Abstract
Canine granulomatous colitis (histiocytic ulcerative colitis) is an uncommon disease, predominantly of young French Bulldogs and Boxer dogs, that manifests from a dysregulated immune response, primarily to adherent-invasive Escherichia coli (AIEC). In conjunction with histopathology and periodic acid–Schiff staining, the diagnosis of granulomatous colitis currently relies on fluorescence in situ hybridization (ISH) or immunohistochemistry to identify and localize AIEC organisms within macrophages in the mucosa and/or submucosa. We investigated the utility of ISH for E. coli using formalin-fixed, paraffin-embedded specimens collected from 29 cases of suspected granulomatous colitis. Most confirmed cases of granulomatous colitis were in French Bulldogs (12 of 20; 60%) and Boxers (3 of 20; 15%), and the mean age was 25 ± 6 mo with no sex predilection. E. coli ISH signal localized bacterial genetic material within the mucosa in 20 of 29 (69%) cases, supporting the diagnosis. ISH signal was limited to the lumen in 8 of 29 (28%) cases, which did not support the identification of these organisms as AIEC. The remaining case had no hybridization signal, and the diagnosis of granulomatous colitis was not supported. Our results revealed that ISH is a quick and specific detection method that can effectively confirm the diagnosis of canine granulomatous colitis.
Granulomatous colitis (GC), also known as histiocytic ulcerative colitis, is an uncommon, severe manifestation of inflammatory bowel disease seen predominantly in young Boxer dogs and French Bulldogs.3,15 Clinically, these dogs exhibit weight loss, hypoalbuminemia, and hematochezia. Although GC is occasionally seen in other breeds, the marked predilection of Boxers and French Bulldogs suggests an immune-mediated component. Microscopically, erosive and/or ulcerative lesions with a mixed inflammatory population are dominated by macrophages that often contain intracytoplasmic periodic acid–Schiff (PAS)-positive material.3,16 Definitive diagnosis of this entity requires the identification of numerous PAS-positive macrophages within the lamina propria or deeper layers of the intestine. These PAS-positive macrophages have been identified as CD204+, lysozyme−, and inducible nitric oxide synth-ase (iNOS)-rich macrophages, suggesting transformation of recruited monocytes. 12 Through histochemical and ultrastructural evaluation, the PAS-positive material within macrophages has been determined to be phagocytized bacteria and bacterial phospholipids. 16
Because of the clinical, gross, and histologic features of canine GC, this disease was originally compared to Whipple disease and Crohn disease in people, both of which have a bacterial component.13,15 After decades of investigation into an underlying cause, adherent-invasive Escherichia coli (AIEC) has been implicated as the cause of canine GC.6,7,9,10,13,16 Interestingly, AIEC is the same pathotype that has been associated with Crohn disease. 4 Moreover, the histologic features and breed predilection suggest that canine GC is the manifestation of an aberrant or exorbitant immune response to AIEC, similar to Crohn disease. 4 Given that AIEC has been implicated as the primary pathogen involved in canine GC, current testing relies on a combination of bacterial culture, fluorescence in situ hybridization (FISH), or immunohistochemistry (IHC) to detect this organism in the affected tissues.2,8,13 FISH is a time-consuming and potentially costly test and may not always be practical in a diagnostic setting. We investigated the potential use of in situ hybridization (ISH) for E. coli in cases of canine GC to further identify the causative agent.
We performed a retrospective search of the biopsy database of the New York State Animal Health Diagnostic Center (AHDC; College of Veterinary Medicine, Cornell University, Ithaca, NY, USA) between 2021 and 2022 using the following terms: canine, granulomatous colitis, ulcerative colitis. We retrieved from the database 29 cases that met the search criteria and further reviewed these cases for the appropriate histologic features. Twenty-one (72%) of the selected cases were sent for a second opinion with a limited number of unstained slides, and H&E staining was not performed (i.e., cases were submitted solely for ISH analysis). Five (17%) cases had PAS staining performed at another institution, and PAS staining was performed on 16 (55%) additional cases. Aerobic culture 13 was performed in-house in 2 cases. Culture results from the submitting institution were included in 2 case submissions, without information regarding culture methods.
An ISH probe for E. coli was designed and validated (RNAscope; Advanced Cell Diagnostics [ACD]). Controls consisted of an E. coli culture–positive intestinal section from a calf and a case of canine GC confirmed with aerobic culture. ISH was performed at the AHDC using a standard protocol and an automated ISH research platform (Discovery Ultra; Roche). 1 After deparaffinization and target retrieval (760-248, mRNA sample prep kit; Roche), 4-μm tissue sections mounted on charged slides were hybridized with 200 μL of ISH probe (433299, RNAscope 2.5 VS Probe- B-E. coli-16SrRNA [Escherichia coli KLY complete genome 16S ribosomal RNA]; ACD). This was followed by signal amplification (760-236, mRNA RED probe amplification kit; Roche), detection (760-234, mRNA RED detection kit; Roche), and counterstaining with hematoxylin. All slides included negative control tissue from a sample of bovine lung with suspect toxic diffuse alveolar disease from which no bacterial organisms were isolated with aerobic culture nor observed histologically. ISH with this probe was performed on 3 samples of histologically normal colon as an additional negative control. ISH for Canis lupus familiaris peptidyl-prolyl isomerase B (PPIB; 437449, RNAscope 2.5 VS Probe- Cl-PPIB, ACD), a reference gene, was performed on 3 selected cases (6, 7, 29) to confirm RNA integrity in the samples. Slides were scanned on a Ventana DP 200 slide scanner (Roche) for digital evaluation.
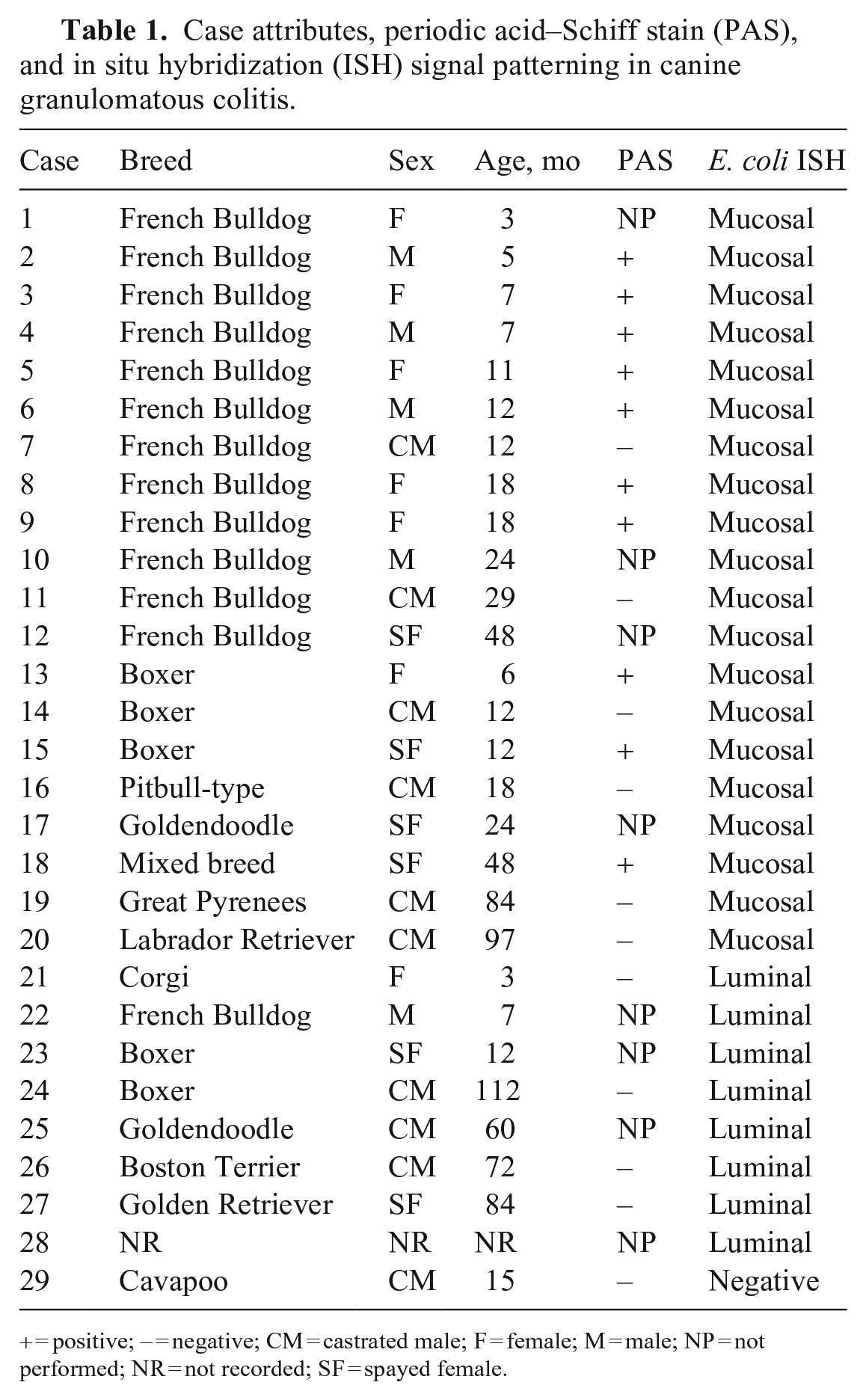
Most of the confirmed cases were in dogs with the expected signalment, with the list predominated by young French Bulldogs and Boxers (Table 1). The remaining confirmed cases included Goldendoodle, Great Pyrenees, Labrador Retriever, and Pitbull-type dogs. The mean age of dogs with confirmed GC was 25 ± 6 mo, and the median age was 15 mo. Of the 20 confirmed cases, there were 4 intact males, 6 castrated males, 6 intact females, and 4 spayed females. Aerobic culture was performed on specimens from 4 cases.
Case attributes, periodic acid–Schiff stain (PAS), and in situ hybridization (ISH) signal patterning in canine granulomatous colitis.
+ = positive; – = negative; CM = castrated male; F = female; M = male; NP = not performed; NR = not recorded; SF = spayed female.
Histologically, the lamina propria of affected colonic specimens had a robust infiltrate predominated by sheets of macrophages, with abundant intracellular material, intermixed with fewer lymphocytes and plasma cells (Fig. 1). The mucosa was multifocally eroded and/or ulcerated, and the inflammatory infiltrate in these regions was accompanied by variably degenerate neutrophils. Twenty-one cases (72%) had PAS results (Table 1). All 10 cases with PAS positivity had positive mucosal ISH signal. All 5 ISH-negative cases were PAS-negative. Of cases with PAS results, 6 of 16 (38%) ISH-positive cases had no PAS staining. In positive cases, the PAS stain highlighted brightly staining intracellular material within macrophages in the lamina propria, which is the characteristic histologic feature of GC.
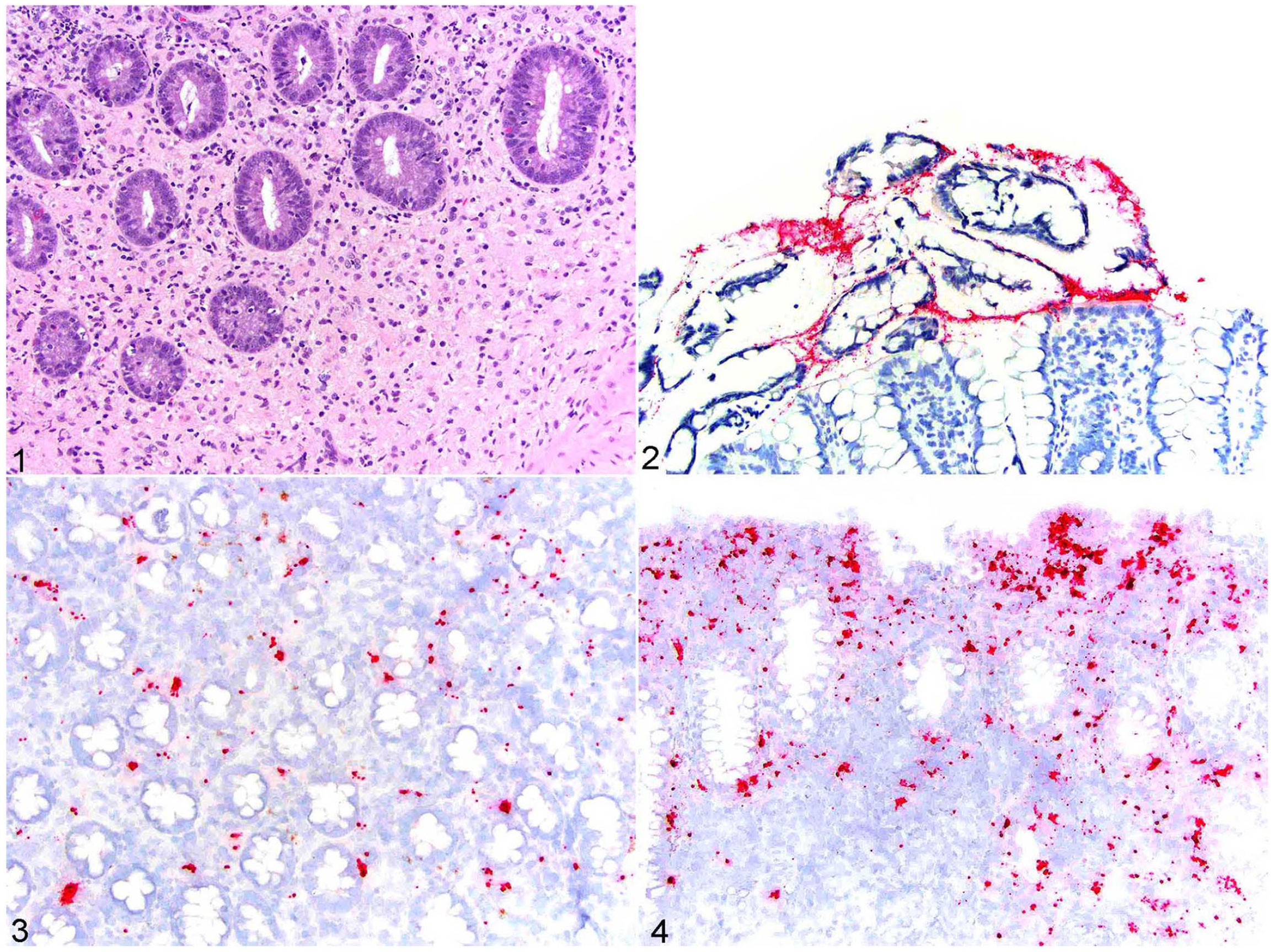
Escherichia coli in situ hybridization (ISH) in canine granulomatous colitis.
The ISH signal for E. coli was detected in 28 of 29 (97%) specimens (Table 1). Hybridization signal was restricted to the luminal material in 8 of 28 (30%) cases (Fig. 2). The signal was localized to the mucosal inflammation in 20 of 28 (71%) cases (Figs. 3, 4). ISH signal was limited to the lumen in 3 of 3 (100%) cases of histologically normal colon. RNA integrity was confirmed in 3 of 3 (100%) randomly selected cases with canine PPIB ISH. In 2 cases with positive mucosal signal, E. coli or coliform bacteria without further specification were isolated on culture. In one case with luminal signal, E. coli and Klebsiella pneumoniae were isolated on culture. In one case with no positive signal, E. coli and Campylobacter upsaliensis were isolated on culture.
Our study confirms that automated E. coli ISH is an excellent ancillary test to augment the diagnosis of AIEC-associated GC in the dog in formalin-fixed biopsy specimens. Positive E. coli ISH results, characterized by mucosal hybridization signal, confirmed the diagnosis of GC in 20 cases. A critical component in the diagnosis of AIEC-induced GC is the localization of E. coli within the mucosal epithelium and macrophages, given that this is the phenotypic colonization pattern of this pathotype. As reflected in the limited culture results from the cases in our study, culture alone is not sufficient to diagnose GC because E. coli can be isolated from cases with strictly luminal hybridization signal or from cases that completely lack hybridization signal. ISH allows localization of AIEC in the colonic mucosa as with IHC, but the specificity of ISH avoids the cross-reactivity seen with IHC.8,11 Similarly, ISH allows evaluation with a light microscope and accessible whole-slide scanning, unlike FISH, which requires a specialized fluorescence microscope and/or fluorescent-capable slide scanner that may not be readily available in a diagnostic pathology setting. Lastly, ISH represents an automated, rapid test that can be performed on formalin-fixed tissues. In some cases of GC, similar to Crohn disease in people, regional lymph nodes may contain PAS-positive macrophages. Further assessment of regional lymph nodes with E. coli ISH may aid in characterizing the extraintestinal impact of colonic infection with AIEC.
The significance of ISH-positive cases without PAS staining is unclear. This result differs from previous studies, in which all cases of GC had PAS positivity8,13; however, this may be the result of selection bias in that the case definition in those studies included PAS-positivity. Additionally, it may represent the multifocal nature of the lesions by which they are absent from subsequent sections cut from a tissue block. Lastly, these results suggest that some cases with mucosal E. coli genetic material lack the characteristic intraphagocytic PAS-positive cytoplasmic material.
Other than the dog, ISH for E. coli can aid in the diagnosis of GC in other species, as has been documented in a single case in a cat. 14 In addition to its use in the diagnosis of GC, ISH can be utilized to further understand the etiology of, as well as to diagnose, other conditions. This includes malakoplakia, which is most commonly described in dogs of similar signalment to those affected by GC and has similar histologic lesions in the urinary bladder as opposed to the colon. 5 Given the breed predisposition, it is likely that GC and malakoplakia reflect an increased susceptibility to AEIC or a dysregulated phagocytic immune response with an unidentified genetic basis. As evidenced by the presence of intraphagocytic material with PAS, ultrastructural evaluation, FISH, and ISH, this dysregulated granulomatous reaction is at least, in part, targeted specifically at AIEC. Whether through increased susceptibility to a virulence factor of this particular agent or a genetic peculiarity of macrophages in affected dogs, phagocytosis of bacterial cell wall and components results in the distinct histologic lesions of these entities.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors declared that they received no financial support for their research and/or authorship of this article.
