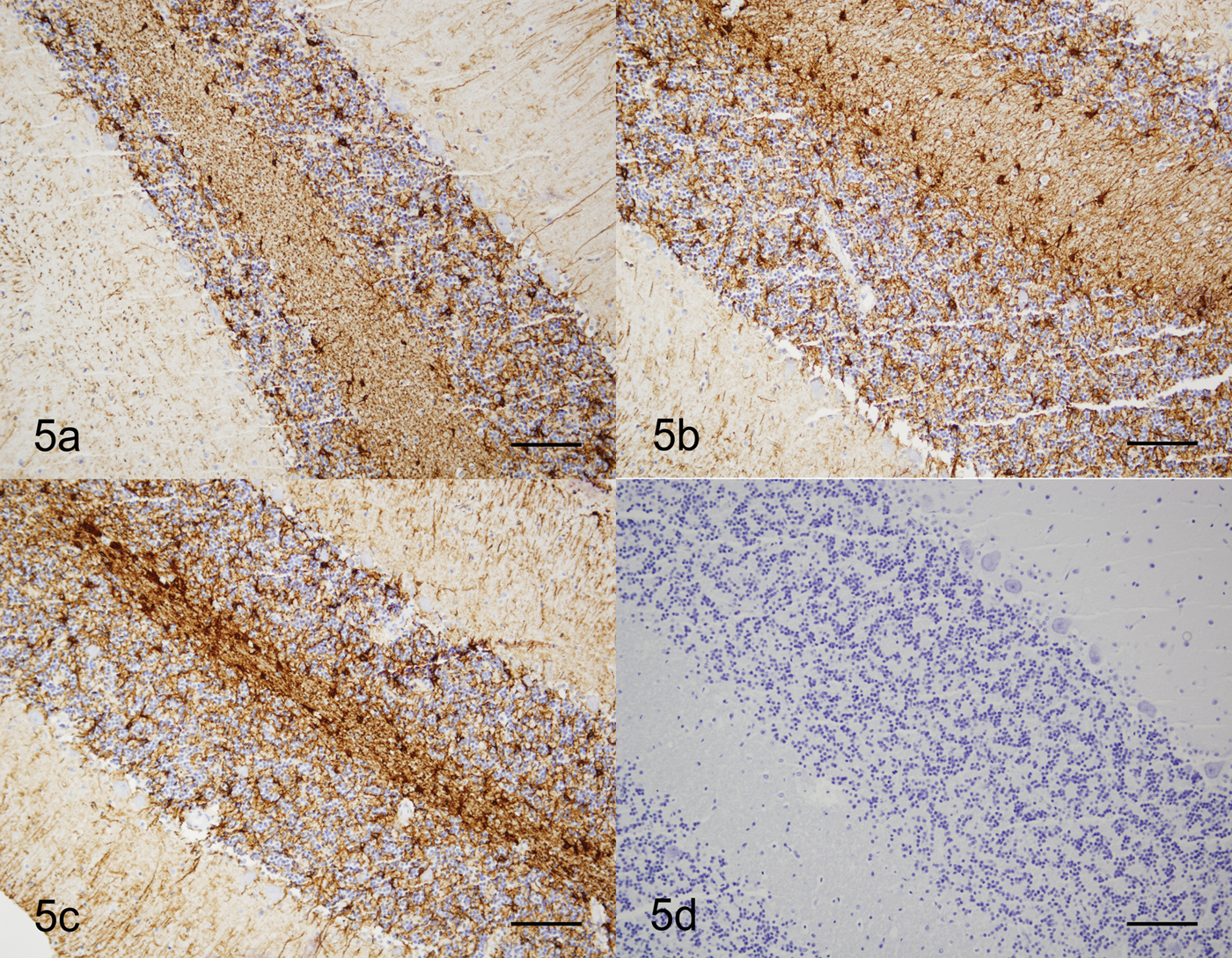
Cerebellum; dog. Samples show strong positive immunoreactivity with glial fibrillary acidic protein. a, sample embedded in HistoGel™ prior to fixation. Bar = 100 µm. b, sample embedded in HistoGel following fixation. Bar = 100 µm. c, sections of cerebellum were not embedded in HistoGel. Bar = 100 µm. d, negative control. Dual link system. Hematoxylin counterstain. Bar = 100 µm.
In summary, the current study demonstrates the successful application of HistoGel to encapsulate a variety of tissues, preserving fluid samples that are traditionally amendable only to cytology, maintaining the integrity of friable samples and appropriate anatomic orientation of small tissues. The results validate the utility of HistoGel in routine veterinary diagnostic cytology and histopathology. Both fresh and formalin-fixed tissues can be embedded into HistoGel with no detectable differences in histologic appearance. This suggests that HistoGel tissue encapsulation can be performed by the surgeon prior to submission such that anatomic orientation of tissue margins is maintained and pathologist reporting of margins correlates to the surgeon’s reference point. Due to the additional labor embedding samples in HistoGel, it is only recommended in cases in which orientation of small samples is critical or for cytologic procedures in which multiple tests (e.g., immunocytochemistry, histochemical stains) will be performed.
Footnotes
Acknowledgements
Part of this study was presented as a poster at the 60th annual meeting of the American College of Veterinary Pathologists in Monterey, California, December 2009. The authors thank the Auburn University College of Veterinary Medicine Diagnostic Histopathology Laboratory and Clinical Pathology Laboratory staff for technical assistance.
a.
HistoGel™, Richard-Allan Scientific, Kalamazoo, MI.
b.
Dual Link System-HRP Envision+ (catalog no. K4061), Dako North America Inc., Carpentaria, CA.
c.
Dako Universal autostaining system (model no. LV-1), Dako North America Inc., Carpentaria, CA.
d.
Cytokeratin (catalog no. IR053), Dako North America Inc., Carpentaria, CA.
e.
Glial fibrillary acidic protein (catalog no. SK200), Dako North America Inc., Carpentaria, CA.
f.
Muscle specific actin (catalog no. IR700), Dako North America Inc., Carpentaria, CA.
g.
Vimentin (catalog no. IR630), Dako North America Inc., Carpentaria, CA.
h.
Heat induced epitope retrieval, target retrieval solution, Pascal S2800, Dako North America Inc., Carpentaria, CA.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
