Abstract
Psittacid alphaherpesvirus 3 (PsAHV-3) is a rarely reported virus that has been associated with pneumonia in psittacine birds. A 5-mo-old Indian ringneck parakeet (syn. rose-ringed parakeet; Psittacula krameri) was euthanized after developing torticollis and lethargy. Postmortem examination of the parakeet revealed severe pneumonia with syncytial cells containing eosinophilic intranuclear inclusion bodies (INIBs) in the respiratory epithelium. Gene sequencing of total DNA extraction detected PsAHV-3 in the frozen lung. The pancreas had severe lytic necrosis, with the remaining parenchymal cells expanded by large basophilic INIBs. Electron microscopy of the pancreas revealed intranuclear paracrystalline arrays of viral particles morphologically resembling adenovirus. Although we found 3 PsAHV-3 cases in birds in a literature search, we found no cases with adenoviral coinfection.
Keywords
Avian herpesviruses have caused considerable morbidity and mortality in various avian species. 10 Among all of the viruses belonging to subfamily Alphaherpesvirinae (Orthoherpesviridae), genus Iltovirus has been identified as a causative agent of significant respiratory disease in birds. 10 Iltoviruses include gallid alphaherpesvirus 1 (GaAHV-1; Iltovirus gallidalpha1; syn. infectious laryngotracheitis virus),10,14 passerid herpesvirus 1 (PaHV-1),10,18 Amazon tracheitis virus, 19 and psittacid alphaherpesvirus 1 (PsAHV-1; Iltovirus psittacidalpha1; syn. Pacheco disease virus). 7
Four disease-associated PsAHVs (-1, -2, -3, -5) have been identified in psittacine birds.1,4,5,8–10,12,15,16 PsAHV-1 is known to cause fatal systemic disease, also known as Pacheco disease, in birds.10,15 Among the psittacids, Amazon parrots, macaws, and cockatoos are highly susceptible to PsAHV-1, which frequently targets the liver and leads to hepatic necrosis with syncytial cells and intranuclear inclusion bodies (INIBs). 14 Although PsAHV-1 appears to have less tropism for the respiratory system, syncytial cells and INIBs have been detected in the respiratory epithelium of affected birds. 10 PsAHV-2 has been linked to choanal and cloacal papilloma in African grey parrots (Psittacus erithacus erithacus), in which viral genetic material was detected via PCR from pooled choanal and cloacal swabs or cloacal tissue.8,12 PsAHV-3 in parrots has been reported in the United States, 11 Australia, 4 and southern Brazil 9 ; these parrots had pneumonia with syncytial cells containing INIBs in the respiratory epithelium, and PsAHV-3 was detected by PCR analysis of lung tissues. A novel pathogenic virus, PsAHV-5, has been described in Australia, 13 Italy, 1 Brazil, 3 and the United States in Indian ringneck parakeets (syn. rose-ringed parakeet; Psittacula krameri). 5 These birds had respiratory lesions similar to those affected by PsAHV-1 or -3, with PsAHV-5 detected in the affected lungs through PCR analysis. 5
Adenoviral infection occurs in a variety of avian species, and the characteristic histologic findings include multifocal necrosis in a range of organs, with large, basophilic INIBs. 20 Liver, spleen, pancreas, intestine, kidney, and lung are reportedly targeted organs. 7 Several species of psittacine adenovirus (PsAdV; Adenoviridae) have been isolated to date: Psittacine aviadenovirus C, which includes PsAdV-1; Psittacine aviadenovirus B, which includes PsAdV-4; Psittacine siadenovirus D, which includes PsAdV-5, -6; and Psittacine siadenovirus E, which includes PsAdV-7. 6 Although PsAdV-2 (Siadenovirus) has yet to be classified by the International Committee on Taxonomy of Viruses (ICTV), the virus has been documented in birds and caused corresponding pathologic findings.7,20
We describe here a case of PsAHV-3–associated pneumonia and coinfection with adenovirus-like particles in the pancreas of an Indian ringneck parakeet, diagnosed by histopathology, PCR assay, in situ hybridization (ISH), and transmission electronic microscopy (TEM).
A 5-mo-old, male Indian ringneck parakeet was presented to the Avian and Exotic Service of Auburn University Small Animal Teaching Hospital (Auburn, AL, USA) for evaluation of acute onset of lethargy, paraparesis, and dull mentation. The parakeet had been acquired from a breeder 6 d before presentation. At admission, the parakeet was mildly responsive and had a body condition score of 1 of 5, bradycardia, and horizontal nystagmus. Lactated Ringer solution and meloxicam were administered as treatment. The animal developed torticollis shortly after being placed in an incubator. Given the rapidly progressive neurologic signs and poor prognosis, the owner elected euthanasia. Tentative differential etiologic diagnoses included lead toxicity, avian bornavirus infection, avian polyomavirus infection, and psittacine beak-and-feather disease (PBFD).
The parakeet was sent to the Department of Pathobiology at Auburn University for postmortem evaluation. Apart from a segmentally congested small intestine, no significant gross abnormalities were noted. Sections of lung and liver were frozen and forwarded for PCR analysis, including pan-herpesvirus, psittacid adenovirus, fowl adenovirus, and avian polyomavirus. Samples of multiple organs were collected, fixed in 10% neutral-buffered formalin, processed routinely, and 3-µm sections of assorted viscera stained with H&E for histologic examination. Histologically, the lung and pancreas were the organs that were affected most severely. The lining respiratory epithelium of many parabronchi was hyperplastic and formed up to 4 layers, within which were many admixed syncytial cells with up to 12 nuclei (Fig. 1). The syncytial cells contained amphophilic to lightly eosinophilic, 5–20-µm diameter INIBs that filled the entire nucleus (Fig. 1, inset). Filling numerous parabronchial lumina were strands of mucus with degenerate and necrotic sloughed respiratory epithelial cells, RBCs, and low numbers of heterophils. Adjacent to the affected parabronchial lumina, increased numbers of lymphocytes and plasma cells were present in the lamina propria of secondary bronchi. The pancreas contained multifocal-to-extensive areas of lytic and coagulative necrosis (Fig. 2). In the less-affected parenchyma, the residual pancreatic cells were severely karyomegalic, with pale-basophilic-to-amphophilic, 25–70-µm diameter INIBs and marginated chromatin (Fig. 3). In addition, the liver contained rare and randomly scattered enlarged hepatocytes and/or Kupffer cells with similar INIBs.
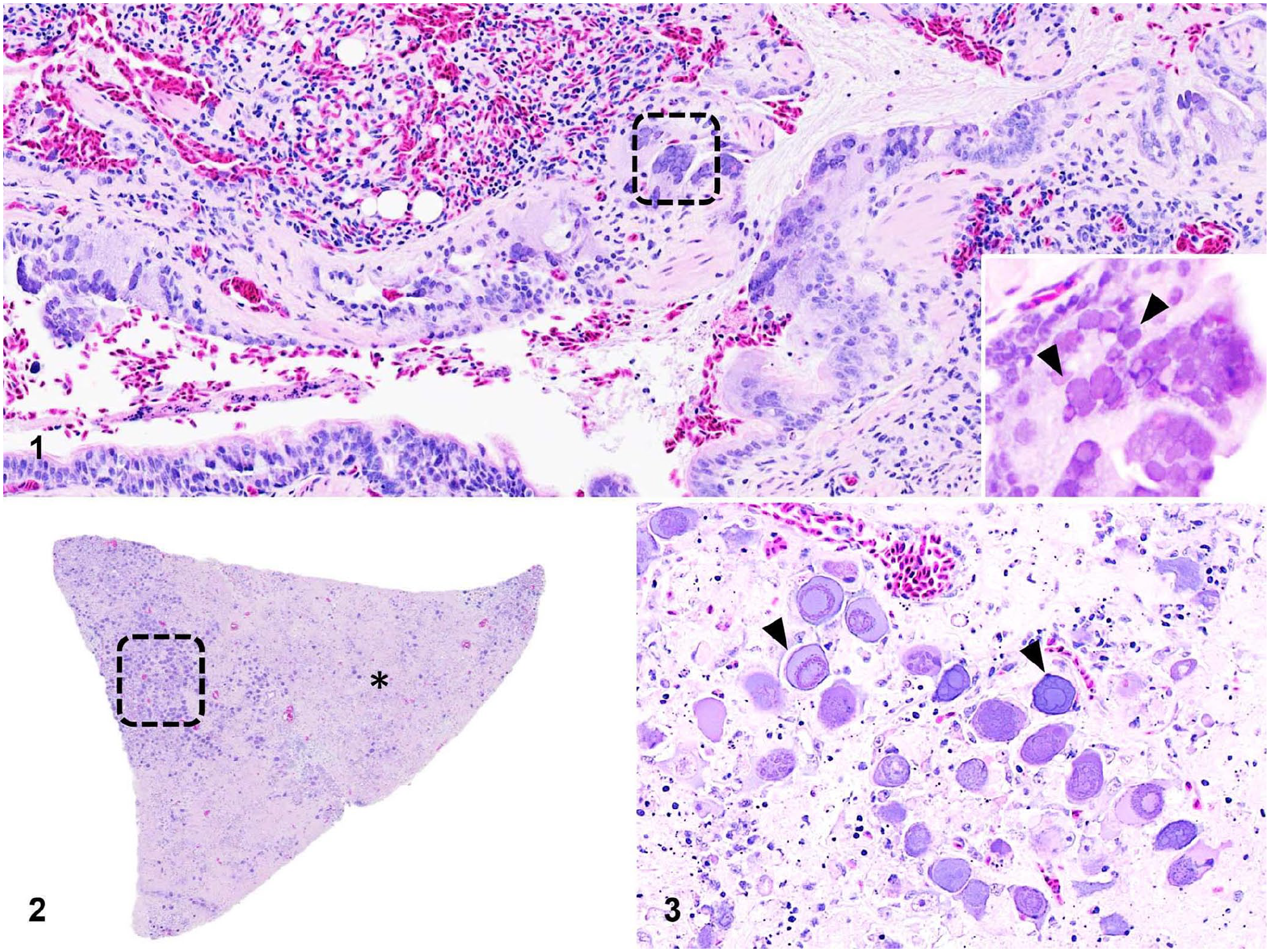
Histopathologic findings of psittacid alphaherpesvirus 3 and probable adenovirus coinfection in an Indian ringneck parakeet.
Our main differential causes in this case were herpesviruses, avian polyomavirus (budgerigar fledgling disease), PBFDV, and avian adenovirus. Ancillary tests, including PCR, ISH, and TEM, were performed at the Alabama State Diagnostic Laboratory (Auburn, AL, USA) and Infectious Diseases Laboratory at the College of Veterinary Medicine at the University of Georgia (UGA; Athens, GA, USA; Table 1). A consensus primer PCR method was performed on the total extracted DNA from the frozen lung to detect herpesvirus (UGA). 17 The pan-herpesvirus degenerate primers used were designed to target a highly conserved region of the herpesviral DNA–directed DNA polymerase gene (primary PCR primer: ILK, 5′-TCCTGGACAAGCAGCARNYSGCNMTNAA-3′, DFA-5′-GAYTTYGCNAGYYTNTAYCC-3′, and KG1, 5′-GTCTTGCTCACCAGNTCNACNCCYTT-3′; secondary PCR primer: TGV, 5′-TGTAACTCGGTGTAYGGNTTYACNGGNGT-3′, and IYG, 5′-CACAGAGTCCGTRTCNCCRTADAT-3′). A 208-bp herpesviral product was amplified, and sequences were aligned and queried against GenBank with BLAST (https://blast.ncbi.nlm.nih.gov/Blast.cgi). The herpesviral nucleotide sequence was identified as 100% identical with 100% query coverage to PsAHV-3 (MK922358.1).
Ancillary test results for infectious agent identification performed on an Indian ringneck parakeet.
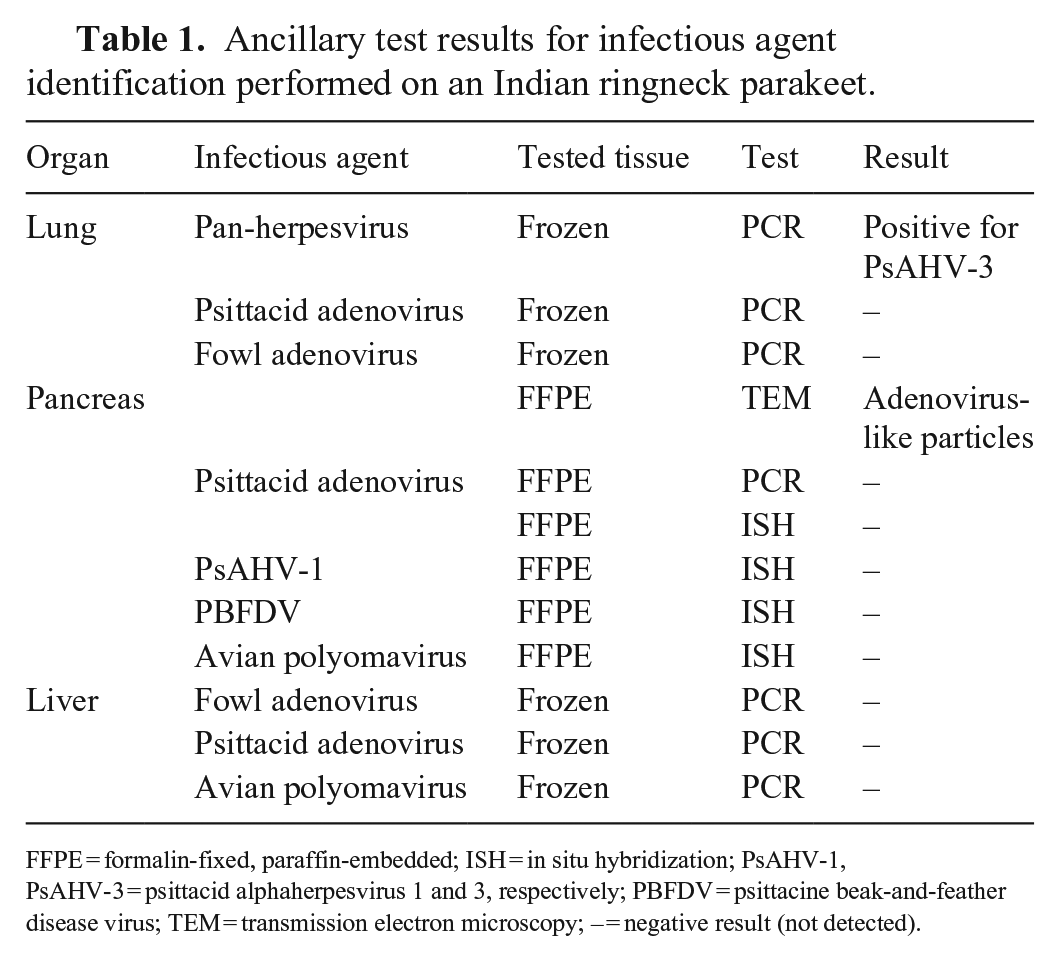
FFPE = formalin-fixed, paraffin-embedded; ISH = in situ hybridization; PsAHV-1, PsAHV-3 = psittacid alphaherpesvirus 1 and 3, respectively; PBFDV = psittacine beak-and-feather disease virus; TEM = transmission electron microscopy; – = negative result (not detected).
Frozen lung tissues were negative in psittacid adenovirus (UGA) and fowl adenovirus (Auburn) PCR assays. Formalin-fixed, paraffin-embedded (FFPE) pancreas was sent to the UGA electron microscopy laboratory to inspect for viral particles. The pancreas had intranuclear viral particles often arranged in paracrystalline array correspondent to the INIBs (Fig. 4). These viral particles were non-enveloped, 65.6–74.4-nm diameter, with a central dense core, surrounded by a halo, and were further encased within a peripheral capsid (Fig. 5). These structures highly resembled adenoviral particles. Although FFPE tissue of the pancreas was submitted for PCR assay and ISH (UGA), no psittacid adenoviral nucleic acid was detected. FFPE tissue of the pancreas was negative for PsAHV-1, PBFDV, and avian polyomavirus with ISH (UGA). No nucleic acid of fowl adenovirus (Auburn), psittacid adenovirus (UGA), or avian polyomavirus (UGA) was detected in the frozen liver sample.
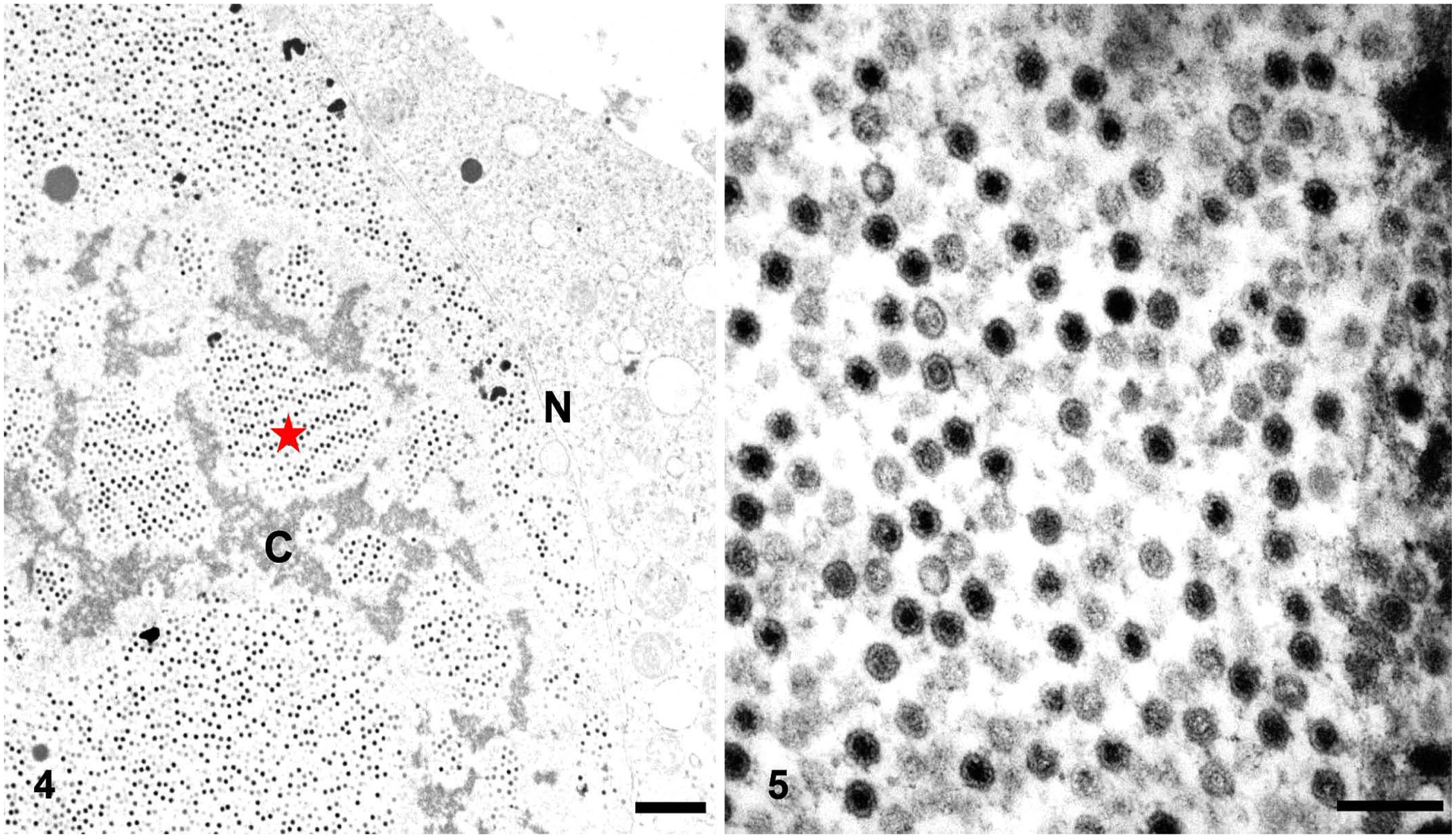
Ultrastructural findings of the pancreas in an Indian ringneck parakeet.
The source and transmission route of the PsAHV-3 and adenovirus remain unclear in our case. In the previous reports, a multispecies aviary with grouping history was mentioned. Therefore, exposure to other sick birds is the most likely route of infection given the fact that the parakeet, in our case, was purchased from a breeder in the United States. Unfortunately, we have limited information of the history of the bird, and we are not aware of whether this animal was imported from other countries or indigenous to the United States, or even the health status of other potential conspecifics.
PsAHV-3 was first described in Bourke’s parrots with an outbreak of respiratory disease in the United States in 2012. 11 Australia and Brazil reported similar sporadic cases in 2013 and 2020,4,9 respectively. In these cases, there were no overlapping avian species, and the birds had emaciation, depression, seizure, and respiratory signs (Table 2). However, the parakeet in our case mainly had neurologic signs with torticollis at admission. Careful examination of the brain was performed; however, the cerebrum and cerebellum were grossly and histologically unremarkable. Pneumonia with syncytial cells containing eosinophilic INIBs in the respiratory epithelium was the predominant lesion shared among the published cases and our case. PsAHV-3 infection appeared to be a fatal systemic disease with several organs involved; however, the parakeet in our case had only lung affected.
Brief literature review of birds with psittacid alphaherpesvirus 3 infection.

PBFD = psittacine beak-and-feather disease.
The listed organs presented variable histopathologic lesions including necrosis, syncytia, and intranuclear inclusion bodies.
The cases in Brazil and Australia of PsAHV-3 infection had Aspergillus as a concurrent infection, in addition to PBFDV in Australia.4,9 Aspergillosis is considered a common fungal infection in birds and PBFDV is ubiquitous in Australian psittacine populations.4,9 In our case, although our attempts to identify the infectious agents in the pancreas at the molecular level failed, abundant viral particles that morphologically resembled adenovirus were easily detected via TEM. Similar single large basophilic INIBs with marginated chromatin were observed in hepatocytes and/or Kupffer cells. Adenovirus and avian polyomavirus were highly suspected; however, the total extracted DNA from frozen liver did not amplify the targeted nucleic acid. The mild hepatic degeneration and necrosis with rare INIBs might be the explanation for negative results with inadequate amounts of nucleic acid. Although psittacid adenovirus is frequently considered an incidental and subclinical disease in parrots that died of other diseases, it has been associated with multisystemic necrosis in red-bellied parrots (Poicephalus rufiventris) and with necrotizing hepatitis in various psittacine birds. 2 Therefore, psittacid adenovirus should not be ignored in psittacids.
Interestingly, there are rare case reports and nucleotide sequences in psittacine birds associated with PsAHV-3. In addition, to our knowledge, this is the second case report 10 y after the first PsAHV-3 case was described in the United States. 11 Before 2012, several reports in parrots described “respiratory herpesvirus of parakeets” with tropism to the lower respiratory tract and lacked significant upper respiratory tract or liver involvement. Although the pathologic and ultrastructural findings supported the diagnosis of avian herpesviral infection, there was no molecular analysis for further confirmation. These cases might not all be associated with PsAHV-3 infection given the lack of molecular confirmation. But PsAHV-3 might cover a wide geographic distribution with cases documented in the United States and Asia. 9 Herpesviruses can evolve and have low pathogenicity in their natural host. 2 Therefore, it is possible that PsAHV-3 is globally ubiquitous; the latent infection, viral evolution, and gradually low pathogenicity led to diverse disease severity ranging from subclinical to fatal.
Our case supports the finding that pneumonia with syncytial cells containing eosinophilic INIBs occurs in birds with PsAHV-3 infection. Although the clinical signs and gross findings may not be specific, PsAHV-3 infection should be considered as an important differential diagnosis in psittacine birds with acute fatal disease, especially with lung involvement. The source, transmission, and importance of PsAHV-3 are uncertain in psittacine birds; however, coinfection with other pathogens may synergize disease development. Coinfection with adenovirus-like viral particles in the pancreas with severe necrosis stresses the importance of electron microscopy in cases in which molecular tests are unrewarding. Examination of pancreatic tissue has important diagnostic value when screening for viral diseases in psittacids.
Footnotes
Acknowledgements
We thank Cynthia Hutchinson and Lisa Parsons (Histology Laboratory, Pathobiology, College of Veterinary Medicine, Auburn University) for making histopathology slides. We thank Mary Brown Ard (Georgia Electron Microscopy, University of Georgia) for helping with transmission electron microscopy.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for research, authorship, and/or publication of this article.
