Abstract
Listeria monocytogenes is a bacterium that can cause disease in many species, including humans, livestock, and wildlife. Increased interactions via shared habitats may promote pathogen transmission among these groups. Our objectives were to evaluate the Southeastern Cooperative Wildlife Disease Study diagnostic data to characterize and compare L. monocytogenes–induced lesions and comorbidities in gray foxes and wild turkeys, and to describe cases of listeriosis in 2 cervids. From 1991–2020, 8 gray foxes, 8 wild turkeys, a neonatal elk, and a white-tailed deer fawn from several eastern states in the United States were diagnosed with listeriosis. All 8 foxes had hepatitis and/or hepatic necrosis with intralesional gram-positive bacilli, and concurrent canine distemper virus (CDV) infection; 2 of the foxes had been vaccinated recently for CDV. L. monocytogenes was cultured from the liver (6 of 8) or lung (2 of 8) of foxes. Lesions in wild turkeys included hepatocellular necrosis (3 of 8), heterophilic hepatitis (1 of 8), heterophilic granulomas (1 of 8), intrasinusoidal gram-positive bacilli without hepatic lesions (1 of 8), granulomatous dermatitis (1 of 8), and/or granulomatous myocarditis (2 of 8). Lymphoproliferative disease viral DNA was detected in 5 of 6 turkeys tested; reticuloendotheliosis viral DNA was detected in 2 of 3 turkeys tested. Both cervids had systemic listeriosis, with L. monocytogenes isolated from liver. Immunohistochemistry for Listeria spp. on select cases revealed immunolabeling in affected organs. Listeriosis was thus established as a cause of morbidity and mortality in 3 wildlife species, which often suffered from concurrent infections and likely immunosuppression.
Keywords
With current and ongoing trends in human population growth and associated changes in landscape use, many wildlife species face higher anthropogenic pressures, come into increased contact with conspecifics, as well as humans, livestock, and other domestic animals, and often must adapt to suboptimal habitat. These factors may facilitate pathogen transmission to and among wildlife, especially when under concurrent stress and immunosuppression associated with decreased habitat quality, diminished resource availability, environmental conditions, or comorbidities. 8 The increased interface between wildlife, humans, and domestic animals warrants vigilance for pathogens of both zoonotic and veterinary health importance.
One such zoonotic agent, Listeria monocytogenes, is a globally distributed, ubiquitous, saprophytic, opportunistic bacterium that is found and can persist in a variety of substrates, including decaying plant material, water, and soil. L. monocytogenes can cause variable, sometimes fatal, disease manifestations (listeriosis) in humans and a wide host range, including many species of domestic and/or wild mammals, birds, fish, and crustaceans. 4 Factors such as age extremes, pregnancy, or immunosuppressive or immunocompromising coinfections or conditions predispose to disease. 5 Transmission can occur via ingestion of contaminated material or via inhalation of contaminated dust and soil. 4 Its ubiquity is evidenced by the detection of L. monocytogenes in many species of apparently healthy, free-ranging wildlife, including both avian and mammalian species.9,11,14,18,24 Despite the frequency of detections of L. monocytogenes in free-ranging wildlife, associated disease in wildlife is documented uncommonly.11,14,15 L. monocytogenes–associated disease has been described in 4 gray foxes (Urocyon cinereoargenteus) with viral comorbidities,1,12,16 as well as in a population of fallow deer. 6
Given the ubiquity and persistence of L. monocytogenes in the environment, numerous possible transmission routes, and the prevalence of potentially immunosuppressive conditions in wildlife, listeriosis should be considered as a potential cause of morbidity and mortality. Additionally, wildlife may represent an important reservoir or source of L. monocytogenes, given their shared habitat with humans. The wild turkey and gray fox are ubiquitous throughout much of North America and are adaptable species that commonly live in juxtaposition with humans and domestic animals. The wild turkey is a popular game bird across North America and typically inhabits wooded areas, but increasingly utilizes agricultural and suburban habitats. 20 The gray fox is a furbearing species trapped for its pelt and often is found in residential areas. 13 White-tailed deer are commonly found in urban, suburban, and exurban areas across much of North America, and their interactions with humans are an important aspect of their management. 22
Individuals, particularly those at greater risk of listeriosis, should practice good hygienic practices and wear appropriate personal protective equipment when handling wildlife carcasses or tissues because clinical disease and gross lesions may be subtle to non-evident. It also is important to note that wildlife may harbor L. monocytogenes without exhibiting clinical signs or gross or microscopic evidence of disease, given that L. monocytogenes has been isolated from apparently healthy wildlife, including wild deer and red foxes.18,24 When gross lesions of bacterial hepatitis are apparent, L. monocytogenes should be differentiated from other potential wildlife-borne zoonotic agents, including Francisella tularensis (the causative agent of tularemia), which can cause similar gross and microscopic lesions in foxes and other wildlife species. 12 Although most commonly considered a foodborne infection, listeriosis in humans has the potential for case-fatality rates of 20–30%, and is the second leading cause of foodborne infection–related mortality in the United States and Europe. 10
A better understanding of differences in the manifestation of disease across wildlife hosts as well as potential significant comorbidities may provide important insights into variations in pathogenesis and epidemiology that shed light on potential risk factors to humans, domestic animals, and wildlife. Our objectives were to describe cases of listeriosis in 2 common North American wildlife species, the wild turkey and gray fox, submitted to the Southeastern Cooperative Wildlife Disease Study (SCWDS; University of Georgia [UGA], Athens, GA, USA), and to characterize and compare L. monocytogenes–induced lesions and comorbidities in these 2 species, along with 1 case each in 2 cervid species, the elk (Cervus canadensis) and white-tailed deer (Odocoileus virginianus).
Materials and methods
Records for all diagnostic cases submitted to SCWDS from January 1975 to December 2021 were reviewed for those with diagnoses of listeriosis or L. monocytogenes infection. Background data for each submission included case history (i.e., field observations); location (county and state) and date the animal was found dead, harvested, or euthanized as a result of clinical disease; species; body weight; sex; and age (i.e., immature or adult, primarily based on weight in addition to plumage characteristics in wild turkeys 19 ). Diagnostic data included gross and histologic findings, ancillary test results, and final diagnoses. The percent occurrence of listeriosis among the gray foxes was estimated based on the number of cases among all those submitted for the species from the earliest diagnosis to study completion (January 1, 1991–December 1, 2021), given that full carcasses of gray foxes generally are received for autopsy. The percent occurrence for wild turkeys was similarly estimated; however, this included some cases for which selected tissues were submitted for specific diagnostic testing rather than exclusively full carcasses or complete sets of major tissues. The percent occurrence was not estimated for cervids given the high numbers of case submissions, which are more often submitted as select, field-collected tissues, often aimed at targeted testing for selected diseases (e.g., chronic wasting disease, hemorrhagic disease).
Case criteria for a diagnosis of listeriosis required microscopic lesions consistent with listeriosis concurrent with detection of L. monocytogenes by immunohistochemistry (IHC) and/or aerobic bacterial culture of affected tissues. All animals were free-living when found sick or dead except for 2 gray foxes that had been trapped recently and held temporarily in captivity, during which time they were vaccinated with a 5-way, modified-live virus canine vaccine (Duramune Max 5; Elanco).
For histologic examination, tissue samples were fixed in 10% neutral-buffered formalin, processed routinely, and 4-µm sections were stained with H&E. Additional histochemical stains, performed in select cases based on histologic findings, included Lillie–Twort or Brown and Brenn Gram stains, periodic acid–Schiff reaction, Ziehl–Neelsen acid-fast stain, modified acid-fast stain, and Grocott methenamine silver stain.
IHC for L. monocytogenes was performed on tissues with lesions consistent with bacterial infection from 8 of 8 wild turkeys and 7 of 8 gray foxes (including the 2 vaccinated foxes). IHC was performed on an automated stainer (Nemesis 3600; Biocare Medical). Rabbit polyclonal antiserum against L. monocytogenes was used as the primary antibody (L2650-09A; US Biological), diluted 1:100, and incubated for 45 min at 37°C with agent-positive control. Antigen retrieval was by proteinase K (S302030-2; Agilent) for 5 min; enzyme blockage was via 3% H2O2 for 5 min; protein blockage was by Power Block (HK085-5K; BioGenex) for 5 min; link was by biotinylated goat anti-rabbit IgG antibody at a dilution of 1:100 (BA-1000; Vector) for 10 min; labeling was 4+ streptavidin horseradish peroxidase (HP604L; Biocare Medical) for 10 min with DAB chromogen substrate (BDB2004L; Biocare Medical) for 12 min. Known L. monocytogenes–positive control tissues were tested alongside each case.
Aerobic bacterial culture was performed on a liver sample from 6 of 8 gray foxes and lung from 2 of 8 gray foxes. Liver from 8 of 8 turkeys with listeriosis was cultured. Intestine also was cultured in two of these, skin and heart in a third, and brain and lung in a fourth. Aerobic culture was performed on liver and spleen samples from the elk, and on the liver and brain from the white-tailed deer. A fluorescent antibody test (FAT) for rabies virus (Lyssavirus rabies) antigen was performed on brain of all 6 non-vaccinated gray foxes and 1 of 2 cervids. Neither of the vaccinated foxes was tested for rabies. Canine distemper virus (CDV; Canine morbillivirus) FAT was performed on brain of 6 of 6 non-vaccinated gray foxes and on brain, lung, stomach, and urinary bladder of the 2 vaccinated foxes. Tissue samples (bone marrow, liver, and/or spleen) from 6 of 8 turkeys were tested by PCR for lymphoproliferative disease virus (LPDV) and 3 of 8 for reticuloendotheliosis virus (REV). Bacterial culture and FAT were performed using standard protocols at the Athens Diagnostic Laboratory (UGA, Athens, GA, USA), an American Association of Veterinary Laboratory Diagnosticians–accredited laboratory, in accordance with protocols established by the Clinical and Laboratory Standards Institute (https://clsi.org/). The LPDV and REV PCR tests were performed in a research virology laboratory at UGA according to validated methods. 17
Results
Number of cases of listeriosis in gray foxes and wild turkeys
From 1991 to 2021, listeriosis was diagnosed in 8 of 197 (4.1%) gray foxes evaluated; 2 of these were most likely infected during a brief captive period prior to death. Listeriosis was diagnosed in 8 of 836 (0.96%) wild turkeys evaluated from 1991 to 2021.
Demographic and seasonal data and geographic distribution of gray foxes and wild turkeys
Four of 8 foxes were female and 4 were male; all 8 were adults. For the naturally infected foxes, 1 each was submitted from Georgia (Hall County), Louisiana (St. Tammany Parish), and Maryland (Anne Arundel County), and 3 were from North Carolina (2 from Caldwell and 1 from Brunswick Counties). The 2 foxes with vaccine-induced CDV originated from Madison County, Georgia and both were adult males. Of the 8 foxes, 3 died in winter, 2 in fall, 2 in spring, and 1 in summer.
Four of the 8 wild turkeys with listeriosis were male and 4 were female; all were adults, except for 1 juvenile female. Two of the turkeys were from Alabama (Macon and Dallas Counties); 3 were from Kentucky (Perry, Bath, and Livingston Counties); and 1 each was from Mississippi (Lowndes County), West Virginia (Upshur County), and Kansas (Leavenworth County). Of the 8 turkeys, 3 died in spring, 3 in fall, and 1 each in winter and summer.
Case histories for gray foxes and wild turkeys
All case submissions for wild turkeys and gray foxes included in our study consisted of whole carcasses, except for one adult male turkey from which only a liver sample was submitted. Abnormal behavior or neurologic signs were reported in all 6 free-living foxes; 5 were killed by gunshot, and manner of death (i.e., natural death vs. euthanasia) was not provided for 1 fox. Two additional foxes were among a group of 25 wild-caught foxes held in a hunting pen. Of these foxes, 4 were found dead 1–8 d following CDV vaccination, and 2 were submitted to SCWDS for autopsy. Two wild turkeys were euthanized after observation of nodular skin lesions (of suspect poxviral origin) on the head and/or neck. Five birds were found dead. Another turkey reportedly was morbid, with final manner of death unreported.
Diagnostic findings in gray foxes
Autopsy revealed that 3 of 8 foxes were in poor nutritional condition, with diffusely decreased adipose stores and variable muscular atrophy. Three foxes were in fair, and 2 were in good, nutritional condition. Three of the foxes had mucopurulent exudate within bronchiolar lumina. One fox had dozens of <1–6-mm, firm, white-to-tan foci throughout the hepatic parenchyma and on the capsular surface (Fig. 1), and fewer similar foci throughout the pulmonary pleura and parenchyma. One fox had a 10 × 10 mm ulcer along the right side of the tongue. Foxes killed by gunshot had associated soft tissue damage and multiorgan hemorrhage, which occasionally hindered gross evaluation of the lungs. Mild, incidental enteric cestodiasis or gastric nematodiasis was noted in 3 of the foxes. Minimal numbers of ectoparasites, including ticks, fleas, and ear mites, were sometimes noted.
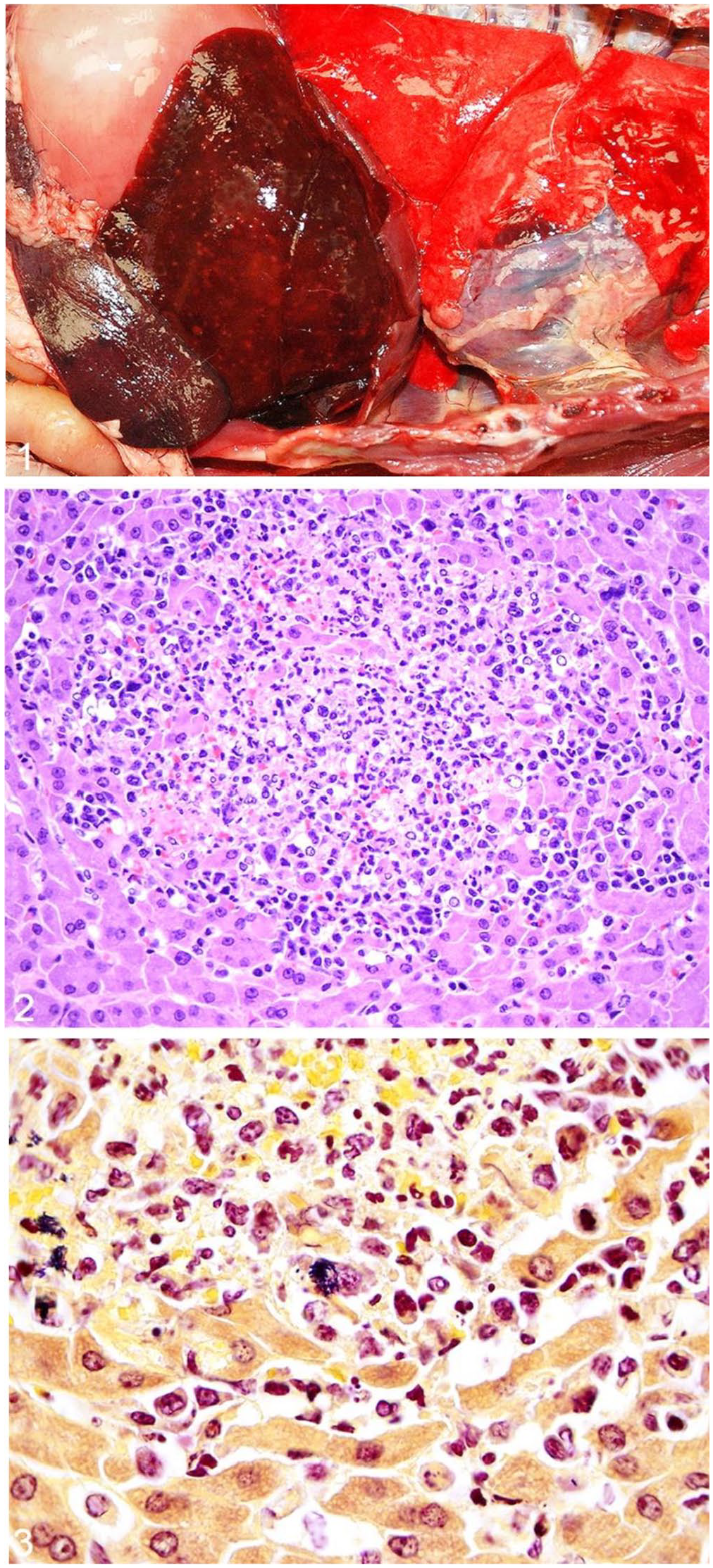
Listeria monocytogenes–associated gross and histologic lesions in a gray fox.
Microscopically, all 8 foxes had hepatic lesions (Suppl. Table 1). Most commonly, these included mild-to-severe, random, multifocal-to-coalescing, necrotizing and/or suppurative hepatitis (6 of 8), although severe, random, multifocal-to-coalescing, lymphoplasmacytic and histiocytic hepatitis affected 2 of 8 foxes (Fig. 2). Intralesional gram-positive bacterial rods were detected in the livers of 7 foxes (Fig. 3), and in the lungs of 1 fox. In a fox with subacute liver changes, no bacteria were detected with H&E or Gram stains. The foxes with vaccine-induced distemper had multifocal necrosis throughout the spleen, and one had additional foci of necrotizing and suppurative inflammation throughout the spleen, lymph node, and adrenal gland. Bronchointerstitial pneumonia was noted in 7 of 8 foxes, and all 8 had variable lymphoid depletion in spleen, lymph node, and/or bone marrow. Eosinophilic, intranuclear and intracytoplasmic inclusion bodies, consistent with morbilliviral (i.e., CDV) infection, were in bronchiolar or bronchial epithelium (6 of 8), biliary epithelium (5 of 8), glial cells (2 of 8), small intestinal and gastric mucosal epithelium (1 of 8), urinary bladder mucosal epithelium (1 of 8), renal urothelium (1 of 8), neurons (1 of 8), and salivary gland ductal epithelium (1 of 8). Incidental lesions, each noted in a single fox, included focal adrenal corticomedullary necrosis and hemorrhage, myofiber degeneration and necrosis, eosinophilic enteritis and esophagitis, focal ulcerative glossitis with thrombosis, splenic hemosiderosis, and mild enteric cestodiasis and nematodiasis.
Aerobic bacterial culture in at least one tissue from each fox (i.e., lung [2 of 8] or liver [6 of 8]) led to isolation of L. monocytogenes (Suppl. Table 1). In 2 foxes, there was concurrent isolation of Escherichia coli, and in 1 fox, there was concurrent heavy growth of Klebsiella pneumoniae and an unidentified Citrobacter species. IHC for L. monocytogenes was performed on tissues from 7 of 8 foxes; immunoreactivity was observed in liver (7 of 7), spleen (4 of 7), and lung (3 of 7). There was variable, dark, globular-to-granular labeling within necrotic foci in the liver and within the cytoplasm of macrophages throughout all 3 organs with immunoreactivity. All 8 foxes (including the 2 vaccinated) had concurrent CDV infection. No rabies viral antigen was detected by FAT in brain of any fox tested.
Diagnostic findings in wild turkeys
Among the 8 wild turkeys diagnosed with listeriosis, nutritional condition was emaciated (2 of 7), poor (1 of 7), fair (1 of 7), and good (3 of 7). Nutritional condition, lesion patterns, and comorbidities were not assessed in one turkey because only liver was submitted for evaluation. Four turkeys had firm, tan-to-yellow, granular, proliferative dermal masses along the head and/or neck. Two of these had crusting lesions along glabrous skin of the limbs. Three turkeys had gross hepatic lesions. The most severely affected turkey had multifocal-to-coalescing, random, firm, white-to-tan nodules (granulomas) dispersed throughout both hepatic lobes. One of these had widely scattered, dark-brown foci (necrosis) and the other had a large (~25% of the liver affected), dark-red focus. One bird had splenomegaly with nodular white masses on cut section (attributable to LPD). One turkey had a large, granulomatous mass expanding the ventricular myocardium (Fig. 4). The turkey with the hepatic granulomas had concurrent cerebral, intramuscular, serosal (along the cecum, with intraluminal cecal core), and pulmonary granulomas, as well as a mild tick infestation and minimal tracheal nematodiasis.
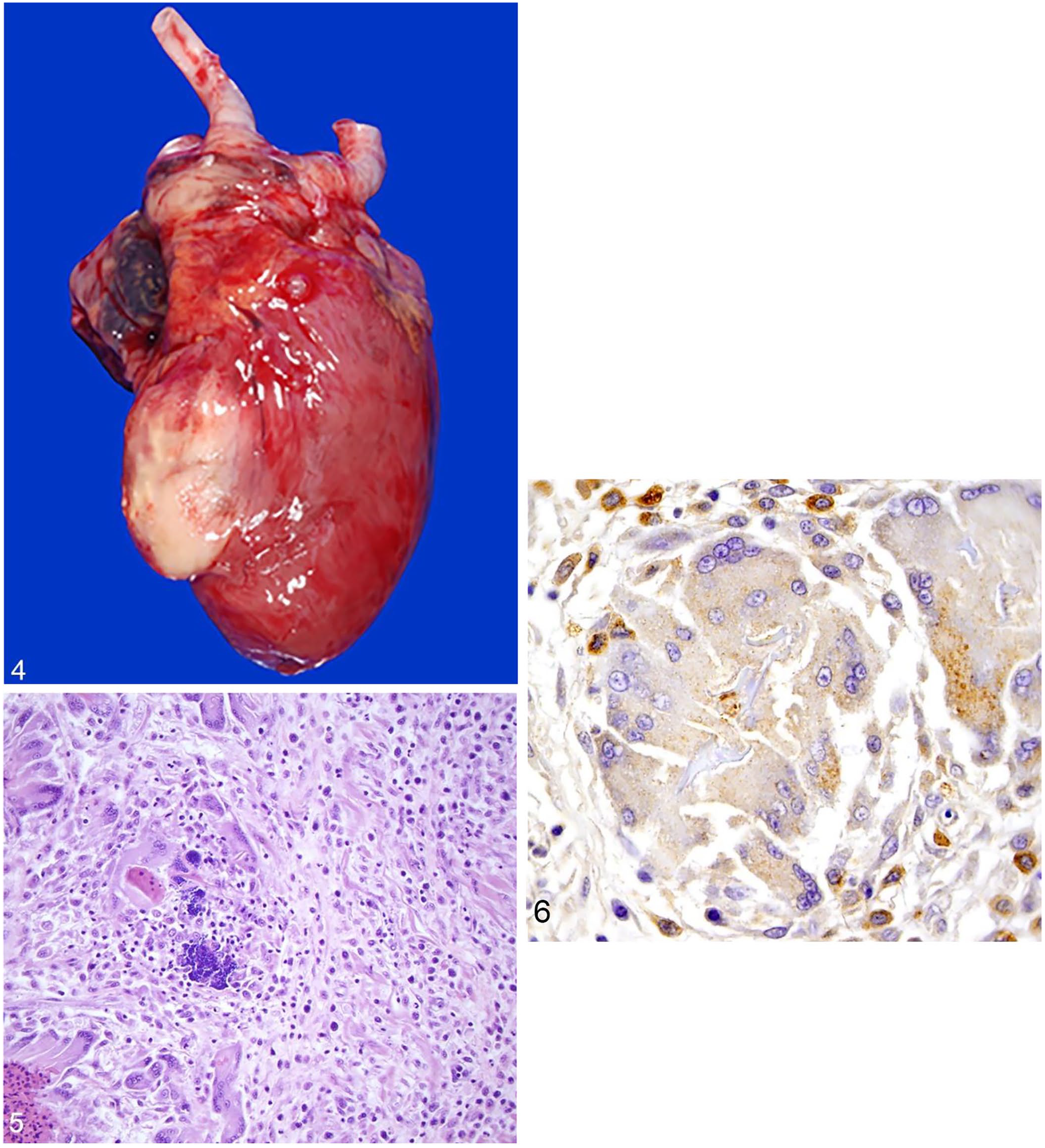
Listeria monocytogenes–associated gross and histologic lesions in a wild turkey.
Microscopic lesions attributable to Listeria spp. infection were variable in the turkeys and included granulomatous myocarditis in 2 of 8 (Fig. 5); 1 of these also had intralesional gram-positive bacilli and concurrent granulomatous dermatitis (Suppl. Table 2). Hepatic lesions included hepatocellular necrosis with gram-positive bacilli in 3 of 8; heterophilic hepatitis with gram-positive bacilli in 1 of 8; heterophilic granulomas with gram-positive coccoid bacteria in 1 of 8; and intrasinusoidal gram-positive bacilli without concurrent hepatic lesions in 1 of 8. One of the turkeys with hepatic necrosis also had splenic and interstitial pulmonary necrosis, and renal tubular and cardiomyocyte degeneration with gram-positive bacilli. Several comorbidities of variable clinical significance were detected in the wild turkeys (Suppl. Table 2). Histologic lesions (n = 1 each) included: multisystemic round-cell infiltrates; protozoal endoarteritis, encephalitis, and myositis; mild cardiac microfilariasis; lymphoplasmacytic proventriculitis or ventriculitis; fibrinonecrotizing enteritis; granulomatous dermatitis; poxviral glossitis; small intestinal mucosal blunting. The turkey with granulomas noted grossly had a fungal cerebral granuloma, bacterial heterophilic pulmonary granulomas, heterophilic and necrotizing rhabdomyositis with fungal hyphae and intra-sarcoplasmic protozoal cysts, severe lymphohistiocytic typhlocolitis with intraluminal necroheterophilic core, chronic heterophilic dermatitis and blepharitis with superficial cocci and serocellular crusting, foreign material within air sacs, moderate ingluvial capillariasis, necrotizing splenitis, and koilin erosion in the ventriculus. Areas of granulomatous inflammation did not contain acid-fast bacteria or fungi. Additional lesions in wild turkeys included lymphoid depletion (n = 2), poxviral dermatitis (n = 2), splenic and hepatic hemosiderosis (n = 2), and incidental intra-sarcoplasmic sarcocysts in skeletal muscle (n = 2).
Aerobic bacterial culture in at least one tissue from each wild turkey (i.e., liver [7 of 8] or heart [1 of 8]) led to isolation of L. monocytogenes (Suppl. Table 2). In 2 animals, there was concurrent isolation of E. coli. In the turkey with multiorgan granulomas, L. monocytogenes was isolated from a hepatic granuloma, but not from granulomas in the brain or lung. IHC for Listeria spp. was performed on tissues from all 8 turkeys. Tissues from 6 birds exhibited strong cytoplasmic immunoreactivity for L. monocytogenes within hepatocytes and/or macrophages throughout areas of inflammation. Two of the birds with hepatic labeling also had strong labeling in the spleen within areas of necrosis, and one also had strong labeling in renal tubular epithelial cells. One of the turkeys with no immunoreactivity in the liver had strong labeling within intravascular macrophages and areas of inflammation in the heart and lung, and the other had labeling in macrophages in the heart and skin (Fig. 6).
Tissues from 5 of 6 wild turkeys tested had LPDV DNA detected by PCR test (Suppl. Table 2). Three turkeys were tested for REV and 2 were positive, 1 of which was the turkey with no LPDV DNA detected. The turkey with multisystemic protozoal infection had Toxoplasma and Neospora spp. antigen detected via FAT. The turkey with multiorgan granulomas had Toxoplasma gondii DNA detected by protozoal PCR test. The bird with severe enteritis had Tetratrichomonas gallinarum DNA detected by PCR test.
Demographic data, geographic distribution, and diagnostic findings in cervids
From 1991 to 2021, 2 cervids (a 4-d-old male elk calf from Carter County, MO, USA and a 6-mo-old female white-tailed deer fawn from Clarke County, GA, USA) were diagnosed with listeriosis. The elk calf had been found dead near its birth site, and select tissues were received for evaluation in May 2016. The white-tailed deer fawn had ptyalism, dyspnea, and reduced activity; it was euthanized, and the carcass was submitted for autopsy in December 2016.
Microscopically, the elk calf had mild, random, multifocal-to-coalescing, suppurative inflammation with variable numbers of macrophages, lymphocytes, and plasma cells in the liver, kidney, lungs, and heart, with intralesional gram-positive bacilli in the liver, kidney, and heart; the latter also had myocardial degeneration, and necrosis. Aerobic bacterial culture of liver and spleen yielded heavy growth of L. monocytogenes from both organs; Pseudomonas aeruginosa (liver and spleen), Enterobacter amunigenus (liver), and E. coli (spleen) were also cultured. Formalin-fixed, paraffin-embedded tissue blocks were not available at the time of our study for IHC.
The white-tailed deer fawn was in fair nutritional condition with ascites and pleural effusion. The liver was mottled with white, linear streaks along the capsular surface. Microscopically, the fawn had mild, neutrophilic meningitis with satellitosis; moderate, neutrophilic and necrotizing hepatitis with gram-positive bacilli; severe, diffuse submucosal and serosal edema and lymphoplasmacytic serositis of the colon; and neutrophilic glomerulonephritis and pneumonia (Figs. 7, 8). In the liver, the bacteria were confirmed as L. monocytogenes via IHC. Other histologic findings included splenic hemosiderosis, steatitis, and renal tubular degeneration. Aerobic culture of brain and liver yielded L. monocytogenes in both samples. Rabies viral antigen was not detected by rabies virus FAT, and the prion that causes chronic wasting disease was not detected by IHC.
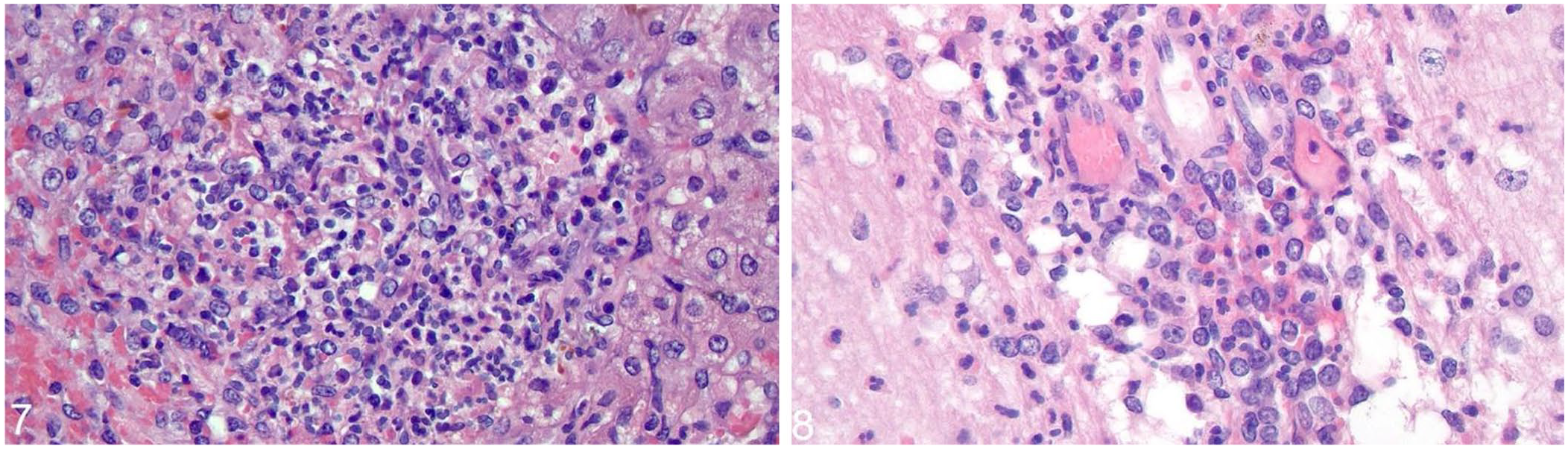
Listeria monocytogenes–associated microscopic lesions in a white-tailed deer fawn.
Discussion
In domestic ruminants, listeriosis is seen most often as meningoencephalitis, abortion, or septicemia, 7 whereas in humans, the most common clinical syndromes include febrile gastroenteritis, maternal-fetal and/or neonatal listeriosis, and sepsis, which may have CNS involvement. 5 Lesion patterns attributed to L. monocytogenes infection among gray foxes, wild turkeys, and wild cervids varied in our study, and sometimes overlapped with forms described in humans and livestock. No consistent gross lesions were seen in gray foxes, and nutritional condition varied from poor to good. When present, gross liver lesions usually were subtle; however, all 7 foxes had microscopic lesions of random, multifocal, hepatocellular necrosis and suppurative hepatitis. These microscopic lesions are similar to those reported in gray foxes with listeriosis that were concurrently infected with CDV.12,16 In wild turkeys, microscopic liver lesions often were similar to those in foxes, and other lesions included granulomatous myocarditis and/or dermatitis, splenic and interstitial pulmonary necrosis, and renal tubular and cardiomyocyte degeneration. Many of these findings are consistent with lesions of listeriosis documented in domestic poultry. 3 Two wild turkeys in our study had granulomatous myocarditis with strong Listeria spp. immunolabeling within inflammatory cells; one of these turkeys also had granulomatous dermatitis with similar Listeria spp. immunolabeling. Lesions of listeriosis in the elk and white-tailed deer were consistent with those seen in young domestic animals affected by the septicemic form of listeriosis. 2
Liver consistently was a useful sample for the detection of L. monocytogenes by aerobic culture in animals with suspected disease. Culture of heart and lungs also was useful in wild turkeys with lesions in these organs. Gram stains helped determine bacterial distribution and guided tissue selection for culture. IHC revealed strong cytoplasmic immunolabeling in inflammatory cells in both wild turkeys and gray foxes and was used to confirm the presence of L. monocytogenes within lesions in more unusual anatomic locations or with variable lesion patterns, such as in cases of granulomatous inflammation in skin and heart of turkeys.
All wild turkeys and gray foxes diagnosed with listeriosis had coinfections or comorbidities. In wild turkeys, this most commonly included LPDV, which is an oncogenic, potentially immunosuppressive retrovirus that has been detected in high proportions of healthy, and fewer diseased, wild turkeys across much of the United States and portions of Canada. 17 Several wild turkey samples contained REV antigen, a virus with oncogenic and immunosuppressive potential in domestic poultry. 23 Another wild turkey had systemic protozoal disease. All gray foxes with listeriosis had concurrent CDV infection, which manifested as mild-to-severe bronchointerstitial pneumonia, sometimes with concurrent, mild gliosis throughout the brain, and included variably severe lymphoid depletion in the lymph nodes and/or spleen. Coinfections have been reported in a small number of gray foxes with listeriosis, including CDV, skunk adenovirus 1 (Skunk mastadenovirus A), and Yersinia pseudotuberculosis infections.1,12,16 Cervids with listeriosis had no evidence of immunosuppression or comorbidities; however, both cervids were juveniles, and likely lacked complete immune system development. Further, lack of complete clinical history and sample evaluation in these individuals may have underestimated this assessment.
Based on the lesion patterns of listeriosis in both wild turkeys and gray foxes, ingestion of L. monocytogenes–contaminated material (food, water, soil, etc.) may represent a portal of entry, with subsequent hematogenous spread. Opportunistic infections also are possible secondary to immunosuppression. There are at least 2 reports of L. monocytogenes septicemia in domestic dogs associated with immunosuppression, which may suggest a similar pathogenesis in gray foxes with CDV infection–induced immunosuppression. 21 Further, immunosuppression from pre-existing or concurrent infections may be required for the development of listeriosis in many wildlife individuals based on the detection of comorbidities in our and past studies12,16 and the relatively high detection without apparent disease. 24 In the neonatal elk, transmission may have occurred transplacentally, given that most cases of neonatal septicemia are thought to develop in utero. 2 The classic L. monocytogenes encephalitis in adult domestic ruminants is associated with feeding of silage and it is thought that L. monocytogenes enters via oral wounds, and spreads to the trigeminal nerve, from which it travels to the brain. 2 Although similar lesions were not noted in either cervid in our study, access to silage may be another mechanism that could affect wild ungulates. The potential for wildlife access to contaminated material in the environment, including silage and discarded bedding or food material, and sewage on farms and other human-altered habitats, should be considered in the field aspects of diagnostic evaluations. Further, farm practices should consider potential risk to surrounding wildlife in waste management practices. Correlation with seasonality data was not present for the gray foxes or turkeys.
Limitations of retrospective studies of diagnostic data, including ours, are the incomplete case histories and inconsistent sample sets and types for evaluation. Postmortem intervals of wildlife cases often are extended as a result of variations in carcass discovery and recovery from the field, as well as the multistep process of field sample collection, transport, storage, and subsequent shipment to a diagnostic laboratory. Listeria spp. as a cause of disease may have been underestimated among wild turkeys, gray foxes, and cervids in our diagnostic database because not all cases involved full autopsies, and autolysis is a frequent challenge that may have obscured more subtle lesion patterns associated with Listeria spp. infection. Despite these challenges, retrospective diagnostic evaluations in wildlife provide unique and valuable data.
Supplemental Material
sj-pdf-1-vdi-10.1177_10406387221104830 – Supplemental material for Listeriosis with viral coinfections in 8 gray foxes, 8 wild turkeys, and 2 young cervids in the southeastern United States
Supplemental material, sj-pdf-1-vdi-10.1177_10406387221104830 for Listeriosis with viral coinfections in 8 gray foxes, 8 wild turkeys, and 2 young cervids in the southeastern United States by Alisia A. W. Weyna, Kevin D. Niedringhaus, Melanie R. Kunkel, Heather M. A. Fenton, M. Kevin Keel, Amy H. Webb, Charlie Bahnson, Rebecca Radisic, Brandon Munk, Susan Sánchez and Nicole M. Nemeth in Journal of Veterinary Diagnostic Investigation
Footnotes
Acknowledgements
We thank SCWDS member state wildlife agencies for diagnostic case submission, including the states of Alabama, Arkansas, Florida, Georgia, Kentucky, Kansas, Louisiana, Maryland, Mississippi, Missouri, Nebraska, North Carolina, Oklahoma, South Carolina, Tennessee, Virginia, and West Virginia, USA. Further support was provided by SCWDS federal wildlife agency partners, including the U.S. Fish and Wildlife Service National Wildlife Refuge System and the U.S. Geological Survey Ecosystems Mission Area. SCWDS diagnosticians and staff contributed to the completion of cases and molecular testing (Kayla Adcock), and Athens Veterinary Diagnostic Laboratory and Poultry Diagnostic Research Center colleagues provided technical laboratory expertise, including bacterial culture, histology processing, and IHC.
Declaration of conflicting interests
The authors declare no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
Funding was provided by the sponsorship of the Southeastern Cooperative Wildlife Disease Study by the fish and wildlife agencies of Alabama, Arkansas, Florida, Georgia, Kentucky, Kansas, Louisiana, Maryland, Mississippi, Missouri, Nebraska, North Carolina, Ohio, Oklahoma, Pennsylvania, South Carolina, Tennessee, Virginia, and West Virginia, USA. Support from the states to SCWDS was provided in part by the Federal Aid to Wildlife Restoration Act (50 Stat. 917).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
