Abstract
Equine enterotyphlocolitis is an inflammatory process of the intestinal tract of horses that is associated with multiple etiologic agents and risk factors. Most clinical cases do not have an etiologic diagnosis. We describe here the pathogens detected and the histologic lesions found in horses with enterotyphlocolitis in Ontario that were submitted for postmortem examination, 2007–2019. We reviewed the medical records of 208 horses that fulfilled inclusion criteria. Cultures were positive in 67 of 208 (32%) equids for Clostridium perfringens, in 16 of 208 (8%) for Clostridioides difficile, and in 14 of 208 (7%) for Salmonella spp.; 6 of 208 (3%) were positive for Neorickettsia risticii by PCR assay. One horse was positive in a Rhodococcus equi PCR assay. All horses tested by PCR assay for equine coronavirus and Lawsonia intracellularis were negative. The histologic lesions were characterized as follows: 6 of 208 (3%) enteritis, 5 of 208 (2%) typhlitis, 104 of 208 (50%) colitis, 37 of 208 (18%) enterocolitis, 45 of 208 (22%) typhlocolitis, and 11 of 208 (5%) enterotyphlocolitis. We strongly recommend standardized testing of diarrheic horses during and/or after postmortem examination, as well as standardized reporting of histologic lesions in enterotyphlocolitis cases.
Acute colitis and enterotyphlocolitis are significant inflammatory diseases of the equine intestinal tract,13,15,19 are the most important cause of diarrhea in horses, and have been associated with significant morbidity and mortality.8,14 Various organisms are capable of causing these diseases, 15 but an etiologic agent is not identified in most cases. Clostridioides difficile, Salmonella spp., and Neorickettsia risticii are the pathogens detected most commonly in adult horses with colitis in North America.9,13,19 Equine coronavirus, large and small strongyles, antimicrobial drug–associated diarrhea, and non-steroidal anti-inflammatory drug toxicity are other conditions associated with colitis in adult horses. 13
Most horses with diarrhea (~60%) are classified as having undifferentiated colitis,8,9 which is defined as a horse with diarrhea and signs of colonic inflammation that tests negative for all of the common pathogens.9,15 Inconsistencies in diagnostic protocols can account for the classification of some cases as undifferentiated colitis. Disturbance of the complex gastrointestinal microbiota of the equine hindgut could also play a role. Our limited understanding of the equine microbial communities in health and disease, associated with the lack of a standardized approach to diagnosis of acute enterocolitis in horses, can contribute to the low number of cases with definitive etiologic diagnoses.
The use of PCR panels in the etiologic diagnosis of acute gastroenteric diseases has increased in human1,16,18 and veterinary medicine.4,5,7,11 A systematic review 16 compiled the findings of 37 articles that evaluated the efficacy and clinical applicability of PCR panels to diagnose and guide the treatment of humans with gastrointestinal infection. Despite some advantages, such as faster turnaround time and an increase in the number of enteropathogens detected, one of the potential disadvantages is the questionable causative association between etiologic agent detection and disease process. 16
Here, we describe retrospectively the pathogens detected during postmortem examination of equids with enterotyphlocolitis in Ontario, and describe the histologic lesions recorded by the case pathologists.
Materials and methods
The electronic postmortem records database was searched for horses > 1-y-old that were presented to the Ontario Veterinary College–Pathobiology Department and the Animal Health Laboratory (AHL), of the University of Guelph in Guelph and Kemptville, Ontario, Canada, between January 2007 and January 2019. The following keywords were entered: “colitis”, “enteritis”, “typhlitis”, “enterocolitis”, and “enterotyphlocolitis”. Horses were included if a full postmortem examination was performed. Cases were excluded if colonic inflammation was secondary to intestinal displacement or gastrointestinal rupture. All of the records that matched were reviewed.
Data recorded included: signalment, area of the intestine affected, histologic diagnosis, and ancillary tests submitted for the etiologic investigation of the enterotyphlocolitis. Ancillary laboratory tests on intestinal content were performed according to AHL protocols and at the discretion of the board-certified pathologist performing the postmortem examination, and included: anaerobic and aerobic bacterial culture of one sample collected during postmortem examination (preceded by enrichment, in aerobic and anaerobic conditions, and in selected enriched media), C. difficile toxins A and B ELISA (C. difficile TOX A/B II; TechLab), C. perfringens enterotoxin ELISA (C. perfringens enterotoxin test; TechLab), N. risticii PCR, Lawsonia intracellularis PCR, Rhodococcus equi PCR, and equine coronavirus PCR. A sample was considered positive if bacterial culture and/or PCR was positive for Salmonella spp., if bacterial culture was positive for C. perfringens and C. difficile, and if PCR was positive for N. risticii, L. intracellularis, R. equi, or coronavirus. Because parasite infestation is uncommon in adult horses in our geographic area, fecal flotation is not performed routinely on the intestinal content of horses submitted for postmortem examination, unless there is gross evidence of adult parasites or encysted cyathostomin larvae. Therefore, it was not included in the tests in our retrospective analysis. Results are presented descriptively, and numerical values are reported as medians and ranges.
Results
Animals
In our postmortem examination records database search between May 2007 and December 2019, we identified 297 equids with a final postmortem diagnosis of “colitis”, enterotyphlocolitis”, “enterocolitis”, or “typhlocolitis”; 208 of 297 cases fulfilled our inclusion criteria.
Signalment
The sex of 197 of 208 equids was recorded; 87 of 197 (44%) were female, and 110 of 197 (56%) were male (gelding or intact male). Age was available for all 208 cases, with a median of 8 y (min. 1-y-old; max. 49-y-old). Of the 187 animals for which breed was available, the most prevalent were Standardbred (n = 46), Thoroughbred (46), American Quarter Horse–related breeds (25), Warmblood (19), and mixed breed (14). The remaining horses belonged to the following breeds: pony (n = 8), Miniature Horse (5), Trakehner (4), Clydesdale (3), Rocky Mountain Horse (2), Arabian (3), Tennessee Walking Horse (1), Haflinger (1), Belgian (1), Andalusian (1), Shire (1), Percheron (1), Black Forest Horse (1), Canadian (1), and donkeys (2).
Distribution of cases
The distribution of the cases throughout the years of the study was as follows: 2007 (n = 13), 2008 (16), 2009 (9), 2010 (16), 2011 (22), 2012 (19), 2013 (13), 2014 (17), 2015 (14), 2016 (22), 2017 (16), 2018 (0), 2019 (11). Cases were presented more often in the summer and early fall (47%; Fig. 1).
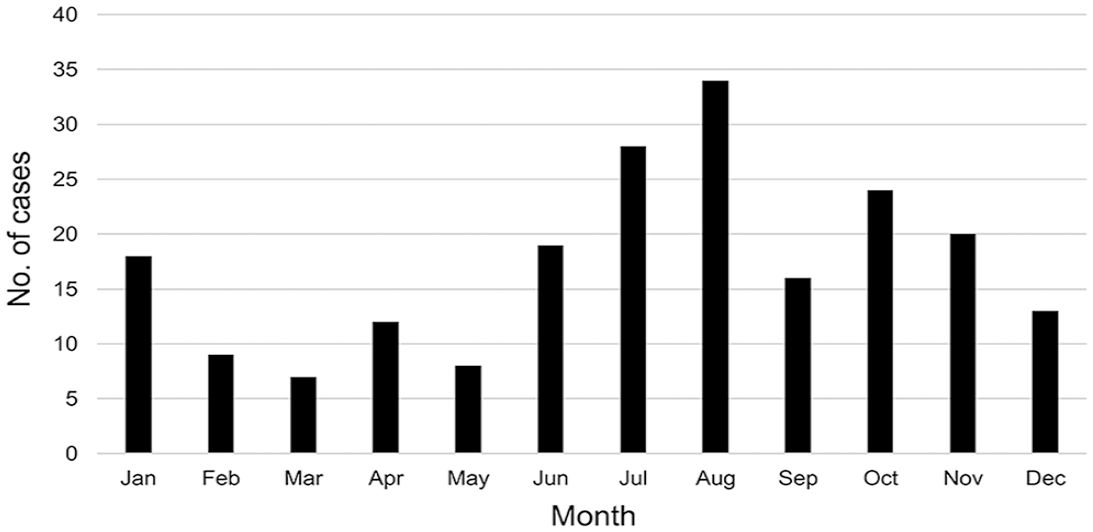
Distribution of enterotyphlocolitis cases by month of presentation for horses > 1-y-old submitted for postmortem examination.
Histologic diagnosis
The histologic lesion was characterized as follows: 6 of 208 (3%) enteritis, 5 of 208 (2%) typhlitis, 104 of 208 (50%) colitis, 37 of 208 (18%) enterocolitis, 45 of 208 (22%) typhlocolitis, and 11 of 208 (5%) enterotyphlocolitis. In 140 of the 208 cases included, a modifier descriptive term was available regarding the type of inflammatory lesion present, with 39 of 140 (28%) described as hemorrhagic, 70 of 140 (50 %) as necrotic, 46 of 140 (32%) as ulcerative, and 21 of 140 (15%) as erosive.
The predominant infiltrative inflammatory cell was described in 66 of 208 (32%) cases. Of those, 34 of 66 (51%) were suppurative or neutrophilic, followed by 15 of 66 (23%) lymphoplasmacytic, 10 of 66 (15%) eosinophilic, and 7 of 66 (11%) histiocytic. Only 48 of 208 (23%) histologic descriptions had a description of the extent of the lesions. A diffuse or generalized lesion was described in 40 of 48 (83%), as multifocal in 6 of 48 (13%), and focal in 2 of 48 (4%). Additionally, of the 80 cases in which severity of the lesion was stated, 6 of 80 (8%) were described as mild, 8 of 80 (10%) as moderate, and 66 of 80 (82%) as severe or marked.
Etiology
In general, a median of 3 (min. = 0; max. = 6) microbiologic tests were performed per postmortem examination as part of the etiologic diagnostic investigation; 165 of 208 (79%) cases were submitted for Salmonella culture, of which 14 of 165 (8%) were positive. The serovars identified were Salmonella enterica subsp. enterica serovar Heidelberg (n = 4), S. Thompson (3), S. Braenderup (1), S. Muenster (1), S. Infantis (1), S. Mbandaka (1), S. Agona (1), and S. Newport (1).
A total of 156 of 208 (75%) samples were submitted for Clostridium perfringens culture, and 61 of 208 (29%) were also tested for C. perfringens enterotoxin ELISA. C. perfringens was isolated in 67 of 156 (43%) of the cultures; 3 of 61 (5%) were positive for enterotoxin by ELISA. Further C. perfringens testing was not carried out and/or was not recorded in the medical records. Samples were submitted for C. difficile culture in 51 of 208 (25%) cases, and 85 of 208 (41%) were submitted for C. difficile toxin ELISA. C. difficile culture was positive in 16 of 51 (31%) of the cases, and 12 of 85 (14%) were positive for toxins on ELISA. Twenty-four cases were submitted for both C. difficile culture and ELISA; 13 samples were negative on both tests, and 8 samples were positive on both tests. One sample was positive on culture and negative on ELISA, and 2 samples were negative on culture and positive on ELISA.
Neorickettsia risticii—the cause of Potomac horse fever (PHF)—was tested by PCR in 44 of 208 (21%) cases; 6 of 44 (14%) were positive. L. intracellularis was tested in 3 of 208 (1%) cases, R. equi in a single case, and equine coronavirus in 13 of 208 (6%) cases, all via PCR. The case tested for R. equi was positive; all cases tested for L. intracellularis and equine coronavirus were negative. A single case was positive in 2 tests (S. enterica subsp. Heidelberg and C. perfringens enterotoxin).
Discussion
Our study highlights the inconsistency of the etiologic diagnosis obtained postmortem in cases of enterotyphlocolitis in equids, as well as the inconsistency of the histologic reporting of these cases. One could argue that the implementation of more recent and/or improved test methods could have played a role in the etiologic diagnosis and pathogen detection in our study. Different methodologies for pathogen detection (e.g., culture, molecular-based tests) may have changed over the years, and therefore the rate of pathogen detection may be affected by this variable. In a study performed in California, the undetermined cases of fatal intestinal inflammatory lesions from 1990 to 1997 were 138 of 202 (68%), and from 2006 to 2013 were 137 of 233 (59%). 9 Although there was a decrease in the proportion of undetermined cases of fatal colitis, this difference appears minimal for a 20-y gap. The need to continue investigating and reviewing diagnostic strategies for enterotyphlocolitis cases is emphasized by both their study and our study.
For many of the cases included in our study, testing was inconsistent and/or incomplete for the most common pathogens and toxins known to cause enterotyphlocolitis in horses, which could have contributed to the lack of an etiologic diagnosis in some of the undiagnosed cases. Economic constraints could have limited the number of tests performed in some cases. In addition, the methodologies of some of the tests performed have low sensitivity and could therefore give false-negative results. For example, although enrichment was performed prior to bacterial culture, only one sample per animal was submitted for Salmonella spp. detection. Additionally, the use of only culture to detect Salmonella spp., C. perfringens, and C. difficile could be another limitation. 2 Standardized validated laboratory protocols would likely have improved the identification of etiologic agents and toxins in these cases.
Testing guidelines for enteric diseases, such as colitis, are lacking in veterinary medicine. We have created proposed etiologic testing guidelines to improve the diagnosis of equine enterotyphlocolitis (Fig. 2). Adult horses with enterotyphlocolitis should be tested for all of the etiologic agents associated with disease, considering factors such as season and geographic location. Although Paeniclostridium sordellii has been associated with enterocolitis in horses, 10 the role of this bacterium as a cause of acute enterotyphlocolitis should be further investigated. Therefore, we included screening for P. sordellii in our proposed guidelines. Similar to the clinical diagnosis of other enteric clostridial disease (e.g., C. difficile) in humans and animals, C. perfringens toxigenic culture or fecal (or intestinal contents) toxin assays should be the gold standard. There are, however, no commercial tests for the detection of C. perfringens major and/or minor toxins, nor studies validating the performance of such tests in clinical specimens, to our knowledge. The role of C. perfringens as the cause of gastrointestinal disease in horses remains an association at best, and prospective studies investigating virulence factors in clinical cases are warranted. We expect that, if more cases are extensively tested, then the number of cases classified as idiopathic or undetermined colitis could decrease. The etiologic diagnosis of these cases is especially important given that they can be infectious, and biosecurity protocols need to be implemented.
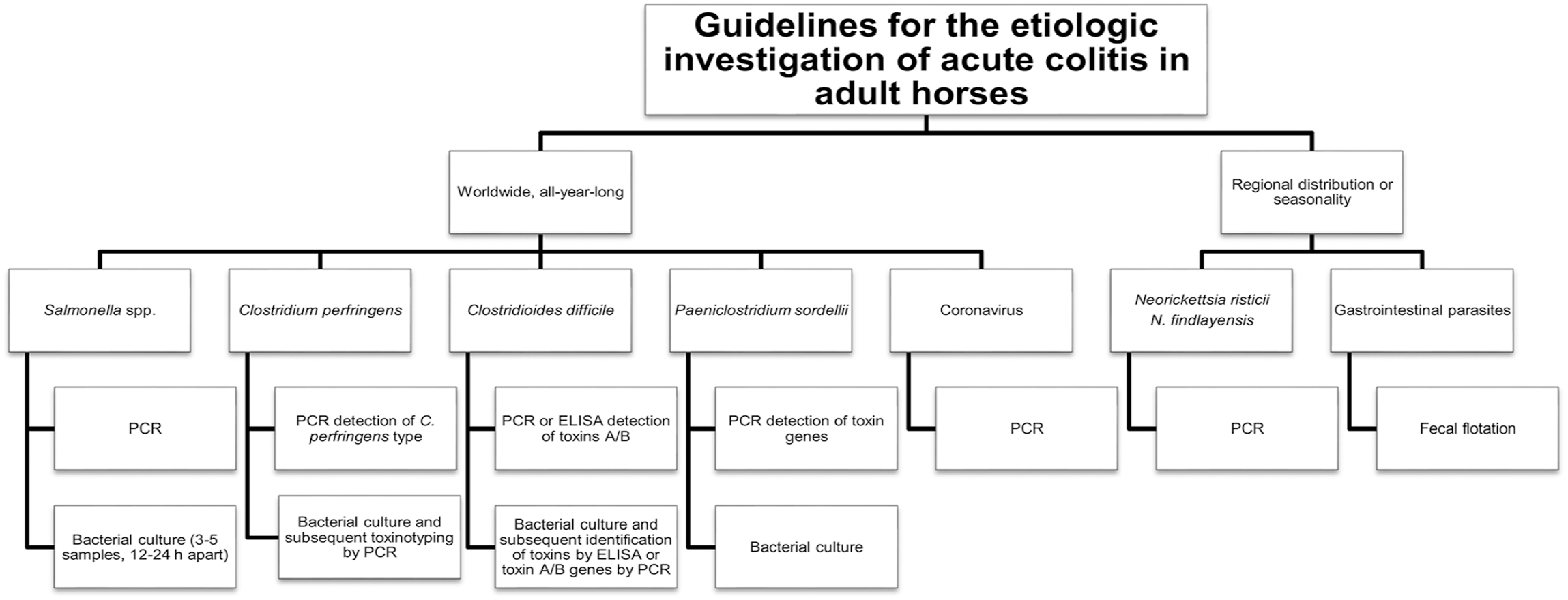
Proposed investigation of the etiology of acute enterotyphlocolitis in adult horses. Testing can be performed on feces and/or intestinal content. Collection of multiple samples from different gastrointestinal tract sites may increase the likelihood of reaching an etiologic diagnosis.
Our retrospective study highlights the lack of standard testing protocols in our locale, which could be a contributing factor to the low number of equine colitis cases with a final etiologic diagnosis. We suggest that any diarrheic adult horse, particularly those hospitalized and that have received antimicrobial drugs, should be tested for common high-risk pathogens such as Salmonella spp., C. perfringens, and C. difficile, as well as pathogens indicated according to the geographic area, age of the animal, and season of the year. Moreover, merely identifying C. perfringens or C. difficile by bacterial culture does not indicate disease causation.12,14 C. perfringens can often be found in the gut contents of horses with or without alimentary tract disturbances.12,14 In addition, the potential role of some C. perfringens types (e.g., type A) as an enteropathogen in adult horses and foals is questionable. Therefore, C. perfringens toxinotyping is imperative to establish causation of gastrointestinal disease. Furthermore, C. perfringens strains have been more recently classified into 7 toxinotypes (A–G) according to the combination of major typing toxins that each type produces. 6 Major toxins include α-toxin, β-toxin, ε-toxin and ι-toxin, enterotoxin (CPE), and NetB, whereas non-typing toxins such as β2-toxin and θ-toxin could be found in any toxinotypes. 6 Since types have been linked to certain host and diseases, 6 toxinotyping of isolates recovered from horses with enterocolitis must be undertaken, otherwise, bacterial culture per se is clinically irrelevant. 6 Moreover, the diagnostic approach for N. risticii, L. intracellularis, and R. equi should take into consideration geographic, seasonal, and age-related factors. Interestingly, the one case tested that was positive for R. equi in our study was a 3-y-old American Quarter Horse mare with ulcerative and suppurative colitis. R. equi is not commonly associated with clinical disease in adult horses. The identification of this pathogen by PCR in this case should be interpreted cautiously; DNA detection alone should not be associated automatically with disease causation.
Multiplex PCR panels have been used widely in human medicine for several years.1,16,18 Among the advantages of these panels are faster turnaround time, rapid identification of potential etiologic agent, aid with antimicrobial therapy choice (e.g., PHF), and higher sensitivity and specificity for detection of organisms from a single specimen. 16 Conversely, disadvantages include increased cost, inability to distinguish between live and dead organisms, unknown clinical significance in some cases, and inability to perform antimicrobial susceptibility testing on bacterial isolates. 16 Multiplex PCR is very useful as part of the diagnostic investigation of acute gastrointestinal diseases; however, clinical judgment is paramount in test interpretation. 16 Although the ultimate detection of etiologic agents and toxins can also vary as a result of sampling and test sensitivity and specificity, we consider that our proposed guidelines could provide consistent data compilation, and therefore improve our understanding of equine colitis.
In addition to the inconsistency of etiologic laboratory tests performed on this series of equids, another diagnostic limitation identified in this dataset was the absence of standardized histologic description reporting. Although the inconsistency in the description of the lesions would not alter the final diagnosis, a standardized approach for the description of the intestinal lesions may be useful to better categorize equine enterotyphlocolitis. Such improvements in the description of gastrointestinal histologic samples were proposed and adopted for small animals more than a decade ago,3,20 and implementation of a similar protocol for equine cases could improve future investigations. The histologic description of all intestinal lesions of horses with typhlocolitis should include distribution, type of inflammatory infiltrate, including the grade of severity for each cell type, description of circulatory disturbances, and description of epithelial changes. A standardized protocol could be extremely important for future comparisons, including multi-center studies, as well as for future correlation with potential pathogens, which could solidify and confirm a diagnosis, as pathogen detection can be equivocal in some cases.
Overall, equine cases that we reviewed were 44% female and 56% male, had a median age of 8-y-old, and were mostly Standardbreds and Thoroughbreds. Although no direct comparison with the overall hospital population admitted during this study time was performed, our hospital caseload consists mostly of racehorses; predominantly Standardbreds and Thoroughbreds, as occurred in our study population. Our observations support the literature, 19 which notes that age, sex, or breed have not been identified as risk factors for the development of colitis in horses.
In our case series, ~50 % of the colitis cases occurred in the summer and early fall (June–September). The higher prevalence of equine colitis during this period is well-known and described. 12 Salmonellosis and PHF occur mostly during this time of the year in the Northern Hemisphere given the increased environmental temperatures, access to water, pasture, and activity of intermediate hosts. 13 Interestingly, only 20 of our cases were positive for Salmonella spp. or PHF. This suggests that many cases were undiagnosed (lack of testing, or, less likely, false-negative cases), that many of these cases survived, or that other pathogens, such as the newly described Neorickettsia findlayensis as a causative agent of PHF, 17 should be further investigated in these cases.
The main limitations of our study include those typically associated with retrospective data, including missing information, which can lead to misclassification bias. The lack of tests available in the earlier years of our study could also have contributed to our limited results.
Footnotes
Declaration of conflicting interests
The authors declare no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
