Abstract
Four 3-month-old Jersey calves and three 3-month-old Holstein calves were inoculated with cervid adenovirus and monitored for clinical signs until necropsied between 10 and 42 days postinoculation. The neonatal Jersey calves had received colostrum, and the Holstein calves were colostrum deprived. Preinoculation and postinoculation serum samples were tested for antibodies to the cervid adenovirus, bovine adenovirus type 6, bovine adenovirus type 7, and goat adenovirus type 1. Virus isolation was performed on kidney, nasal secretion, and/or lung homogenates in fetal white-tailed deer lung cells. Negatively stained preparations of feces from Jersey calves were examined weekly using an electron microscope, and weekly blood samples were collected for complete blood counts. Full necropsies were performed on all calves. A complete selection of tissues was evaluated for microscopic changes, and immunohistochemistry was performed on all tissues using a polyclonal antibody to deer adenovirus. No clinical signs were observed in the calves during the study period. Following inoculation, colostrum-deprived calves developed low antibody titers to deer adenovirus, while the Jersey calves that received colostrum did not. Calves that received colostrum had high antibody titers to bovine adenovirus type 7 and goat adenovirus type 1. No consistent gross or microscopic lesions were seen. Adenovirus was not observed in negatively stained preparations of feces. Immunohistochemistry results did not demonstrate virus in all tissues examined microscopically, and virus was not isolated from lungs, nasal secretions, and kidneys.
Introduction
A novel adenovirus was the cause of an epizootic of hemorrhagic disease that caused high mortality in mule deer (Odocoileus hemionus) in 17 counties of central and northern California in 1993 and 1994. 14 Since first identified in 1993, the deer adenovirus (Odocoileus adenovirus; OdAdV) has been diagnosed as the cause of death in moose (Alces alces) in Canada, 8 white-tailed deer (Odocoileus virginianus) in Iowa, 9 and deer and/or moose in Wyoming, Washington, Oregon, and Idaho (Williams, Bildfell, Cornish, Drew; personal communication) and has been diagnosed yearly in black-tailed deer (Odocoileus hemionus columbianus) in California (Woods, personal observation). Infected animals develop systemic vasculitis resulting in pulmonary edema and/or hemorrhagic enteropathy (systemic infection), necrotizing lesions confined to the upper alimentary tract (localized infection), or both. 1,11–14 This novel adenovirus is a distinct member of the Atadenovirus genus of the Adenoviridae family but is closely related antigenically to ovine adenovirus type 7 (OAdV-7), goat adenovirus type 1 (GAdV-1), and bovine adenovirus type 7 (BAdV-7). 5,15 Although OdAdV has never been identified as the cause of death in any traditional domestic livestock species, it is important to rule out the possibility of OdAdV clinical and subclinical infection in livestock. Lesions of the localized infection in deer (stomatitis) can be strikingly similar to lesions caused by the foot-and-mouth disease virus; thus, it is crucial to determine if clinical localized infection can occur in livestock. Subclinical infection could affect interstate and international movement of domestic livestock during an adenovirus die off in deer. The purpose of this study was to determine if colostrum-deprived calves and calves receiving colostrum can become infected with OdAdV, develop clinical signs and lesions associated with infection, and replicate and shed the virus.
Materials and methods
Viral inoculum
Deer adenovirus was isolated in fetal white-tailed deer (Odocoileus virginianus) lung (FOdvL) cells, as previously described. 5 Briefly, lung from a clinical case (California strain D94–2569) 14 was homogenized in Eagle's minimum essential medium (10 ml) supplemented with gentamicin sulfate (50 μg/ml) and amphotericin B (10 μg/ml) and clarified by low-speed centrifugation. Supernatant fluid was used to inoculate tissue culture flasks (25 cm 2 ) containing subconfluent FOdvL cells. The inoculum (1.5 ml) was allowed to adsorb for 2 to 4 hours and then was replaced with fresh medium. Flasks were examined daily for viral-induced cytopathic effect (CPE) and subpassaged after 7 to 10 days if no CPE was observed. Cultures with CPE were subjected to a freeze-thaw cycle, and the supernatant was inoculated into flasks containing subconfluent FOdvL cells. The isolated virus was biologically cloned by inoculating 6 wells of a 24-well tissue culture plate containing subconfluent FOdvL cells with 0.1 ml of serial 10-fold dilutions of the virus. The plates were examined with an inverted microscope to identify wells with a single plaque, the plate was subjected to a freeze-thaw cycle (2×), and the identified wells were used to make dilutions to repeat the process through 3 successive plaque purifications. Biologically cloned OdAdV was used to produce stock virus for calf inoculum. When viral-induced CPE was maximal, the FOdvL cultures were subjected to 2 freeze-thaw cycles, clarified by centrifugation at 2000 × g for 15 minutes, pooled, aliquoted, and stored at —80°C. The inoculum contained 10 4,8 50% tissue culture infective dose (TCID50)/ml OdAdV.
Animals
Group A. Five 1-day-old Jersey bull calves (nos. 1–5) were purchased from a local dairy after receiving colostrum from their dams, and they were transported to an indoor isolation facility at the California Animal Health and Food Safety Laboratory. Calves were bottle-fed milk obtained from the University of California, Davis, dairy twice daily during the first 2 weeks and then once daily for the remainder of the study. Alfalfa hay and water were available ad libitum after the first 2 weeks.
Group B. Three 3-month-old, clinically healthy, colostrum-deprived Holstein calves (nos. 6–8) were housed in an isolation facility at the National Animal Disease Center. Ames, Iowa. The calves were provided with calf starter and alfalfa hay and water ad libitum. Studies were done in compliance with the Animal Welfare Act, US Public Health Service Policy on the Humane Care and Use of Laboratory Animals, and the Guide for Care and Use of Agriculture Animals in Agriculture Research and Teaching for all study groups.
Group C. Three 2-month-old orphaned mule deer fawns (Odocoileus hemionus) and 1 white-tailed deer fawn (Odocoileus virginianus) were housed in an isolation facility at the Wyoming State Veterinary Laboratory. University of Wyoming. Orphaned fawns were obtained at 2 to 4 days of age from Wyoming counties. Fawns were initially bottle-fed commercial kid replacer 3 times daily for the first 3 weeks and then twice daily for the remainder of the study and supplemented with alfalfa hay. Water was available ad libitum.
Inoculation
Group A. At 3 months of age, 4 of the Jersey calves (nos. 2–5) were inoculated with 2 ml of OdAdV (10 4,8 TCID50/ml) in cell culture fluid via mucous membranes of the eyes, nose, mouth, and trachea and housed in 1 isolation room. One calf (no. 1; negative control) was inoculated with 2 ml of cell culture fluid prepared from a control flask via the mucous membranes and housed in a separate isolation room. Calves were observed daily for clinical signs and body temperature; pulse and respiration were monitored at weekly intervals for 6 weeks until the end of the study at which time all calves were humanely euthanized by intravenous administration of pentobarbital and necropsied.
Group B. Three 3-month-old Holstein calves were inoculated transtracheally with 15 ml of OdAdV (10 4,8 TCID50/ml) and observed daily for clinical signs for 10 days (nos. 6, 7) and 21 days (no. 8) postinoculation, at which time the animals were humanely euthanized using intravenous pentobarbital and necropsied.
Group C. At 2 months of age, 3 fawns were inoculated with 1 ml of OdAdV (10 4,8 TCID50/ml) via mucous membranes of the eyes, nose, mouth, and trachea and housed in 1 isolation room. One mule deer fawn was not inoculated but housed in the same room as the inoculated fawns. The inoculum used in group C was the same stock virus used for the calf inoculation study and was stored at −80°C until fawns could be obtained. Fawns were monitored 3 times daily for clinical signs and were humanely euthanized using tranquilization (xylazine) and intravenous pentobarbital when clinical signs appeared or at the end of the study. Necropsies were performed on all fawns.
Serology, complete blood counts, and direct electron microscopy
For group A, preinoculation sera, sera collected at weekly intervals postinoculation, and sera collected just prior to necropsy were stored at −80°C until they were tested for antibody to BAdV-6, BAdV-7, and GAdV-1 using the serum virus neutralization test, as previously described. 4 Preinoculation and weekly postinoculation sera were tested for antibodies to OdAdV. The viruses were diluted to provide 100 TCID50 per well. Serial 2-fold dilutions of the calf and serotype-positive control sera were made starting at 1:16. Other controls included back titration in the reference virus, a fetal calf negative control serum, and a positive control rabbit polyclonal antiserum. Serum antibody titers were expressed as the reciprocal of the highest dilution of serum preventing CPE in 50% of the wells after 7 days of incubation. The serum virus neutralization test was performed on all serum samples at the same time. Complete blood counts were performed on blood collected from each calf preinoculation with virus and at weekly intervals postinoculation for the first 4 weeks. Fresh feces were collected from the room that housed the inoculated calves and from the room with the negative control calf at 1, 2, and 3 weeks' postinoculation. Feces were suspended in distilled water, centrifuged, negatively stained with 2% phosphotungstate, and examined with a Zeiss 10 C electron microscope. For group B, preinoculation sera and sera collected just prior to necropsy were tested for antibody to OdAdV, BAdV-6, BAdV-7, and GAdV-1 in a serum virus neutralization test.
Pathology
Group A. Calves 3, 4, and 5 were euthanized at 6 weeks postinoculation, and complete necropsies were performed. One calf (no. 2) that developed aspiration pneumonia prior to the study (during bottle-feeding) was necropsied at 5 weeks postinoculation. Portions of a standard set of tissues were collected from each calf including brain, heart (right and left atria, septum, and right and left ventricular free wall), liver, kidneys, lungs (right and left cranial, middle and caudal and accessory lobes), lymph nodes (retropharyngeal, parotid, mandibular, mesenteric, hilar), small intestine (duodenum, jejunum, ileum), large intestine (cecum, colon), forestomach (reticulum, rumen, omasum), abomasum, skeletal muscle, spleen, adrenal glands, tongue, urinary bladder, thymus, tonsil/pharynx, lip, nasal mucosa, trachea, esophagus, and salivary gland. Tissues were immersed in 10% buffered neutral formalin and processed routinely for histologic examination.
Group B. Calves were euthanized at 10 days (nos. 6, 7) and 21 days (no. 8) postinoculation, and complete necropsies were performed. Portions of brain, ileum, abomasum, urinary bladder, thymus, skeletal muscle, adrenal gland, pancreas, lymph node, trachea, spleen, kidney, liver, heart, lung, and forestomach were immersed in buffered neutral formalin and processed routinely for histologic examination.
Group C. Necropsies were performed on fawns soon after natural acute death from the virus, on fawns euthanized after showing clinical signs, or at the end of the study. Portions of a standard set of tissues were collected including brain, heart (right and left atria, septum, and right and left ventricular free wall), liver, kidneys, lungs (right and left cranial, middle and caudal and accessory lobes), lymph nodes (retropharyngeal, parotid, mandibular, mesenteric, hilar), small intestine (duodenum, jejunum, ileum), large intestine (cecum, colon), forestomach (reticulum, rumen, omasum), abomasum, skeletal muscle, spleen, adrenal glands, tongue, urinary bladder, thymus, tonsil/pharynx, lip, nasal mucosa, trachea, esophagus, and salivary gland. Tissues were immersed in 10% buffered neutral formalin and processed routinely for histologic examination.
Immunohistochemistry
Immunohistochemistry was performed on all tissues collected from all calves (groups A and B) and from select tissues from the fawns (group C) using rabbit polyclonal antibody. A streptavidin-biotin peroxidase method was used for immunohistochemistry, as previously described. 12 Briefly, tissue sections on adhesion-coated Shur/Mark slides a were digested for 10 minutes with pepsin and stained on a Dako Autostainer Plus. b The rabbit polyclonal antibody directed against OdAdV diluted 1:2,000 in phosphate-buffered saline-Brij 35 containing 1% casein c was applied to sections for 1 hour at room temperature. Casein served as the only blocking reagent. Sections were rinsed between incubation steps with tris-buffered NaCl solution with Tween 20. b A biotinylated goat antirabbit IgG b diluted 1:200 in the above antibody diluent was applied to the sections for 30 minutes, followed by peroxidase-conjugated streptavidin for 30 minutes. The substrate for color development was aminoethylcarbazole. followed by a Mayer hematoxylin counterstain. Duplicate sections treated in the same manner as above received normal rabbit serum d diluted 1:2,000 as a substitute for the OdAdV antibody and served as negative controls. Tissue from a deer experimentally inoculated with OdAdV served as the positive control. 12
Antibody titers in calves preinoculation and postinoculation with OdAdV.
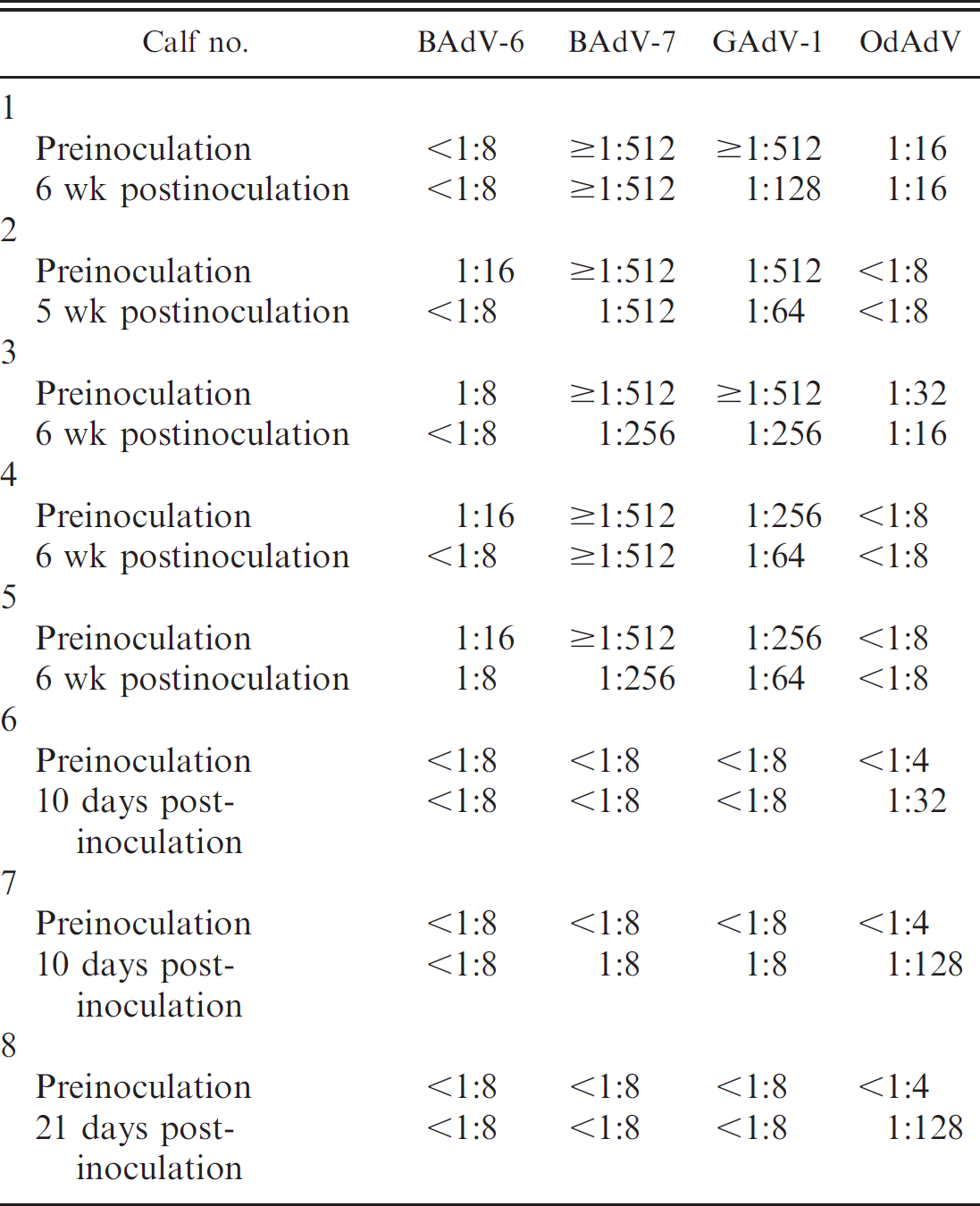
Virus isolation
Tonsil, nasal, and/or kidney tissues were collected from calves 3, 4, and 5 for virus isolation on FOdvL cells as described in the previous section. Nasal swabs and lung were collected from calves 6, 7, and 8 for virus isolation.
Results
Group A. Calves (nos. 1–5) had preinoculation antibody titers to GAdV-1 and BAdV-7 as listed in Table 1. All 5 had antibody titers ≥1:512 to BAdV-7 and 1:256 or ≥1:512 to GAdV-1. Most antibody titers declined during the study period. Calves inoculated with OdAdV (nos. 2–5) either had no or low (1:16) antibody titers to deer adenovirus postinoculation. Calves were clinically normal during the study period (5–6 weeks), except calf no. 2, which developed aspiration pneumonia as a neonate prior to inoculation. Complete blood counts were mostly within reference ranges, except for consistently high neutrophil counts in calf 2 preinoculation and postinoculation, which peaked 2 weeks postinoculation (12,593/μl) and then declined. There were no gross lesions or microscopic lesions in the 4 inoculated calves (nos. 2–5) and 1 control calf (no. 1) that would be consistent with gross and microscopic lesions previously described in cervid adenovirus-infected deer 11–14 and as seen in the positive control fawns in the present study. All calves including the negative control (no. 1) had a mild diffuse lymphocytic interstitial pneumonia. Calf 2 was necropsied at 5 weeks postinoculation and had a chronic, necrotizing bronchopneumonia from which Pasteurella multocida was isolated on aerobic culture of the lungs. Deer adenovirus was not isolated in FOdvL cells from the tissues of calves 1 to 5. No viral antigen was detected by immunohistochemistry in any of the tissues examined microscopically.
Group B. No antibodies were detected to GAdV-1 and BAdV in calves 6, 7, and 8 preinoculation and postinoculation. Calves 6, 7, and 8 were seronegative for OdAdV preinoculation and developed antibody titers of 1:32, 1:128, and 1:128, respectively, 10 days postinoculation. The calves were clinically normal during the study period (up to 21 days). Virus isolation results on nasal sections and lung homogenates from calves 6 to 8 were negative. There were no gross lesions or microscopic lesions in the 3 inoculated calves that would be consistent with gross and microscopic lesions previously described in cervid adenovirus-infected deer 11–14 and as seen in the positive control fawns in the present study. Immunohistochemistry results did not demonstrate OdAdV antigen in any of the tissues examined microscopically.
Group C. One mule deer fawn was found dead in the morning 6 days postinoculation with the cervid adenovirus. No clinical signs were seen the previous night. Necropsy findings included pulmonary edema and hemorrhage throughout the small and the large intestines. Subcutaneous edema was evident in the neck and head. The brain was wet and swollen with flattened gyri. The left retropharyngeal lymph node was enlarged and dark red, and the mandibular lymph nodes were bilaterally enlarged and red. Both adrenal glands were dark red. Microscopically, endothelial cells were hypertrophic with smudgy amphophilic to eosinophilic intranuclear inclusion bodies that were most prominent in the lungs and intestines but were also present in other organ systems. Immunohistochemistry demonstrated cervid adenovirus within endothelial cells in all size vessels in select tissues. The fawn that was not inoculated (but exposed to the inoculated fawns) developed localized infection with necrotizing cheilitis 2 weeks after exposure.
Discussion
Results of the present study demonstrate that deer adenovirus does not produce clinical disease or lesions in calves with and without antibodies to bovine adenovirus when infected via mucosal membranes. In addition, negative virus isolation results on select tissues and negative immunohistochemistry test results on all tissues examined microscopically from inoculated calves suggest virus replication did not occur. Stock virus used for the calf inoculum was stored at −80°C for several years until fawns for the positive control group could be obtained and housing was available. Despite the extensive period of storage and the smaller dose of inoculum (1 ml) used in the fawns, systemic adenovirus hemorrhagic disease was reproduced in 1 fawn and localized infection (cheilitis) developed in an exposed fawn, demonstrating that the virus did not attenuate either during biological cloning or storage. OdAdV has not been replicated in bovine cell culture systems in vitro, 5,14 which supports findings in the present study that cervid adenovirus does not replicate in bovine tissues in vivo. Immunohistochemistry was performed on all tissues examined microscopically despite the lack of clinical signs in the calves and the absence of lesions because it is well known that viruses can replicate or infect tissues without associated clinical signs or lesions. 6 In addition, since this is a newly recognized virus, there is much to be elucidated regarding the pathogenesis of this disease. It was essential to examine all tissues to rule out the possibility of a target tissue that might harbor the virus during latent infection.
Because BAdV-7 is widespread in the U.S. cattle population 2,3,7,10 and antibodies to BAdV-7 neutralize OdAdV 5 in vitro, the authors used both colostrum-deprived and colostrum-fed calves to determine if the deer adenovirus would replicate in calves with or without antibodies to BAdV-7. High antibody titers to BAdV-7 in preinoculated calves that received colostrum (nos. 1–5) with gradual decline during the study indicated that these antibodies were maternal antibodies. Calves with high titers to BAdV-7 did not respond immunologically to inoculation with OdAdV, yet calves without maternal antibodies did respond with titers up to 1:128. Maternal antibodies to BAdV-7 may have neutralized the OdAdV inoculum. Since antibodies to BAdV-7 are neutralizing antibodies to OdAdV in vitro, 5 they are likely neutralizing antibodies in vivo as well. The antibody titers elicited in the colostrum-deprived calves were relatively low compared with experimentally inoculated deer, despite a much higher viral inoculation titer in the calves. 13 Negative virus isolation and immunohistochemistry test results suggest that virus replication did not occur and that the low antibody response in these calves resulted from the virus inoculum in the lung (antigenic load).
The routes of inoculation of calves in groups A and B were slightly different. Both groups were inoculated via the trachea. Group A calves were inoculated via the eyes, nose, and mouth in addition to the trachea. Both methods have been used to inoculate deer with no difference in response (virus distribution, clinical signs, and incubation period). 11–13 Therefore, the slight difference in the additional inoculation sites does not change the conclusion that calves do not become infected with and shed OdAdV when infected through natural means of exposure to the cervid adenovirus.
All calves from group A had a mild lymphocytic interstitial pneumonia that was not a result of the cervid adenovirus inoculum because similar changes were seen in the negative control calf. These changes were nonspecific, and no etiologic agent was identified on diagnostic workup. The calf from group A (no. 2) that contracted aspiration pneumonia as a neonate prior to inoculation with the virus was not eliminated from the study to determine if angiogenesis associated with fibroplasia would make proliferating endothelial cells more susceptible to the deer virus. In a diagnostic case of a calf with chronic bacterial bronchopneumonia (natural infection), bovine adenovirus infection was demonstrated by immunohistochemistry and limited to the endothelium in newly formed vessels associated with fibroplasia (Woods, personal observation). Deer adenovirus antigen was not present in endothelial cells of any vessels, including those within newly forming fibrous connective tissue tracks in the lungs of calf 2.
Results of the present study demonstrate that colostrum-fed and colostrum-deprived calves do not develop clinical disease or lesions when inoculated with OdAdV. OdAdV could not be isolated from, or demonstrated in, the tissues of inoculated calves, suggesting that the virus does not likely replicate and amplify in the respiratory tract or in the kidneys or at least is rapidly eliminated in calves experimentally infected with cervid adenovirus.
Acknowledgements
The authors gratefully acknowledge the Center for Food Animal Health, School of Veterinary Medicine, University of California, Davis, for support of this study; Karen Sverlow, Jim Koobs, and Sa Yang, California Animal Health and Food Safety Laboratory System, School of Veterinary Medicine, University of California, Davis; and the Wyoming Game and Fish Department for providing fawns for the study.
Footnotes
a.
Triangle Biomedical Sciences, Durham, NC.
b.
Dako, Carpinteria, CA.
c.
Sigma Chemical Co., St. Louis, MO.
d.
Vector, Burlingame, CA.
