Abstract
Over a period of ~2 wk, 5 adult beef cows developed severe necrotizing skin lesions on the face and neck shortly after being moved to a field with a cover crop of Brassica spp. turnips. We describe here the clinical signs, hematology, and serum chemistry results, as well as gross and histopathologic findings, from this outbreak. We made a presumptive diagnosis of Brassica-associated liver disease (BALD) based on the similarities in the history and diagnostic findings observed with previously reported cases of BALD from elsewhere in the world. BALD has not been reported previously in cattle in North America, to our knowledge, despite the increasing usage of cover crops to improve soil conditions and provide forage for livestock. After the presumptive diagnosis of BALD was made, the cattle were removed from the turnip field and no additional cases were noted by the producer. BALD is well known in other parts of the world, and veterinarians and diagnosticians should be aware of this condition given that the usage of cover crops is likely to continue to increase.
Cases of photosensitivity dermatitis in livestock species are traditionally classified into 1 of 3 (and sometimes 4) categories based on the underlying mechanism of lesion development. 9 Primary photosensitization (type I) is most commonly the result of ingestion of plants containing photoreactive compounds that are absorbed into circulation and deposited near the skin surface. Congenital photosensitization (type II) is rare, and is the result of a deficiency in one of several enzymes responsible for heme synthesis. Hepatogenous photosensitization (type III) is the result of the accumulation of phylloerythrin in tissues as a result of impaired biliary excretion of the compound. The inability to excrete phylloerythrin is most often attributed to primary hepatocellular damage, but is also suspected to be caused by cholestasis to a lesser degree. Type III photosensitization is the most common form in domestic species worldwide, accounting for 68.5% of all cases in a review of the prevalence of the different categories. 3 Type IV photosensitization is occasionally used to classify cases in which the pathogenesis or etiology is unknown.
Various plants that are suspected or proven to cause type III photosensitization in domestic animals include Panicum spp., Brachiaria spp., Narthecium ossifragum, and Tribulus terrestris, among others.3,9 The mycotoxin sporidesmin is also well characterized as a cause of facial eczema of cattle and sheep in locations throughout the world.2,9 Hepatogenous photosensitization in cattle grazing Brassica spp. of forage is well documented, particularly in Australia and New Zealand, although the precise toxic principle and mechanism remain unknown.3,5,8,9 The term Brassica-associated liver disease (BALD) has been proposed for this condition in cattle. 8 We retrieved no reported cases of BALD in North America in a search of Google, PubMed, CAB direct, Web of Science, and Scopus when using the search terms “brassica”, “turnip”, “photosensitization”, “cattle”, suggesting that this condition has not been reported in cattle in North America. Additionally, we consulted with 2 researchers (United States and New Zealand) who have published papers on plant-induced photosensitization and BALD in other parts of the world, and neither was aware of a presumptive BALD case being reported in North America in cattle.
The most prominent clinical sign associated with BALD is dermatitis of variable severity ranging from mild erythema to necrosis of areas of skin that may slough. 5 Areas of non- or poorly pigmented skin around the face, ears, or teats are affected most commonly. As with other photosensitization conditions, there are no direct treatments, and therapy is focused on supportive care, including limiting sunlight exposure, corticosteroids, and antibiotics to mitigate risk of secondary skin infections. Clinical chemistry findings tend to include elevations in some or all of gamma-glutamyl transferase (GGT) and glutamate dehydrogenase (GDH) activities and bilirubin concentrations in serum.4,5
The clinical signs and serum chemistry findings of BALD are very similar to those described in cases of facial eczema. 4 Differentiation between these conditions requires either a strong suspicion given the forage being consumed by the animals, a spore count, or known porphyrin levels. The toxic principle in cases of BALD is unknown; therefore, no targeted analyses exist. Differences have been described in bile duct lesions between BALD and facial eczema, although the study that described these differences examined tissues from only 2 BALD and 3 facial eczema cases. 4
Tissue, water, free-choice mineral, and turnip samples were submitted to the Nebraska Veterinary Diagnostic Center (University of Nebraska–Lincoln, Lincoln, NE, USA) in December 2021 from a herd of ~300 cow–calf pairs in Polk County, NE that had recently experienced 2 unexplained deaths (cows 1, 2) with 2 additional cattle (cows 3, 4) with clinical signs. Skin lesions began several days after the cattle were moved to a newly harvested corn field that had been planted with a Brassica spp.–containing cover crop blend. Initial sample submission occurred 2 wk after the onset of disease. The submission form noted that cows had intense pruritus of the face and head as evidenced by excessive rubbing on pivot wheels, fence posts, and herdmates. Affected cattle developed alopecia, primarily on the face and neck. The affected cows were also photophobic and isolated themselves in the shade during daylight hours.
Cow 2 was the second animal to die and was the source of the submitted tissue samples. A field autopsy was performed ~24 h after death; the carcass was partially frozen. The carcass was found within a small tree line at the edge of the field. The submitting veterinarian noted coalescing areas of alopecia on the face and neck (Fig. 1), blood within the nares and mouth, consolidation of the left cranial lung, unclotted blood filling the tracheal lumen, severely autolyzed kidneys, and a diffusely gray liver with advanced autolysis. Histologically, the lung had chronic, mild, multifocal, lymphocytic and eosinophilic bronchiolitis with emphysema. Sections of haired skin had chronic, mild, multifocal, lymphocytic, perivascular dermatitis with mild hyperkeratosis. Sections of liver were markedly autolyzed, which inhibited meaningful histologic examination. At the time of submission, the producer was concerned about polioencephalomalacia (PEM) from excess sulfur given losses attributed to PEM in years prior. Sulfur analysis of the pond water and turnip samples determined a total dietary sulfur concentration of 0.01%, which is well below the maximum tolerable dietary sulfur concentration of 0.4% for cattle.7,11
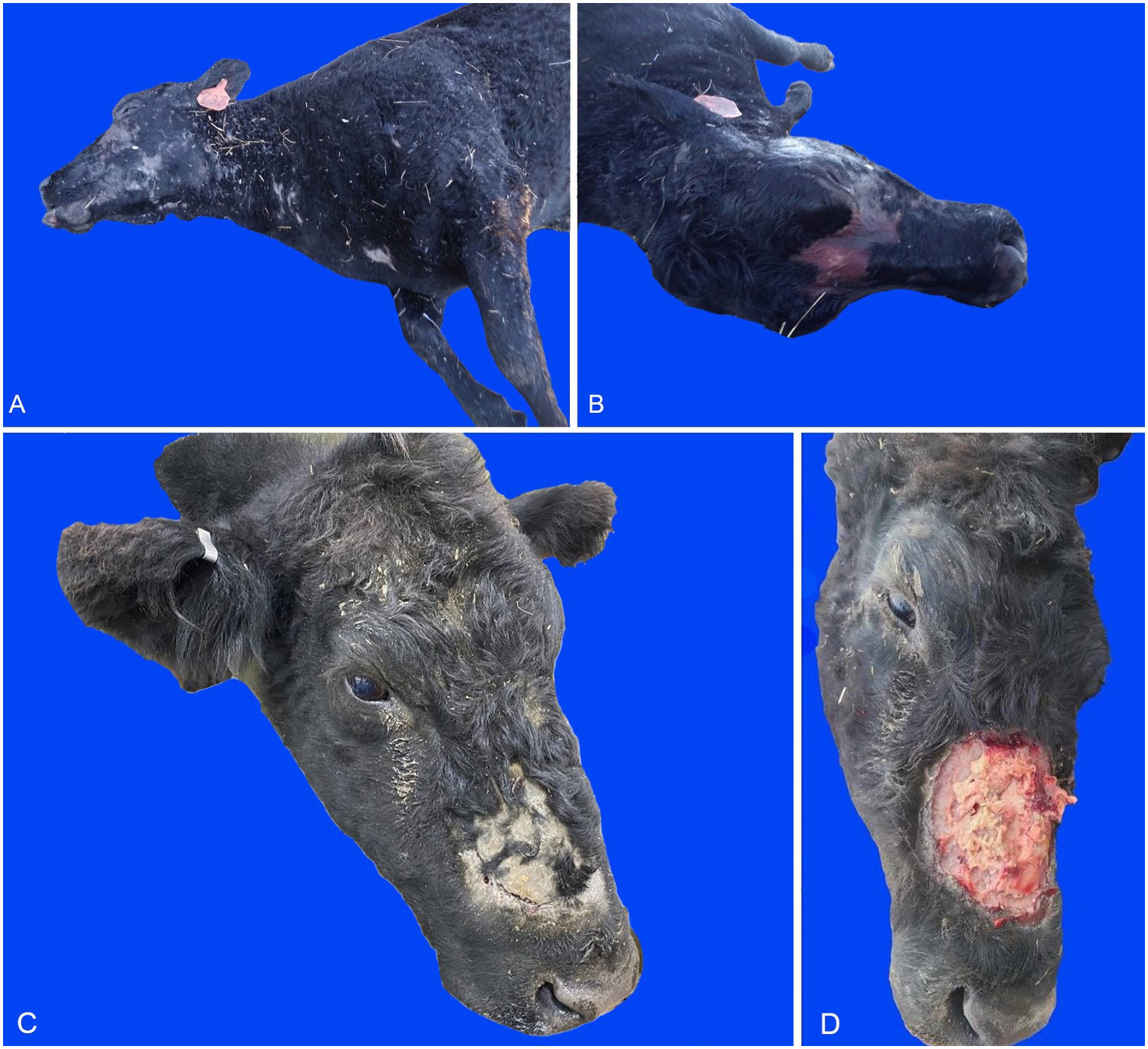
Presumed Brassica-associated liver disease in 2 beef cows.
Approximately 2 wk later, cow 3 was presented with recent severe weight loss, severe respiratory distress, bilateral mucopurulent nasal discharge, and multifocal alopecia of the face and neck. The skin of the alopecia area overlying the face was thick, and bloody purulent exudate was present along the margins. The lesion margins were easily lifted to expose a large amount of underlying purulent material. The blood was watery and took ~48 h after collection to clot. The venipuncture site continued to bleed throughout the physical examination. Cow 3 was treated with ceftiofur and furosemide. CBC and serum chemistry results indicated that the cow was anemic, neutropenic, thrombocytopenic, hyperglobulinemic, and hypocholesterolemic (Table 1). Despite initial improvement in respiration, the cow’s condition worsened, and the owner elected euthanasia and autopsy.
Hematology and serum chemistry results from cow 3 obtained at initial clinical presentation (January 10) and immediately before euthanasia (January 12).
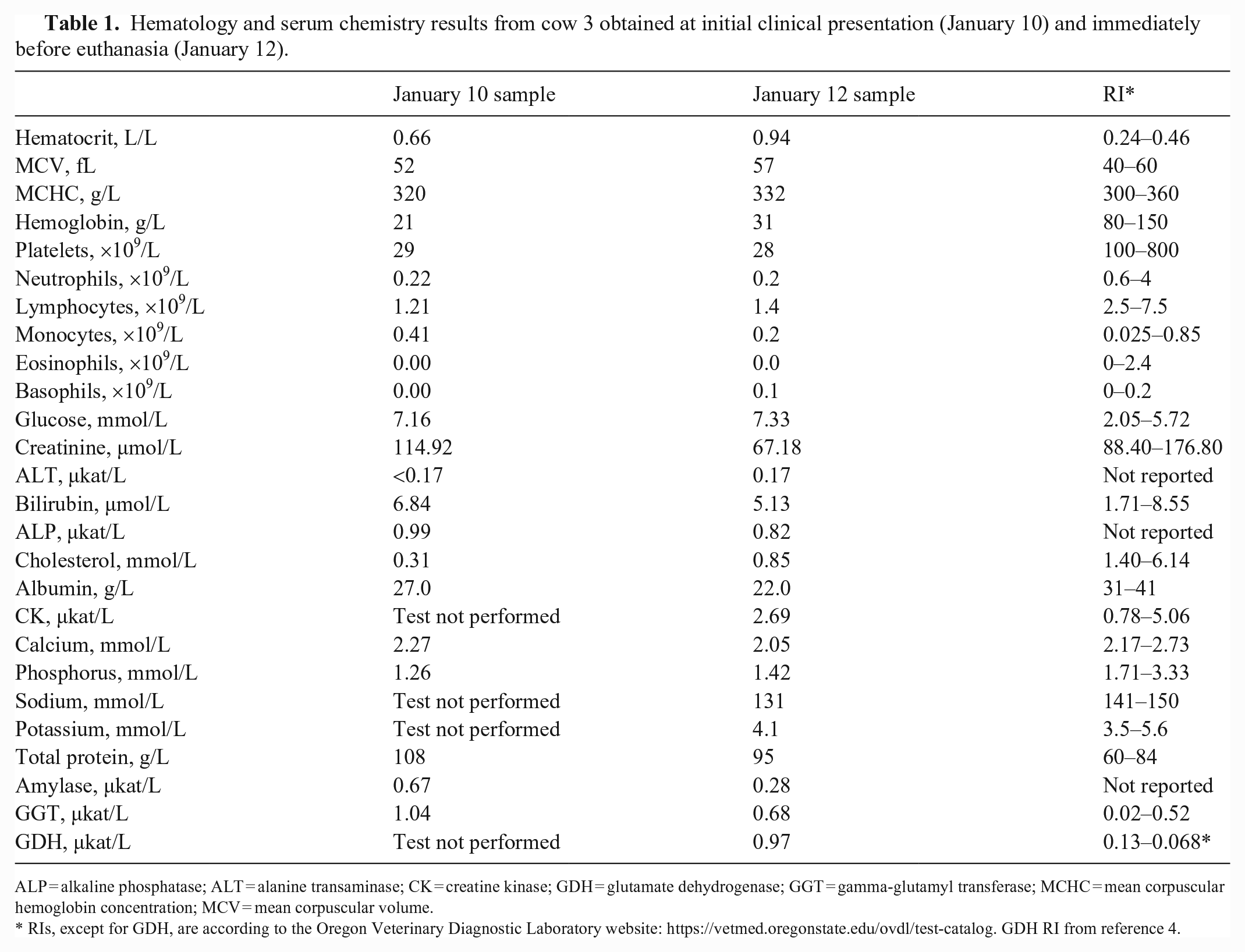
ALP = alkaline phosphatase; ALT = alanine transaminase; CK = creatine kinase; GDH = glutamate dehydrogenase; GGT = gamma-glutamyl transferase; MCHC = mean corpuscular hemoglobin concentration; MCV = mean corpuscular volume.
RIs, except for GDH, are according to the Oregon Veterinary Diagnostic Laboratory website: https://vetmed.oregonstate.edu/ovdl/test-catalog. GDH RI from reference 4.
Cow 3 was estimated to be 5–7-y-old with a body condition score of 3 of 9. Skin over the nasal bridge was thickened, with a single large and smaller coalescing areas of alopecia (Fig. 1). The affected area was 10 × 15 cm and exuded blood-tinged purulent material from the margins. The lesion was easily elevated and removed by hand, exposing the underlying periosteum of nasal and maxillary bones covered in copious purulent material. Removal of the material exposed an ~2-cm linear fracture of the nasal bone near the midline. Blood was pale, watery, and failed to clot. Subcutaneous, omental, perirenal, and pericardial fat was moderately icteric. Serosanguineous fluid (~150 mL) was present within the pericardial sac. Petechial and ecchymotic hemorrhages covered the epicardial surface but did not extend into the muscle. Adhesions on the surfaces of both lungs were interpreted as a previous pleuritis unrelated to the current clinical signs. The liver was diffusely pale. The cranial nasal turbinates were hemorrhagic. No gross lesions were observed in the remainder of the autopsy, which included examination of the brain, gastrointestinal tract, kidneys, urinary bladder, uterus, joints, and skeletal muscle. Blood sampled immediately before euthanasia was marked by severe anemia, hypocholesterolemia, hypoalbuminemia, lymphopenia, and neutropenia, as well as elevated activities of GDH and GGT (Table 1). These results were interpreted as consistent with hepatic disease. PCR testing on fresh spleen for anaplasmosis was negative.
Samples of formalin-fixed liver, lung, skin, spleen, lymph node, cerebrum, and cerebellum were examined histologically. Abundant plasma cells and fewer lymphocytes were observed consistently within hepatic portal areas (Fig. 2). Although a few lymphocytes and plasma cells are commonly observed in portal areas in adult cattle, a panel of 5 veterinary pathologists (4 ACVP-certified) all agreed that the number of periportal inflammatory cells was increased in cow 3. When present, bile duct hyperplasia was mild. Centrilobular and midzonal cells exhibited vacuolar hepatopathy with moderate numbers of variably sized, clear, distinct cytoplasmic vacuoles, consistent with micro- and macrovesicular hepatic lipidosis. Portal fibrosis was not observed. Some sections contained telangiectatic foci, a routine incidental finding in aged cattle.
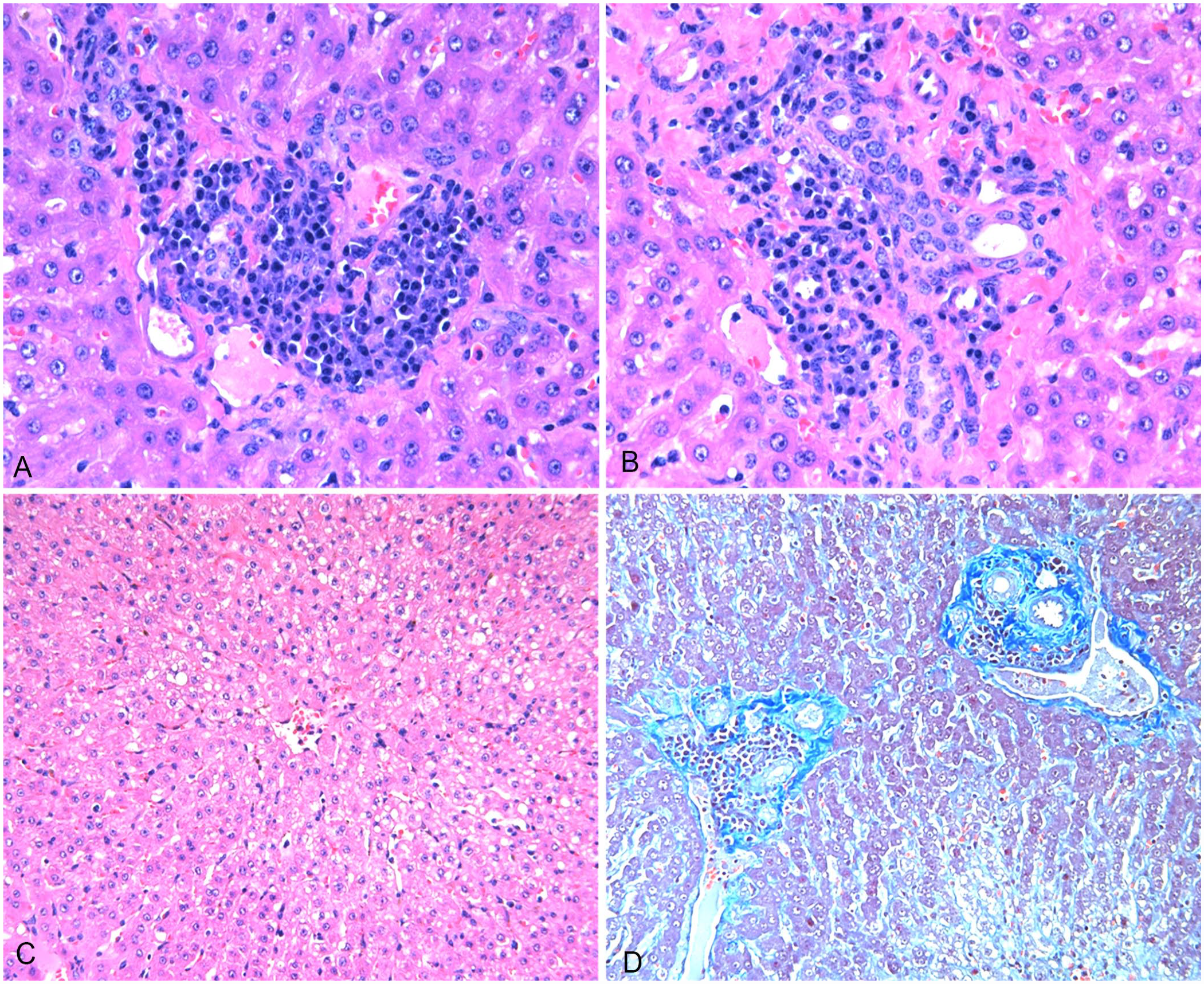
Hepatic lesions in cow 3 with presumed Brassica-associated liver disease.
Skin from the ulcerative lesions in cows 2 and 3 had severe diffuse necrosis throughout all epidermal layers, including adnexal structures, all levels of the dermis, and extending through the subcutis to the section margins (Fig. 3). Collagen bundles were largely degenerate amorphous aggregates of brightly eosinophilic material without fibrillar structure mixed with pyknotic nuclei of fibroblasts and leukocytes. Aggregates of coccobacilli were often present overlying the areas of ulceration. Intact skin adjacent to the ulcerative lesion was moderately acanthotic. There was mild-to-moderate, multifocal superficial dermal edema. The deeper portions of the dermis in the sections of adjacent intact skin had edema, marked lymphoplasmacytic perivasculitis, and variably sized areas of hemorrhage mixed with fibrin. Blood vessels within the deeper dermis were degenerate, with endothelial cells swollen and vacuolated, and the architectural integrity of the vessel walls disrupted by infiltrating leukocytes.
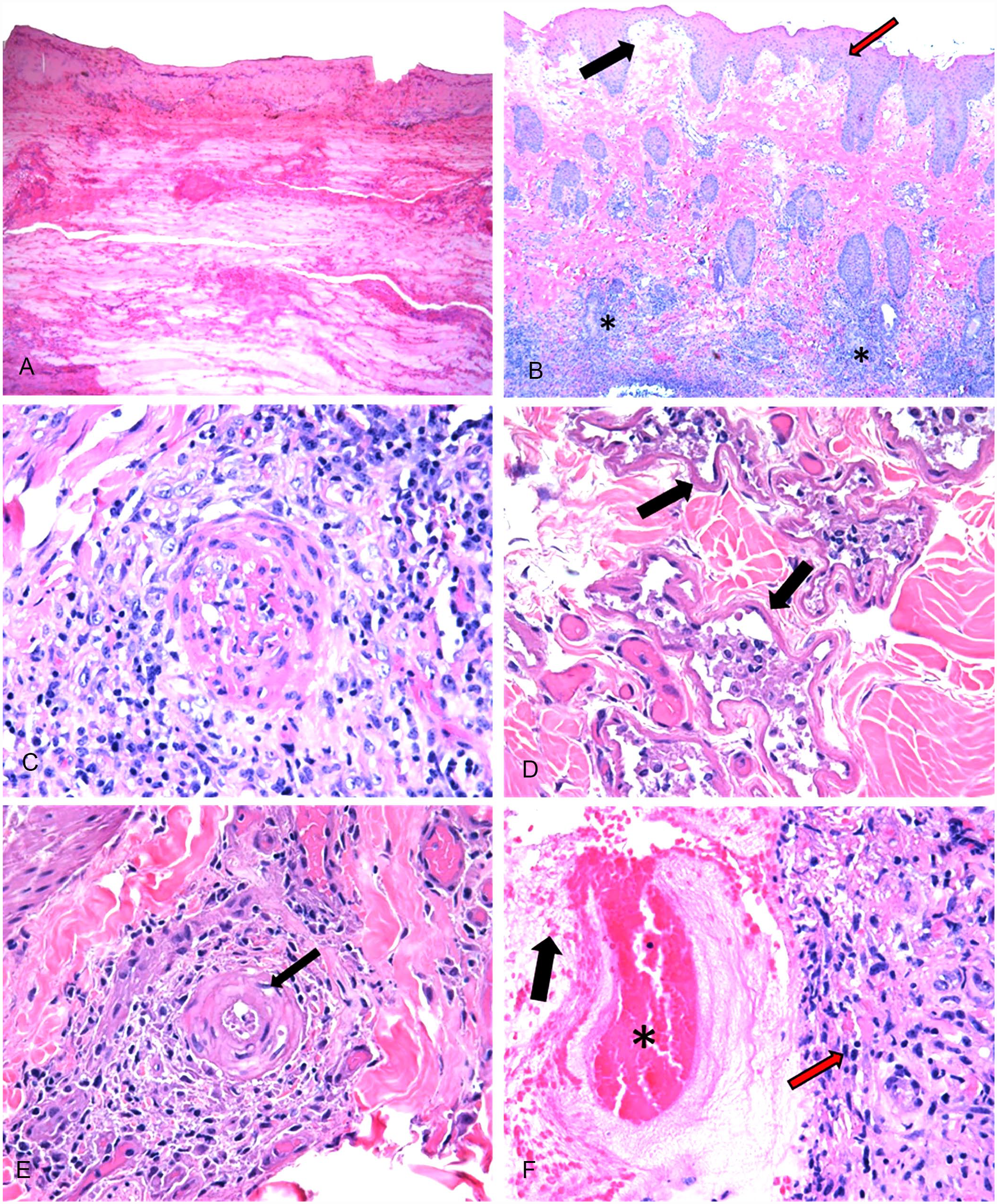
Skin lesions in 2 cows with presumed Brassica-associated liver disease.
Four days after the autopsy of cow 3, cow 4 was presented with alopecia of the head and neck, dramatic recent weight loss, mucopurulent nasal discharge, and photophobia. Cow 4 was exceptionally aggressive, particularly when placed in direct midday sunlight. During the day, the cow continually rubbed her face and neck on the sides of the pen and did not eat or drink until after sunset when she became noticeably calmer. The clinical signs in cow 4 were consistent with the previous 3 cases, but no testing was performed to evaluate skin or liver enzymes, and cow 4 was discharged after 3 d.
The hepatotoxic component in Brassica spp. is unknown. Therefore, cases of BALD diagnosis are, at best, presumptive. Criteria recommended to make a presumptive diagnosis of BALD include: 1) a sudden transition to Brassica forage without allowing gradual adaptation of rumen microflora; 2) hungry or dominant, and often pregnant, cows; 3) clinical signs of photosensitization and/or jaundice; 4) elevated liver enzyme activities (particularly GGT); 5) pale liver on autopsy; 6) typical histologic hepatic lesions, which may be subtle or absent; and 7) no new cases within several days of the cattle being taken off the turnip field. 6 We favor the presumptive diagnosis of BALD in our case given the onset of clinical signs correlated with turnip exposure, the location and severity of skin lesions, jaundice, and photophobia, as well as the histologic lesions and serum chemistry results indicating liver disease. In our case, the cow herd had been exposed to bulls the previous summer. The cows were not verified pregnant after the breeding season, but ~85% were assumed to be pregnant at the time of lesion development. Of the animals autopsied, cow 2 carried a fetus consistent with ~150 d gestation, and cow 3 was open.
All of these cows had been exposed to a bull during the breeding season, and therefore the majority were presumed to be pregnant. The owner agreed to move the cattle after the autopsy results of cow 3 led to the suspicion of BALD. While moving the cattle, cow 5 was observed to have alopecic lesions on the face and neck as well as avoiding sunlight. Once the animals were moved to a new pasture, cow 5 recovered, and the owner reported no additional cases. The recovery of cow 5 and the lack of any new cases after the cattle were moved further support the presumptive diagnosis of BALD. Facial eczema was not pursued as a potential differential given the strict temperature range (13–20°C) and high levels of humidity (≥ 90%) required for sporidesmin production by the fungus Pithomyces chartarum. 1 Our case took place in January in Nebraska, with an average temperature of –1.8°C during that month.
Interestingly, in contrast with previous reports of suspected BALD, our case did not affect only white-faced cattle. The vast majority of this herd were all black, with very few (~5 of 300) white-faced. Of the 5 most severely affected cows, the only white-faced cow was cow 5, which was noticed by the owner when moving the cattle to a new field, and cow 5 recovered quickly thereafter.
Some of the skin and vascular lesions in our case were exceptionally severe, particularly in cow 3, which had deep necrotizing skin lesions and a bone fracture resulting from the severe pruritus. The degenerative vascular changes observed in the dermis of both cows are similar to those described previously, 5 and suggest that the toxic principle is directly toxic to endothelial cells leading to thrombosis and ischemia. Despite being elevated, the GGT activities observed were lower than the extreme elevations reported in other cases of suspected BALD.4,5 The liver from cow 3 did not have fibrosis and/or attenuated bile duct epithelium described previously, although these characteristics have been known to be subtle or absent in some cases of BALD.4,6
It is important for producers, practitioners, and diagnosticians to be aware of the association of Brassica spp. cover crops with hepatogenous photosensitization in cattle. This is particularly true given the increasing popularity of the use of cover crops in North America to reduce erosion, increase organic matter in the soil, and inhibit weeds, as well as provide forage for grazing livestock. 10 It is possible that other unknown factors promoted hepatogenous photosensitization related to the cover crop in our case. Regardless, veterinarians, diagnosticians, and producers presented with cattle with lesions of the face and neck, photophobia, and intense pruritus after being introduced to a Brassica spp.–containing cover crop, should consider BALD as a differential diagnosis and seek a new source of forage. To date, liver biopsy samples in cattle with clinical signs of acute BALD have been unrewarding given that no consistent microscopic lesions have been identified in such samples. 4 Therefore, elevated GGT and/or GDH activities on serum chemistry, vascular changes on skin biopsy, and appropriate clinical signs after exposure to Brassica spp. of plants appear to be the most useful tools to make the presumptive diagnosis.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was supported by operational funds of the Nebraska Veterinary Diagnostic Center and the Nebraska Institute of Agriculture and Natural Resources.
