Abstract
Cryptosporidiosis is an intestinal protozoal disease of public health importance caused by Cryptosporidium spp. Despite the high synanthropism of raccoons, studies describing the pathology of Cryptosporidium spp. infections in this species are lacking. Therefore, we characterized the pathology of cryptosporidiosis in 2 juvenile raccoons. In addition, we conducted a retrospective search of the database of the California Animal Health and Food Safety laboratory for 1990–2019 and found 6 additional cases of cryptosporidiosis in raccoons. Sequencing of cryptosporidia was performed in one autopsied raccoon, and PCR on formalin-fixed, paraffin-embedded tissues in archived cases. The Cryptosporidium skunk genotype (CSkG), a strain of zoonotic relevance, was detected in 6 of 8 cases (75%). Frequently, cryptosporidiosis was associated with enteritis, eosinophilic infiltrates, villus atrophy or blunting and/or fusion, and crypt abscesses or necrosis. In 7 of the 8 cases, there was confirmed concurrent coinfection with canine distemper virus; 1 case was coinfected with canine parvovirus. Although crypt necrosis is considered a classic lesion of canine parvoviral infection in mesocarnivores and not a hallmark of cryptosporidiosis, results suggest that canine distemper virus is capable of mimicking such lesions in combination with cryptosporidia and immunosuppression.
Cryptosporidiosis is a protozoal disease of public health importance, caused by Cryptosporidium spp., a group of apicomplexan parasites that cause intestinal disease and are found in >150 vertebrate species, including mammals, reptiles, and birds. 9 Given ongoing habitat encroachment associated with urbanization of forested land, wildlife species are increasingly coming into contact with human and domestic animal pathogens, becoming accidental carriers and potential pathogen amplifiers. 38 Given the propensity of raccoons (Procyon lotor) to roam within or around human dwellings (synanthropism), there has been an interest in determining the carrier status of these animals for humans pathogens including Cryptosporidium spp.12,33 Synanthropic wildlife can be sentinels of environmental pathogens as well as sources of infection for other animals and humans. 40 Although Cryptosporidium spp. appear to have a strong host adaptation, some genotypes can be detected in numerous host species when a common source of infection exists (e.g., contaminated water). 42 In addition, all procyonids, including raccoons, can become infected with pathogens such as canine parvovirus (CPV; Carnivore protoparvovirus 1) and canine distemper virus (CDV; Canine morbillivirus).5,12,19,20,25,30,32 However, information about Cryptosporidium spp. infection of raccoons in the literature is scant. Hence, the purpose of our study was to describe the pathology of cryptosporidia infections in raccoons.
Materials and methods
Postmortem examination
Two juvenile raccoons (raccoons 1 and 2) were submitted for postmortem examination and diagnostic workup to the San Bernardino branch of the California Animal Health & Food Safety (CAHFS) Laboratory System, University of California–Davis. An autopsy was performed in both cases. Samples of brain (cerebrum, thalamus, hypothalamus, midbrain, pons, cerebellum, medulla oblongata), lung, heart, diaphragm, spleen, liver, stomach, intestine, pancreas, adrenal gland, kidney, and urinary bladder were collected, fixed in 10% neutral-buffered (pH 7.2) formalin for 48 h, and processed routinely to produce 4 µm-thick hematoxylin and eosin sections.
Ancillary tests and molecular identification
Several ancillary tests were performed for the retrospectively sought and prospectively collected raccoons following CAHFS standard operating procedures (SOPs), including immunohistochemistry (IHC) for CDV in small intestine and/or lung, CPV in the small intestine, and Toxoplasma spp. in brain sections, and a fluorescent antibody test for rabies on brain, per the SOP of the San Bernardino County Public Health Department. Modified Ziehl–Neelsen staining was performed on fecal smears of both raccoons. Samples of small intestine collected during autopsy of raccoon 1 were placed in half-strength Karnovsky fixative, and thin sections were processed for transmission electron microscopy (TEM). Following aldehyde fixation, tissues were washed in 0.2 M sodium cacodylate, fixed again in 2% osmium tetroxide reduced with 2.5% potassium ferrocyanide, dehydrated in ethanol, and infiltrated and embedded in Spurr epoxy resin. Sections of 1 μM were placed on glass slides and stained with toluidine blue. Thin sections were then mounted on 150-mesh copper grids, stained with 6% methanolic uranyl acetate, followed by Reynold lead citrate stain, and observed in a transmission electron microscope (LEO 906E; Zeiss) at 60 kV accelerating voltage. Aerobic cultures, coprologic examinations, and rabies testing were also performed on both raccoons.
Mucosal scrapings were collected from the small intestine of raccoon 1 during autopsy. A sample (~200 mg) was processed for DNA extraction (QIAamp DNA stool mini kit; Qiagen) following the manufacturer’s instructions. Cryptosporidium spp. identification was determined by a nested PCR assay targeting a fragment of the 18S ribosomal RNA, based on a method described previously. 39 The first PCR assay targeted an ~1,325-bp fragment using the primers 5′-TTCTAGAGCTAATACATGCG-3′ and 5′-CCCATTTCCTTCGAAACAGGA-3′. In the second PCR assay, an ~850-bp fragment was amplified using the primers 5′-GGAAGGGTTGTATTTATTAGATAAAG-3′ and 5′-AAGGAGTAAGGAACAACCTCCA-3′. 39 DNA extracted from C. parvum oocysts obtained from The Cryptosporidium Production Laboratory, University of Arizona (Tucson, AZ), was used as positive control in all reactions. PCR products were fractionated on 2% agarose gel and visualized by ethidium bromide staining. Amplicons obtained in the second PCR assay were purified using a QIAquick gel extraction kit (Qiagen) following the manufacturer’s instructions. The extracted products were then sequenced (Prism 3730 genetic analyzer; Applied Biosystems). The obtained sequences were aligned and subjected to homology searching using BLAST (https://blast.ncbi.nlm.nih.gov/Blast.cgi). A phylogenetic tree was constructed using the neighbor-joining method, with evolutionary distances computed using the Tamura–Nei model, and 1,000 bootstraps replicated using MEGA7 software. 21
Retrospective analysis
The database of CAHFS was searched for all raccoon submissions to any of the branches of the system (Davis, Turlock, Tulare, San Bernardino) between January 1990 and December 2019. Inclusion criteria included raccoon submissions of any age and sex, in which an autopsy and histopathology were performed. This search was further refined to identify submissions with a diagnosis of cryptosporidiosis, CPV, and/or CDV infection.
Retrospective molecular identification of Cryptosporidium skunk genotype
A PCR assay was developed to amplify a specific, short (~116 bp) fragment of the gp60 gene of Cryptosporidium skunk genotype (CSkG) on formalin-fixed, paraffin-embedded (FFPE) small intestinal sections from raccoons. The following set of primers was used: 5′-TCATCGAGTGATCAAGTGGGT-3′ and 5′-TTGCCGGTCTCCTGGGTA-3′. The reaction was performed in a total volume of 25 µL containing 0.5 µL of each primer (0.5 µM), 2 µL of extracted DNA, 10 µL of nuclease-free water, and 12 µL of PCR master mix 2× (Promega), which contains Taq DNA polymerase (pH 8.5, 50 U/mL), dNTPs (400 µM), and MgCl2 (3 mM). Thermocycler profiles were as follows: 95°C for 8 min, 35 cycles of 95°C for 35 s, 52°C for 35 s, and 72°C for 35 s, and a final extension step at 72°C for 6 min.
Results
Clinical history and macroscopic findings
Raccoon 1 was a 3-mo-old, wild-caught female with a history of diarrhea, lethargy, dysphagia, and ocular and oral discharge. This raccoon was held for an undisclosed length of time in a wildlife rehabilitation facility with its sibling, who displayed similar clinical signs. The condition of the raccoon declined rapidly, and it was euthanized. The carcass was in fair nutritional condition with moderate muscling and fat stores. The perianal fur was matted with light-yellow fecal material. Both lungs were diffusely and moderately edematous and had multifocal-to-coalescing areas of pale pink discoloration and increased consistency. The lumen of the distal portion of the trachea and main bronchi contained fibrin deposits. Minimal contents were found in the small intestine, and the large intestine contained small amounts of light yellow-green pasty contents.
Raccoon 2 was a wild-caught male juvenile cared for at a wildlife rehabilitation facility for ~1 mo, developing upper respiratory signs, hyporexia, and weight loss during the first week of capture. The raccoon was euthanized given its unresponsiveness to treatment. The nutritional condition was good, with adequate muscling and ample visceral fat stores. About 30–40% of both lungs were multifocally dark red and had increased consistency; these lesions were most marked in the caudal lobes. No other significant gross lesions were observed in the carcass.
Microscopic findings
Histologically, the mucosa of the small intestine in raccoon 1 had diffuse villus atrophy and multifocal infiltration of eosinophils in the lamina propria. Attached to the apical membrane of the intestinal epithelial cells were multiple, 5–10 µm, round, basophilic structures (Fig. 1). Multifocally, the crypts were dilated, and often contained sloughed epithelial cells, cellular debris, and small numbers of neutrophils and eosinophils in the lumen (Fig. 2). Intestinal and splenic lymphoid follicles were mildly to moderately depleted. The pulmonary interstitium was multifocally infiltrated by lymphocytes and plasma cells, and further expanded by edema causing a reduction of alveolar spaces. Nearby bronchi, bronchioles, and peribronchial glandular lumens were filled with sloughed epithelial cells, often containing intranuclear and intracytoplasmic, 5–15 µm, oval-to-round, smudgy, eosinophilic inclusion bodies (Fig. 3). Microscopic findings in the intestine and lungs of raccoon 2 were similar to those described for raccoon 1. In the brain of raccoon 2 there were small, multifocal lymphocytic infiltrates throughout the neuropil, and neurons contained intranuclear and intracytoplasmic inclusions. These inclusions were also observed within macrophages, tracheal epithelium, renal pelvic urothelium, and within biliary epithelial cells of the liver in raccoon 2.
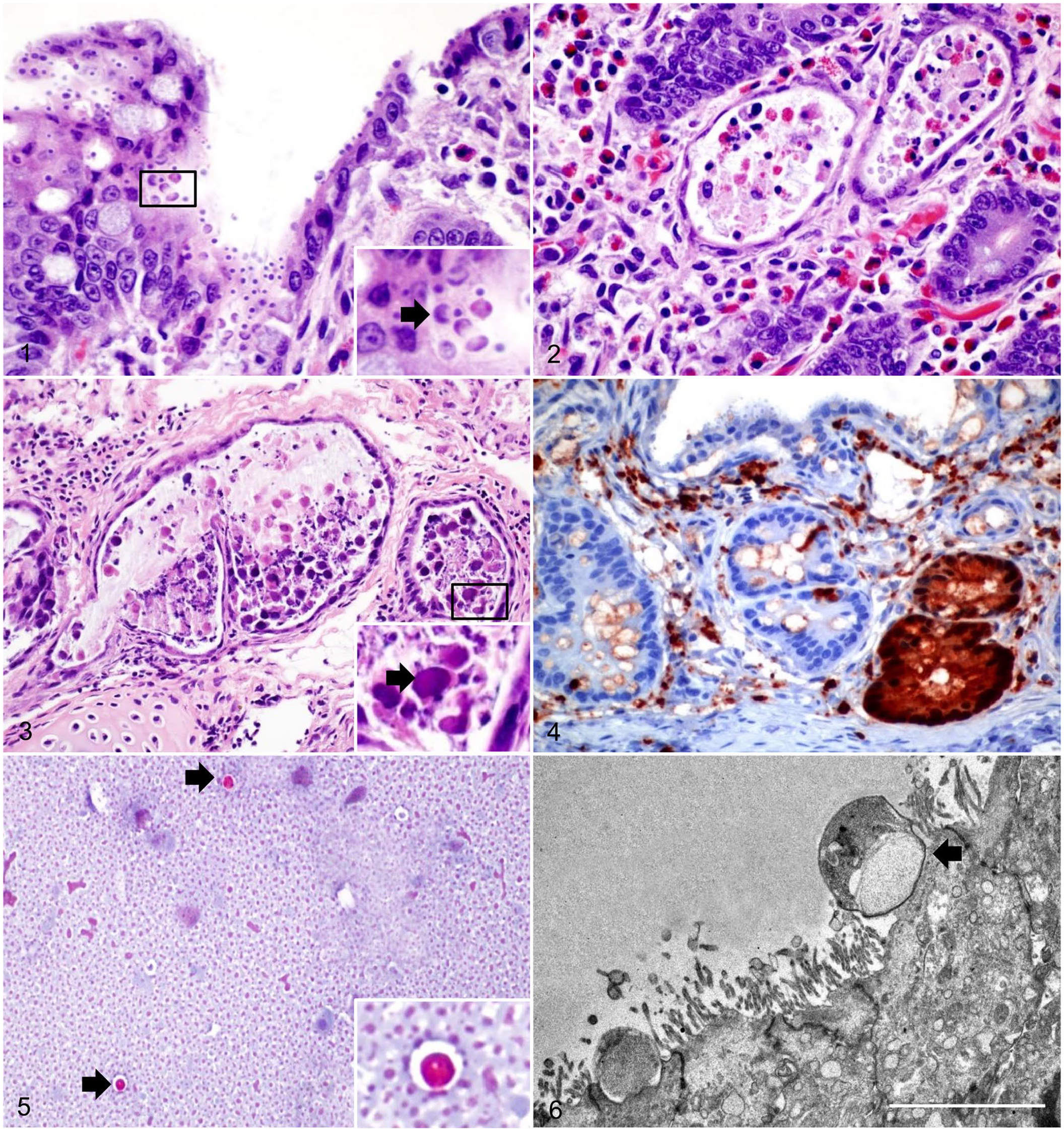
Cryptosporidiosis in raccoons.
Ancillary tests and molecular identification
There was strong cytoplasmic and nuclear immunolabeling in bronchial, bronchiolar, and peribronchial glandular epithelial cells for CDV in lung and in crypt epithelial cells in the intestine for both raccoon 1 and 2 (Fig. 4). No immunolabeling was detected for Toxoplasma in brain tissue (raccoon 1) or CPV in the intestine of either animal. Rare numbers of Cryptosporidium spp. were observed on fecal smears of both raccoons stained with modified Ziehl–Neelsen (Fig. 5). In the small intestine, TEM demonstrated small, round protozoa compatible with Cryptosporidium spp. (Fig. 6). These organisms were observed attached to the apical portion of enterocytes, causing loss of microvilli. In raccoon 1, moderate numbers of Escherichia coli in pure culture were isolated from the lungs. No aerobic bacteria were isolated from the lung of raccoon 2. Rabies testing was negative for both raccoons. Cryptosporidium sp. was detected in the small intestine of raccoon 1 by PCR, and the sequenced amplicon clustered with CSkG (Fig. 7).
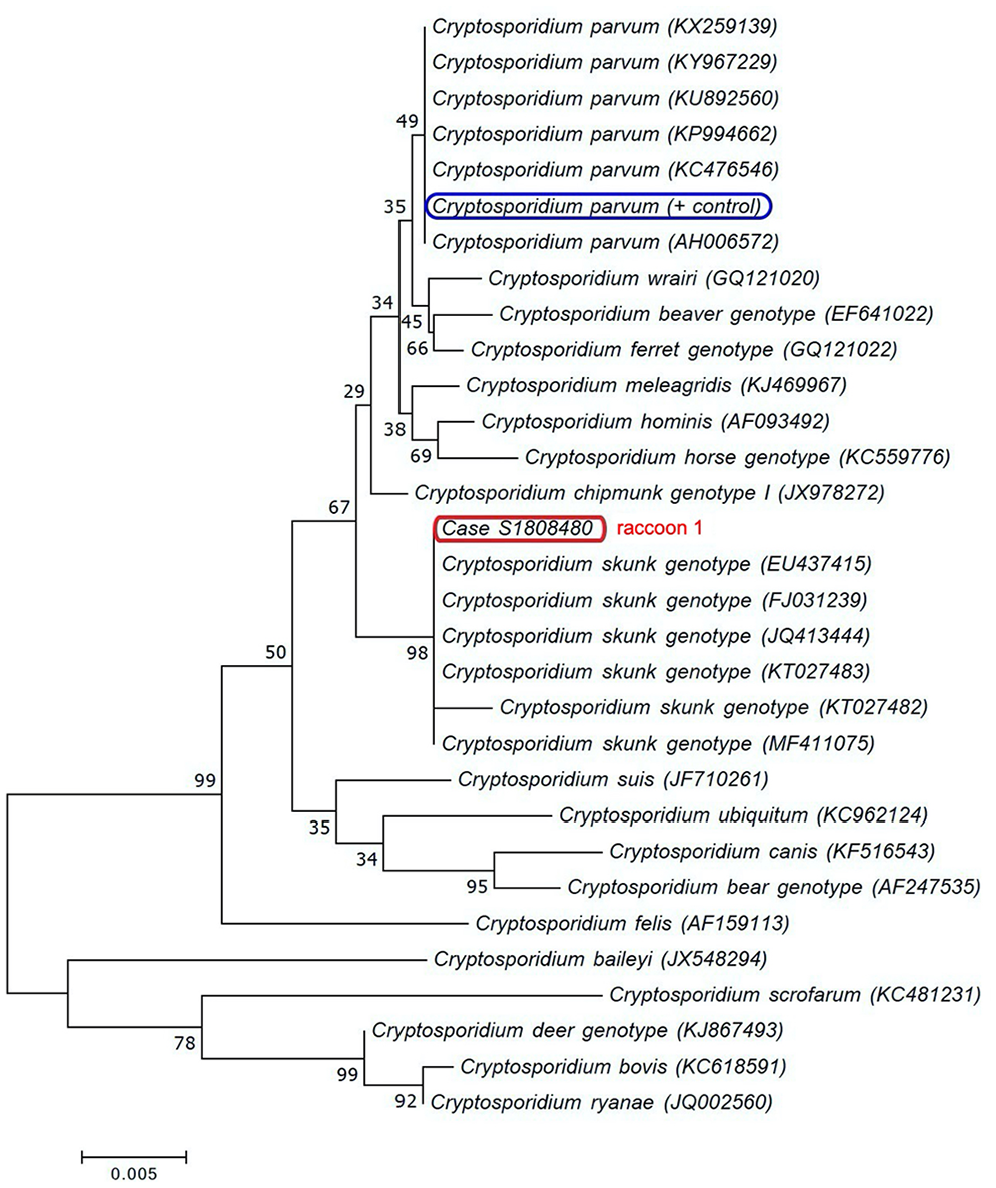
Phylogram of Cryptosporidium species and genotypes, inferred by the neighbor-joining method using partial 18S rRNA gene sequences, for species and genotypes affecting the small intestine. The apicomplexan detected in raccoon 1 clustered with the Cryptosporidium skunk genotype. Scale bar represents substitutions per nucleotide, and numbers below the branches indicate bootstrap values (>1,000).
Retrospective analysis
CAHFS laboratories received 318 raccoon submissions for autopsy between 1990 and 2019; cryptosporidiosis was diagnosed in 8 raccoons (including the 2 described above). Relevant clinical history details of retrospectively collected raccoons indicate that all were wild-caught, with 5 found severely sick with ocular-nasal discharge, extreme lethargy, and/or neurologic signs. Only 1 was held at a wildlife rehabilitation facility for 5 wk prior to developing clinical signs. A total of 64 (33.2%) and 13 (6.7%) raccoons were diagnosed with CDV and CPV, respectively, including 6 CDV and 5 CPV presumptive, unconfirmed infections. Also, the vast majority of the cryptosporidia-positive raccoons were received in CAHFS San Bernardino toward the end of the study period assessed (1 each in 2000, 2008, 2011, 2015, and 2017, and 3 in 2018). A similar trend was observed for the CDV and/or CPV diagnoses made at CAHFS Davis. Most of the raccoons were juveniles; no sex predilection was observed. Seven of 8 raccoons with cryptosporidiosis were coinfected with CDV, confirmed either via IHC or virus isolation. The remaining raccoon was positive for cryptosporidiosis and CPV. CSkG was identified in 6 of 8 positive samples from CAHFS San Bernardino when targeting the gp60 gene by PCR on FFPE sections (Table 1).
Summary of relevant histopathologic findings associated with raccoon cryptosporidiosis associated with Cryptosporidium skunk genotype (CSkG), and infection status by canine distemper and parvovirus for raccoons submitted for autopsy to the California Animal Health and Food Safety Laboratory, 2000–2018.

CDV = canine distemper virus; CPV = canine parvovirus; F = female; J = juvenile; M = male; NP = not performed; SA = subadult; U = unknown. Dash (–) = negative or not observed histologically.
Raccoon 1.
Raccoon 2.
Confirmed via virus isolation.
Discussion
We confirmed cryptosporidia, specifically CSkG, in synanthropic raccoons in southern California submitted to CAHFS, 1990–2019. CSkG was first detected from striped skunks in California and was given this name given its presumed host-specificity. 40 Through multi-locus characterization and phylogenetic analyses, it was determined that host adaptation was a general phenomenon of Cryptosporidium given that specific genotypes tend to be detected in similar groups of animals, and that genetic heterogeneity might be an inevitable result of host-parasite coevolution. 40 To date, there are 20 valid species recognized for Cryptosporidium, with most mammalian infections caused by C. parvum.4,7 Molecular studies have determined the existence of non–C. parvum genotypes, such as CSkG, a wildlife-adapted genotype previously thought to only infect wild or zoo animals,6,8,10,22,29 and to lack zoonotic significance. 44 However, CSkG was later determined to have a broader host range after being isolated from multiple squirrel species, river otters, and opossums in the United States,35,45 from raccoons in Japan, 15 and even in cases of human diarrheal disease in the United States and United Kingdom.8,29 Just as with C. parvum, CSkG may act more in concordance with a group of subtypes (14 to date) and subtype families (4 families) without evidence for host adaptation. 40 In our study, different attempts at molecular identification failed to detect C. parvum or other protozoal species in the 2 raccoons negative for CSkG, which is in part attributed to the limitations of FFPE tissues for genotyping purposes. Despite this issue, C. parvum should still be considered in the differentials given its high prevalence in other mammalian species.4,7
All CSkG cases originated from southern California, one of the most densely populated regions of the state and of the country. 37 Although this might be the reflection of a truly broad distribution and high prevalence of CSkG infection among North American raccoons (within both native and introduced ranges),15,23,41 other potential explanations for high infection frequency were considered. Environmental factors that may have contributed to intrinsically skew or spatially restrict our CSkG findings to southern California raccoons include increased habitat comingling or shared water sources with other known synanthropic reservoirs, population density leading to increased reporter bias, and proximity to a laboratory in densely populated regions of the state.
We also considered that permanence at a wildlife rehabilitation facility facilitates pathogen transmission among wildlife. 17 Nonetheless, 6 of 8 raccoons in our study displayed severe signs at the time of capture, strongly suggesting that at least distemper infections were natural and occurred prior to captivity. CSkG has a broad host range, a factor that appears to facilitate the persistence of this parasite in surface water in the United States and Canada.34,42 The large proportion of desert or barren biomes in southern California may also influence transmission, where limited watersheds could boost artificial concentration of wildlife, enhancing parasite shedding and water-borne transmission compared to the northern part of the state.
In our study, almost all of the CSkG-positive raccoons were also infected with CDV, a highly pathogenic virus that is spread by body excretions and secretions (urine). Its fatality rate is only overtaken by rabies in domestic dogs, making it an important veterinary pathogen. 36 CDV is capable of inducing virulent disease in a range of mammals, particularly in juveniles, exhibiting epithelio-, lympho- and neurotropism causing systemic infection in almost all organ systems. 24 Clinical presentation in procyonids is similar to that of domestic dogs, and includes cystitis with pyuria, 26 as well as diarrhea, dyspnea from severe pneumonia, neurologic signs, jaundice, and profound immunosuppression.3,20 We hypothesize that the pantropism of CDV combined with severe CDV-induced immunosuppression enhances the vulnerability of raccoons to acquire cryptosporidia and suffer severe disease from a parasitic infection that would otherwise be self-limiting. In addition, the combination of young age 11 and infection with an enteropathogen is likely to shorten the onset, delaying the cessation, and prolonging the duration of CSkG shedding given impaired immune status, as has been demonstrated in calves. 43
As reported previously, opportunistic pulmonary infections are common, 5 such as in raccoon 1 in which Escherichia coli was isolated. If animals overcome the CDV infection, they can develop lifelong immunity or die within a short time post-infection. 1 Natural and vaccine-induced CDV infections have been reported in all families of terrestrial carnivores.2,31 Spillover of domestic animal viruses to competent wild hosts is difficult to control and leads to almost inevitable establishment in the population as well as epidemics. In the United States, there are regular epidemics represented as 4-y distemper cycles in free-ranging raccoons, indicating that the infection is endemic in some North American raccoon populations.16,32 Thus, epidemics in raccoons play a role in the CDV epidemiology in domestic dogs and non-domestic zoo populations.5,26 CDV epidemics in wildlife occur given that environmental factors (cold temperatures, high moisture) positively influence the persistence of the virus outside susceptible hosts; once infected, these hosts either overcome the infection or die. Spillovers of CDV and CDV-like viruses into canid and non-canid carnivores also carry significant conservation impacts, such as the large-scale, fatal epidemics in free-ranging African felids. 31 CDV prevalence varies widely in raccoon populations across the United States, ranging from 22% in New York, 23% in Illinois, 55% in Florida, and 84% in Maryland.16,18,25,27
Six of 8 raccoons were positive to CSkG in our study, the majority (5 of 6) of which were also coinfected with CDV. Common histologic correlates in cases of infection by CSkG and CDV included enteritis and/or crypt necrosis, encephalitis, pneumonia, and lymphoid depletion. The literature documents important infections concomitant with CDV infections in wildlife, including toxoplasmosis in martens, listeriosis in a badger, and rabies in raccoons.3,14
Another documented infection concomitant with CDV is cryptospodiosis, 5 which appears to exacerbate gastrointestinal signs. This further supports the hypothesis that increased vulnerability to infection by other pathogens is likely secondary to profound CDV-driven immunosuppression. Frequently, there was an underlying viral disease in all of our cryptosporidiosis cases, often CDV and rarely CPV. Changes to the crypts similar to those observed in our cases (e.g., crypt necrosis) are considered classic for CPV infection. However, there was evidence of CPV infection in only one of our cases. At least for the raccoons tested in our study, findings suggest that Cryptosporidium infection worsened by a concomitant CDV can mimic lesions thought to be classical for other agents, such as CPV. Although the literature states that solely Cryptosporidium infection can cause marked crypt dilation with necrotic debris or crypt necrosis and/or worsened by concomitant CDV infection,7,13,28 crypt necrosis is still not generally considered a classic morbilliviral or cryptosporidial lesion; thus, we believe the search for an underlying enteric virus is justified in cases in which crypt necrosis is found along with apicomplexan infections.
Footnotes
Acknowledgements
We thank Joseph Pesavento for the electronic microscopy images, J. Beingesser for histology and immunohistochemistry support, and P. Yant for microbiologic and coprologic analyses.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
