Abstract
A 6-wk-old managed male Arctic wolf with lethargy, drooling, dehydration, elevated temperature, and acute onset of seizures was submitted for autopsy. The wolf had been vaccinated with a multivalent vaccine exactly 2 wk prior to presentation. Grossly, long bones were brittle and easily fractured under pressure; the intestinal contents were mucoid and yellow. Histologically, there was widespread lymphoid and hematopoietic necrosis, failure of endochondral ossification within long bones, as well as intranuclear and intracytoplasmic inclusions in various tissues and cell types. Canine distemper virus was detected in numerous tissues by IHC and confirmed by RT-rtPCR and sequencing as an American-4 strain, an emerging strain in domestic dogs and wildlife species in the southeastern United States. The clinical and pathologic findings associated with this emergent CDV strain have not been reported previously in wolves, to our knowledge. Canine parvovirus 2 (CPV-2b) was also detected in the spleen by IHC and confirmed by conventional PCR as a wild-type strain. The exact impact of CPV-2b on the clinical course is unknown. Early vaccination in this case may have predisposed this Artic wolf to developing clinical disease.
The Arctic wolf (
CDV infects all terrestrial carnivores and causes variable disease involving particularly the respiratory, gastrointestinal, and nervous systems. 8 Infection can be subclinical, and the development of clinical signs depends on viral strain virulence, environmental factors, and host immune function. 8 Although modified-live and recombinant vaccines exist for CDV in domestic canine species, few studies have focused on the safety and efficacy of administering commercial vaccines developed for the domestic dog in nondomestic canids.1,9,12,17 Vaccine-induced disease following administration of modified-live CDV vaccine has been described in several canid species, including wolves and African wild dogs, but vaccines are often used in these nondomestic species without adverse effects.3,8
Herein we describe a dual infection with an emergent strain of CDV concurrently with CPV in a managed juvenile Arctic wolf. Although previous work provided molecular characterization of this emergent CDV strain, the associated gross and histologic changes have not been described in wolves, to our knowledge.19,20 A 6-wk-old male Arctic wolf developed lethargy, drooling, dehydration, and elevated temperature. The owner had acquired the pup 5 d before the onset of clinical signs and was providing for it a diet of milk, ground turkey, and ground beef. While en route to the clinic, the pup had 2 seizures. The pup had been vaccinated 2 wk before presentation with a multivalent vaccine that included CDV, CPV, canine adenovirus, parainfluenza, and coronavirus. An in-house test for CPV (SNAP parvo test; IDEXX, Westbrook, ME) was negative. Despite medical intervention, the pup died and was submitted for autopsy at the Athens Veterinary Diagnostic Laboratory (University of Georgia, Athens, GA).
Grossly, the carcass was in thin body condition with scant internal adipose stores. There was a moderate amount of perianal fecal staining. The long bones were brittle and easily broken with little pressure. The intestinal lumen contained yellow, mucoid-to-liquid feces. Histologically, the trabeculae within the epiphysis and metaphysis had not completely ossified, had persistence of the primary spongiosa, retention of cartilage spicules, scant osteoid production, and frequent microfractures (Fig. 1). The trabeculae were separated by necrotic cellular debris and hemorrhage with severe necrosis and depletion of hematopoietic tissue, osteoblasts, and osteoclasts (Figs. 2, 3). The spleen, lymph nodes, and Peyer patches of the ileum were severely necrotic with a few infiltrating macrophages (Supplementary Figs. 1–4). Similarly, the mucosa of the ileum was occasionally collapsed because of severe necrosis, with few remaining intact crypts. The liver contained multifocal areas of hepatocellular necrosis and portal infiltrates of lymphocytes and macrophages (Supplementary Fig. 5). The bronchioles occasionally contained sloughed, necrotic epithelium. Intranuclear, eosinophilic viral inclusions were frequently detected in a wide variety of tissues and cell types, including lymphocytes and crypt epithelium within the intestine; astrocytes within the brainstem and cerebellum; and leukocytes within the spleen, lymph nodes, and liver. Intracytoplasmic, eosinophilic viral inclusions were also detected within astrocytes and the bronchiolar-lining epithelium in the lungs, as well as rarely within chief cells in the stomach and transitional epithelium of the urinary bladder (Supplementary Fig. 6). Lesions and inclusions within the intestine, brain, spleen, liver, lymph node, lung, and bone labeled strongly positive for CDV on immunohistochemistry (IHC; Fig. 4, Supplementary Figs. 7–11). Additionally, segments of the choroid plexus and individual leukocytes within the thyroid, parathyroid, adrenal gland, and pancreas were strongly positive with CDV IHC (monoclonal, IgG2B, catalog CDV-NP; Veterinary Medical Research and Development, Pullman WA). Parvoviral antigen was detected by IHC within necrotic regions of splenic lymphoid follicles and infrequently within the cytoplasm of macrophages located in necrotic Peyer patches of the intestine (monoclonal, IgG2A kappa, catalog CPV1-2A1; Custom Monoclonals International, Sacramento CA; Supplementary Fig. 12). Finally, conventional and reverse-transcription, real-time PCR (RT-rtPCR) assays targeting the
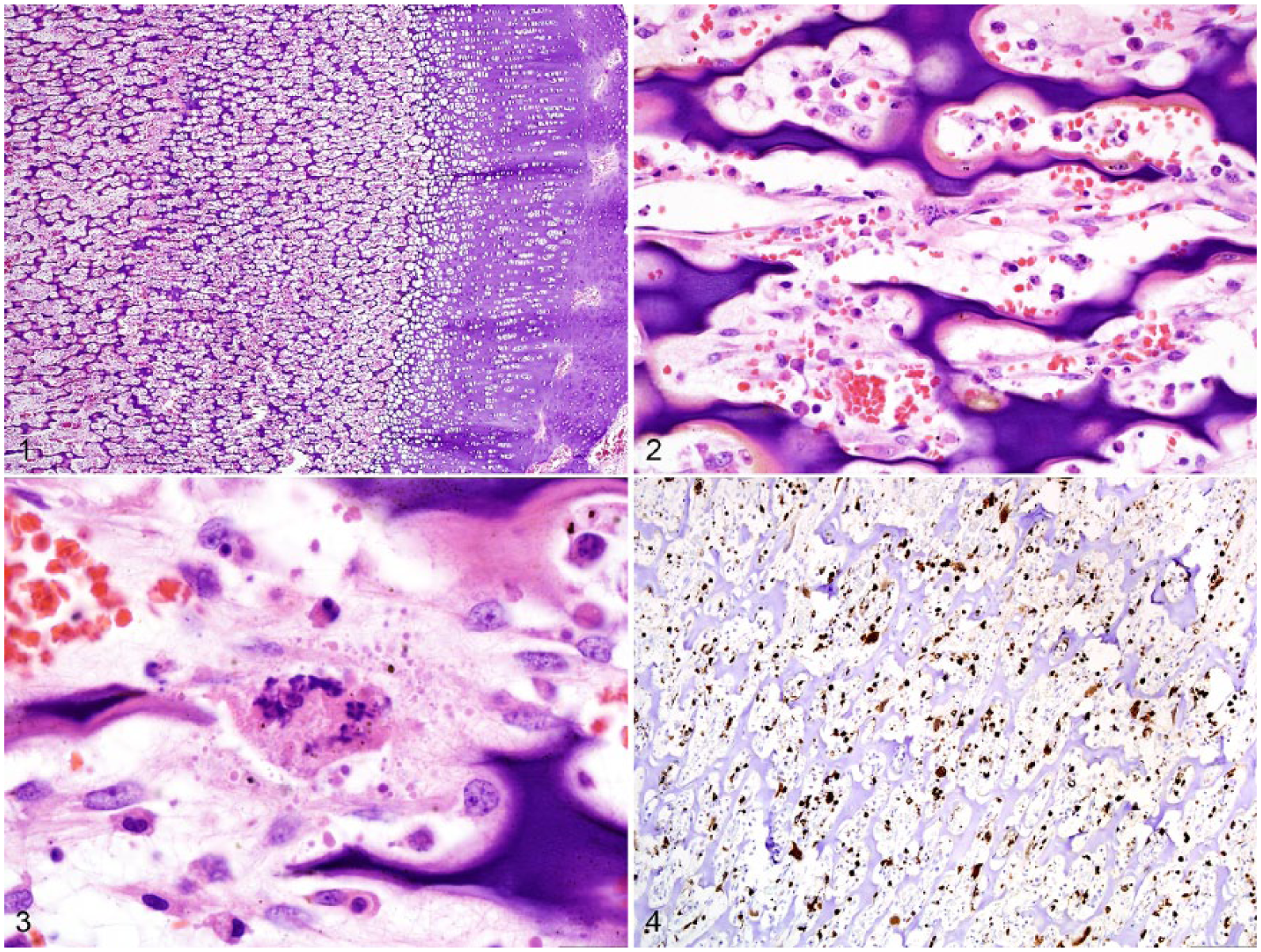
Failure of endochondral ossification in the femur of a wolf pup infected with canine distemper virus (CDV) and canine parvovirus.
Systemic CDV infection was determined to be the cause of death in this wolf pup, as supported by clinical and autopsy findings, and confirmed by IHC and PCR. All lesions and viral inclusions were associated with CDV infection; lesions in the spleen were also associated with CPV based on IHC and PCR. The incubation period for CDV, which is 14–18 d, is consistent with the reported clinical timeline of vaccine administration in this case 8 ; however, a vaccine-specific RT-rtPCR was negative, and sequencing of the M-F intergenic region confirmed the virus as an American-4 strain, a recently characterized emergent strain of CDV in wildlife and domestic species in the southeastern United States.19,20,24 This strain has been identified in domestic dogs, gray foxes, and raccoons, suggesting the existence of wildlife reservoirs.19,20 Serum antibody titer responses to this strain did not increase following vaccination in domestic dogs despite increased responses to the vaccine strain. 20 Cross-protection was titer-dependent with higher titers required for adequate protection, suggesting decreased cross-protection against this emergent strain, likely as a result of antigenic strain differences. 2 Furthermore, this strain was detected in clinically diseased, previously vaccinated dogs, supporting the possibility of vaccine escape. 20
Although general vaccination guidelines do not exist for all nondomestic canids, Association of Zoo and Aquarium (AZA) Canid Species Survival Plan (SSP) programs have created species-specific recommendations. 3 The American Association of Zoo Veterinarians (AAZV) recommends using the Merial PUREVAX ferret distemper recombinant canary pox vector vaccine for all nondomestic canids either annually or after serologic titer measurement; other vaccine forms are also considered acceptable for use in certain species. 3 Similarly, parvoviral vaccine recommendations are determined on a species-specific basis. 3 For canid species with no established vaccination recommendations, such as the Arctic wolf, protocols for domestic dogs are typically followed, despite being potentially inappropriate for nondomestic species.1,23 Although the exact brand of vaccine was not reported in our case, commercial multivalent canid disease vaccines are usually recommended for use in individuals aged at least 6 wk. 23 Considering that administration of multivalent vaccines often occurs without adverse effects in nondomestic canid species, vaccine misuse may have contributed to morbidity given that the pup in this case was 4-wk-old at the time of reported vaccination, although species-specific susceptibility to this strain remains a possibility.3,8 The status of maternal antibody production and delivery to the pup is unknown and, if insufficient, potentially further increased the risk of disease. Additionally, early administration of vaccine could have consumed maternal antibodies resulting in failure to generate immunity and decreased protection against CDV. 23 Even if the bitch was adequately vaccinated, maternal antibodies transferred to the pup successfully, and antibody levels not impacted by early vaccine administration, the antibodies generated against vaccine strains may react in a titer-dependent manner.2,20
Although parvoviral antigen was rarely detected in the intestinal tract, necrotic lymphoid follicles within the spleen labeled strongly positive using CPV IHC. Considering that CPV has a relatively short incubation period of 3–7 d, the splenic distribution of antigen likely indicates a late stage of infection, fitting with the timing of vaccine administration.
21
In contrast, PCR amplification and sequencing of a portion of the
Lesions associated with CDV infection are frequently reported in the brain, respiratory tract, and gastrointestinal tract. 8 The lesions described in the bone in our case were striking and highly similar to Paget disease and metaphyseal osteopathy of young dogs, which are associated with CDV infection.4,5,16 In experimental challenges, CDV antigen was detected most frequently in the metaphyseal bone cells of the primary spongiosa.4,5 Similar to our case, CDV infection resulted in persistence of the primary spongiosa and osteoclast, osteoblast, and bone marrow necrosis while sparing chondrocytes and the physis.4,5,16 Although the history provided regarding the diet of the wolf pup suggests the possibility of a nutritional component to the bone lesions, the severe loss of osteoblasts and osteoclasts, persistence of the primary spongiosa, lack of osteoid production, and unaffected growth plate indicate a primary infectious cause targeting the osteogenic and hematopoietic cells.4,5,16
Although the husbandry and maternal antibody status of this wolf pup is unknown, inappropriate administration of a modified-live vaccine potentially contributed to CDV and CPV antibody consumption, immunosuppression from CPV, and subsequent infection with an emerging, wild-type strain of CDV. In addition to more frequently reported respiratory, gastrointestinal, and neurologic lesions, we report widespread lymphoid and hematopoietic necrosis, failure of endochondral ossification, and systemic viral dissemination and inclusion body formation in numerous tissues. Vaccine recommendations from an authoritative body, such as an AZA Canid SSP program or the American Animal Hospital Association, should be followed to maximize immune function and minimize complications with modified-live vaccines in nondomestic canids. In cases of suspected vaccine-induced disease, viral strains must be molecularly characterized to confirm the origin of the agent.7,24
Supplemental Material
DS1_JVDI_10.1177_1040638719851832 – Supplemental material for Dual infection with an emergent strain of canine distemper virus and canine parvovirus in an Arctic wolf under managed care
Supplemental material, DS1_JVDI_10.1177_1040638719851832 for Dual infection with an emergent strain of canine distemper virus and canine parvovirus in an Arctic wolf under managed care by Justin M. Stilwell, Eman Anis, Rebecca P. Wilkes and Daniel R. Rissi in Journal of Veterinary Diagnostic Investigation
Footnotes
Acknowledgements
We thank Dr. Grazieli Maboni and Ingrid Fernandez (Athens Veterinary Diagnostic Laboratory) for their support with CPV and CDV molecular characterization.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplementary material
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
