Abstract
We describe here a novel peeling skin condition (PSC) in 2 neonatal Pacific walruses (Odobenus rosmarus subsp. divergens). Macroscopically, calves had various degrees of peeling skin exacerbated by mechanical trauma. Lesions occurred in areas subject to friction: ventrum, fore- and hindflippers, and associated joints. Histopathologic features included pseudocarcinomatous epithelial hyperplasia with orthokeratotic hyperkeratosis. Bacterial cocci were present within the stratum corneum. A few intraepidermal clefts were present. Inflammation, epidermolysis, and vasculopathies were not observed. PCR assays were negative for vesivirus and for Staphylococcus aureus exfoliative and toxic shock syndrome toxins. Tissue samples were cultured and bacteria isolated and identified by MALDI-TOF MS as Carnobacterium maltaromaticum, Psychrobacter phenylpyruvicus, Globicatella sanguinis, Streptococcus phocae, Pseudomonas spp., Rahnella aquatilis, and Escherichia coli. Given the young age of the calves and their clinical presentation, congenital ichthyosis was suspected. No genetic differences were detected for sequenced portions of keratin genes (keratin gene K10) between diseased and normal walrus skin. This rare PSC in neonatal Pacific walruses is recognized as novel by indigenous Alaskan marine mammal hunters of the Bering Strait region. A comprehensive diagnostic work-up of future case materials is needed to characterize the underlying biochemical defect(s).
Walrus skin is characterized by unusual thickness of the epidermis, strength, and durability; only a few skin diseases have been reported in free-ranging neonatal and young-of-the-year Pacific walruses (Odobenus rosmarus subsp. divergens). Most skin lesions are related to physical trauma (e.g., lacerations, punctures, and bruising from tusk jousting among walruses 6 ). Other reported skin-associated lesions unrelated to physical trauma among free-ranging Pacific walruses include frostbite, localized dermatomycosis, and an ulcerative dermatitis syndrome of unknown etiology.4,6,7 For walruses under human care, a variety of inflammatory skin conditions associated with various microbial agents have been reported. 22
We report here a novel peeling skin condition (PSC) among neonatal Pacific walruses. Pacific walruses breed in January and February with calving and nursing occurring on sea ice from April to June the following year. 6 The index case, a subsistence-hunted calf, was reported 2012 May 18, by indigenous marine mammal hunters from St. Lawrence Island, AK, USA during their spring walrus hunt. Three additional cases, 2 in 2014 (May 4, 5), and 1 in 2021 (May 11) have since been reported by hunters. Of the 4 calves with reported PSC, hunters submitted 1 subsistence-hunted and 1 found-dead calf, both from 2014 for postmortem examination and diagnostic workup (our study). The 2 other subsistence-hunted calves, 1 each in 2012 and 2021, were left on the ice because of food safety concerns.
This newly observed abnormal skin condition in walrus calves, clinically characterized by peeling skin and cleft formation within the epidermis, most closely resembles ichthyosis, a rare congenital keratin formation defect reported in humans, domestic animals, and other species.1,14,18 Inherited ichthyosis is usually apparent during the first year of life, often at birth, and continues to affect animals throughout life. The keratin-10 (K10) gene codes for keratin production in the outer layer of the skin; defects in this gene are known to produce skin conditions in humans, such as epidermolytic hyperkeratosis (characterized by thick, blistering skin), ichthyosis with confetti (red, scaly skin), and cyclic ichthyosis (thick, blistering skin). 21 We describe here characteristic skin features, associated histopathologic findings, and outcomes from a preliminary investigation into the genetic basis of this novel PSC.
During 2014, 2 frozen neonatal Pacific walrus carcasses (1 partial, case 1; 1 whole, case 2) were submitted for autopsy to the North Slope Borough, Department of Wildlife Management (NSB DVM; Utqiagvik, AK, USA). Carcasses were submitted by marine mammal hunters on Saint Lawrence Island, AK for further scientific investigation of the unusual PSC. This abnormal skin condition in Pacific walrus calves had not been observed by elders and experienced marine mammal hunters on Saint Lawrence Island, nor was it referenced by regional traditional ecologic knowledge.
The Pacific walrus is an essential cultural and nutritional subsistence resource to Saint Lawrence Island Yupik peoples. The U.S. Fish and Wildlife Service (USFWS) has management authority of Pacific walruses in the United States (https://www.mmc.gov/priority-topics/arctic/co-management-and-alaska-native-tribal-consultation/) and works closely with their co-management partner, the Eskimo Walrus Commission (EWC) through a cooperative agreement (1997). Marine mammal tissue collection occurred under USFWS permit MA 80164B-0.
Case 1 was subsistence hunted on sea ice (2014 May 5) near Gambell, AK. Case 2 was found dead on the sea ice (2014 May 4) with an attending live adult female (subsequently hunted) north of Savoonga, AK. Skin abnormalities were present at the time of discovery in both calves. Hunters also reported that skin peeling was exacerbated by mechanical trauma associated with moving the carcasses on the ice. The main birth period for Pacific walruses occurs April 15–May 25, and we estimated calf 1 to be ~3-wk-old at the time of harvest. The 2nd calf was full-term but discovered dead. A limited autopsy (2014/06/09, R. Stimmelmayr) was conducted on case 1 because all organs and head had been removed by the hunter in the field. The hunter commented on the appearance of removed organs: “the lungs were red and shriveled and the intestines were off-color and of smaller diameter than normal.” An autopsy was conducted on case 2 (2014/06/08, R. Stimmelmayr) following standard postmortem examination procedures for pinnipeds. 5
Carcass condition code for the 2 calves, both females, was classified as 2 (freshly dead). 5 Cutaneous abnormalities, including peeling and intraepidermal clefts, with similar anatomic distribution of lesions, were present in both calves. Multifocal, single-to-confluent peeling epidermis (Fig. 1A) and single-to-grouped 1–3-cm intraepidermal clefts (Fig. 1B) were present along 30–80% of the ventrum, involving the lower neck region, axillary region, sternum, and lower abdomen to tail. Lesions were also present on 30–100% of the dorsal and ventral surfaces of the foreflippers and the dorsolateral surfaces of the hindflippers. Case 2 also had focal peeling skin on the back of the head and lower lip; additionally, the upper and lower conjunctivae of the right eye were missing. Case 2 was a more severe case with a greater surface area of skin peeling. In areas with ongoing skin peeling (e.g., hindflippers), the epidermis (outer layers) could be peeled away by gentle pulling. In areas in which the epidermal top layer had peeled off, the exposed deeper epidermal layer was speckled gray with a red tint in the non-pigmented areas. Nails were within normal limits: long, pointed, and curved for the hindflippers and short on the foreflippers. Hindflipper nails did not break when bent. Short, dense, pale-blond hair (natal coat at full-term) was present in normal and abnormal skin areas. Additional gross findings for case 2 included serosanguineous fluid in the thoracic, abdominal, and pericardial cavities (200 mL). Lungs were small, dark-red, collapsed, and sank in formalin. Significant findings were not evident in other organs.
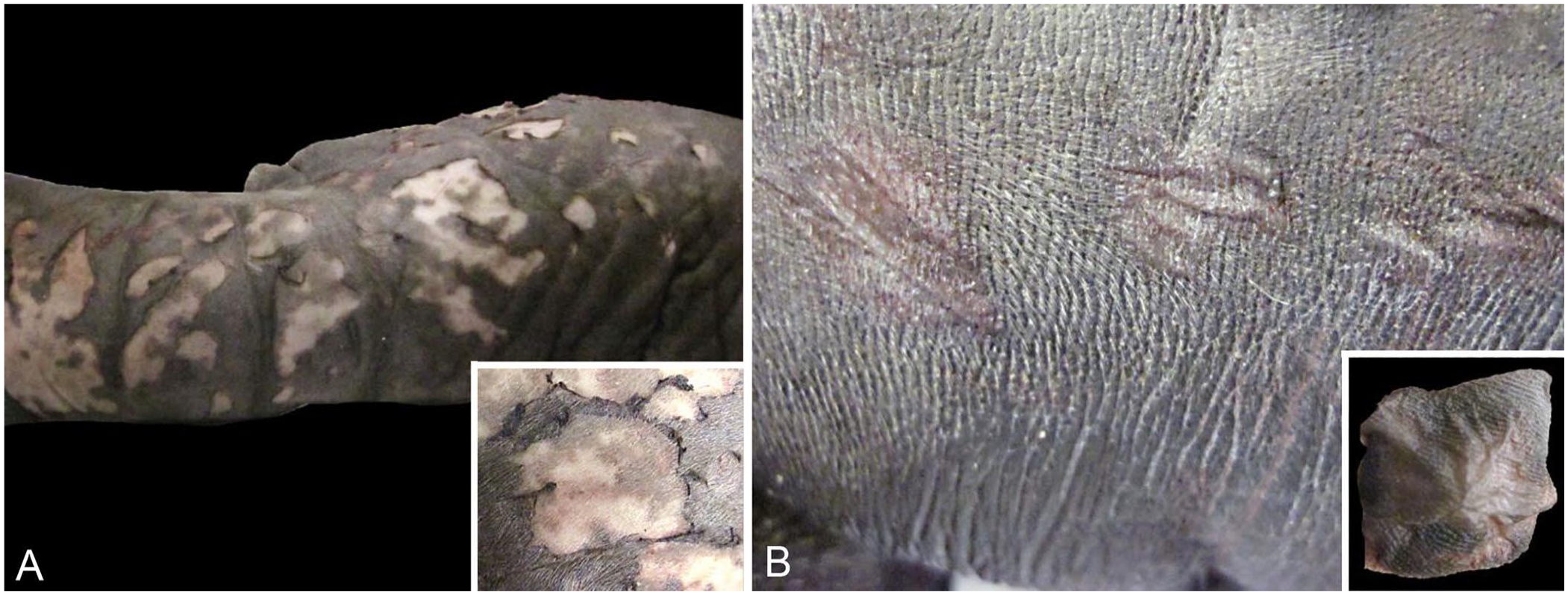
Characteristic gross features of a novel peeling skin condition in neonatal Pacific walruses (Odobenus rosmarus subsp. divergens) near Saint Lawrence Island, AK.
Tissues from both calves were fixed in 10% neutral-buffered formalin for histologic examination, and for case 2 (intact carcass) brain, visceral and endocrine organs, muscle, blubber, and numerous skin sections were evaluated. Full-thickness skin sections were collected from the neck, lip, ventrum, and fore- and hindflippers. For case 1, only skin and muscle samples were collected. Formalin-fixed samples were processed routinely, sectioned at 4–6 µm, and stained with H&E. Following the initial postmortem examination, frozen full-thickness sections of skin with representative lesions, including peeling skin and clefts, were submitted to the Athens Veterinary Diagnostic Laboratory (Athens, GA, USA) for microbiologic analysis (macerated skin tissue culture, PCR) and to the University of Houston–Downtown (Houston, TX, USA) for further molecular and/or genetic analyses. For bacterial culture using standard techniques, frozen skin sections were thawed, washed twice in sterile water, and then macerated. Generic vesivirus (Caliciviridae) and Staphylococcus aureus toxic shock syndrome and exfoliative toxins (S. aureus [TSS-T1] toxin, S. aureus exfoliative toxins A and B) were investigated by PCR assays.3,9,10,19 DNA was extracted from skin tissue (cases 1 and 2; control case 3 [healthy subadult walrus]) using standard techniques. Using the human genome KRT10 gene, we performed a BLAST search against the Pacific walrus genome (BioProject PRJNA167474) to locate the KRT10 gene in the Pacific walrus. We used the program Primer31111,12,20 to develop primers to amplify portions of the KRT10 gene in walruses, focusing on exons, assuming that any expressed mutation would occur there. Four sets of PCR primers were designed (Table 1). We amplified each of these regions separately for samples from the 3 cases. PCR reactions for each primer set were run using the following thermocycler conditions: initial denaturation at 95°C for 2 min; 35 cycles of 95°C denaturation, 55°C annealing, a 72°C extension step, and a final 72°C extension of 7 min. Each 23.5-μL PCR reaction contained 3 μL of DNA (10–30 ng/μL), 1.5 μL of each primer (10 μM), 6.5 μL of ultra-pure water, and 12.5 μL of master mix (GoTaq; Promega). PCR products were visualized on 1% agarose gel and purified before sequencing at an external facility using the same PCR primers (Table 1). Sequences were edited using Geneious v.9.0.5 (Dotmatics). Our samples were aligned to the Pacific walrus genome sequence using the “map to reference” option in Geneious and examined for mutations with respect to the genome sequence.
Primer pairs and their sequence of the KRT10 gene in Pacific walrus (Odobenus rosmarus subsp. divergens).

Cutaneous histopathologic findings (Fig. 2A, 2B) included orthokeratotic hyperkeratosis and a few clefts within the stratum corneum to the stratum granulosum. Regions of epithelial invagination were filled with retained keratin with occasional evagination of the stratum granulosum into the retained keratin. Small-to-moderate numbers of bacterial cocci were present within the stratum corneum. Inflammatory cells and vascular changes were not observed. For case 2, there was pulmonary atelectasis. No other significant histopathologic findings were observed in non-cutaneous organs. For comparison, normal walrus skin is depicted in Fig. 2C.
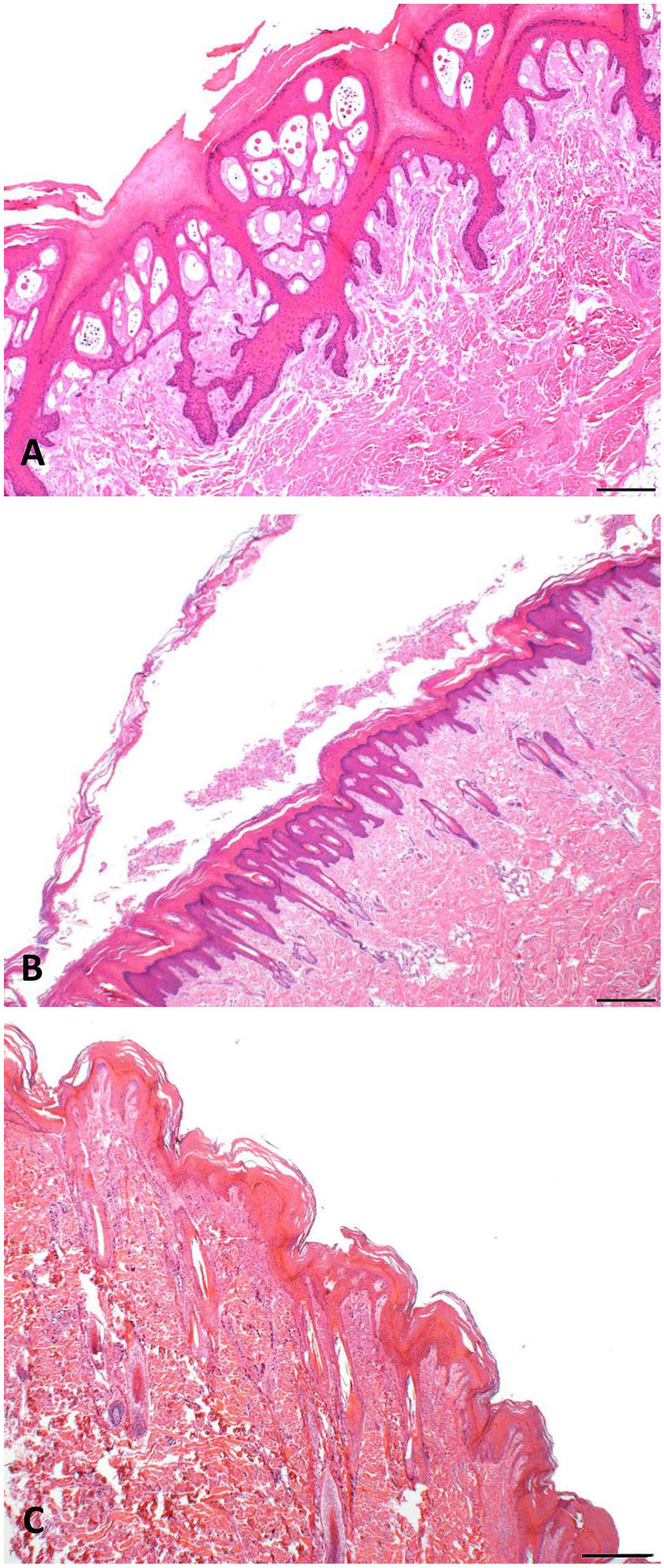
Novel peeling skin condition in neonatal Pacific walruses.
PCR assays were negative for vesivirus and S. aureus toxic shock syndrome and exfoliative toxins. A mix of bacteria was cultured and identified by PCR as Carnobacterium maltaromaticum, Psychrobacter phenylpyruvicus, Globicatella sanguinis, Streptococcus phocae, Pseudomonas spp., Rahnella aquatilis, and E. coli. None are unusual for the sample type (skin) cultured.
We successfully obtained DNA sequences of the KRT10 gene for the regions covered by primer pairs KRT10F2/KRT10R2 and KRT10F4/KRT10R4 (corresponding to exons 3 [part], 4–6) for all samples (Table 1). The KRT10F5/KRT10R5 primer pair amplified all samples but did not give quality sequence results. The KRT10F1/KRT10R1 primer pair only amplified in samples from the control case (3). Of the regions of KRT10 sequenced, there were no sequence differences among the 3 walrus samples, nor were there any differences in our samples compared to the published walrus genome sequence; they were genetically identical at this locus.
The clinical presentation of epidermal skin peeling, with and without intraepidermal cleft formation, and the histopathologic findings of orthokeratotic hyperkeratosis in both calves are compatible with ichthyosis.14,21 The early onset suggests a congenital form of ichthyosis. Case 2 was found dead. Stillbirth and neonatal mortality are not well documented among free-ranging Pacific walruses. 6 Although not a definitive test, the uninflated lung together with the histologic finding of atelectasis suggest that case 2 was likely stillborn or died shortly after birth. The underlying cause(s) of death of this neonate was not determined.
Differential diagnoses that we considered other than congenital ichthyosis included retained lanugo and exfoliative dermatitis. Lanugo, the white hair of first pelage, is shed in utero by the time fetuses are at near- or full-term development (April–June). 6 The pelage appearance in both calves was typical of the second (natal) pelage, with curly blond hairs extending several millimeters above the skin surface. Exfoliative dermatitis occurs as localized or generalized erythema and skin peeling (scaling), the latter often associated with hair loss, but is typically associated with an infectious disease, such as leishmaniasis in dogs, neoplasia, and other insults. 16 Characteristic histopathologic features of exfoliative dermatitis include lymphocytic interface dermatitis, hydropic degeneration of basal keratinocytes and apoptotic keratinocytes, hyperkeratosis (orthokeratotic or parakeratotic), desquamation, atrophic or absent sebaceous glands, and various degrees of perivasculitis. 8 Although desquamation and orthokeratotic hyperkeratosis were characteristic features in affected skin sections, we did not observe other histopathologic features of exfoliative dermatitis. Thus, we ruled out exfoliative dermatitis. In addition, hereditary zinc deficiency, as reported in dogs and cattle, was also excluded based on histopathologic features including no perivasculitis or parakeratotic hyperkeratosis. 13 PCR testing for various exfoliative toxins was also negative. However, culture was positive for bacteria that can be opportunistic pathogens (e.g., S. phocae) in marine mammals. 15 In sea otters, skin trauma is significantly associated with S. phocae detection and subsequent localized or systemic bacterial spread. 2 In our case material, inflammation and vasculopathies were absent in skin sections, and we observed no significant histopathologic findings in non-cutaneous organs. Thus, we do not consider that the isolated bacteria were pathologic in our case material.
We did not detect any mutations of the KRT10 gene; hence, we are unable to state equivocally that these cases are ichthyosis based on the available genetic information. However, because we did not successfully sequence all regions of the KRT10 gene, we cannot rule out mutations of the KRT10 gene playing a role in this novel PSC in Pacific walrus. Additionally, different keratin genes (e.g., KRT1, KRT2) may be responsible.
A conservative estimate of overall and annual prevalence of this novel skin condition in Pacific walrus calves is 4 of 636 (0.006%); 1 of 481 (0.002%) for 2012, 2 of 64 (0.031%) for 2014, and 1 of 91 (0.011%) for 2021. Calf harvest estimates are based on observed cow:calf ratios on Saint Lawrence Island and corrected for adult female non-reporting compliance and “struck and loss” based on an estimate of 42%. 6 Clinically speaking, we can only speculate on the negative implications of this novel PSC on calf survival in the long run. In other species with genodermatoses, negative energetic consequences caused by the ongoing needs of skin repair, heat and fluid loss, and secondary infections have been observed. 17 Given their challenging Arctic nursing habitat characterized by various sea ice substrates with cold ambient temperatures, and their greater vulnerability to increased heat loss as a result of their large surface area and limited blubber resources, we speculate that neonatal mortality of affected calves is likely higher.
Footnotes
Acknowledgements
We thank the marine mammal hunters and their communities on Saint Lawrence Island for sharing their indigenous knowledge and observations about walrus health and diseases with us. Special thanks to Vera Metcalf, Executive Director, and Charlie Brower, Chair of the Eskimo Walrus Commission, for their support and continuing encouragement of walrus health–related studies. We also want to thank Yosty Storm for postmortem examination assistance, and Anna Bryan with the Alaska Department of Fish and Game for logistical support in carcass shipment.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article. The findings and conclusions of the authors are their own and do not necessarily represent the views of the U.S. Fish and Wildlife Service.
Funding
Our study was partially funded with qualified outer continental shelf oil and gas revenues by the Coastal Impacts Assistance Program, Fish and Wildlife Service U.S. Department of Interior, USFWS walrus disease and mortality assessment grant (F12AC01607; F20AP10932), and the North Slope Borough, Department of Wildlife Management.
