Abstract
A deceased 9-wk-old male gray fox (Urocyon cinereoargenteus) with a history of decreased ambulation and diarrhea was submitted to the Texas A&M Veterinary Medical Diagnostic Laboratory. No significant gross findings were evident on postmortem examination. Histologically, the cerebrum and brainstem had mild necrotizing meningoencephalitis with protozoal schizonts and merozoites. Additionally, glial cells contained intracytoplasmic and intranuclear viral inclusion bodies. Sections of the cerebrum were positive for canine distemper virus (CDV) and negative for Sarcocystis neurona on immunohistochemistry. Bayesian analysis revealed that this Sarcocystis sp. clustered most closely with a clade of unnamed Sarcocystis sp. found in viperid snakes, with a posterior probability of 99%. CDV likely played a significant role in the expression of clinical sarcocystosis in this gray fox.
Sarcocystis spp. are apicomplexan parasites with an obligatory 2-host life cycle.6,9 A sexual life cycle is completed in a canid definitive host, and asexual development is completed in a wide range of herbivorous and omnivorous animal intermediate hosts. 37 Definitive hosts are usually clinically unaffected, harbor the agent in the intestine, and shed oocysts and sporocysts in their feces. 18 Sporulation occurs in a favorable environment, and intermediate hosts ingest these infective sporulated oocysts or sporocysts. Sporocysts rupture in the intestine of the intermediate host and release sporozoites, which penetrate endothelial cells of small arterioles and convert into first-generation schizonts by schizogony. Schizonts are transformed into merozoites, invade small capillaries, and convert to second-generation merozoites that invade muscle cells to become sarcocysts containing numerous bradyzoites. The encysted sarcocyst is infective for a definitive host. Sarcocystis spp. may also cause clinical infection in sporocyst-consuming hosts that are unlikely to be eaten by the definitive host, such as S. neurona in horses and S. suihominis in humans. 11
Clinical disease caused by Sarcocystis spp., or sarcocystosis, is not reported commonly in canids. Various species, including S. neurona, S. canis, S. caninum, and S. svanai, can cause disease in domestic dogs. 10 S. svanai, which uses an unknown definitive host, causes muscle sarcocystosis in Pampas foxes.28,29 S. arctica, which uses corvid definitive hosts, causes muscle sarcocystosis in red foxes, Arctic foxes, and gray wolves.4,15,18,26 S. lutrae, which uses otter definitive hosts, causes muscle sarcocystosis in Arctic foxes. 14 An unidentified Sarcocystis causes muscle sarcocystosis in red foxes in Japan. 22 We did not retrieve any cases of sarcocystosis involving other body systems in a fox using Google, PubMed, CAB Direct, Web of Science, and Scopus, suggesting this condition has apparently not been reported in a fox.
A 9-wk-old male gray fox (Urocyon cinereoargenteus) was presented to a private veterinary hospital by a wildlife rehabilitator. The fox had a 3-wk history of decreased ambulation and diarrhea. Other young foxes were also affected. The animals had received 2 rounds of canine distemper virus (CDV; Paramyxoviridae, Canine morbillivirus), adenovirus 2, parvovirus, and parainfluenza (DA2PP) vaccination, although the timing of the vaccinations was unclear. All of the affected foxes died; however, only one fox was submitted to the Texas A&M Veterinary Medical Diagnostic Laboratory (College Station, TX, USA). A postmortem examination was performed, and no significant findings were noted. Brain, lung, liver, kidney, spleen, adrenal gland, stomach, and intestine were collected and placed in 10% neutral-buffered formalin. Tissues were processed, embedded, and stained with H&E for microscopic examination. For immunohistochemistry (IHC), 4-µm thick sections of the brain were stained by the avidin–biotin–peroxidase method (Vectastain ABC kit; Vector Laboratories), following the manufacturer’s instructions. The IHC staining was performed at the University of Georgia (Athens, GA, USA), as described previously. 35 S. neurona IHC was performed with a rabbit polyclonal antibody (1:500 dilution; David Lindsay, Michigan State University, E. Lansing, MI, USA), and CDV IHC was performed with a mouse monoclonal antibody (1:1,500 dilution; VMRD).
Histologically, the cerebral cortex contained areas of increased cellularity, pallor, and prominent blood vessels (Fig. 1). At higher magnification, these areas contained increased numbers of astrocytes and microglia, fewer lymphocytes, karyorrhectic debris, and fine vacuolation of the edematous neuropil. Within these foci were occasional immature protozoal schizonts that were oval or irregularly round, had thick walls, and were up to 20 µm in diameter (Fig. 2). Rare protozoa had ovoid merozoites radiating around a residual body (Fig. 3). A few glial cells within inflammatory foci contained intranuclear (Fig. 3) and intracytoplasmic (Fig. 4) viral inclusion bodies (INIBs, ICIBs, respectively). Multifocally, the meninges of the brainstem contained small numbers of macrophages and fewer lymphocytes. The lung was marked by interstitial pneumonia with numerous intralesional yeast consistent with Histoplasma capsulatum. Cerebellum, liver, kidney, spleen, heart, adrenal gland, stomach, and intestine were within normal limits. A few glial cells were positive for CDV on IHC from sections of the cerebrum. IHC for S. neurona was negative. Electron microscopy performed on formalin-fixed, paraffin-embedded brain sections did not identify the protozoa. Meningoencephalitis may have been the cause of the clinically observed decreased ambulation.
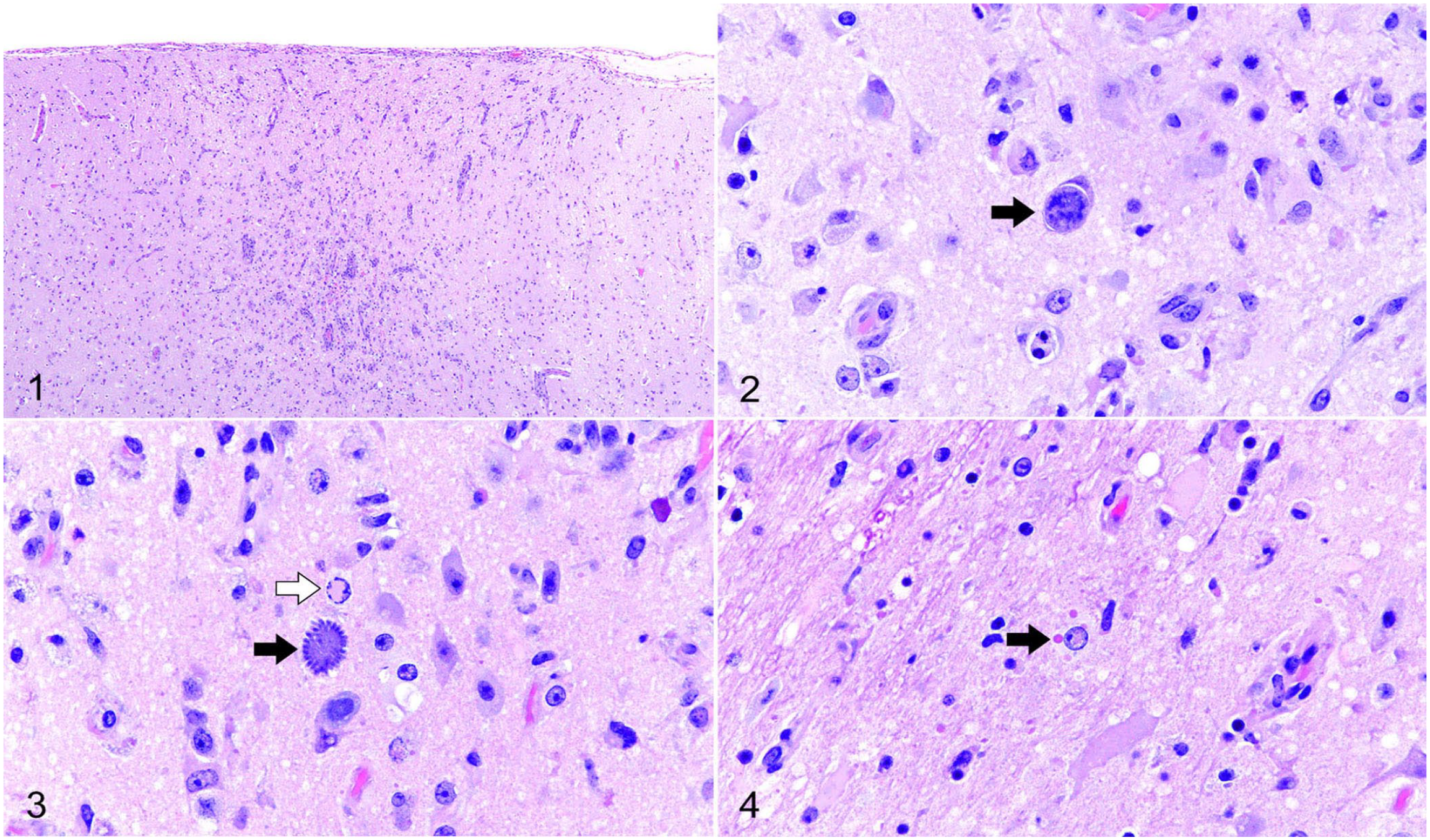
Microscopic lesions in the of brain of a gray fox with canine distemper virus and Sarcocystis sp. infection.
For molecular characterization of the protozoan agent, DNA was extracted from the formalin-fixed, paraffin-embedded brain (DNeasy blood and tissue kit; Qiagen). Previously described pan-Eucoccidia consensus PCR methods were used to amplify the 18S rRNA gene.13,28 The product was identified by Sanger sequencing and was 354 bp after primers were edited out. Additional protocols targeting a larger internal transcribed spacer 1 region resulted in no amplification. 10 This Sarcocystis sp. was distinct from all available homologous sequences in GenBank. We submitted the sequence to GenBank (OQ230473).
Predicted homologous 18S nucleotide sequences of related coccidian species were aligned using MAFFT, as described previously. 19 Bayesian analyses of each alignment were performed using MrBayes on the CIPRES server with gamma-distributed rate variation, a proportion of invariant sites, and a general time-reversible model.24,27 Eimeria arnyi was used as an outgroup. Four chains were run, and statistical convergence was assessed by looking at the SD of split frequencies and potential scale-reduction factors of parameters. The first 25% of 2,000,000 iterations were discarded as a burn-in. Two independent analyses were performed to avoid entrapment on local optima. In the Bayesian analysis, our unknown Sarcocystis sp. clustered most closely with a clade of unnamed Sarcocystis sp. that use viperid snakes as definitive hosts, with a posterior probability of 99% (Fig. 5). Although further confirmation is needed, we hypothesize that the definitive host of this organism may be a snake, and the fox may have been an aberrant intermediate host.
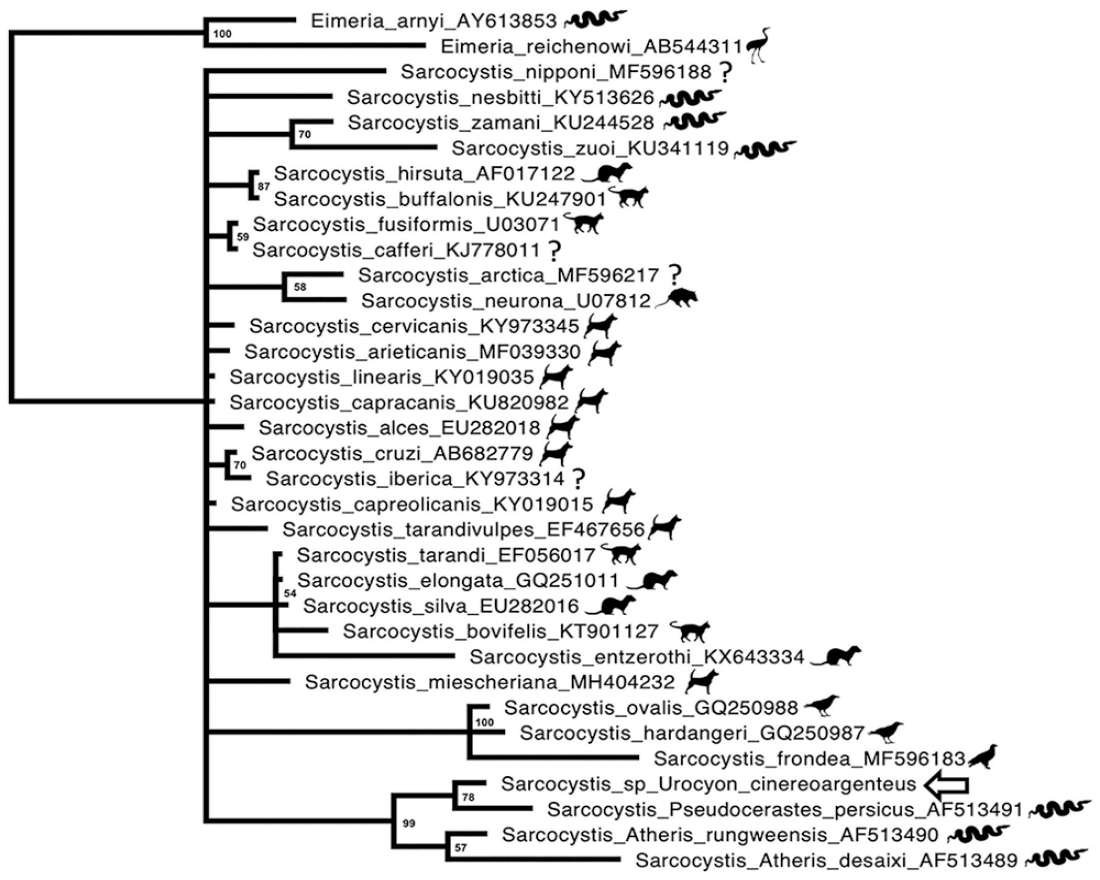
Bayesian phylogenetic tree of predicted nucleotide sequence of Sarcocystis sp. of gray fox based on MAFFT sequence alignment. Bayesian posterior probabilities of clusters as percentages are given at the nodes. The gray fox Sarcocystis sp. is indicated by the arrow. GenBank accessions are given after the names.
Canids are the definitive host for various species of Sarcocystis, and as definitive hosts they are usually subclinical and shed sporulated oocysts. For some other Sarcocystis spp., canids can also rarely serve as intermediate hosts, with sarcocysts embedded within skeletal muscle. Clinical sarcocystosis in domestic dogs is rare and can involve the brain, liver, eye, lung, and skeletal muscle.2,7,8,16,36 In the reported cases of Sarcocystis encephalitis in dogs, the lesions were necrotizing, but unlike in our case, the degree of inflammatory cell infiltration was more severe.6,9,25 Severe granulomatous and necrotizing meningoencephalitis has been reported in the cerebrum and cerebellum.6,7,24 Systemic infection with necrotizing granulomatous inflammation and intralesional schizonts and merozoites has also been reported in the eye, tongue, myocardium, pancreas, small intestine, liver, and nasal turbinates. In our case, mild necrotizing encephalitis with minimal inflammatory cell infiltration was evident. This may have been the result of CDV-induced immunosuppression. The lesions were seen in the cerebrum and brainstem, but the cerebellum appeared to be spared. We did not observe Sarcocystis lesions in the other tissues examined.
Immunosuppression induced by CDV infection likely predisposed this gray fox to meningoencephalitis caused by Sarcocystis. Microscopic findings in this case favor CDV as the primary cause of meningoencephalitis rather than Sarcocystis given the lack of typical protozoal necrotizing lesions. Immunosuppression is a significant effect of all members of the genus Morbillivirus, including CDV. Morbilliviruses enter host cells via the signal lymphocyte activation molecule expressed on activated B and T lymphocytes, thereby damaging lymphocyte populations critical for immune response and causing immunosuppression in the host. 38 Immunosuppression caused by CDV may be the result of lymphocyte lysis, interference with interferon function, and reduction of interleukin release.1,21 Viral infection can also suppress monocyte function by inhibiting the production of interleukin-1 and enhancing the release of excess prostaglandin E2, leading to immunosuppression in dogs.34,37 Additional evidence of immunosuppression in our case was the presence of pulmonary histoplasmosis.
The source of CDV in our case may have been the vaccine. Modified-live CDV vaccines are virulent in a variety of nontarget species in the suborder Caniformia, and their use is not recommended in nontarget species.3,5,12,23,31,32 Gray foxes are a highly CDV-susceptible species, and vaccine-induced CDV has been reported.17,33 Recombinant CDV vaccines are preferable for use in nondomestic carnivores. The specific type of vaccine used on this fox is unknown.
Clinical disease associated with concurrent Sarcocystis and CDV infection has been reported in raccoons, skunks, and dogs.2,20,24,34 Concurrent S. neurona and CDV infection has been reported in 4 raccoons and a skunk. 20 These animals had severe cavitating necrosis with numerous eosinophils, abundant mineralization, many merozoites and meronts, and ICIBs and INIBs in glial cells. Another report of encephalitis in a raccoon caused by Sarcocystis sp.–like protozoa and concurrent CDV infection described severe, locally extensive, neutrophilic and necrotizing encephalitis with numerous intralesional protozoa and perivascular nonsuppurative meningoencephalitis. 30 In our case, the fox had similar necrotizing meningoencephalitis, but the degree of inflammatory cell infiltration was milder. The relative lack of inflammatory cell infiltration compared to other reported cases of canine Sarcocystis encephalitis may have been the result of CDV-induced immunosuppression.
To our knowledge, the Sarcocystis sp. that we are reporting here has not previously had sequence characterization and likely represents a novel species. CDV likely played a significant role in the expression of clinical disease. Further study is needed to characterize the life cycle and clinical implications of this organism.
Footnotes
Acknowledgements
We thank Dr. Jitender P. Dubey for a helpful consultation and the Athens Veterinary Diagnostic Laboratory at the University of Georgia for performing the immunohistochemistry.
Declaration of conflicting interests
The authors declared no potential conflict of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
