Abstract
A 4-y-old neutered male German Shepherd Dog was presented with a 3-d duration of lethargy, restlessness, and vomiting. Physical examination revealed generalized lymphadenopathy, pale mucous membranes, systolic heart murmur, dehydration, and fever. Hematologic abnormalities included moderate-to-marked leukocytosis, characterized by neutrophilia with a left shift to progranulocytes and 2% presumptive myeloid blasts, marked anemia that was nonregenerative, and marked thrombocytopenia. Dysplasia was evident in neutrophils and platelets. Bone marrow examination revealed marked myeloid and megakaryocytic hyperplasia with 7% blasts, erythroid hypoplasia, and trilineage dysplasia. Flow cytometric analysis confirmed that bone marrow cells were mostly of neutrophil lineage, with reduced expression of common leukocyte antigens (CD45, CD18) and neutrophil-specific antigen. Bone marrow cells were cytogenetically analyzed for the breakpoint cluster region–Abelson oncogene using multicolor fluorescent in situ hybridization. The genetic aberration was present in 7% of cells, which was a negative result (>10% of cells is considered positive). Euthanasia was elected. Histologic examination showed extensive infiltration of multiple organs by neoplastic myeloid cells, with effacement of lymph node and splenic architecture. The final diagnosis was atypical chronic myeloid leukemia (aCML), an uncommon myeloproliferative disorder with features of myelodysplastic syndromes (dysplasia) and chronic leukemia (neutrophilic leukocytosis with <20% marrow blasts, extramedullary infiltrates). The trilineage dysplasia, lack of monocytosis, and supporting cytogenetics distinguish aCML from CML, chronic neutrophilic leukemia, and chronic myelomonocytic leukemia.
Keywords
A 4-y-old neutered male German Shepherd Dog weighing 44.3 kg was presented to the University of Pennsylvania because of lethargy, restlessness, and vomiting of 3-d duration. Physical examination revealed pale, tacky mucous membranes, generalized peripheral lymphadenopathy, mild dehydration (~5%), and fever (40.2°C). A left systolic heart murmur (grade 2 of 6) was detected on thoracic auscultation. Organomegaly was detected on cranial abdominal palpation.
Initial laboratory testing consisted of a complete blood count (CBC), serum biochemical profile, urinalysis of a voided urine sample, thoracic radiography, abdominal ultrasonography, and fine-needle aspiration of the right and left prescapular and left popliteal lymph nodes. Abnormalities on the CBC included marked anemia (Hct: 0.16 L/L, reference interval [RI]: 0.40–0.60 L/L) that was nonregenerative (reticulocyte percentage 0.7%, RI: 0.0–1.6%); marked thrombocytopenia (platelet estimate: <30 × 109/L, RI: 177–398 × 109/L); and moderate to marked leukocytosis (72.0 × 109/L, RI: 5.3–19.8 × 109 cells/L). The leukocytosis was characterized by neutrophilia (56.6 × 109 cells/L, RI: 3.1–14.4 × 109 cells/L) with an orderly left shift to progranulocytes (7.2 × 109 band neutrophils/L, RI: <0.3 cells/L; 2.9 × 109 metamyelocytes/L; and low numbers of myelocytes and progranulocytes). There were also low numbers of mononuclear cells (2% of the differential leukocyte count; 1.4 × 109/L) that were 15–18 μm diameter containing an oval-to-round nucleus with a finely reticular chromatin pattern, a single nucleolus, and a scant amount of pale blue cytoplasm. These mononuclear cells were interpreted to be myeloblasts. Dysplasia was noted in several neutrophils (giant forms, elongated hypochromic nuclei, and hypersegmentation; Fig. 1) and platelets (large forms and hypogranulation; Fig. 1). Rare micromegakaryocytes were noted (Fig. 1, inset). Low numbers of neutrophils were pyknotic or karyorrhectic. Abnormal erythrocyte morphology included a few echinocytes, schistocytes, keratocytes, and polychromatophilic erythrocytes.
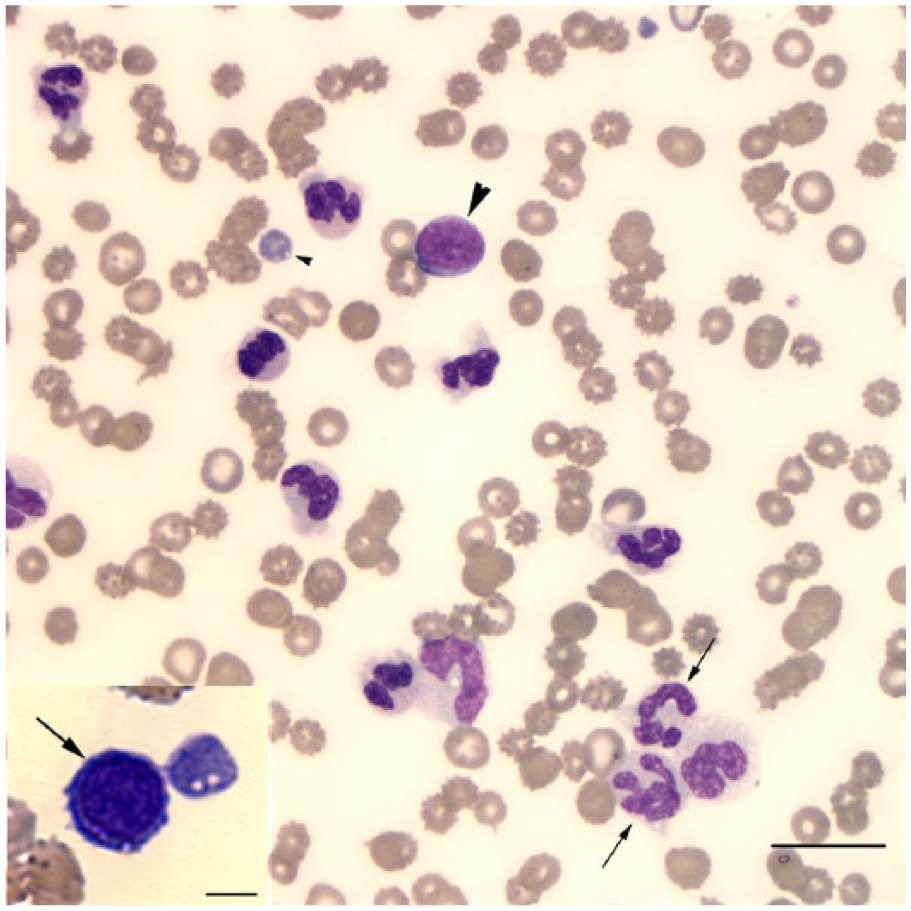
Representative image of a peripheral blood smear from a German Shepherd Dog with atypical chronic myeloid leukemia showing dysplastic neutrophils (arrows), a presumptive myeloblast (large arrowhead), and dysplastic platelet (small arrowhead). Wright stain. Bar = 20 μm.
Abnormalities in the serum biochemical profile included mild hyponatremia (137 mmol/L, RI: 140–150 mmol/L), mild hypokalemia (3.5 mmol/L, RI: 4.0–5.2 mmol/L), mildly increased alkaline phosphatase activity (182 U/L, RI: 20–155 U/L), and mildly increased total bilirubin concentration (10.3 μmol/L, RI: 1.7–8.6 μmol/L). The electrolyte abnormalities were attributed to losses with vomiting. The increased alkaline phosphatase activity and total bilirubin concentrations were suggestive of cholestasis. Increased concentrations of endogenous corticosteroids (“stress”) could have contributed to the high alkaline phosphatase activity. No abnormalities were present in the urinalysis.
Thoracic radiography revealed sternal and tracheobronchial lymphadenopathy, a small amount of pleural effusion, and a diffuse bronchial pattern with bronchiectasis that could be seen with chronic lower airway disease, infectious bronchitis, or neoplasia. Abdominal ultrasonographic findings included marked hypoechoic hepatomegaly; rounded splenic margins with mottled, hypoechoic foci; mild-to-moderate hypoechoic abdominal lymphadenopathy; irregular margins and marked hypoechoic thickening of the pancreas; and a small amount of anechoic abdominal effusion.
Cytologic examination of Wright-stained smears of all lymph nodes revealed a predominance of segmented neutrophils, with fewer band neutrophils and metamyelocytes, and low numbers of macrophages in a mildly hemodilute and proteinaceous background with nonstaining spaces that were interpreted to be free lipid. A few dysplastic and pyknotic neutrophils, as observed in peripheral blood, were noted. Moderate numbers of neutrophils contained variably sized, light-to-dark blue, intracytoplasmic globules that were interpreted to be phagocytosed material. Similar material was noted in some of the macrophages. Low and moderate numbers of mixed lymphocytes were present in smears from the popliteal lymph node and both prescapular lymph nodes, respectively. The lymphocytes were mostly small cells with low numbers of plasma cells. The lymphocytes were interpreted to be a residual normal lymphoid population. The cytologic interpretation was marked suppurative lymphadenitis; however, the presence of immature and dysplastic neutrophils was concerning for a concurrent neoplastic infiltrate.
A presumptive diagnosis of myeloid leukemia was made based on CBC results and cytologic findings of the lymph node aspirates. To further classify the leukemia, a bone marrow aspirate was taken from the proximal end of the left humerus on the same day as the CBC and lymph node aspirates. Examination of Wright-stained smears revealed a markedly hypercellular bone marrow, consisting primarily of myeloid cells with rare erythroid progenitors (9:1 myeloid-to-erythroid ratio). Differentiating and mature granulocytes were the dominant myeloid cells, with low numbers of monocytes. Myeloid blasts comprised 7% of a differential cell count. Dysplasia was evident in the neutrophil lineage (giant cells, hypochromic or hypersegmented nuclei, focal or abnormal granulation). Dysplasia was also noted in the rare erythroid progenitors (hypochromic nuclei, nuclear fragments, megaloblasts). There were numerous megakaryocytes, several of which were dysplastic (micromegakaryocytes, nuclear hypolobulation; Fig. 2). Immature and dysplastic megakaryocytes outnumbered mature megakaryocytes. The bone marrow results were compatible with chronic myelomonocytic leukemia (CMML) or atypical chronic myeloid leukemia (aCML), both of which are characterized by trilineage dysplasia and <20% blasts. A diagnosis of aCML was favored, based on the lack of monocytosis in peripheral blood.
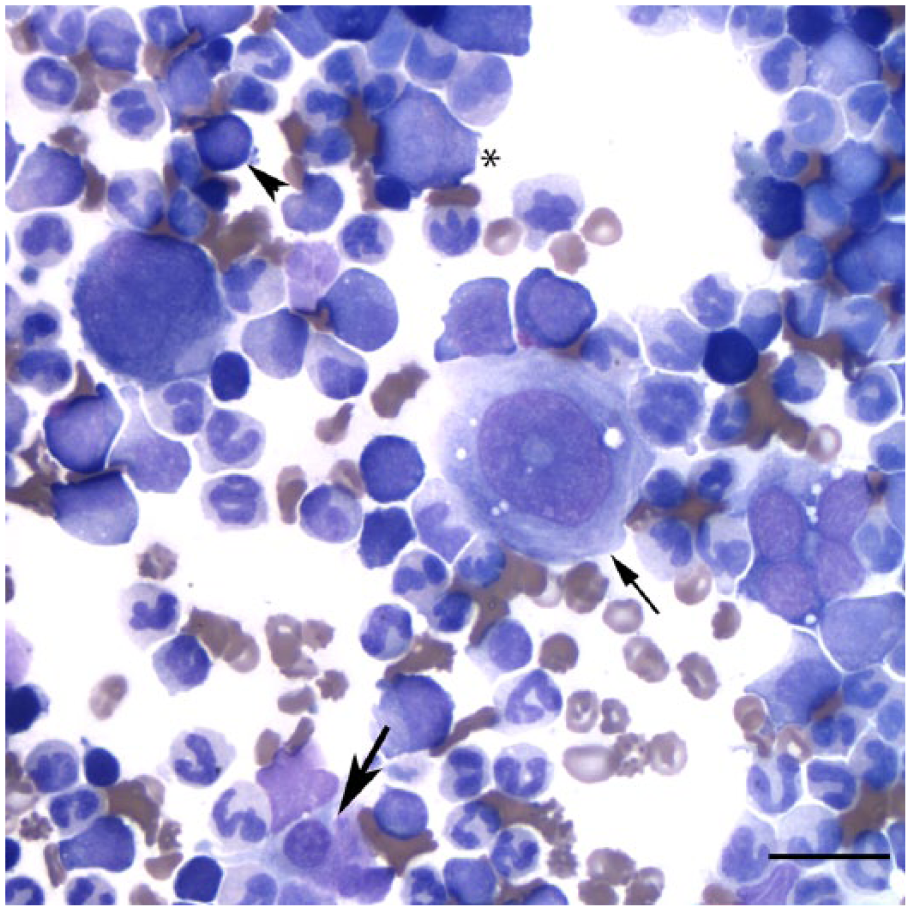
Representative image of a bone marrow aspirate from a German Shepherd Dog with atypical chronic myeloid leukemia showing a predominance of neutrophils, some dysplastic; dysplastic megakaryocytes including a mature form (thin arrow) and a micromegakaryocyte (thick arrow); and low numbers of presumptive myeloid blasts (*) and erythroid precursors (arrowhead). Wright stain. Bar = 20 μm.
Flow cytometric analysis was performed on the bone marrow aspirate as described previously. 25 A forward versus side scatter plot revealed 1 main population (R1), along with a less distinct second population with higher forward and side scatter (R2; Fig. 3A). The cells in both gated populations stained positively with antibodies against myeloid markers (CD45, CD18, CD11b, CD11c), and the majority were also positive for the mature neutrophil markers, CD4 and neutrophil-specific antigen. Expression of the leukocyte common antigens (CD18, CD45) and neutrophil-specific antigen was weaker than that seen in blood leukocytes from healthy animals or animals with lymphoid neoplasms infiltrating the marrow (aberrant expression; Fig. 3B). Approximately 8% of events in the R2 gate were monocytes (positive for CD14). The cells did not label with antibodies against major histocompatibility complex class II (aberrant for monocytes, but normal for neutrophils) or stem cell antigens (CD34 or CD90). The flow cytometric results indicated that most of the bone marrow cells were maturing neutrophils with few blasts and monocytes, supporting the cytologic diagnosis of aCML. Reduced or aberrant expression of some markers was also considered to reflect a neoplastic process.
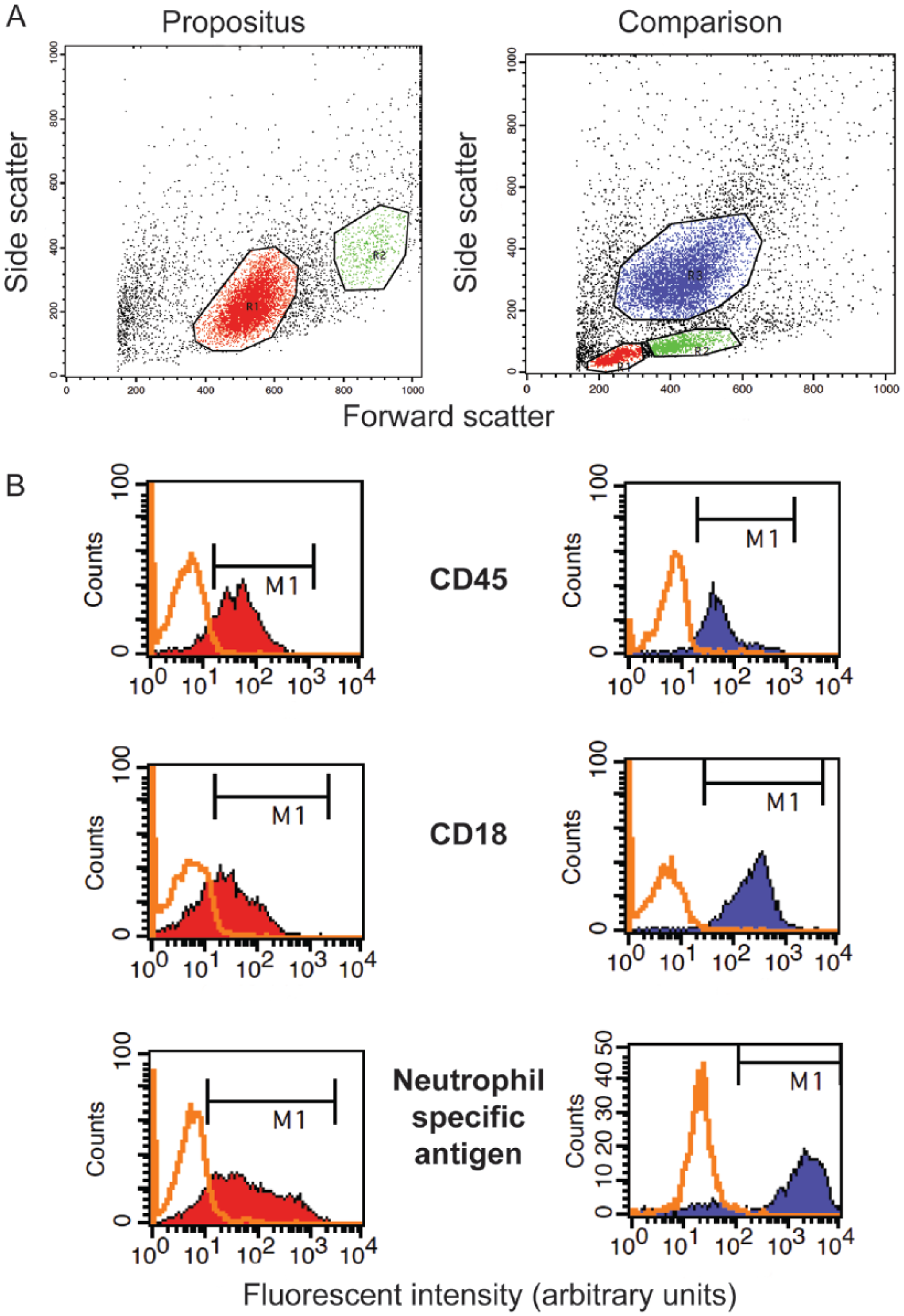
Flow cytometric plots of a bone marrow aspirate from a German Shepherd Dog with atypical chronic myeloid leukemia (left panels) and a dog with B-cell lymphoma for comparison (right panels).
Cytochemical staining for α-naphthyl butyrate esterase (a marker of platelets, monocytes, and some lymphocytes), alkaline phosphatase (a marker of monoblasts), and chloroacetate esterase (a marker of neutrophils) was performed on bone marrow smears, as described. 25 The cytoplasm of the megakaryocytes (normal and dysplastic forms, including those identified as micromegakaryocytes) stained positively with α-naphthyl butyrate esterase, with 2–4% positively stained monocytes. Chloroacetate staining was most prominent in neutrophil precursors (myelocytes and earlier forms) with more mature forms being negative or weakly positive (aberrant staining pattern). There were <5% alkaline phosphatase–positive monoblasts in the bone marrow. Similar to the flow cytometric results, cytochemical staining supported the cytologic diagnosis of aCML (low percentage of blasts and maturing monocytes, with aberrant staining in maturing neutrophils).
The bone marrow smears were sent to North Carolina State University to screen for the breakpoint cluster region–Abelson (BCR-ABL) oncogene translocation. Bacterial artificial chromosome (BAC) clones labeled the BRCA1 (breast cancer 1) and ABL (Abelson murine leukemia viral oncogene homolog 1) sequences on canine chromosomes 9 and 26, and colocalization was analyzed by multicolor fluorescent in situ hybridization as described previously.6,8 A total of 226 cells were counted, and 16 (7%) were positive for the BCR-ABL translocation. In dogs with leukemia screened for this genetic lesion, a positive diagnosis of BCR-ABL is considered when >10% of cells in interphase have the translocation.
The owner elected to euthanize the dog, and an autopsy was performed. Gross findings included generalized mild-to-moderate lymphadenopathy of internal and peripheral lymph nodes, diffuse liver enlargement (5% of body weight), mild splenomegaly, and a diffusely firm pancreas with enhanced lobulation. Histologic examination of formalin-fixed, paraffin-embedded, hematoxylin and eosin–stained sections from bone marrow, peripheral and visceral lymph nodes, tonsils, spleen, liver, pancreas, small intestine, and lung revealed neoplastic myeloid cells within vessels or infiltrating the parenchyma. Neoplastic myeloid cells were predominantly immature and segmented neutrophils with fewer large blasts. The bone marrow was the most severely affected organ and was highly cellular (>80% cells), consisting mainly of differentiating myeloid cells with a few scattered hemosiderophages, small lymphocytes, and plasma cells. Erythroid cells were rare, and there were ~2–4 megakaryocytes per 20× objective field, some of which were dysplastic (hypolobulated nuclei, dwarf cells). There were also focal accumulations of 2–3 blasts (Fig. 4A). Neoplastic cells replaced the splenic red pulp (Fig. 4B) and extensively infiltrated multiple lymph nodes, often effacing the normal architecture (Fig. 4C). In the small intestine, the tunica muscularis was the most severely affected location, with neoplastic cells separating muscle bundles. In the liver, myeloid cells and megakaryocytes (including dysplastic forms) were observed in vessels and hepatic sinusoids (Fig. 4D). Additionally, sections of pancreas and visceral lymph nodes contained intravascular fibrin thrombi and vascular wall necrosis, suggesting a systemic hypercoagulable state. Within the parenchyma of the pancreas and visceral lymph nodes and extending into the peripancreatic and perinodal adipose tissue, there were multifocal-to-coalescing areas of necrosis (infarction), fibrin, and degenerate neutrophils. In the lung, there were multifocal areas of alveolar septal fibrosis and bronchiolar smooth muscle hypertrophy associated with reactive type II pneumocyte hyperplasia, intra-alveolar fibrin, neutrophils, and foamy macrophages. The final morphologic diagnosis from the autopsy was myeloid leukemia with concurrent fibrinosuppurative lymphadenitis, steatitis, and necrosuppurative pancreatitis with multifocal thrombosis. The steatitis and potentially fibrinosuppurative lymphadenitis were considered secondary to the pancreatitis, although infarction could have contributed to the lymphadenitis. The lesions in the lung most likely represented vascular damage but other concurrent causes of chronic pulmonary disease could not be completely excluded. Based on morphologic features, immunophenotyping, and cytogenetic analysis, a diagnosis of aCML was made.7,12,16,29
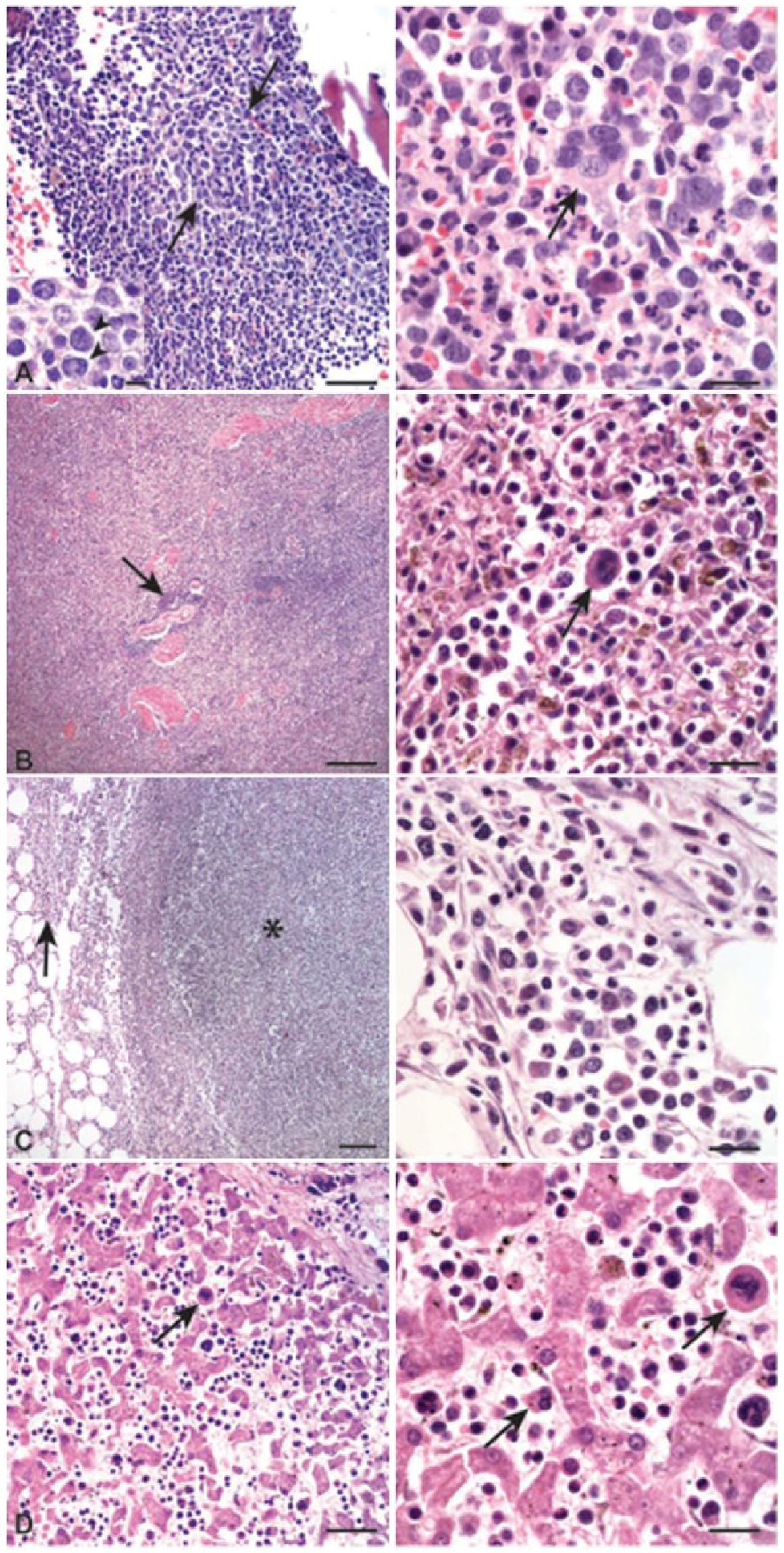
Representative images of formalin-fixed tissue from a German Shepherd Dog with atypical chronic myeloid leukemia. Hematoxylin and eosin.
Leukemias can be broadly classified into myeloid or lymphoid categories based on the originating cell line and then categorized as acute or chronic based on the degree of neoplastic cell differentiation. The World Health Organization (WHO) divides myeloid neoplasms in humans into 3 general categories: acute myeloid leukemia (AML), myeloproliferative neoplasm (MPN), and myelodysplastic syndrome (MDS).2,20 AML is defined as a neoplasm of immature myeloid precursors, with >20% myeloid blasts in the bone marrow at the time of diagnosis.2,20,28 MPNs, in contrast, are characterized by effective hematopoiesis, with high cell numbers of the involved cell lineage in blood and bone marrow.1,2,27,28 The MPNs include CML, chronic neutrophilic leukemia, chronic eosinophilic leukemia, polycythemia vera, essential thrombocythemia, primary myelofibrosis, and MPN, unclassifiable.2,4,20,28 Dysplasia in hematopoietic cells is not a distinguishing feature of these neoplasms, although extramedullary tissue infiltrates are commonly seen. Myelodysplastic syndromes are characterized by clonal ineffective hematopoiesis with morphologic evidence of dysplasia in >10% of cells in 1 or more lineages, resulting in solitary or multiple cytopenias despite a hypercellular marrow.2,20,28 Extramedullary tissue infiltrates are not typically observed with MDS. Myeloid blasts comprise <20% of the cells in both MPN and MDS, helping distinguish these disorders from AML (which can show features of dysplasia, particularly in acute myelomonocytic leukemia in the dog).2,10,20,25,28 A fourth category of myeloid neoplasms was created to encompass those disorders showing features of both MPN (extramedullary tissue infiltrates, high peripheral counts) and MDS (dysplasia in hematopoietic cells) with <20% blasts—myelodysplastic syndrome/myeloproliferative diseases (MDS/MPN).2,3,19,28,29 This category includes aCML, CMML, juvenile myelomonocytic leukemia, MDS/MPN with ring sideroblasts and thrombocytosis, and MDS/MPN, unclassifiable.2,4,12,16,20,28,29
Atypical CML was initially described in humans as a subtype of CML lacking the “Philadelphia” chromosome, which results from the reciprocal translocation of the ABL and BCR genes between chromosomes 9 and 22, respectively. Colocalization of these 2 genetic loci create the constitutively active BCR-ABL tyrosine kinase leading to cellular activation and proliferation.2,16,29 With the development of molecular cytogenetics, some evolutionarily conserved genetic loci between humans and dogs in naturally occurring hematopoietic tumors have been found. A reciprocal translocation between canine chromosomes 9 and 26, leading to the colocalization of the BCR and ABL genes, termed the “Raleigh” chromosome, has been identified in dogs.6,7 The BCR-ABL translocation has been described in several dogs with CML, 7 chronic monocytic leukemia,8,21 CMML, 9 and AML. 10 Dogs with CML, in which the diagnosis was made using CBC, cytopathologic, histopathologic, and immunophenotyping results according to the WHO classification of leukemias, are reported to express the translocation in 11–34% of cells. 7 Up to 5% of cells from healthy dogs have also been shown to have this translocation, which resulted in the laboratory cutoff of >10% for a designation of a positive result for the “Raleigh” chromosome (Breen, pers. com., 2015). Similarly, 9–32% of healthy human volunteers are positive for the BCR-ABL transcript. 17 Potential explanations for the presence of the translocation in clinically healthy people include a defect in terminal cells not capable of self-regeneration and immunocompetence of certain HLA types to the BCR-ABL antigen. 18 Given the presence of the “Raleigh” chromosome in healthy dogs, it is difficult to determine the pathologic relevance of the translocation in 7% of cells in the dog of our report; however, in conjunction with the other data, the low percentage of positive cells would favor a diagnosis of aCML over CML in this dog.
In addition to cytogenetic differences, aCML and CML have distinguishing hematologic and morphologic findings. In humans with CML, there is marked leukocytosis (usually >50 × 109 cells/L) with mature neutrophils predominating, although immature forms (myelocytes and promyelocytes) are frequently seen.1,27 There is also characteristic basophilia and eosinophilia. Monocytes are proportionally low, but absolute numbers can be above reference intervals. The platelet count is variable, but often decreased, and nonregenerative anemia is also characteristic of CML. Bone marrow aspirates reveal numerous mature and differentiating neutrophils and a low blast count (<20%).1,27 Although the neoplastic neutrophils can show features of dysplasia, this is uncommon and not a pronounced feature in human patients with CML.1,16,27,29 In dogs, CML is quite rare with isolated reports in the literature, and it is essentially a diagnosis by exclusion (ruling out other causes of leukocytosis).15,21,23,26 Basophilia is inconsistently seen, and dysplastic changes in neutrophils, such as giant band neutrophils, hypersegmentation, cytoplasmic basophilia, and pyknotic nuclei, have been reported.15,21,23,26 In these reports, dysplasia was not present in the erythroid or megakaryocytic lines, and cytogenetics were not used to help classify the leukemia. Some studies report that the canine disorder more closely resembles chronic neutrophilic leukemia in humans given the infrequent presence of basophilia, which is almost always present in human patients with CML.1,27 In both humans and dogs, CML has an initial prolonged, chronic phase, and often progresses to an AML.1,15,23,26,27
Although the hematologic findings in aCML closely resemble CML in humans, subtle differences exist. Cases of aCML have characteristic dysplasia in all 3 hematopoietic cell lines. Deficient or absent granulation, nuclear abnormalities, or bizarre hypersegmentation can be seen in mature neutrophils and neutrophil precursors in the bone marrow and blood.4,12,16,20,28,29 Dysmegakaryocytopoeisis is a hallmark of aCML, leading to the presence of micromegakaryocytes, which are not seen in CML in humans12,16,29 and dogs15,20 -23 or in chronic neutrophilic leukemia.11,19 Dyserythropoeisis is often present, and patients are usually anemic.12,16,27,29 Unlike CML, basophilia is uncommon, and basophils are frequently absent in affected human patients. Monocytosis is minimal to absent, comprising <1 × 109/L cells or <10% of total leukocytes, which differentiates aCML from the other MDS/MPN, CMML, and juvenile myelomonocytic leukemia.1,12,16,29
The dog in our report had leukocytosis consisting of neutrophilia with an orderly left shift. Dysplasia, including the presence of micromegakaryocytes, was evident in all hematopoietic lineages in blood and bone marrow, and there was <20% blasts (based on morphologic features and flow cytometric immunophenotyping) in the bone marrow. The low blast count ruled out an AML, and the lack of monocytosis in blood and bone marrow was not compatible with a CMML.1,12,16,29 The leukocytosis (indicating effective granulopoiesis) and extramedullary tissue infiltrates are features that are inconsistent with MDS. The prominent trilineage dysplasia in addition to the supportive cytogenetics in this dog mimics aCML in humans. It is interesting that the neutrophils, albeit neoplastic, appeared functional and responded to areas of necrosis in the pancreas and visceral lymph nodes. The lymph node necrosis was attributed to fibrin thrombi, which were apparent in blood vessels in these tissues and suggest a hypercoagulable state. The leukocytosis and other blood cytopenias cannot be solely attributed to the inflammation in these organs, because inflammation does not cause the prominent dysplasia in all 3 hematopoietic lineages, effacement of tissue architecture by sheets of myeloid cells as evident in the spleen and lymph nodes, or the unusual location of the myeloid infiltrates in the intestine (tunica muscularis).
In humans, CML and aCML have different prognoses and treatments. Prognostic implications were the basis for replacing the French-American-British Cooperative Group categorization of hematopoietic malignancies, particularly MDS, with the WHO classification in humans.4,17,20,28 Atypical CML has an inferior survival pattern to CML, with an overall median survival of 14–56 mo,13,29 with 1 case series reporting only 27% of patients alive at 3 y 16 versus a 5-y overall survival rate of 89% with imatinib treatment in CML. 24 With aCML, there is a high risk for developing acute leukemia (20–45%), with risk factors including hepato- or splenomegaly, 3–8% monocytes in peripheral blood, >5% bone marrow blasts, marked dyserythropoiesis, and transfusion requirements. 5 Poorer survival times were noted in older patients 5 or patients with high leukocyte counts (>50 × 109 cells/L), 5 high numbers of circulating blasts,5,29 higher bone marrow blast counts, and lower platelet counts. 29 With CML, progression to acute leukemia is often delayed and typically happens 4–5 y after initial diagnosis. 24 Treatment of CML in humans relies on drugs inhibiting the BCR-ABL tyrosine kinase, such as imatinib mesylate, nilotinib, and dasatinib. 24 In contrast to CML, where treatment is aimed against the BCR-ABL tyrosine kinase, a variety of mutations have been found in humans with aCML, making targeted treatment less effective.13,14,29 A few small retrospective case studies on hematopoietic stem cell transplantation have demonstrated long-term, disease-free survival in human patients with aCML.13,24
Reported survival in dogs with CML is variable depending on the stage of disease at the time of diagnosis and if treatment is elected by the owners. The reported rate of progression is also variable, and prognostic criteria have not been identified because of the paucity of published clinical cases. 25 In dogs with CML, hydroxyurea is a common treatment and has been documented to delay onset to the acute leukemic phase11,15,19,21,22,26 with survival after diagnosis varying greatly from days to 4.5 y.15,21,23,26 There is 1 report of a BCR-ABL–positive canine chronic monocytic leukemia case entering remission following toceranib (a tyrosine kinase inhibitor) and prednisone therapy. 22 CML is rare in dogs, which makes evaluating treatment options difficult, but we will continue to receive guidance from the human literature. Based on the human literature, we presume treatment options would be limited and the prognosis poor in any future cases.
Footnotes
Authors’ contributions
CL Marino drafted the manuscript. All authors contributed to conception and design of the study; contributed to acquisition, analysis, and interpretation of data; critically revised the manuscript; gave final approval; and agreed to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
