Abstract
The Western honey bee (Apis mellifera) is economically important as the primary managed pollinator of many agricultural crops and for the production of various hive-related commodities. Honey bees are not classically or thoroughly covered in veterinary pathology training programs. Given their unique anatomic and biological differences from the other species more traditionally evaluated by veterinary pathologists, establishing routine and consistent methods for processing samples for histology ensures accurate diagnostic and research conclusions. We developed and tested several field protocols for the sampling of honey bees. We compared the tissue-quality outcomes for worker bees fixed, collected, and/or softened under the following protocols: 1) routine formalin fixation; 2) softening chitin via exposure to Nair for 2 d or 3) 5 d; 4) shortened times between formalin submersion and trimming of body segments to enhance penetration of formalin into internal tissues; 5) ethanol submersion of specimen prior to formalin fixation; 6) indirect dry ice exposure; and 7) prolonged −80°C storage. Routine formalin fixation, exposure to Nair for 2 d, indirect dry ice exposure, and trimming body segments within 2 h of formalin submersion resulted in the highest quality histologic tissue sections. The poorest quality sections resulted from softening of chitin by exposure to Nair for 5 d, submersion in ethanol for 3 d before formalin fixation, and prolonged storage at −80°C. Our results indicate that routine formalin fixation is adequate, and that immobilizing bees with indirect dry ice exposure aids in sample collection without negatively impacting the quality of histologic sections.
The Western honey bee (Apis mellifera; hereafter, honey bee) is one of the most globally widespread organisms, economically important for its role as the pollinator of a range of agricultural crops, in addition to the production of honey, propolis, beeswax, and other related products.5,22,28 Although honey bees and other invertebrate species are not thoroughly covered in veterinary pathology training programs, the agricultural and economic importance of honey bees warrants greater attention by veterinarians, including veterinary pathologists, as has been the case recently.13,15,27 There has also been a general increase in interest in invertebrate pathology by veterinary pathologists, as evidenced by the release of new textbooks and journal articles on these subjects directed particularly at veterinary pathologists.13,15,16 This growing interest follows the relatively new U.S. Food and Drug Administration (FDA) rule that aims to reduce the use of antimicrobials in food-producing animals, preventing the contribution to widespread development of antimicrobial resistance; the rule now requires beekeepers to obtain a veterinary feed directive or a prescription from a veterinarian for antimicrobial use to manage bacterial diseases such as American foulbrood and European foulbrood in honey bee colonies. 33 Research and management efforts to ensure the health of honey bees is crucial to ensure the sustenance of healthy and functioning agricultural and natural ecosystems.
Honey bee populations have been declining because of challenges from biotic and abiotic factors, including climate change–related stressors, habitat loss–mediated compromises to nutrition, exposure to environmental toxins, and debilitating pests and pathogens.8,19,29,31 The effects of stressors have been exacerbated in California as the demand for commercial pollination services in fruit and nut orchards continues to grow, requiring colonies to be brought into the state from across the United States, and creating a perfect opportunity for pest and pathogen spread. 32 Health of honey bees has been challenged by the prevalence of devastating pests such as the parasitic mite, Varroa destructor, and pathogens that include a host of different debilitating viruses, bacteria, fungi, and microsporidians.8,29 The increase in pathogens and related disease incidents, and the importance of honey bee pollination services for the successful production of economically important agricultural crops, validates the need to develop and standardize techniques for identifying diseases and other stressors in bees to enable the prompt development of appropriate mitigation strategies. We investigated the use of histologic techniques to evaluate normal honey bee tissues and identify the presence of pathogens, specifically Nosema spp., which cause premature aging in individual bees. In the past, colonies were often able to cleanse themselves of Nosema spp. following a nectar flow with flowering plants blooming as the season progressed. 24 However, Nosema spp. infections have become chronic, potentially compounding the impact of other biotic and abiotic stressors on colonies.21,25
Although many fixatives are used for histologic applications, they all carry advantages and disadvantages.1,3 Fixatives other than 10% neutral-buffered formalin (NBF, “formalin”) described in the honey bee literature used for histologic purposes include other formaldehyde-based formulations, such as Bouin fixative.12,34 Considerations in selecting a fixative include access or availability, safety of the fixative, and post-fixation applications (e.g., light vs. electron microscopy; immunofluorescence; immunohistochemistry). For example, Bouin fixative produces crisp staining of delicate tissues with superior staining quality. However, Bouin fixative is not adequate for tissues intended for electron microscopy or immunohistochemistry; additionally, this fixative contains picric acid, which is highly explosive.3,4
The tissue fixative used most routinely by veterinary pathologists is 10% NBF. 9 There are unique challenges in the histologic evaluation of insects, including the whole bodies of honey bees. Insects have a chitinous exoskeleton that may impede formalin penetration of internal tissues.2,26 Further, the chitinous exoskeleton can result in artifactual fracturing of tissue when sectioned after paraffin embedding given that the exoskeleton is the first structure through which the microtome blade must pass. Depending on the goal of the research, artifactual fracturing of tissues could be avoided by dissecting out individual target organs for microscopic evaluation. However, dissection is not always practical and does not allow whole-body processing and evaluation. Minimizing tissue-fracture artifact may be achieved by softening the chitinous exoskeleton, but there are limited descriptions of approaches to processing whole honey bees (and other invertebrates) with softening agents, and many methods may require reagents that are uncommonly used or stocked. The protocols may also be complex and often impractical for most routine histology and research laboratories.2,10,20 This challenge extends beyond honey bees; larger invertebrates present more substantial challenges with increasingly thick cuticles, as in, for example, tarantulas, for which the commercially available Nair, an at-home hair removal agent, has been used to successfully soften the chitinous exoskeleton. 20
Field manipulation of honey bees often requires placing bees on dry ice immediately upon collection as a method of anesthesia or euthanasia before transport to the laboratory. This routine technique is used by both researchers and beekeepers, and does not generally impact other downstream applications, including molecular protocols, such as PCR and gene expression analyses. However, exposing tissues to freezing temperatures is typically avoided in histology because freezing results in variable “freeze artifact,” characterized by vacuolation, loss of cellular detail, and empty spaces within tissues.30,35 The degree of freeze artifact in honey bees, secondary to brief and indirect dry ice exposure, and the utility of specimens treated in this manner are not well understood nor reported in the literature.
After field collection is completed, honey bees are frequently stored in freezers at −80°C until further processed, and research facilities may have years of collected and stored honey bees that may hold extremely valuable historical records of honey bee health. However, this practice poses a risk of tissue damage secondary to freeze artifact. Histology is used increasingly in honey bee research, and determining the impact of freezing on histology is warranted.
An alternative and equally common practice in the field is the collection of honey bees in ethanol (typically 70–100%) to preserve specimens for DNA extraction and analyses. 33 Ethanol could potentially render specimens brittle or otherwise difficult to handle during processing for histology.6,23 Although storage in ethanol may be superior for certain molecular applications, formalin is the preferred fixative utilized by pathologists for routine fixation and is superior to alcohol-based fixatives because of cellular and tissue artifacts created by fixation in ethanol, including cell shrinkage, condensation of chromatin, and morphologic alterations to stromal tissue. 17 Despite unavoidable ethanol-induced tissue artifact, there may still be utility in these specimens collected into ethanol in the field, particularly if transported back to a laboratory within a reasonable timeframe, where they can be transferred to formalin before processing to prevent further development of tissue artifacts.
To address some of these unknowns and develop protocols to be used in future research efforts, we tested several common and novel collection, preservation, and fixation approaches for honey bees and their impact on overall quality of routine histologic sections stained with H&E. We also demonstrated that some of the specific protocols allowed for the identification of Nosema spp. spores, indicating the potential for use in pathogen and pest identification.
Materials and methods
Animals
We used honey bees (Apis mellifera ligustica) sourced from colonies headed by open-mated Italian queens purchased from Tauzer Apiaries (Davis, CA, USA) and reared at the U.S. Department of Agriculture–Agriculture Research Service (Davis, CA, USA) in May 2022. Specimens for all groups were collected from a single standard Langstroth hive and managed consistent with industry standards. 18
Field collection, fixation, softening, and processing
To evaluate and standardize sample collection and processing for histologic sections, collected honey bees were allocated into 7 different groups:
Group 1 (“24-h formalin”) bees were collected into a 50-mL conical tube (Corning Falcon 50-mL conical centrifuge tubes, Fisher Scientific; Fig. 1), capped with a modified cap with 8-mesh hardware cloth, and then fixed for 24 h in 10% NBF before trimming into tissue cassettes (Tissue-Tek Uni-cassette; Sakura).
Group 2 (“2-d Nair”) bees were collected as in group 1. After 24 h of NBF fixation, the specimens were submerged in 10% Nair (Church & Dwight), diluted in PBS (Gibco) for 48 h, thoroughly rinsed with PBS, and subsequently trimmed into cassettes. The Nair solution was diluted with PBS in a glass beaker using a stir bar and magnetic stir plate to agitate the Nair. A 15-mL syringe was used to measure the Nair and gradually add it to the PBS while increasing the speed of the stir bar as needed to achieve the consistency of “thin yogurt” based on a protocol described previously. 20
Group 3 (“5-d Nair”) bees were collected and treated as in group 2 but submerged in the 10% Nair solution for 5 d before being rinsed thoroughly with PBS.
Group 4 (“Indirect dry ice”) bees were anesthetized (immobilized) via indirect exposure to dry ice for 1 min and then treated as in group 1. Bees were placed on 2 layers of thin cotton towels covering a block of dry ice within a rotomolded cooler (Yeti; Fig. 1A). The towel barrier was created to minimize development of freeze artifacts in the tissues. Both the sublimation of CO2 and the cold temperature act to immobilize the bees making it easier to subsequently submerge bees in 10% NBF.
Group 5 (“2-h trim”) bees were collected into formalin as in group 1, trimmed into cassettes within 2 h of initial submersion into formalin, and then fixed for the remaining 22 h (total of 24 h) and stored in formalin until paraffin embedding and sectioning onto glass slides.
Group 6 (“3-d ethanol”) bees were collected directly into 70% ethanol in a 50-mL conical tube where they remained for 3 d, transferred to formalin for 24 h, and then trimmed into cassettes.
Group 7 (“−80°C”) bees were stored at −80°C for ~9 mo, removed from the freezer, and immediately placed into ice-cold formalin to thaw.

Field methods for the collection of honey bees for histologic evaluation.
We collected 10–15 bees per group from a frame with developing larvae of various ages known to be attended by nurse bees (as per commonly used beekeeping and research practices),11,14 thus controlling for any minor anatomic variations. Bees were collected by moving the 50-mL conical tube up and down the frame and gently manipulating the bees into the tube before closing the lid, which was modified by cutting out the solid flat portion and replacing it with 8-mesh hardware cloth to facilitate filling of tubes with fixative (Fig. 1B). All collected specimens, except for groups 4 and 7, were immediately submerged in 10% NBF by pouring it through a small funnel into the collection tubes. After immobilization and cessation of all movement (presumed death), 4 × 4 surgical gauze (Dealmed) was placed in the conical tubes, and tubes were closed with intact lids to keep bees submerged in the fixative and ensure contact of the 10% NBF with the individual honey bees.
Five honey bees per group were trimmed into cassettes by a single technician (L. Rivera). The head, thorax, and abdomen were separated by utilizing spring scissors (World Precision Instruments [WPI], WPI-14111-G). The head and thorax were sectioned transversely, and the abdomen was sectioned in sagittal orientation along the midline using a carbon-steel surgical blade (Miltex) that was replaced every 3 samples to prevent dulling; all tissues from one bee were placed in a single cassette (Fig. 2). All cassettes were stored in 10% NBF and shipped to a histology laboratory (SpecialtyVETPATH, Seattle, WA, USA) for processing: routine paraffin embedding, sectioned at 4 μm, mounted on glass slides, and stained with H&E. Two consecutive sections of all 3 body segments were mounted on a single glass slide.
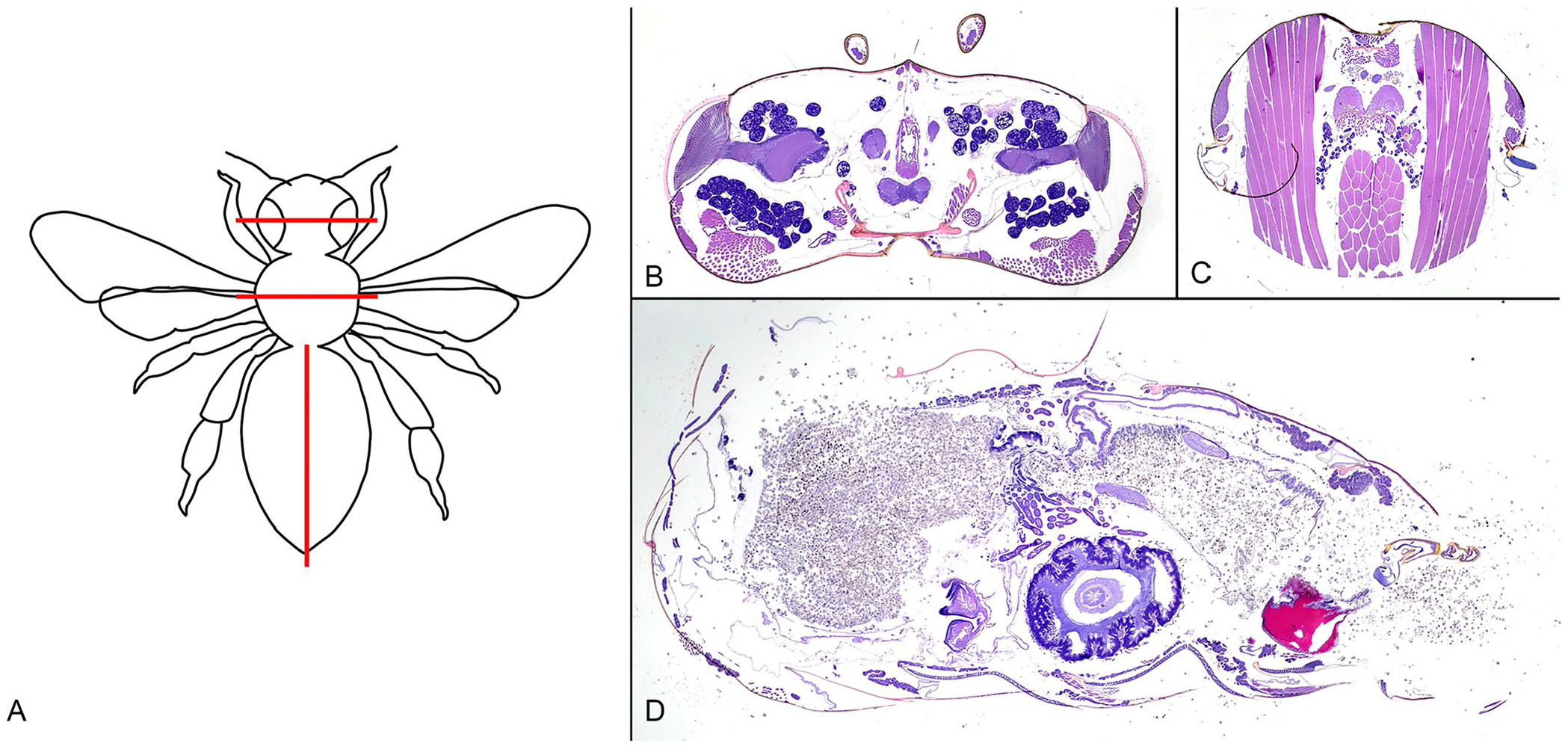
Honey bee sectioning.
Histologic evaluation
The honey bee sections were evaluated by 2 board-certified veterinary pathologists (S.E. Cook, C.E. Alex) for overall quality, tissue fragmentation, and cellular integrity and detail for each treatment group. All slides were initially evaluated independently by each pathologist, one of whom was anonymized to the treatment groups. Subsequently, both pathologists evaluated each slide together and, for any disagreement in histologic scoring, reached a consensus. Interobserver variation was low and, when there were interobserver differences, they almost exclusively differed by only a single scoring unit (scale 0–4).
Histologic assessment was carried out via a systematic scoring system based on the principles of valid histologic scoring in research. 7 Each body segment was scored independently for 1) degree of tissue fragmentation and 2) loss of cellular detail and integrity. Tissue fragmentation was defined as fragmentation or fracturing of the tissue section and/or displaced or absent tissue. Loss of cellular detail and integrity was defined as shrinkage and/or loss of cytoplasmic or nuclear detail. These categories were given a histologic score of 0–4. Scores were defined as follows: 0 = no displacement or loss of tissues and distinct cellular and nuclear detail, 1 = a single focus or a few minor foci of either tissue fragmentation or loss of cellular/nuclear detail, 2 = moderately affected regions (< 33% of tissue affected), 3 = severely affected or large coalescing regions (33–66% of tissue affected), and 4 = markedly affected to nearly entire section affected (> 66% of tissue affected).
A combined score (tissue fragmentation combined with cellular and/or nuclear detail across each body segment) was calculated for each bee, with higher combined scores indicating overall worse histologic tissue quality. Kruskal–Wallis and post-hoc Dunn multiple comparisons tests were performed to determine the significant effect of groups on the variance and pairwise differences between groups, graphically depicted using box plots. All statistical analyses and graphing were performed using Prism 9 (GraphPad). Any finding of Nosema spp. infection was noted for each bee evaluated.
Results
Histologic scoring and tissue artifacts
There were statistically significant effects of treatment groups on the combined histologic score (Kruskal–Wallis, p = 0.0002, H = 26.5) with specific statistical differences identified with a Dunn multiple comparisons test (Fig. 3).
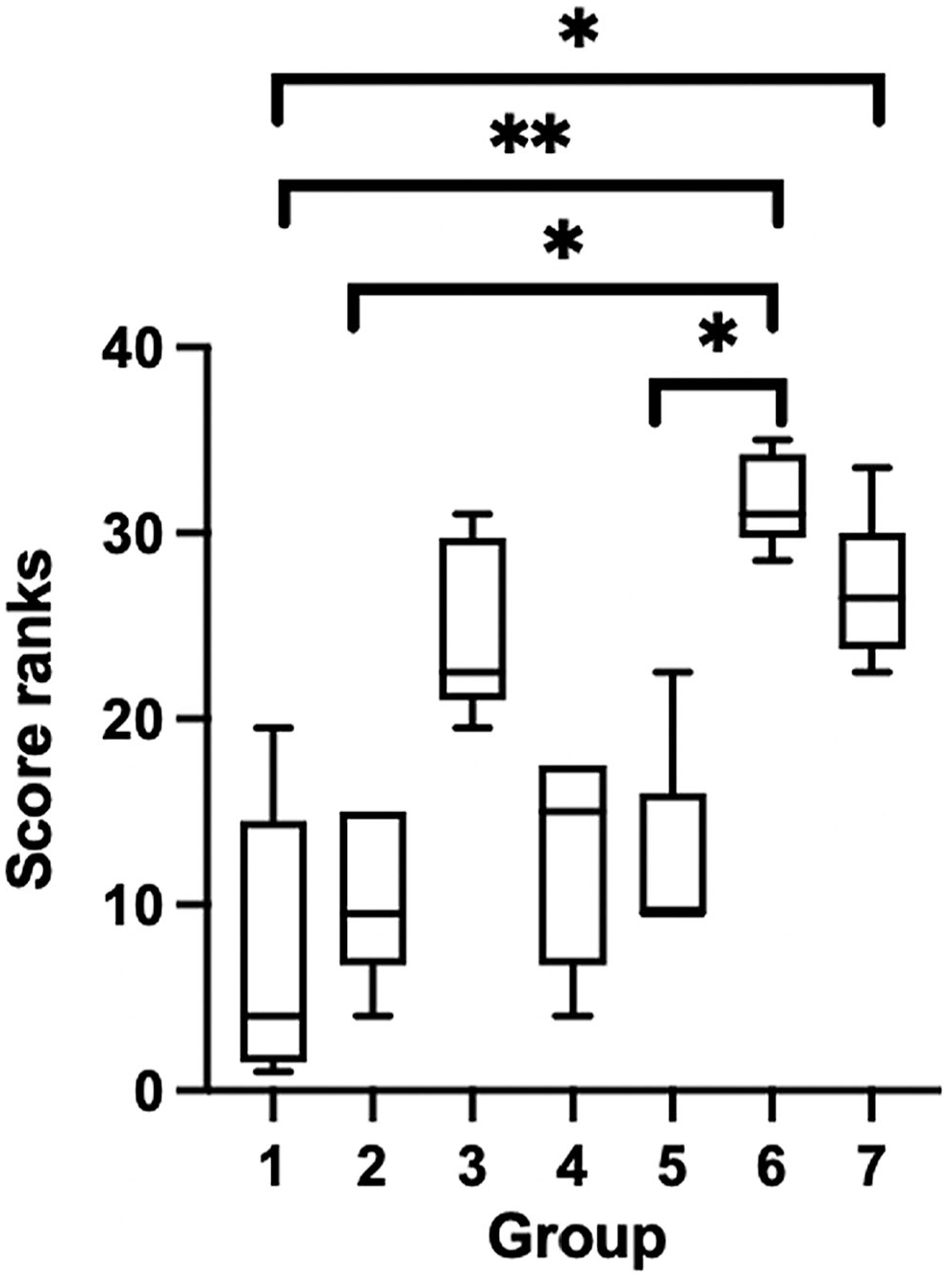
Box plots of combined histologic score ranks across treatment groups. Groups: 1 = routine formalin fixation; 2 = 2-d Nair; 3 = 5-d Nair; 4 = indirect dry ice; 5 = 2-h trim; 6 = 3-d ethanol; 7 = −80°C. Upper and lower whiskers represent the maximum and minimum rank; the interior bar is the median. Bars represent the 25th and 75th percentiles.
When evaluating the degree of tissue fragmentation across groups and by individual body segment, there was overall statistical significance between treatment groups with regards to the sections of head (Kruskal–Wallis, p = 0.03, H = 13.9), although no specific statistical differences were identified with the post-hoc Dunn multiple comparisons test. There was no statistical significance between groups for degree of tissue fragmentation in sections of thorax or abdomen (Fig. 4).
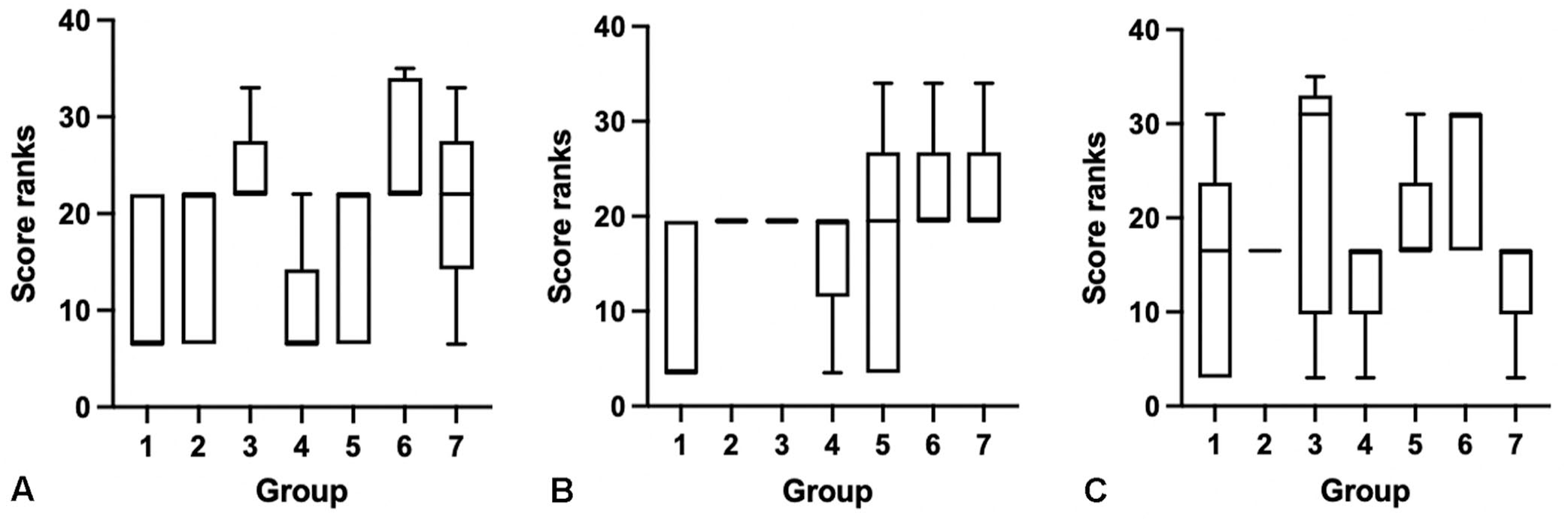
Box plots of tissue fragmentation score ranks across treatment groups: (
A spectrum of histologic changes was identified consistent with loss of cell detail and loss of integrity. With decreasing cell detail, there was shrinkage and separation of cells, vacuolation, cell lysis, and shrunken, hyperchromatic-to-indistinct nuclei (Fig. 5).
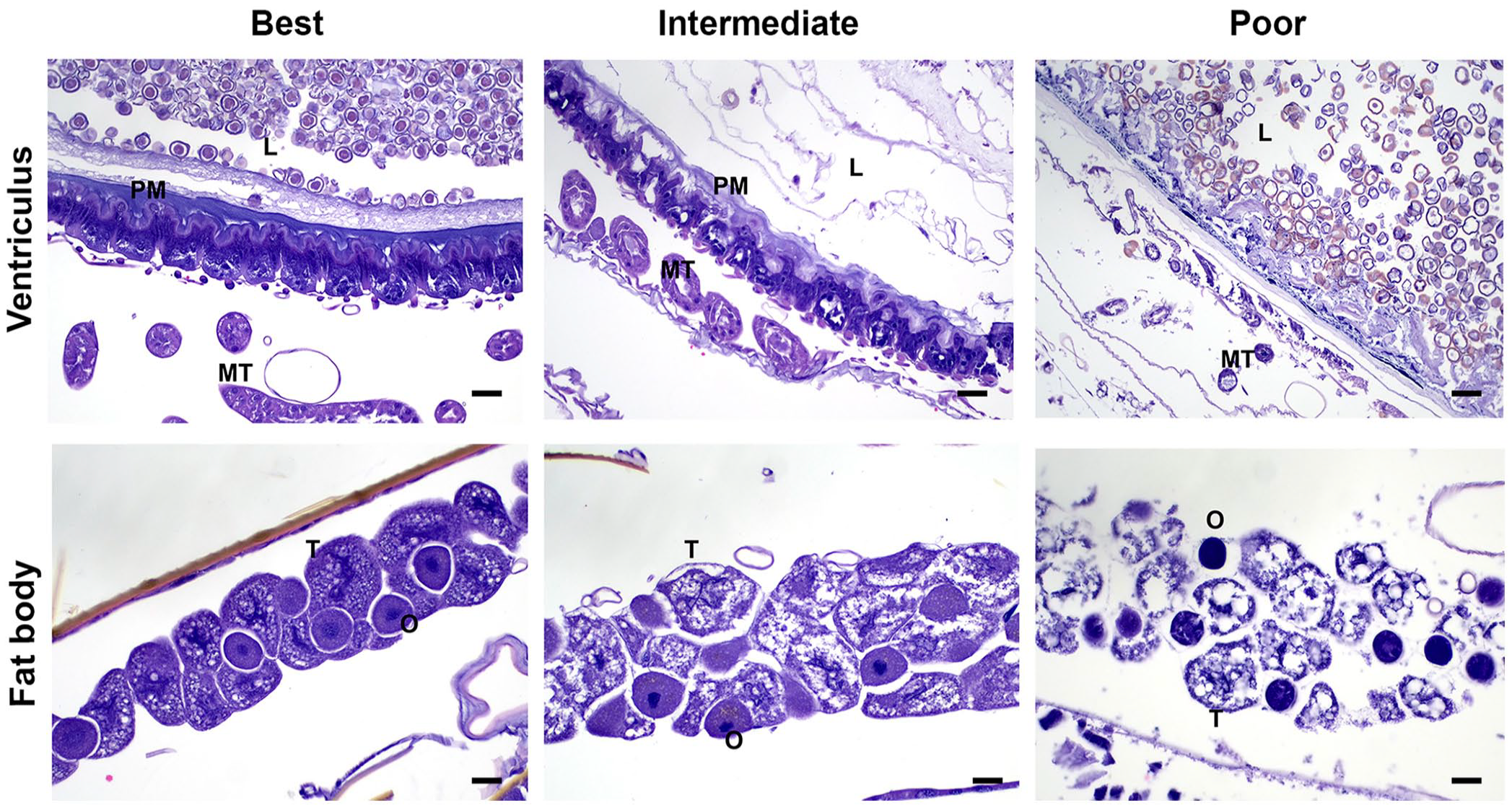
Images of cellular detail and integrity of highest to lowest quality (left to right) for sections of ventriculus and fat body. The left column depicts images of best quality from group 1 (upper) and group 5 (lower). The middle column depicts examples of intermediate quality from a bee in group 7. The right column depicts poor tissue quality images from group 6. Note the increasing shrinkage of cells, vacuolation and lysis of cells, and shrunken, hyperchromatic nuclei with increasingly poor tissue quality.
For cell detail and integrity, there was statistical significance between treatment groups with regards to the head (p = 0.004, H = 24.9), thorax (p < 0.0001, H = 28.8), and abdomen (p = 0.0003, H = 25.1) using a Kruskal–Wallis test (Fig. 6). The Dunn post-hoc multiple comparisons test identified a number of specific statistical differences between groups when evaluating sections of head, thorax, and abdomen (Fig. 6). Group 6 (3-d ethanol) was most commonly significantly different from other groups by stepwise comparisons.
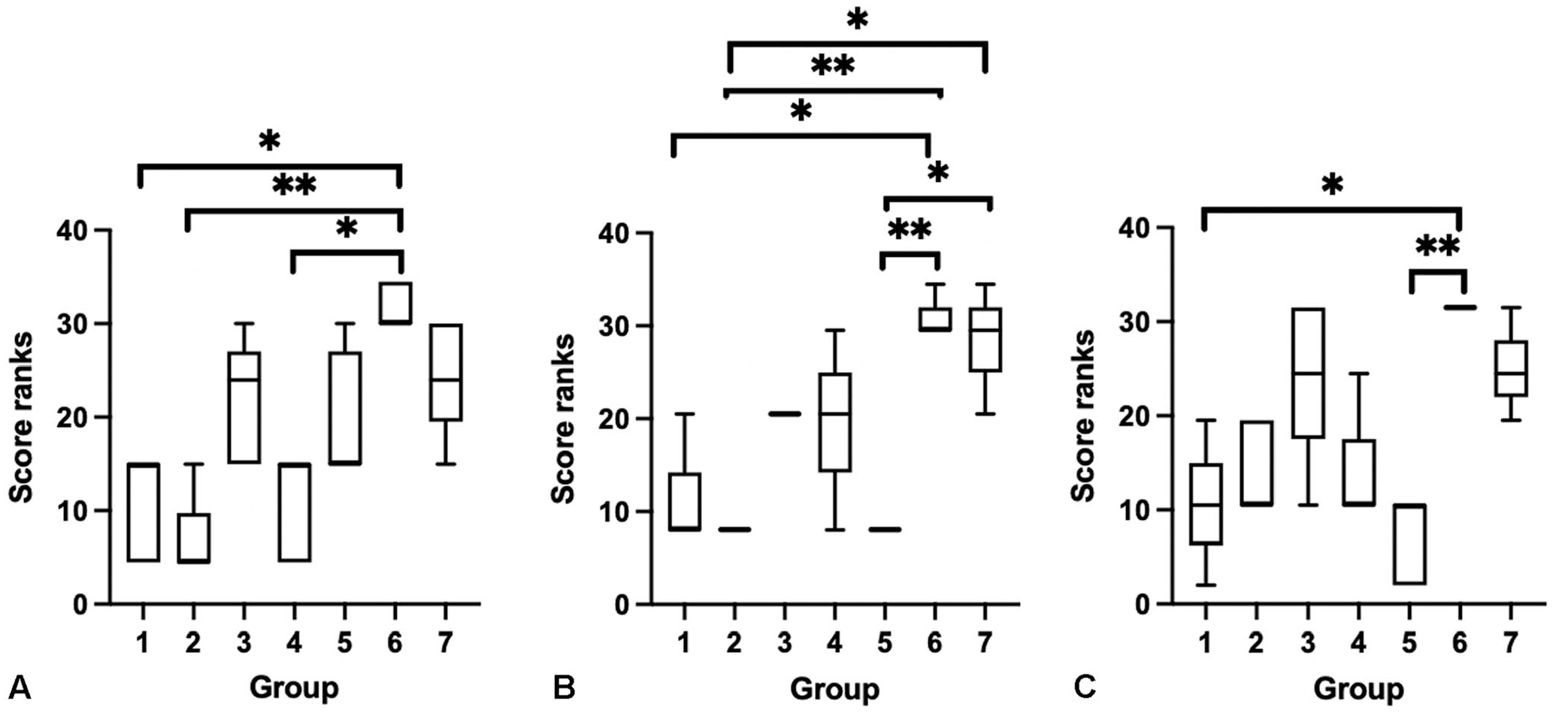
Box plots of cell detail and integrity score ranks across treatment groups: (
Overall, the groups with the best cell detail and integrity were routine formalin (group 1), 2-d Nair (group 2), and indirect dry ice (group 4), and the worst quality groups were 3-d ethanol (group 6) and −80°C (group 7), with 5-d Nair (group 3) trending toward, but not statistically significant, qualifying as one of the poorest quality treatments.
Nosema detection
Nosema spp. organisms were found in 4 bees: 1 in group 1, 1 in group 4, and 2 in group 7 (Fig. 7). Bees in group 6 were not of adequate quality to detect Nosema spp. Ventricular epithelial cells in affected bees were distended with abundant spores along with occasional free luminal spores.
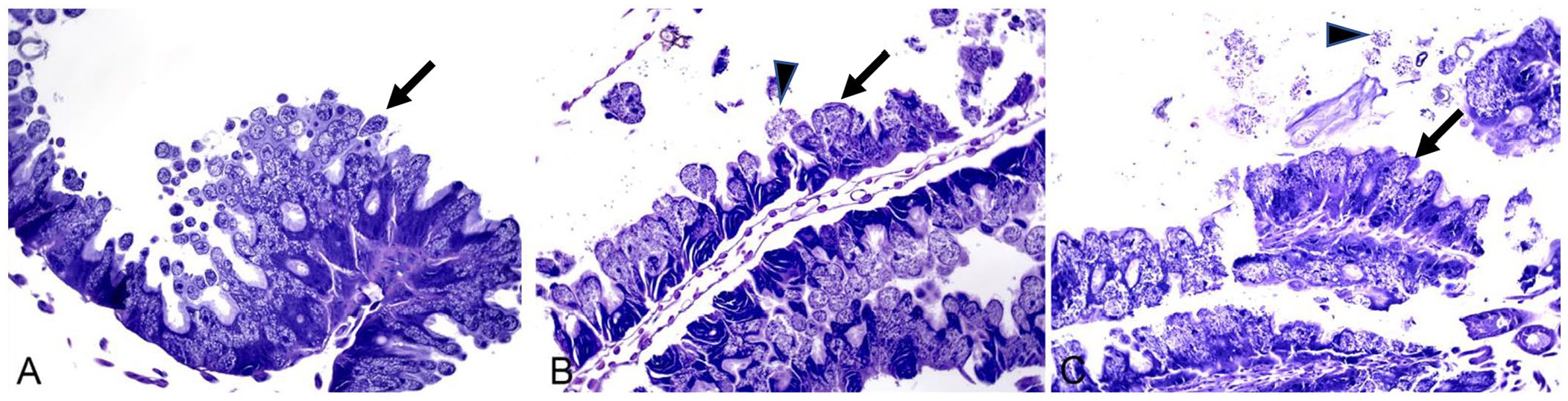
Sections of ventriculus, with epithelial cells, are distended with abundant Nosema spp. spores (arrows), as well as ventricular luminal spores (arrowheads) in (
Discussion
We found that the histologic quality outcomes for the processing and microscopic evaluation of whole honey bees were dependent on specific sample collection and fixation methods. We also established a systematic scoring system for assessing quality of histologic tissue sections based on the severity of tissue fragmentation and cellular detail and integrity. This resulted in limited interobserver variation between 2 veterinary pathologists, and the successful scoring of bees across the designated study groups. Finally, we documented the ability to detect Nosema spp. life stages across different study groups, including those with overall poor tissue quality but not those initially fixed in ethanol, which lacked adequate cellular detail.
Our finding of tissue damage by Nair confirms that the use of Nair as a softening agent for Western honey bees is not only an unnecessary additional step but in fact may reduce the quality of histologic tissue sections. Conversely, others have found that prolonged submersion in Nair solution, a similar formulation to that used in our study, has been used with other invertebrates, such as arachnids (specifically tarantulas), with improved histologic outcome quality, likely as a result of size differences. 20 Thus, our result should not be extrapolated to other invertebrates, and processing and softening approaches should be individually optimized for different species and sizes of invertebrates.
In addition, the lack of significant difference in the quality of histologic tissue sections between routine formalin fixation and exposure to indirect dry ice for immobilization or anesthesia is of particular interest because dry ice is used commonly in the field during collection of honey bees to manipulate bees more easily and potentially more safely into collection tubes.
One concern regarding the fixation of invertebrates is the potential for delayed fixation of internal soft tissues as a result of delayed penetration of fixatives through the chitinous exoskeletons.2,26 Minimizing the time between submersion in formalin and transection of body segments (e.g., trimming body segments into cassettes) may enable faster penetration of internal tissues by the formalin and, thus, result in higher quality tissue sections and cellular detail. We found no difference in sections from honey bees that were trimmed into cassettes within 2 h of submersion into formalin and those routinely fixed for a minimum of 24 h (as whole honey bees) before trimming into cassettes. Therefore, we consider trimming within 2 h of submersion to be unnecessary.
We evaluated the effect of initial exposure to ethanol (3 d) prior to transferring bee samples into formalin for histology processing to duplicate the field scenario in which honey bees are collected in ethanol and then either shipped or transferred into formalin within 3 d. Ethanol is more commonly accessible for use in honey bee fieldwork; formalin is not always available nor is it stocked routinely by honey bee research laboratories. Initial exposure to ethanol for 3 d resulted in a significant loss of quality in histologic tissue sections, including increased tissue fragmentation, with the worst overall histologic score. The increase in tissue fragmentation is suspected to be secondary to increased brittleness, which is known to occur variably with ethanol fixation. 23 Tissue sections were considered nondiagnostic for the ethanol group, and although Nosema spp. could be detected in each group, the very poor quality of the ventriculus in ethanol group bees precluded detection of this agent. We, therefore, do not recommend storage of bees in ethanol if histologic evaluation is intended.
Honey bee researchers have commonly collected and stored whole honey bees in −80°C freezers for future applications, such as molecular analyses. Although overall quality was reduced, as expected, Nosema spp. remained detectable in bees from this treatment group, which implies potential use toward documenting the historical prevalence of this pathogen.
We found that manipulation of samples beyond routine fixation with 10% NBF for a minimum of 24 h did not result in improved quality of histologic tissue sections, and the use of indirect dry ice, which aids in handling of honey bees, did not reduce tissue quality. Ethanol produced tissue sections of nondiagnostic quality. Prolonged storage of bee samples at ultra-low temperatures caused the predicted freeze artifact, with reductions in tissue quality, although not beyond the ability to diagnose nosemosis. Lastly, use of Nair or other softening agents on honey bees is not necessary and can lead to reduced tissue quality in the event of prolonged exposure, which can impede accurate histologic analysis.
Footnotes
Acknowledgements
The statements made in the article represent the authors’ views and should not be interpreted as endorsement from their respective employers or the funding agencies. Mention of tradenames or commercial products in this publication is solely for the purpose of providing specific information and does not imply recommendation or endorsement by the University of California–Davis or the U.S. Department of Agriculture (USDA). USDA is an equal opportunity provider, employer, and lender.
Editor’s note
Declaration of conflicting interests
The authors declare no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
Funding for our research was in part provided by various donors to Elina L. Niño and the USDA-ARS cooperative agreement 58-2030-0-040 to E.L. Niño, as well as USDA-ARS 2030-21000-055-000-D funds to Bernardo D. Niño, Laura Rivera, and Arathi Seshadri.
