Abstract
Focal symmetrical encephalomalacia (FSE) is the most prominent lesion seen in the chronic form of enterotoxemia caused by Clostridium perfringens type D in sheep. However, this lesion has not been reported in goats. The current paper reports a case of FSE in a goat from the state of Paraíba in the Brazilian semiarid region. As reported by the farmer, 30, 4–48-month-old animals from a flock of 150 goats died after showing nervous signs, including blindness and recumbence, for periods varying between 1 and 14 days. The flock was grazing native pasture supplemented with wheat and corn bran. Additionally, lactating goats were supplemented with soybeans. A 4-month-old goat with nervous signs was examined clinically and then necropsied 3 days after the onset of clinical signs. Bilateral, focal, and symmetrical areas of brown discoloration were observed in the internal capsule and thalamus. Histologic lesions in these areas consisted of multifocal, bilateral malacia with a few neutrophils; endothelial cell swelling; perivascular edema; and hemorrhages. The etiology of these lesions was not determined. However, FSE is considered pathognomonic for C. perfringens type D enterotoxemia in sheep, and it is speculated that this microorganism was the etiologic agent in the present case. The flock had been vaccinated against type D enterotoxemia only once, approximately 3 months before the beginning of the outbreak. Insufficient immunity due to the incorrect vaccination protocol, low efficacy of the vaccine used, and a diet including large amounts of highly fermentable carbohydrates were suspected to be predisposing factors for this outbreak.
Enterotoxemia is a frequent disease of sheep and goats caused by Clostridium perfringens type D, 9,10,19 which is a bacterium found as a normal inhabitant of the intestine in a small number of healthy animals. 9,10 Alterations in the intestinal environment caused by sudden changes in diet, mainly consisting of the ingestion of highly fermentable carbohydrates, are associated with proliferation of C. perfringens type D and production of large quantities of epsilon prototoxin, which is then activated by intestinal trypsin and other proteases. Epsilon toxin is responsible for the clinical and pathologic findings of enterotoxemia. 9,11,17,19
In sheep, enterotoxemia is most frequently acute or subacute, and the main target organs of epsilon toxin are the lungs and brain, where it causes increased vascular permeability leading to edema. Perivascular proteinaceous edema of the brain is a characteristic and diagnostic lesion of the acute and subacute forms of enterotoxemia in sheep. 19 The chronic form of enterotoxemia in sheep is rarely observed, and it is mainly characterized by focal symmetric encephalomalacia (FSE), a lesion consisting of bilateral and roughly symmetrical foci of malacia affecting mainly the corpus striatum, thalamus, and cerebellar peduncles. 1,2,6,7,12,19,20
In goats, type D enterotoxemia can be acute, subacute, or chronic. The acute and subacute forms of the disease can be similar to the corresponding forms in sheep, although perivascular edema of the brain is not as frequently observed, and enterocolitis or colitis can be also observed in the subacute form. Chronic enterotoxemia in goats is characterized by enterocolitis or colitis without brain or other systemic clinical signs or lesions. Focal symmetric encephalomalacia has not been described in any form of caprine enterotoxemia, nor has it been described associated with any other disease in this animal species. The current study describes an outbreak of chronic neurologic disease in goats and reports lesions of FSE in a goat originating from that outbreak.
An outbreak of neurologic disease occurred on a farm in the Municipality of Boa Vista, state of Paraíba, the semiarid region in northeastern Brazil, in April–June (considered to be the rainy season) 2009. Thirty-four of 150 goats died in a period of 3 months. The age of the animals in the flock varied from 1 day to 7 years. Affected animals were 4–48-month-old Nubian, Saanen, and American Alpina cross-breeds. Most goats had been vaccinated against type D enterotoxemia 3 months before the beginning of the outbreak. Booster vaccinations were given later in April (after the first deaths occurred), May, and June (after the last death occurred). The flock was grazing a native pasture and supplemented ad libitum with a concentrate of wheat and corn bran. Lactating goats were also supplemented ad libitum with soybeans. The farmer reported that the goats that died on the farm showed recumbence and other neurologic signs, including blindness, for up to 2 weeks before death. In the previous year, 25 goats of different ages died with abdominal pain and diarrhea less than 24 hr after clinical signs appeared.
A 4-month-old male Saanen goat that had been recumbent for 24 hr was submitted to the Veterinary Hospital at the Federal University of Campina Grande (Patos, Paraíba, Brazil). On clinical examination, the goat had rigidity of all 4 legs, opisthotonos, absence of menace response, and blindness, followed by marked depression and decreased swallowing reflex. The animal was treated with 10 mg/kg of intramuscular thiamine and 0.2 mg/kg of intravenous dexamethasone. This treatment was repeated 12 hr later, but the goat did not show clinical improvement and died 3 days after the onset of disease.
A necropsy was performed soon after death. The carcass was in good nutritional condition. The serosa and mucosa of the colon were slightly red, and an excess of clear pericardial fluid was observed in the pericardial sac. Multifocal, bilateral, and roughly symmetrical brownish areas approximately 0.1–1.0 cm in diameter were observed in the internal capsule and thalamus. No other significant gross abnormalities were observed in the rest of the carcass. Samples of small intestine, colon, fore stomachs, abomasum, mesenteric lymph nodes, adrenal gland, spleen, liver, heart, lung, trigeminal ganglion, and pituitary gland were taken and fixed in 10% buffered formalin. The whole brain and spinal cord were also fixed in 10% buffered formalin, and subsamples were obtained from the cervical, thoracic, and lumbar spinal cord; medulla oblongata; pons; rostral and caudal colliculi; thalamus; corpus striatum; cortex; cerebellar peduncles; and cerebellum.
All tissues were processed routinely, embedded in paraffin, sectioned at 4–5 μm, stained with hematoxylin and eosin, and coverslipped. Histologic lesions in the brain were observed in areas where gross abnormalities were present. These histologic lesions consisted of roughly symmetrical, bilateral, multifocal areas of malacia characterized by pallor and rarefaction of the neuropil (Fig. 1A–C) with necrosis of glial cells and neurons (Fig. 1D). Axonal swellings (spheroids; Fig. 1D) were also observed. Occasionally, perivascular proteinaceous edema and/or hemorrhages were observed, and the vascular endothelial cells were hypertrophic (Fig. 1C). A few neutrophils were also observed within the necrotic lesions. Congestion of blood vessels in the intestinal mucosa and lung was also observed. No other significant histologic abnormalities were observed in any of the tissues examined.
The present study describes a case of FSE in a goat. However, FSE is considered a misnomer, because the lesions are generally multifocal, 19 which was the case in this goat. A more appropriate name would be multifocal symmetrical encephalomalacia. In sheep, FSE is pathognomonic for the chronic form of C. perfringens type D enterotoxemia, and this lesion is considered diagnostic for the disease. In goats, however, FSE has not been previously described in cases of enterotoxemia or any other disease. 15,18 The etiology of FSE in the goat in the current study was not determined, although it is speculated that it was produced by epsilon toxin of C. perfringens type D, because FSE is considered a pathognomonic lesion of chronic C. perfringens type D enterotoxemia in sheep. A similar situation occurs in cattle, a species in which natural FSE has been described in animals with neurologic signs but for which a definitive causal association between C. perfringens type D and FSE has not been demonstrated (Buxton D, Macleod NSM, Nicolson TB: 1981, Focal symmetrical encephalomalacia in young cattle. Vet Rec 108:459. Abstract). 4 However, natural FSE in cattle has long been suspected to be produced by C. perfringens type D. Recently, FSE was experimentally produced by infusion of C. perfringens type D in the duodenum of a calf, 5 which lends support to the hypothesis that the natural cases of FSE described previously in cattle were indeed produced by C. perfringens type D.
Detection of epsilon toxin in intestinal content is considered diagnostic for type D enterotoxemia in the acute and subacute, but not in the chronic, form of the disease in sheep. However, FSE is a manifestation of the chronic form of type D enterotoxemia in this species, and epsilon toxin is usually not found in intestinal contents of animals with this form of the disease. 11 A diagnosis of chronic enterotoxemia in sheep is made by the observation of gross and histologic manifestations of FSE, located mainly in the internal capsule, thalamus, mesencephalon, and/or cerebellar peduncles. The diagnosis is further confirmed by excluding other diseases of the nervous system, including polioencephalomalacia, listeriosis, rabies, and pregnancy toxemia, all of which were excluded in the current case based on the histologic findings. Furthermore, isolation of C. perfringens type D from the intestine is of little, if any, diagnostic value for cases of enterotoxemia in any species, because this microorganism can be a normal inhabitant of the intestinal tract in healthy animals. Therefore, it is unlikely that attempts to detect epsilon toxin detection or culture C. perfringens would have been of much diagnostic help in the present case, even if the FSE lesions were produced by C. perfringens type D. Other causes of symmetric focal necrosis in the brain in other animal species include intoxication by Centaurea spp. in horses (nigropallidal encephalomalacia) 15 and by Aeschynomene indica in pigs (symmetrical necrosis of cerebellar and vestibular nuclei). 13 These intoxications have not been previously reported in goats, and those plants were not present in the region where the disease reported herein occurred.
In the outbreak reported in the current study, the farmer reported the death of 30 other goats with neurologic signs similar to those observed in the single goat examined in the present case, suggesting that those animals were also suffering from FSE. The reason some sheep survive long enough to develop the chronic form of enterotoxemia and FSE is not fully understood. It has been speculated that FSE occurs in animals in which there are constant low concentrations of epsilon toxin in the gut or in animals that have been only partially immunized and that have, therefore, low epsilon toxin antibody levels in the blood. 15 The latter situation can occur in animals that are improperly vaccinated (e.g., only 1 dose of vaccine). In the outbreak in the present study, the flock had been vaccinated against type D enterotoxemia with only 1 dose of a commercial mixed clostridial vaccine before the occurrence of the disease. The goats were vaccinated again 1 month after the onset of the outbreak and after the end of the 3-month outbreak. The deaths stopped only after the third vaccination was given to the flock. It is possible that the first vaccine induced low antitoxin serum concentrations, which protected against acute and subacute enterotoxemia but not against FSE. In Brazil, an evaluation of type D enterotoxemia vaccines showed that only 2 of 6 commercial vaccines evaluated induced protecting levels of antibodies to epsilon toxin. 9 Also, goats vaccinated with commercial vaccines have lower and less persistent titers of epsilon antitoxin than sheep 16 and should be revaccinated every 4 months, a recommendation that was not followed in this flock. 3
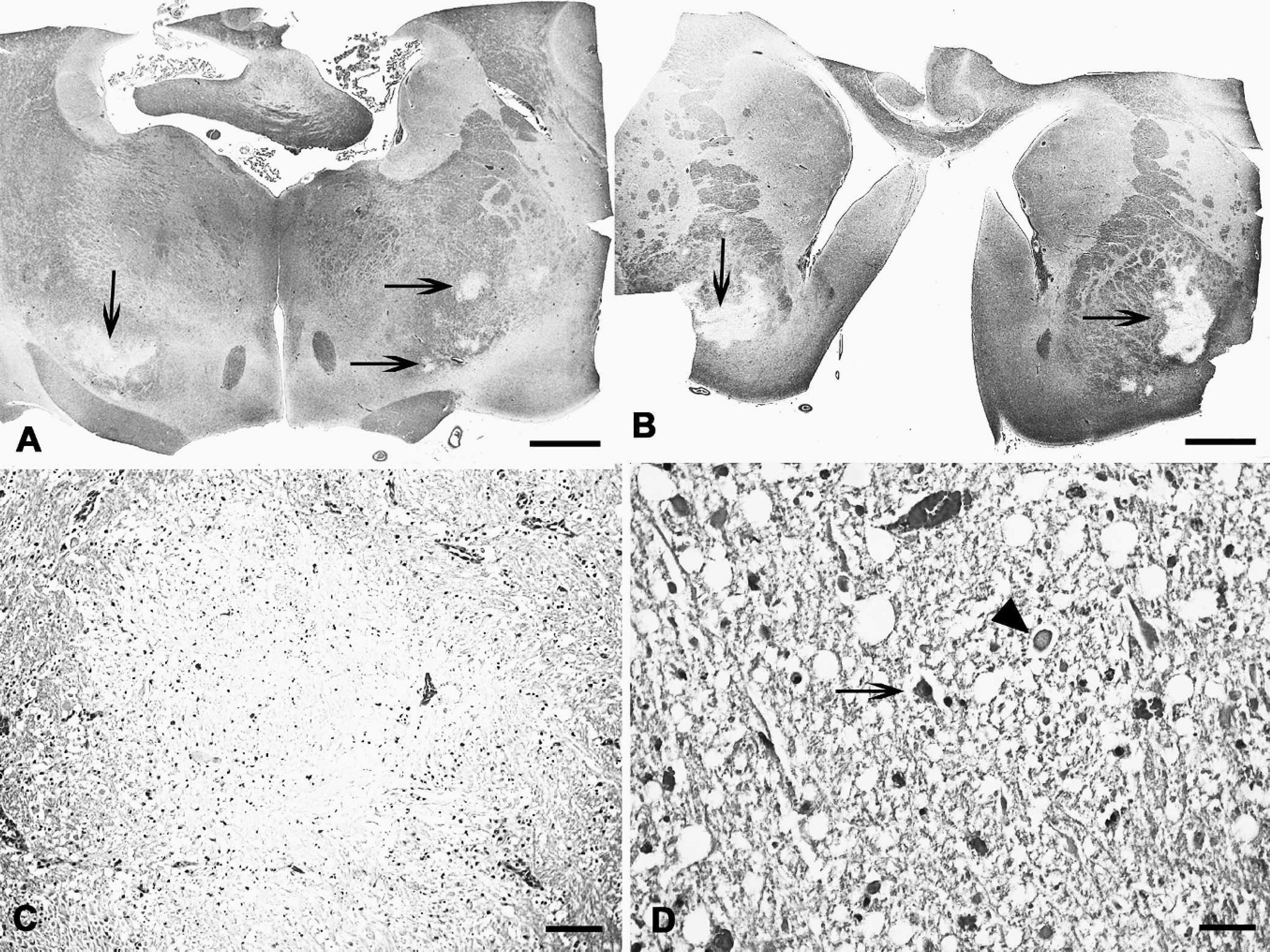
Goat; multifocal symmetric encephalomalacia. Submacroscopic photograph showing areas of malacia in the thalamus (
Another factor associated with the outbreak was the high nutritional levels due to the good pastures during the rainy period and supplementation with grains or their byproducts containing highly fermentable carbohydrates. The ingestion of highly fermentable carbohydrates is a well-known risk factor for the occurrence of enterotoxemia and FSE (Hazlett M, Reid S: 2002, Focal symmetrical encephalomalacia in two lambs. AHL Newsletter 6:30. Available at http://www.uoguelph.ca/labserv/units/ahl/files/ANwsl6-3.pdf. Accessed on April 2, 2010). 6,11,14,20 Future research should be performed to determine the etiology and prevalence of FSE in goats in the Brazilian semiarid region and to understand the pathogenesis of this condition.
Acknowledgements
The authors acknowledge the Co-ordenação de Aperfeiçoamento de Pessoal de Nivel Superior (CAPES) scholarship given to the first author.
