Abstract
Between 2008 and 2024, fowl adenovirus (FAdV) genotypes were determined by hexon gene sequencing for 1,362 samples: 1,234 from 9 Canadian provinces and 128 samples from the United States. Most genotyped samples were from Ontario (681), followed by Alberta (243), Nova Scotia (116), British Columbia (77), Quebec (58), Saskatchewan (21), Manitoba (20), Newfoundland (16), and Prince Edward Island (2). Most samples (1,285) were related to inclusion body hepatitis (IBH); 77 samples were submitted for other reasons. Four FAdV genotypes (FAdV2, FAdV8a, FAdV8b, FAdV11) were associated with IBH-related submissions. Between 2008 and 2014, the most common strains associated with IBH outbreaks were FAdV11 and FAdV8a. However, since 2015, the identity of FAdVs involved in IBH outbreaks has shifted, with FAdV8b becoming the most frequent IBH-associated strain, largely displacing FAdV8a and FAdV11. In a much smaller group of 77 samples from non-IBH submissions, 10 FAdV genotypes were detected: FAdV1, FAdV2, FAdV3, FAdV4, FAdV6, FAdV7, FAdV8a, FAdV8b, FAdV9, and FAdV11. Although FAdV4 is a recognized causative agent of hepatitis–hydropericardium syndrome worldwide, no association with clinical disease was reported in the birds included in our study. Our comprehensive 17-y analysis of FAdV circulation patterns will support the development of control measures and vaccination programs to reduce the impact of FAdV-related outbreaks.
Keywords
Fowl adenoviruses (
IBH can be a devastating disease in broiler chickens, often causing high mortality and significant economic losses for poultry producers. IBH was first described in Connecticut in 1963 as a hepatitis with distinctive intranuclear inclusion bodies in hepatocytes. However, an etiologic agent related to pathology findings could not be isolated in embryonated chicken eggs at that time.
10
Only a decade later, agents present in the livers of affected chickens provided conclusive evidence—based on analyses of their physical, chemical, and biological properties—that adenovirus was the cause of IBH.
26
In Canada, IBH submissions from flocks with high mortality increased markedly from 2000 to 2006; ~40% of those were caused by FAdV11, 40% by FAdV8a, and the remaining 20% involved FAdV8b and FAdV2.
21
We describe FAdV detections at the Animal Health Laboratory (
Materials and methods
Samples
From 2008 Jan 1 to 2024 Dec 31, we tested 5,719 samples by FAdV PCR at the AHL. Of these, 1,810 samples were from research projects and species other than chickens and were excluded from our study. From the remaining 3,909 samples submitted for diagnostic and surveillance purposes, 1,362 FAdV-positive samples were genotyped upon submitters’ requests. These included 1,234 samples from 9 Canadian provinces and 128 from the United States (
Geographic origin of samples used for fowl adenovirus genotyping.
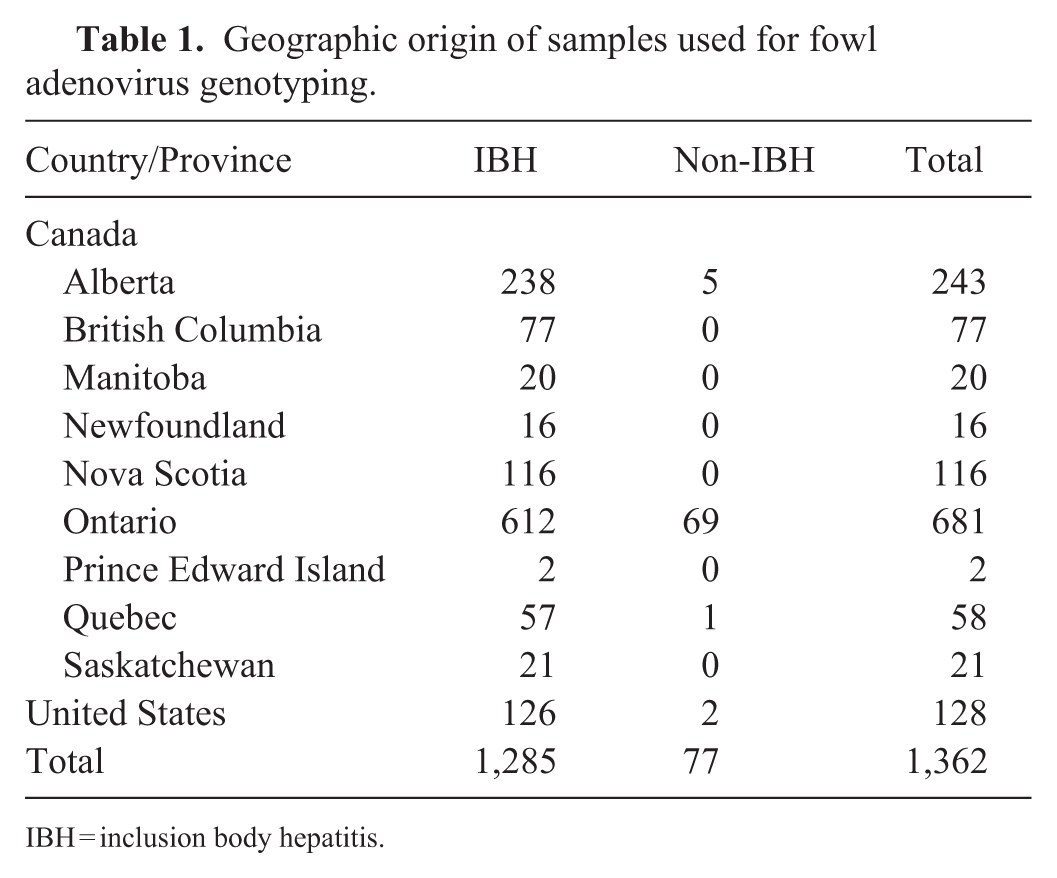
IBH = inclusion body hepatitis.
Criteria
We classified 1,285 genotyped samples as IBH-related, given that they were PCR-positive liver-derived samples (e.g., liver tissue, liver swabs, virus isolates) accompanied by IBH-compatible clinical history and/or gross or histologic lesions. We classified 77 samples as non-IBH because, although FAdV PCR-positive, they lacked histologic lesions or clinical history consistent with IBH, and were derived from non-hepatic tissues (e.g., cloacal swabs, lung, ventriculus, meconium). One sample per submission was genotyped.
Nucleic acid extraction and PCR
We used 2 commercial kits for nucleic acid extraction (MagMAX-96 viral RNA isolation kit, with a MagMAX Express-96 processor, ThermoFisher; MagNA Pure 96 DNA and Viral NA small volume kit, with a MagNA Pure 96 system, Roche). From 2008 to 2012 Aug, we utilized a conventional PCR to detect all FAdVs.
16
From 2012 Aug to 2024 we used a triplex real-time PCR (
Triplex real-time PCR primer and probe sequences for fowl adenovirus PCR assays.

conc. = concentration.
Cycling parameters for fowl adenovirus PCR assays.
Genotyping
We conducted PCR amplification of the hexon protein L1 loop (One step RT-PCR kit, Qiagen; Biometra T3 thermocycler, Analytik Jena) with primers described previously. 17 Nucleotide (nt) sequences of amplicons were determined at the Laboratory Services Division (University of Guelph, Guelph, Ontario, Canada). For sequence assembly, we utilized the SeqMan Pro module of Lasergene software (v.17.5.0; DNAStar) and Geneious Prime software (v.2025.1.2; GraphPad) to calculate nt sequence identities.
Results
The number of samples we tested by FAdV PCR varied, from only 16 samples in 2008 to 1,237 samples in 2023. Of 3,909 samples, 2,741 (70.1%) were positive. Among the 295 samples tested using conventional PCR, 215 (72.3%) were positive, while 2,526 (69.9%) of the 3,614 samples tested using triplex PCR were positive. Signals positive only for FAdV PCR E were found in 1,196 (33.1%) samples, while 249 samples (6.9%) were positive only for FAdV PCR D, and 96 (2.7%) were positive only on the cross-reactive FAdV 52k PCR. Mixed signals were detected in 985 (27.3%) of the samples tested.
We genotyped 1,362 samples and submitted the nt generated sequences to GenBank (PV880996–PV882357). Of the samples, 1,285 (94.3%) were from flocks affected by IBH; 77 (5.7%) were from flocks without IBH. Between 2008 and 2024, infection with 4 genotypes of FAdVs was associated with IBH: 992 (77.2%) samples were FAdV8b, 241 (18.8%) were FAdV11, 48 (3.7%) were FAdV8a, and 4 (0.3%) were FAdV2 (
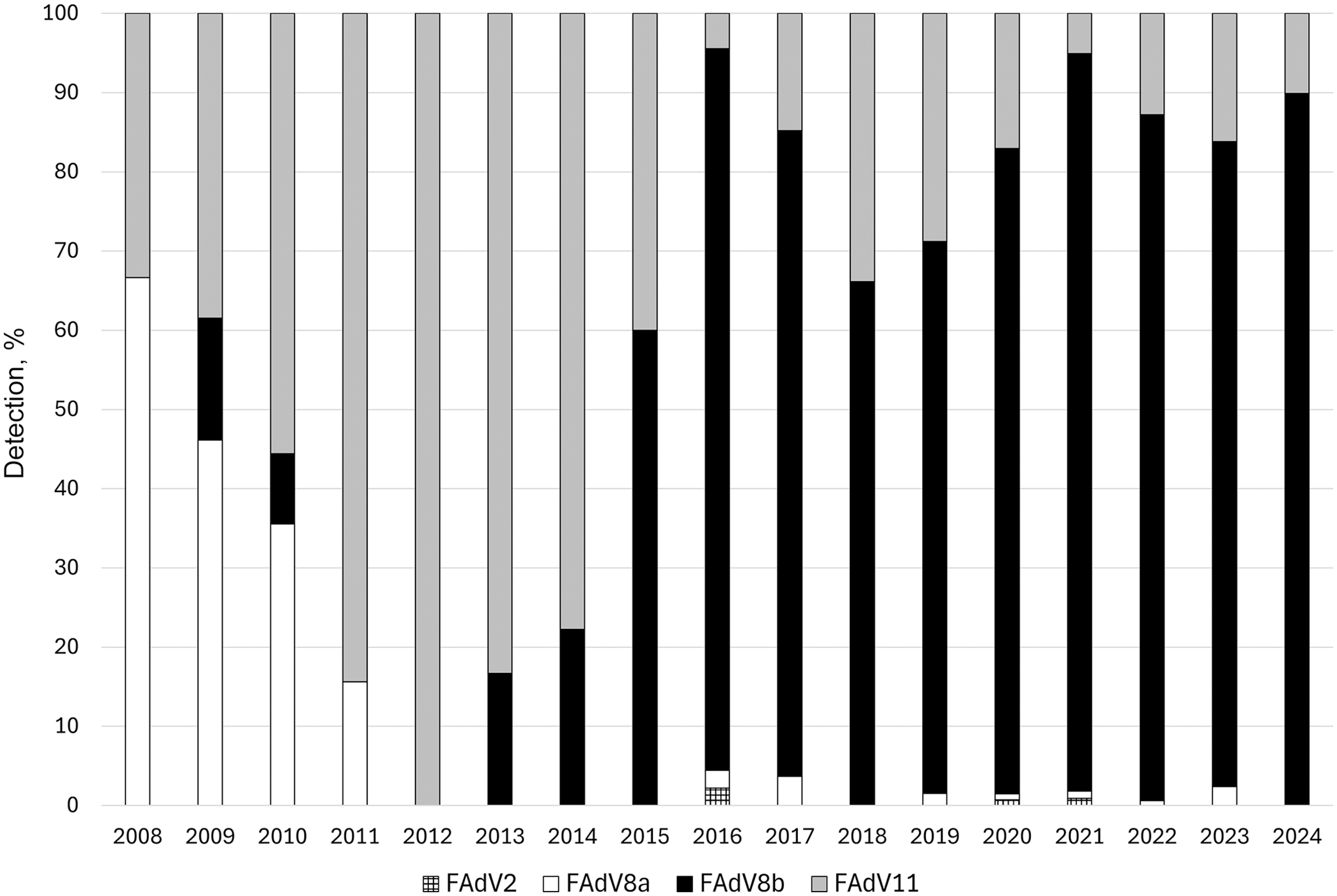
Detection of inclusion body hepatitis-related fowl adenovirus (FAdV) genotype, 2008–2024.
Fowl adenovirus genotypes and strains detected in samples related to inclusion body hepatitis.
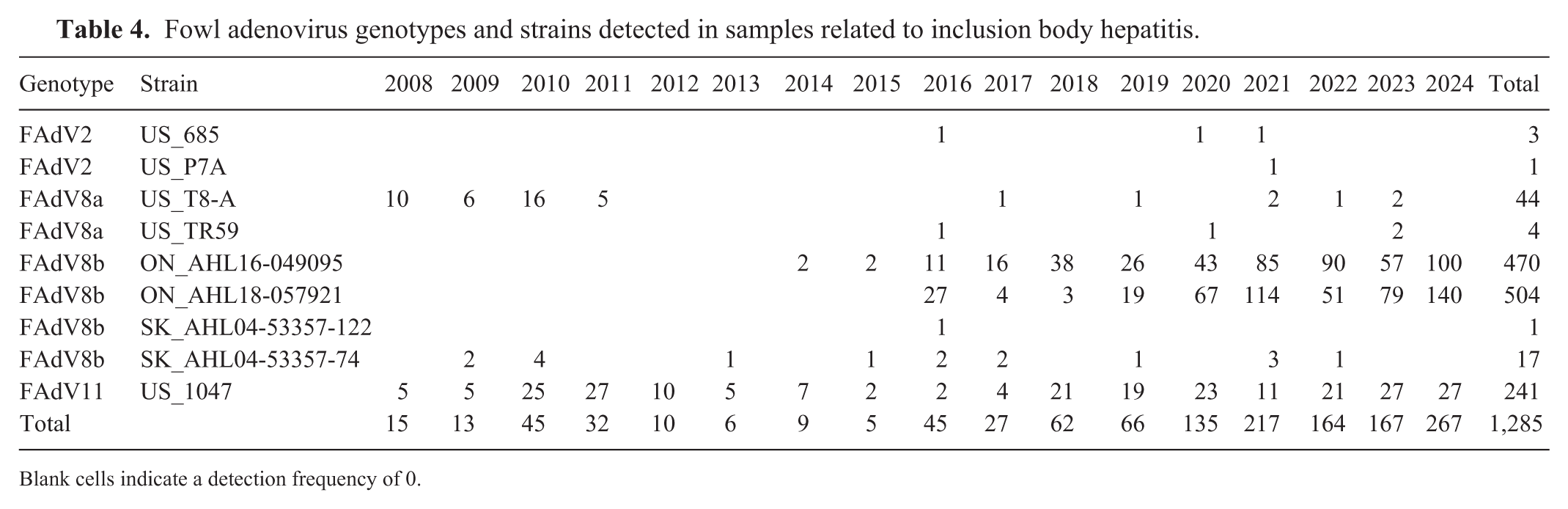
Blank cells indicate a detection frequency of 0.
On the other hand, in 77 non-IBH samples, we detected 10 FAdV genotypes. Most samples, 23 (29.9%), were FAdV1; 13 (16.9%) were FAdV8a; 12 (15.6%) were FAdV4; 10 (13.0%) were FAdV11; 6 (7.8%) were FAdV3; 6 (7.8%) were FAdV8b; 3 (3.9%) were FAdV9; 2 (2.6%) were FAdV7; and 1 (1.3%) each of FAdV2 and FAdV6 (
Fowl adenovirus genotypes and strains detected in samples not related to inclusion body hepatitis.

Blank cells indicate a detection frequency of 0.
Discussion
An increase in incidence of IBH outbreaks caused by FAdV infection was reported by the AHL across 9 provinces in Canada between 2004 and 2006. 21 In 2010, widespread vaccination of broiler breeders was introduced in Ontario with bivalent, autogenous vaccines containing FAdV8a and FAdV11. In the years following the introduction of vaccination, the incidence of IBH outbreaks declined. However, since 2015, an increase in IBH outbreaks has been observed in Canada and worldwide.22,29 We genotyped FAdVs in Canada from samples that were submitted to the AHL from 2008 to 2024 and noted a shift in genotype representation. Between 2008 and 2014, FAdV11 and FAdV8a were the most common strains associated with IBH outbreaks. However, in 2014, a new FAdV8b strain was first detected in 2 samples from Nova Scotia, Canada. Since 2015, FAdV8b has largely displaced FAdV8a and FAdV11 in IBH-related submissions. Of 128 U.S.-origin samples that we received from 2016 to 2021, most, 117 (84.7%), were FAdV8b. Reports describing the introduction of new FAdV8b strains and/or the involvement of FAdV8b as a main IBH-associated strain have been emerging globally—from multiple continents and countries, including China, Iran, Spain, Brazil, Japan, Hungary, Türkiye, South Korea, Malaysia, Egypt, Canada, Poland, and Morocco.1,2,4,5,12,14,18 –20,22 –24,27,28
In 28 samples from non-commercial flocks, 10 FAdV genotypes were detected, but none were from birds with IBH and were not considered primary disease-causing agents. Of 49 non-IBH samples from commercial flocks, only 2 were clearly associated with clinical disease or gizzard erosions. Detection of FAdV4 has been reported in both commercial and non-commercial flocks in Canada and the United States.3,7,22,31 However, FAdV4 infection has not been associated with clinical presentation of HHS in broilers in Canada. An experimental inoculation with a high dose of FAdV4 (2 × 108 pfu/chick) did not result in disease in 10-d-old specific-pathogen-free Leghorn chicks. 8 We detected FAdV4 infection in subclinical broiler breeders and in non-commercial flocks with no signs of HHS or IBH. This suggests that the FAdV4 strains circulating in Canada are either non-hepatopathogenic or that other (yet unrecognized) co-factors necessary for disease induction are absent.
To date, there are no commercial, licensed live vaccines against IBH in North America, highlighting the need for alternative and adaptive strategies to control and reduce the severity of IBH outbreaks in broilers. 9 Although autogenous inactivated vaccines have been used widely in broiler breeders throughout Canada, they sometimes provide inconsistent levels of passive immunity. Increased biosecurity measures that limit the natural exposure of broiler breeders may inadvertently reduce the generation and transmission of passive maternal antibodies, thereby increasing the risk to their offspring. 30
Our findings are subject to limitations from sample collection bias, given that collection was passive and most samples came from Ontario poultry producers—reflecting both the size of the poultry industry in Ontario and the location of our laboratory. Nevertheless, we anticipate that the samples generally reflected the presumed impact of IBH-related field issues in Canada. The IBH situation in Canada may also be compared, to some extent, with that in the United States. From 2016 to 2021, we received 128 samples from the United States, However, no samples were received between 2022 and 2024, reportedly because of the introduction of genotyping services by local U.S. laboratories. Given the size of the poultry industry in Canada and the geographic distribution of sample origins, we believe that our study offers robust data on FAdV circulation patterns across Canada. Our findings may inform effective strategies to minimize and control FAdV-associated outbreaks.
Footnotes
Acknowledgements
We gratefully acknowledge the technical and support staff at the Animal Health Laboratory for their work in processing and testing these submissions.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
We acknowledge financial support from the Ontario Ministry of Agriculture, Food and Agribusiness.
